Abstract
Traumatic experiences and fetal development influence tryptophan (TRP) and its neuroactive byproduct, kynurenic acid (KYNA). Maternal TRP metabolite levels during pregnancy vary by fetal sex, with higher concentrations in mothers carrying male fetuses. This pilot study aimed to explore the relationship between offspring sex, maternal childhood trauma, and maternal salivary KYNA and TRP levels during pregnancy. Liquid chromatography-tandem mass spectrometry (LC-MS/MS) was used to determine KYNA and TRP levels in maternal saliva samples collected from 35 late-pregnancy participants. Maternal childhood trauma was assessed using the Childhood Trauma Questionnaire, including subscales for emotional abuse, physical abuse, sexual abuse, emotional neglect, and physical neglect. Among mothers pregnant with boys, salivary KYNA significantly correlated with physical and emotional neglect, and salivary TRP with emotional neglect. No significant correlations were found in mothers who delivered female offspring. Significant associations of childhood trauma and offspring sex were found for salivary KYNA but not TRP concentrations. Mothers with higher trauma levels who delivered boys exhibited higher levels of salivary KYNA compared to those with lower trauma levels. Moreover, mothers with higher trauma levels who delivered boys had higher salivary KYNA levels than those with higher trauma levels who delivered girls. This pilot study provides evidence of an association between maternal childhood trauma and TRP metabolism, measured in saliva, especially in mothers pregnant with boys. However, longitudinal studies with larger sample sizes are required to confirm these results.
Introduction
Maternal childhood trauma measures the presence of emotional abuse, physical abuse, sexual abuse, emotional neglect, and physical neglect in childhood until 18 years of age, 1 affecting approximately one-third of pregnant women2 -4 with adverse effects on fetal development, child growth, and behavioral problems, such as internalizing and externalizing difficulties.4,5
In the last decade, a growing body of literature has explored the impact of fetal sex on various biological processes in pregnant women. For instance, studies have shown differences in the production of pro-inflammatory cytokines with higher levels of maternal serum cytokines in male pregnancies,6,7 an increased risk of adverse pregnancy outcomes in male pregnancies, 8 higher risk of diabetes mellitus 9 and increased levels of maternal salivary cortisol in women carrying male fetuses between weeks 24 and 30, 10 and higher concentrations of kynurenine and kynurenic acid (KYNA) in serum of mothers pregnant with boys compared to mothers pregnant with girls. 11 Additionally, it has been observed that exposure to adverse psychological events may have different effects on male and female fetuses, with male fetuses exhibiting greater vulnerability to prenatal adversity.12 -14 Despite the increasing relevance of sex differences in perinatal medicine, studies investigating fetal sex as a moderator for the associations between maternal biological processes and negative psychological experiences, such as maternal childhood trauma, are still lacking.
The importance of the kynurenine branch of tryptophan metabolism during pregnancy and its relationship to stress and inflammation for the psychological functioning of the mother and the newborn are not sufficiently elucidated. It has been suggested that stress during pregnancy induces changes in tryptophan (TRP) metabolism and could have long-term effects on behavior.15 -17 Our previous studies have shown that TRP, kynurenine, and KYNA are present at different concentrations in the human placenta, fetal membranes, and umbilical cord, indicating their local compartmentalization, which may have physiological and pathological significance. 18 In addition, our recent study has shown that administering kynurenine to pregnant female rats at embryonic days 1 to 7 or exposure of zebrafish larvae to kynurenine leads to changes in neurobehavioral development. 19 The effects of kynurenine administration may have long-term consequences, as animals exposed to kynurenine during the embryonic period or lactation showed behavioral abnormalities in adulthood.19,20
