Abstract
Frailty is defined as a syndrome of physiological decline in late life, characterized by marked vulnerability to adverse health outcomes. A robust biomarker for frailty is still lacking. Tryptophan (TRP) metabolism through the kynurenine pathway (KP) plays essential roles in aging, the musculoskeletal system, and physical performance. In this study, we quantified 7 KP metabolites, including kynurenine (KYN), kynurenine acid (KYNA), quinolinic acid (QUIN), picolinic acid (PIC), 3-hydroxykynurenine (3-HK), 3-hydroxyanthranilic acid (3-HAA), and anthranilic acid (AA) using ultra-high-performance liquid chromatography and gas chromatography-mass spectrometry in the serum of 85 participants (median age 75; 65% female; 28 non-frail, 29 pre-frail, and 28 frail) at the Nepean Osteoporosis and Frailty (NOF) Study. We looked at the association between TRP metabolites and physical performance, sarcopenia, and frailty. After adjusting for age and sex, our results showed that KYN and KYN/TRP were associated with higher interleukin (IL)-6 levels (r = .324 and r = .390, respectively). KYNA and its ratios to other products (mainly KYNA/KYN, KYNA/QUIN, and KYNA/PIC) were associated with a lower likelihood of frailty by Fried’s criteria (OR 0.93 [0.88, 0.98], P = .009) and Rockwood index (r = −.241, P = .028) as well as a lower likelihood of sarcopenia (OR 0.88 [0.78, 1.00], P = .049). QUIN and QUIN/KYN showed an association with increased IL-6 (r = .293 and .204 respectively), higher likelihood of frailty (OR 1.02 [1.00, 1.04], P = .029 and OR 6.43 [2.23, 18.51], P = .001 respectively) and lower physical function (r = −.205 and r = −.292). In conclusion, different TRP metabolites have various associations with physical performance, frailty, and sarcopenia. Defining the underlying mechanisms may permit the development and validation of new biomarkers and therapeutics for frailty and musculoskeletal conditions targeting specific metabolites of the TRP catabolic pathway.
Introduction
The involvement of tryptophan (TRP) metabolism in aging and musculoskeletal diseases has recently riveted a lot of attention. TRP is a nutritionally essential amino acid for humans and animals, which must be provided by diet and then metabolized in multiple organs, including the gut. 1 TRP metabolism occurs via the 5-hydroxy indole and the kynurenine pathways (KP). Major metabolic products from the 5-hydroxy indole pathway are serotonin (5-hydroxytryptamine) (5-HD) and melatonin (N-acetyl-5-methoxytryptamine). 2 After several intermediate steps (Figure 1), and via indoleamine 2,3-dioxygenase (IDO) activity, TRP is directly converted into kynurenine (KYN) and then, after several intermediate steps, into several end products, amongst them quinolinic acid (QUIN) or picolinic acid (PIC), which have a substantial effect on the musculoskeletal system.3-6

The kynurenine pathway of tryptophan metabolism.