KYNA, a byproduct of kynurenine degradation, has been identified as a marker that directly correlates with stressful events.21,22 KYNA is present both in the peripheral and central nervous systems and plays a role in the metabolic activity of microglia. 23 It is relevant for the psychological stress response. 21 Furthermore, recent studies have demonstrated a correlation between salivary KYNA levels and acute stress response in humans, 22 suggesting that changes in KYNA levels in response to stress can be measured in saliva.22,24 Regarding previous negative experiences and chronic stressors, such as childhood trauma, only one pilot study to date has reported significant associations between serum KYNA concentrations and scores of the Childhood Trauma Questionnaire (CTQ). 25 Although maternal stress has also been associated with alterations in the TRP breakdown pathway,26,27 there are no human studies that examine the role of previous traumatic experiences, such as maternal childhood trauma, as chronic stressors on the TRP breakdown pathway in maternal saliva during pregnancy. Regarding the assessment of metabolites of the kynurenine pathway, previous studies reported correlations between plasma and salivary TRP concentrations. 28 Subsequently, individual reports were published in which the determination method was improved, and the relevance of measuring TRP metabolites in saliva was confirmed, including also in children and infants.29 -31
To this end, the present study aimed to explore the correlations between salivary tryptophan metabolites (KYNA and TRP) and maternal childhood trauma, including emotional abuse, physical abuse, sexual abuse, physical neglect, and emotional neglect, as assessed by the CTQ. Additionally, the study aimed to examine how these correlations might be moderated by fetal sex in pregnant women. Finally, the present study explored the associations between maternal childhood trauma and salivary TRP metabolite concentrations, with fetal sex as a moderator and age as a covariate, given the potential age-related variations in TRP metabolites. 32 Firstly, we hypothesized that salivary KYNA and TRP levels are associated with maternal childhood trauma assessed by the CTQ (factors for each CTQ subscore: physical abuse, emotional abuse, sexual abuse, physical neglect, and emotional neglect) when controlling for fetal sex. Furthermore, we hypothesize that maternal childhood trauma, when inferred by fetal sex, associated with TRP metabolite concentrations in saliva, with an expected difference observed in the group with male offspring.
Materials and Methods
Study design, selection criteria, and study participants
The following pilot study was conducted at Marienhaus Klinikum Hetzelstift (Neustadt an der Weinstraße, Germany) and included adult pregnant women undergoing planned primary cesarean sections. Inclusion criteria required participants to be between 18 and 50 years old, capable of providing consent, and fluent in German. Exclusion criteria were significant genetic findings (eg, trisomy), major child malformations or disabilities, inability to provide consent, pregnancy under legal supervision, multiple pregnancies, and maternal infection with HIV, hepatitis B, or hepatitis C. Detailed information about the study objectives was provided to all participants, who gave both written and oral consent. CTQ was administered to participants in the days leading up to the scheduled cesarean section.
Maternal saliva samples were collected using a Salivette® (Sardstedt Laboratories, Nuernberg, Germany) at 10 p.m. on the evening before the cesarean section. These samples were immediately stored at approximately 4°C, and the next day, they were frozen at −80°C and transferred to the Central Institute of Mental Health for further analysis.
The study adhered to the principles outlined in the Helsinki Declaration and obtained ethical approval from both the Ethics Committee of the Medical Faculty Mannheim of the University of Heidelberg and the Ethics Committee of the State Chamber of Medicine in Rhineland-Palatinate.
Assessment of maternal childhood trauma: Childhood Trauma Questionnaire
Maternal childhood trauma was evaluated using the German version of the CTQ. 33 This self-administered questionnaire comprises 28 items and assesses 5 distinct aspects of childhood trauma: physical abuse, emotional abuse, sexual abuse, physical neglect, and emotional neglect. Each item is rated on a 5-point Likert scale (1 = never true; 5 = very often true), with higher scores indicating greater levels of abuse or neglect. 33 Moreover, the subscales of the CTQ have different cut-offs (physical abuse = 8, emotional abuse = 10, sexual abuse = 8, physical neglect = 8, and emotional neglect = 15).2,33 The CTQ has good validity and internal consistency (Cronbach’s alpha ⩾.80). The construct validity is supported by positive correlations with measures of anxiety/and depression and negative correlations with life satisfaction. The German version of the CTQ is reliable and valid for evaluating childhood trauma.
To analyze the severity of maternal childhood trauma stress in our sample, we dichotomized the total scores of the CTQ into 2 categories based on a median split (median = 31 points), as recommended and used in the literature.34 -36 The 2 categories were defined as lower (⩽31 total CTQ points) and higher maternal childhood trauma (>31 total CTQ points).
LC-MS/MS assessment of TRP and KYNA in saliva
Salivary KYNA and TRP concentrations of 35 pregnant women were analyzed using liquid chromatography with tandem mass spectrometry (LC-MS/MS) at the Department of Bioanalytics of the Medical University of Lublin (Poland). Analytical stable isotope-labeled standards of TRP [13C11, 15N2] and KYNA [ 2 H5] (Alsachim, Illkirch-Graffenstaden, France) were prepared as stock solutions, achieving a concentration of 1000 mg/L. Internal standard working solutions were prepared in methanol. Ultra-pure water was obtained from the Millipore Direct-Q3-UV purification system (Merck KGaA, Germany).
The collected saliva samples were stored initially at −80°C. On the analysis day, 25 µL of the working solution of isotopically labeled internal were added to 200 µL of each sample. The mixture was vortexed with 575 µL of chilled methanol/ethanol (1:1 v/v) for 30 seconds. Subsequently, the samples were placed at −20°C for 15 minutes and centrifuged at 12 100×g for 30 minutes. The resulting supernatants were diluted with ultrapure water (1:1 v/v) and filtered through a 0.2 μm Titan Syringe Filter RC (Thermo Fisher Scientific, Waltham, USA) into autosampler vials.
The determination of salivary TRP and KYNA was carried out using an Agilent Technology liquid chromatograph 1290 Infinity II series coupled with an Agilent Technologies triple quadrupole mass spectrometer 6470 QQQ LC/MS equipped with a Jet Stream ion source. A detailed description of technical aspects of the LC-MS/MS analysis is published elsewhere. 18
Statistical analysis
Quantitative variables were presented as mean (standard deviation) or median (interquartile ranges) in the tables, while categorical variables and count data were presented as numbers or fractions. Data were rounded to the nearest decimal point, and values smaller than 0.01 were presented as <0.01. As the data of the TRP metabolites were not normally distributed, we log-transformed the variables KYNA and TRP.
Spearman rank correlations (Spearman’s ρ) were computed to assess the relationships between variables. Pairwise correlations between the concentrations of TRP metabolites and all other variables were calculated separately for each group (ie, male and female offspring). This approach was chosen to avoid potential incorrect or misleading conclusions regarding the underlying relationship between the variables, as examining the total correlation coefficient alone may yield inaccurate results. 37 Regarding significance tests, adjustments for multiple testing were performed for each TRP metabolite by comparing the corresponding P-values with a threshold value of α = .05, which was Bonferroni-adjusted based on the number of CTQ subscales. Consequently, a significance threshold of Pb ⩽ .010 was established for this analysis to define statistical significance.
Two separate generalized linear models were conducted to determine the relationship between maternal childhood trauma (higher vs lower trauma) and offspring sex (male and female offspring), as well as the 2-way interaction (maternal childhood trauma × offspring sex) on salivary KYNA and TRP concentrations, using maternal age as a covariate. If significant, 2-way interactions were adjusted for multiple comparisons (Bonferroni) in the generalized linear model settings. In this case, significance tests were calculated for these associations, defining statistical significance with a two-tailed P ⩽ .050.
Although this study is an exploratory pilot study, a priori power analysis was conducted using the G-Power, 38 indicating a required sample size of n = 35 to overcome the threshold 1−β = 84.74% recommended in the literature 39 with a large effect (f2 = 0.40), and a significance criterion of α = .05. The sample size of this pilot study also fulfills the general rule of 30 patients to estimate a parameter.40,41
Finally, statistical analyses were conducted using SPSS version 25.0 (Statistical Package for the Social Sciences, International Business Machines Corporation, New York, United States of America). Model graphs were generated using Prism 8 GraphPad (GraphPad Software Inc., California, United States of America).
Results
General characteristics of the sample
Pregnant women near term (n = 35), with a mean age of 32.46 ± 4.65 years, who underwent elective cesarean section at a mean of 38.83 ± 0.68 gestational weeks, were analyzed in this study. The mean log-transformed salivary concentrations of KYNA were 0.02 ± 0.41 ng/mL, while TRP concentrations were 2.75 ± 0.49 ng/mL. Among the participants, 48.60% (n = 17) gave birth to male offspring, and 51.40% (n = 18) gave birth to female offspring. Regarding childhood trauma levels, 54.30% (n = 19) of the pregnant participants exhibited lower levels, while 45.70% (n = 16) showed higher trauma levels. More details concerning the general baseline characteristics of the 35 study participants are presented in Table S1.