There is growing evidence that changes in TRP metabolism with aging are involved in the pathogenesis of multiple age-related diseases.7,8 Levels of KYN are increased with aging due to high levels of IDO-1 activity induced by the chronic inflammatory condition associated with the aging process itself (known as inflammaging), which increases serum levels of interleukin-6 (IL-6) and -1β, and interferon-γ, all potent activators of this enzyme. 9 In addition, age-related changes in the microbiota affect its capacity to produce anabolic metabolites such as PIC and QUIN; thus, serum levels of these metabolites are decreased in older persons. 10
Elevated serum levels of KYN and other metabolites have been associated with osteoporosis via their effect on mesenchymal stem cells (MSC) differentiation by inducing oxidative stress and cell senescence in bone marrow stromal cells (BMSC) and affecting their differentiation into bone-forming cells (osteoblasts).7,11 Human studies have reported that, additionally to high levels of KYN, other TRP metabolites (eg, 3-hydroxykynurenine [3-HK], kynurenic acid [KYNA], and anthranilic acid [AA]) have a detrimental effect on bone, decreasing bone mineral density (BMD) and increasing fracture risk. In contrast, high serum levels of 3-hydroxyanthranilic acid (3-HAA), PIC and QUIN, and nicotinamide adenine dinucleotide (NAD+) increase BMD and are associated with lower fracture risk. 7 In sarcopenia (loss of muscle mass, strength, and function), KYN induces muscle atrophy and lipid peroxidation affecting muscle mass and function. 12
Frailty is a complex clinical condition characterized by decreased physiological reserve and marked vulnerability to adverse health outcomes such as disability, falls, fractures, and mortality.13,14 Physical frailty is predominantly associated with poor physical performance, usually involving sarcopenia and elevated disability risk. Considering that frailty is associated with poor outcomes such as falls, fractures, disability, social isolation, and high mortality, early identification of this syndrome is paramount. 14 The lack of a unique operational definition for frailty and the complex underlying pathophysiology make the development of biomarkers for this condition extremely challenging. Indeed, the current definitional ambiguities of frailty impact the accuracy, specificity, and sensitivity of individual biomarkers proposed so far. The most popular circulating markers are those related to the inflammatory response (eg, C-reactive protein, IL-6, and tumor necrosis factor α). 15 Other proposed biomarkers include clinical parameters (eg, hemoglobin and albumin), hormones (eg, dehydroepiandrosterone sulfate, testosterone, insulin-like growth factor-1, and vitamin D), products of oxidative damage (eg, advanced glycation end products, protein carbonyls, and oxidized low-density lipoproteins), or antioxidants (eg, sirtuins and α- tocopherol). 16 Overall, these proposed biomarkers are only able to capture single aspects of the condition, are weak predictors of disease progression (eg, from prefrail to frail), or are poorly associated with clinically meaningful outcomes (ie, disability); therefore, there is still a need to identify new robust biomarkers with solid biological plausibility, reliability, and clinical predictive value. 16
KP metabolites seem to be an attractive possibility as biomarkers of frailty. Recently, Jang et al, 17 reported the association of circulating KYN with frailty in older adults. In that study, the odds ratio (OR) per increase in serum KYN level for phenotypic frailty was approximately 2.62 (95% confidence interval = 1.22-5.65, P = .014). However, other metabolites generated by the KP pathway were not assessed.
Aiming to identify other metabolites of the KP as potential biomarkers of frailty and predictors of poor physical performance in older persons, in this study, we performed a comprehensive analysis of the association between serum levels of the most relevant TRP metabolites downstream of KYN.
Methods and Materials
The Nepean Osteoporosis and Frailty (NOF) Study is a cross-sectional study of community-dwelling older participants, aged >65 years, recruited between March and December 2013 in Western Sydney. 18 Exclusion criteria included: a Barthel scale <40%, Mini-Mental State Examination (MMSE) <18/30, Geriatric Depression Scale (GDS) >10/15, history of fracture within the last 3 months, use of any osteoporosis treatment, and previous history of myelodysplastic or myeloproliferative disorder. A clinician investigator recorded participants’ background variables and medical history and performed a detailed anamnesis and clinical examination. The clinical research assistant performed an extensive assessment of functional status, including performance testing, questionnaires, and technical examinations. A blood sample was collected in the morning while fasting. The Nepean Blue Mountains Local Health District Human Research Ethics Committee approved the research protocols. Written informed consent was obtained from all participants or their legally appointed proxy decision-maker.
Clinical assessment
Regarding comorbidities, Charlson Comorbidity Index was calculated according to the comorbidities reported in the participants’ hospital/physician’s files following ICD-10 codes. Depression was assessed using the geriatric depression scale (GDS). Height was measured with a digital stadiometer. Nutritional assessment was performed by body mass index calculation and by completing the Mini-Nutritional Assessment tool. Cognition was assessed using the MMSE.
Disability assessment
The Barthel Index and the Older Americans Resources and Services (OARS) scales identified disability in the participants, testing their performance in activities of daily living (ADLs) and instrumental ADLs, respectively.