Correlations between CTQ subscales and tryptophan metabolites
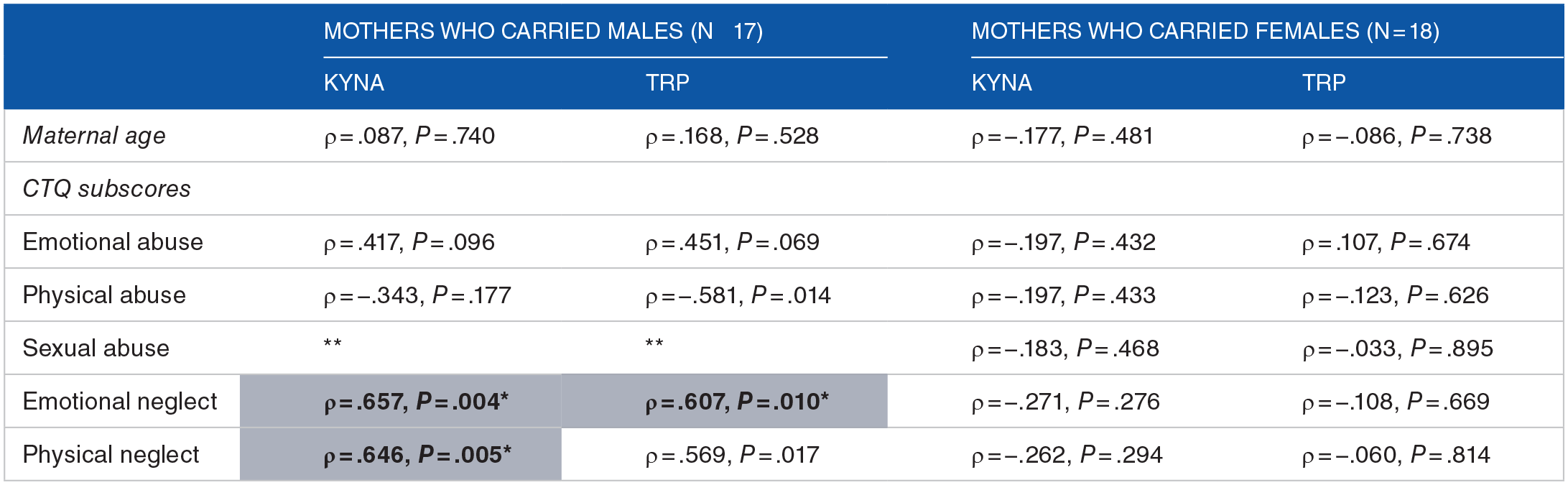
In the group of women who delivered boys, significant correlations were observed between KYNA concentrations and physical neglect scores (ρ = .646, Pb = .005), as well as between KYNA concentrations and emotional neglect scores (ρ = .657, Pb = .004). Regarding salivary TRP, a significant correlation was found only with emotional neglect scores (ρ = .607, Pb = .010). No correlations were observed between the remaining CTQ subscales and salivary KYNA or TRP concentrations in pregnant women who delivered male offspring (Table 1). No significant correlations were found between salivary KYNA concentrations, TRP concentrations, and the CTQ subscales in women who delivered girls (Table 1). Finally, no significant correlations were found between maternal age and salivary KYNA or TRP concentrations.
Spearman correlations table between CTQ subscales and tryptophan metabolites measured in maternal saliva. Salivary KYNA and TRP values were log-transformed. *Significant P-values were bold-marked, with a star tagged and gray-shaded after Bonferroni correction (significance threshold for the 5 subscores: P = .010). **Pairwise comparison had no variability, becoming the denominator for the correlation formula zero. In this case, all mothers who carried males (n = 17) had a total score for CTQ-sexual abuse of 5, that is, all 5 questions for this factor were rated with the lowest grade (1).

Abbreviations: CTQ, Childhood Trauma Questionnaire; KYNA, kynurenic acid; TRP, tryptophan.
Relationship of childhood trauma and sex of the offspring on tryptophan metabolites
Concerning the generalized linear model for KYNA, the 2-way interaction of maternal childhood trauma × offspring sex was statistically significant (Table S2). Generalized linear model post hoc Bonferroni comparisons indicated that, within the mothers who delivered boys, the salivary KYNA of the participants with higher trauma levels was significantly higher than those with lower trauma levels (Figure 1, Tables S3 and S4). In addition, among the mothers with higher trauma levels, salivary KYNA was significantly higher in mothers who delivered boys than those who had girls. Finally, the generalized linear model for salivary TRP also showed an association with the 2-way interaction of maternal childhood trauma × offspring sex on TRP (Table S5). However, there were no significant differences after correcting for multiple comparisons (Figure 1, Tables S6 and S7).