Physical performance assessment
Grip strength was measured following the Groningen Elderly Test using a Smedley Hand Dynamometer. The best of 3 attempts (with 30 seconds rest between them) was recorded. Gait was assessed using a GAIT Rite (CIR Systems Inc., Havertown, PA) instrumented walkway (810 cm × 89 cm × 0.625 cm, sample rate = 80 Hz) positioned along a straight section of the walkway to record spatiotemporal gait data. In addition, sarcopenia was defined using the Sarcopenia Definitions and Outcomes Consortium (SDOC) definition for sarcopenia. 19
Determination of frailty status
Participants were categorized as non-frail, prefrail, or frail according to validated and widely utilized Fried’s frailty screening criteria. 14 These criteria are based on the presence or absence of 5 measurable characteristics: slowed motor performance (by walking speed), weakness (by grip strength), low physical activity level (by Physical Activity Scale for the Elderly), weight loss, and self-reported exhaustion on Center for Epidemiological Studies-Depression scale. Individuals fitting 3 or more criteria were defined as frail, those fitting 1 or 2 criteria, as prefrail, and those with none of the criteria were deemed non-frail.
In addition, we used the Rockwood’s Frailty Index as a second clinical measure of fitness and frailty in this population. 20 To identify the 70 clinical deficits included in this index, we used a similar approach to the Canadian Study of Health and Aging clinical assessment. 20 Items included the presence and severity of current diseases, ability in performing ADLs, and physical and neurological signs from clinical examinations. Each deficit was dichotomized or trichotomized and mapped to the interval 0-1 to represent the severity or frequency of the problem with a higher index indicating frailty.
Laboratory analyses
Venous blood was collected from resting participants. Serum IL-6 levels were measured by chemiluminescence immunoassay (ROCHE Modular Analytics E170).
TRP metabolites quantification
TRP and TRP metabolites (KYN, KYNA, QUIN, PIC, 3-HK, 3-HAA, and AA) were assayed from serum samples using ultra-high-performance liquid chromatography and gas chromatography-mass spectrometry as previously described. 21 Briefly, supernatant samples were filtered with 0.22 μm syringe filters before injecting into analyzers. An Agilent 1290 infinity ultra-high performance liquid chromatography system coupled with temperature-controlled auto-sampler and column compartment, diode array detector and fluorescence detector was used to analyze these metabolites with a 20 μL sample injection volume. Separation of metabolites was performed under stable temperature of 38°C for 12 minutes, using 0.2 mM sodium acetate (pH 4.65) as mobile phase, with an isocratic flow rate of 0.75 mL/minute in an Agilent Eclipse Plus C18 reverse-phase column (2.1 mm × 150 mm, 1.8 μm particle size). 3-HK and KYN were detected using UV wavelength at 365 nm with a 1.2 and 3.1 minutes retention time, respectively. TRP, 3-HAA and AA were detected using fluorescence intensity set at Ex/Em wavelength of 250/438 for neopterin, 280/438 for TRP, and 320/438 for 3HAA and AA. The retention times for neopterin, TRP, 3-HAA and AA are 1, 7.4, 3.3, and 9.8 minutes, respectively. Mixed standards of all metabolites were used for a 6-point calibration curve to interpolate the sample readout quantity. Agilent Open LAB CDS ChemStation (Edition C.01.04) was used to analyze the chromatogram. The inter- and intra-assay coefficient of variation was within the acceptable range of 3% to 7%. PIC and QUIN were detected using GC-MS according to our method previously described. 22
Statistical analysis
Continuous variables are presented as median (interquartile range), and categorical variables are presented as frequency (percentage). Pearson’s correlation coefficient was used for univariable associations between TRP metabolites and continuous outcomes (grip strength, gait speed, Rockwood score, IL-6, and COP cells), followed by linear regression adjusting for age and sex. Results are expressed as correlation coefficients (univariable) and partial correlations (multivariable) with P values. Correlation coefficient of .10 was interpreted as a weak correlation, .30 as a moderate correlation, and .50 as a strong correlation. 23 Some TRP metabolites, ratios, and outcomes (IL-6, Rockwood index) were transformed using natural logarithm to enable better model fit. Logistic regression was used for association with sarcopenia, and ordinal logistic regression was used for frailty. Results are expressed as odds ratios—OR (95% confidence intervals—CI). All analyses were performed using Stata 16.1 (StataCorp. 2019. Stata Statistical Software: Release 16. College Station, TX: StataCorp LLC).