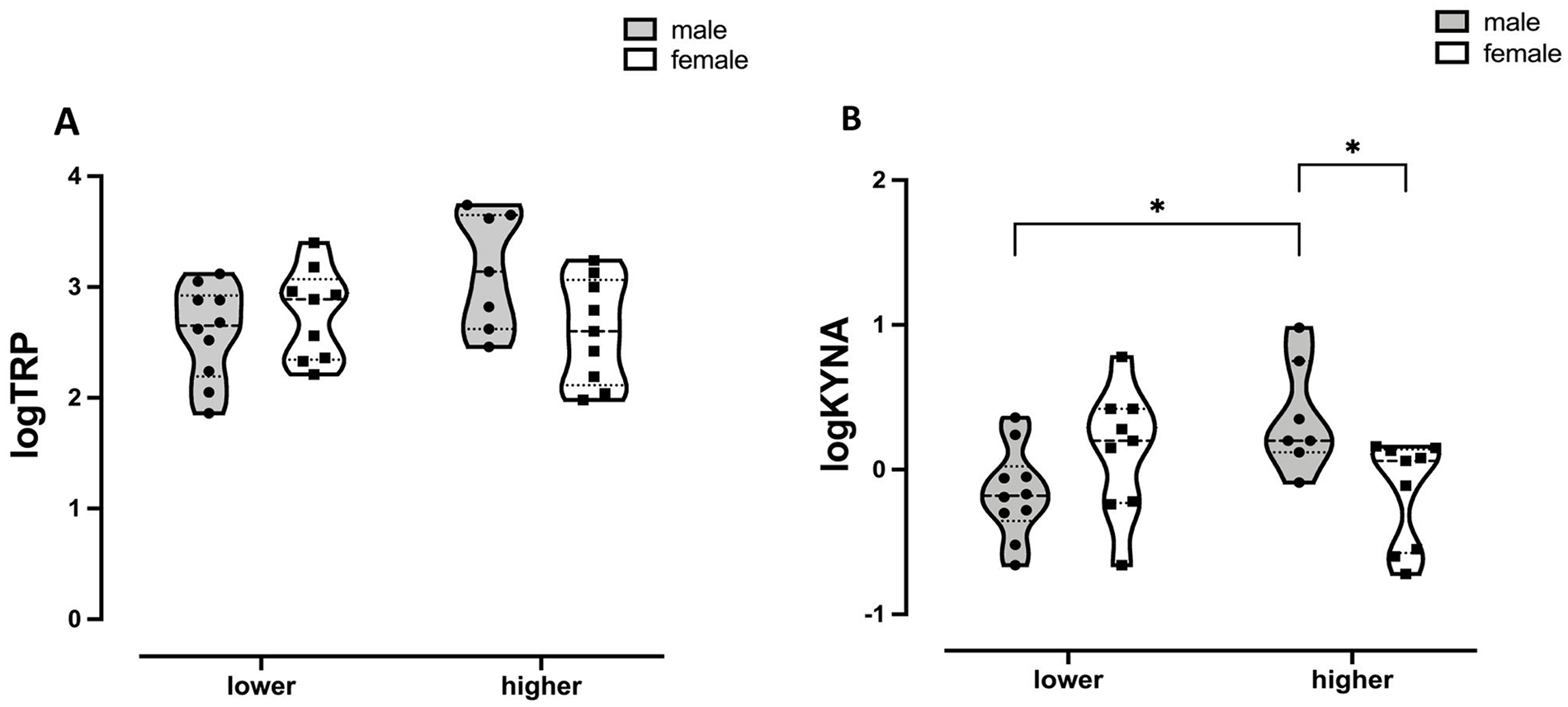
Violin plot of the log-transformed maternal salivary TRP concentrations (A, left) and (B, right) maternal salivary KYNA concentrations with post hoc comparisons. The interactions fetal sex × maternal childhood trauma levels were significant for both tryptophan metabolites (KYNA: P = .002; TRP: P = .02). Maternal childhood trauma levels were calculated using the median split for the total CTQ scores and represented in the x-axis of the bar graph as “lower” (⩽31 total CTQ points) and “higher” (>31 total CTQ points).