Results
Participant characteristics
Eighty-five community-dwelling participants were included in the study, with median age of 75 years, 65% females. One-third of participants were frail (n = 28), and 15% were sarcopenic (n = 13). Their demographic, clinical, and biochemical characteristics are presented in Table 1, with more detailed data presented in Supplemental Tables 1 and 2.
Participants baseline demographic, clinical, and biochemical characteristics.

Association between TRP products and gait speed and grip strength
After adjusting for age and sex, there was no association between TRP products and grip strength, except for KYNA/QUIN ratios which showed a weak positive correlation (r = .262) and QUIN/KYN, which showed a negative correlation (r = −.292). KYNA/QUIN was also correlated with gait speed (r = .270), while 3-HAA and QUIN were both negatively associated with gait speed (r = −.230 and r = −.205, respectively). (Table 2).
Association between tryptophan products and gait speed and grip strength.

Variable was transformed using natural logarithm.
Correlation after removing the effects of age and sex.
Association between TRP metabolites and sarcopenia
We investigated the relationship between TRP metabolites and sarcopenia. After adjusting for age and sex, serum KYNA was associated with a lower likelihood of sarcopenia (an increase in KYNA for 1 nM decreases the odds of sarcopenia by 12%, P = .049). A similar relationship was observed with KYNA/QUIN and KYNA/KYN ratios (OR 0.72 and 0.77, respectively, P = .014 and .018 respectively) (Table 3).
Association between TRP metabolites and sarcopenia.

Variable was transformed using natural logarithm.
Adjusted for age and sex.
Association between TRP metabolites and frailty
After removing the effects of age and sex, higher KYNA, PIC, KYNA/KYN, KYNA/QUIN, and PIC/QUIN were associated with a lower likelihood of frailty by Fried’s criteria and a lower Rockwood index. TRP showed a negative correlation with the Rockwood index only (r = −.220). Higher QUIN/KYN was associated with a higher likelihood of frailty (OR 6.43) and a higher Rockwood index (r = .236) (Table 4).
Association between TRP metabolites and frailty.

Variable was transformed using natural logarithm.
^Correlation after removing the effects of age and sex.