Discussion
The association of maternal childhood trauma with KYNA and TRP remains poorly understood and underreported. Additionally, there is a lack of human studies that explore how fetal sex links to the modulation of maternal biological processes, including TRP metabolite levels, taking into account previous maternal burdens, such as traumatic experiences. Identifying modulating factors associated with pre-conceptional maternal chronic stress, such as past abuse or neglect, could facilitate early identification of groups at risk. This pilot study represents the first investigation of the modulation of fetal sex on the relationship between salivary KYNA and TRP concentrations and maternal childhood trauma in pregnant women.
Our exploratory analysis revealed significant associations between KYNA concentrations with emotional neglect and physical neglect and significant correlations between TRP concentrations and emotional neglect in pregnant women carrying male fetuses but not in women expecting female fetuses. To the best of our knowledge, this is the first study to report correlations between maternal childhood trauma and KYNA, as well as TRP concentrations during pregnancy, showing significant correlations in participants carrying male fetuses. Previous studies have shown that TRP metabolism along the kynurenine pathway is altered by stress and inflammation during pregnancy and postpartum. However, studies examining mood-related changes in TRP metabolism have yielded mixed results.42 -46 The literature on adverse childhood events, including maternal childhood trauma and salivary TRP metabolites, is limited, with a particular lack of studies during pregnancy, which is a particularly critical period for offspring development.
Among the few existing studies with childhood trauma using the CTQ and KYNA, Shovestul et al conducted an exploratory analysis that described a positive correlation between KYNA concentrations and emotional abuse in individuals with diagnosed schizophrenia, which was not replicated in a healthy control sample. 25 Additionally, Bonsignori et al reported a higher tryptophan breakdown index in affective disorders, specifically for the subscales of physical abuse, emotional neglect, physical neglect, and emotional abuse, but not in healthy control subjects. 47 It is well known that childhood trauma is associated with alterations in the serotonin system, which may contribute to an increased risk of suicide. 48 Moreover, KYNA is a biomarker strongly associated with stressful events.21,22 Animal models have demonstrated, for instance, that stress leads to increased KYNA concentration in the body over time, resulting in a generalized KYNA-dependent biological stress response.22,24 Therefore, it is plausible that maternal childhood trauma, acting as a chronic stressor, could dysregulate the serotonin system in pregnant women, leading to elevated KYNA levels.
We hypothesize that the differences observed in our data between mothers pregnant with male and female fetuses, with higher concentrations of KYNA found in male pregnancies, may be associated with a greater vulnerability to prenatal adversity, as has been previously suggested. 13 However, further studies with larger sample sizes must confirm our findings.
This study revealed sex-dependent differences. Only in women pregnant with male fetuses salivary KYNA concentrations were higher in women with higher levels of trauma compared to women with lower levels of trauma. Women with higher levels of trauma showed higher levels of salivary KYNA when carrying male fetuses compared to women with higher levels of trauma carrying female fetuses. A study by Egorova et al reported sex-dependent variances in TRP metabolism, showing higher peripheral KYNA levels in mothers pregnant with boys compared to those pregnant with girls. 11 Male pregnancies are characterized by elevated metabolic ratios in the TRP breakdown pathway, and the authors of the cited work suggested that increased TRP consumption does not explain the higher concentrations of peripheral TRP metabolites in male pregnancies. 11 However, Egorova et al did not investigate maternal childhood trauma in the sample of pregnant participants, nor did they explore fetal sex-dependent differences in maternal KYNA concentrations among mothers with previous traumatic experiences. Therefore, to our knowledge, this pilot study represents the first report indicating significant fetal sex differences in mothers with a history of traumatic experiences. Our findings suggest that previous traumatic experiences, including physical and emotional neglect, could be related to increased enzymatic activity in the TRP breakdown pathway (eg, through indolamine-2,3-dioxygenase 1 -IDO1- or kynurenine aminotransferases). This aligns with observed sex differences in TRP metabolism during pregnancy in previous animal models with chronic stress.49,50 Finally, we observed differences in salivary KYNA concentrations between mothers with higher and lower levels of trauma in male pregnancies. To the best of our knowledge, no studies have reported such sex-specific differences. Chronic stress and past traumatic events have been shown to increase the production of pro-inflammatory cytokines, which upregulate the enzyme IDO1, leading to increased production of KYNA and quinolinic acid, both of which are involved in NMDA transmission and neurotoxicity.51,52 Animal models have demonstrated that exposure to stressors increases KYNA levels in rats.53,54 Inhibiting KYNA synthesis has been shown to reduce fear discrimination impairment caused by stressors, highlighting the role of KYNA in stress response and traumatic experiences. Additionally, sex differences in KYNA production have been observed in pregnant women, with male pregnancies associating with increased KYNA production compared to female pregnancies. Given the association between traumatic experiences and male pregnancies, it is plausible that male pregnancies with higher trauma levels exhibit higher KYNA concentrations. Further studies are necessary to validate these findings and explore the influence of previous traumatic experiences on enzymatic activity in the TRP breakdown pathway, specifically concerning fetal sex differences, particularly in male pregnancies.
Although this pilot study has shed light on the association between perinatal maternal stress and KYNA concentrations, several limitations must be considered. Firstly, the study is a pilot study with small sample size. While the findings of this study cannot be extrapolated to larger population groups, the power obtained from this study with 35 participants was 1−β = .84, which surpasses the recommended threshold for pilot studies. 39 Secondly, the use of salivary KYNA may have limitations. Although stress reactions affect most organs, peripheral KYNA concentrations might not reflect cerebral levels. 22 The group with lower trauma levels had very low CTQ scores, which led to skewed distributions (see Supplemental Material). Including and analyzing other TRP breakdown enzymes or metabolites, such as kynurenine or quinolinic acid, could have provided more information on perinatal maternal stress. Finally, the study did not include other stress measurements (eg, through the Trier Social Stress Test) as a measure of perinatal maternal stress.
In light of our results, we conclude that the association of childhood traumatic experiences with salivary KYNA concentrations is moderated by fetal sex. Our findings demonstrate a significant correlation between emotional and physical neglect in TRP metabolites of mothers carrying males but not females. This highlights the importance of considering fetal sex in studies on maternal stress. Male fetuses are known to exhibit a greater vulnerability to prenatal adversity12 -14; the mechanisms behind this are not clearly understood. Our findings may represent one mechanism by which maternal stress links to altered vulnerability depending on fetal sex. Further studies are needed to explore the longitudinal consequences of changes in maternal KYNA and TRP concentrations on offspring’s development and investigate differences regarding sex and the effect of early maternal life stress on later psychopathology.
Supplemental Material
sj-docx-1-try-10.1177_11786469241244603 – Supplemental material for Fetal Sex as Moderating Factor for the Relationship Between Maternal Childhood Trauma and Salivary Kynurenic Acid and Tryptophan in Pregnancy: A Pilot Study
Supplemental material, sj-docx-1-try-10.1177_11786469241244603 for Fetal Sex as Moderating Factor for the Relationship Between Maternal Childhood Trauma and Salivary Kynurenic Acid and Tryptophan in Pregnancy: A Pilot Study by Bruno Pedraz-Petrozzi, Eva Kathrin Lamadé, Marta Marszalek-Grabska, Alicja Trzpil, Ole Lindner, Pascal Meininger, Emilia Fornal, Waldemar A Turski, Stephanie H Witt, Maria Gilles and Michael Deuschle in International Journal of Tryptophan Research
Footnotes
Acknowledgements
We thank Sara Montgomery for her valuable input and advice.
Author Contributions
BP-P: Data curation, formal analysis, writing - original draft, visualization. This author shares the first authorship with EKL. EKL: Data curation, formal analysis, writing - original draft, visualization. This author shares the first authorship with BP-P. MM-G: data curation, formal analysis, methods, writing – review and original draft. AT: data curation, methodology, and analysis. OL: data curation, writing – original draft. PM: data curation, writing – original draft. EF: data curation, methodology, writing – review and editing. WAT: data curation, methodology, writing – review and editing. SHW: conceptualization, methodology, resources, data curation, writing- review and editing, supervision. MG: data curation, conceptualization, writing – review, and editing. MD: conceptualization, writing – review and editing, supervision, project administration.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The following project was partially supported by the Ministry of Education and Science in Poland, within the statutory activity of the Medical University of Lublin (Project No. DS 670, granted to the author E.F.). Other authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.