OR associated with an increase of 100 in KYN metabolite.
Association between TRP products and IL-6
After removing effects of age and sex, there was a weak positive correlation between KYN, 3-HK, QUIN, KYN/TRP, and QUIN/KYN with IL-6 (r ranging from .204 to .324). In addition, PIC/KYN and PIC/QUIN showed a moderate negative correlation with IL-6 (r = −.226, r = −.311) (Table 5).
Association between tryptophan products and IL-6.
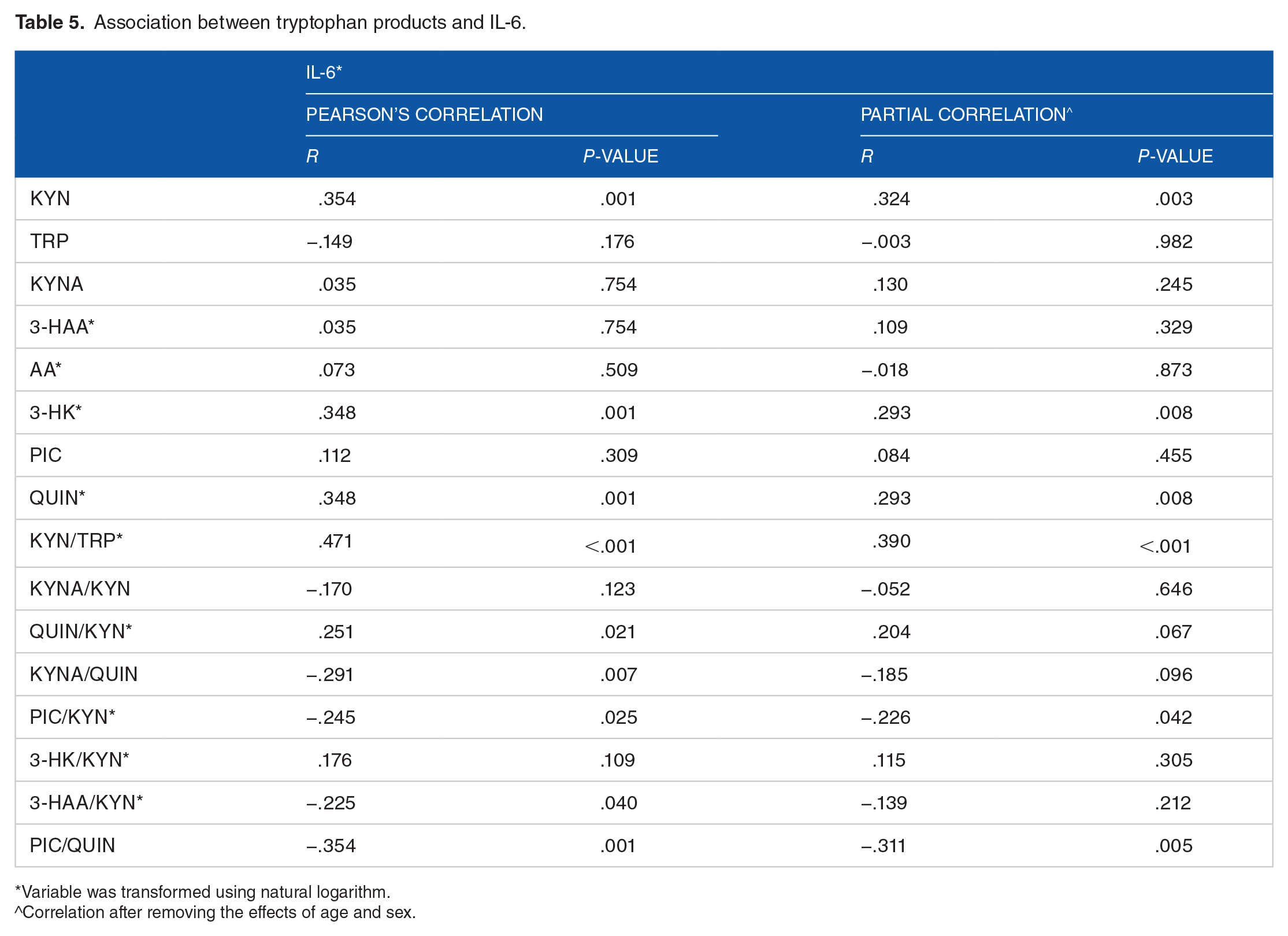
Variable was transformed using natural logarithm.
^Correlation after removing the effects of age and sex.
Discussion
This study explored the relationship between TRP metabolites and physical performance in an older population. Our results showed that serum KYNA, KYNA/KYN, KYNA/QUIN, PIC/QUIN, and KYNA/PIC were associated with a lower likelihood of frailty and sarcopenia. In comparison, QUIN and QUIN/KYN were associated with a higher likelihood of frailty. Lower physical function was observed with increased levels of 3-HAA (gait speed), QUIN/KYN (grip strength), and QUIN (gait speed), while higher grip strength and gait speed were observed with higher levels of KYNA/QUIN. More elevated serum IL-6 was observed in participants with higher KYN, KYN/TRP, 3-HK, QUIN, and QUIN/KYN.
Our results align with previous studies investigating the correlation between circulating KYN and TRP with frailty in older adults.24,25 These studies showed meaningful implications of KYN with frailty diagnosed using Fried’s criteria but not Rockwood’s index. Interestingly, our results indicate that some of those metabolites (KYNA, PIC, KYNA/KYN, and KYNA/QUIN) are associated with the presence of frailty regardless of the definition used. Westbrook et al. 26 reported that frail human subjects showed significant dysregulation in the TRP degradation pathway, including decreased serotonin and increased levels of some neurotoxic kynurenines. They also noted that, in humans, serum KYN strongly correlated with age, frailty status, TNF-αR1 and IL-6, weaker grip strength, and slower walking speed.
Increased inflammatory cytokine levels might drive the strong associations found between some TRP metabolites, frailty, and sarcopenia. Lustgarten and Fielding 27 showed that TRP metabolites were associated with circulating IL-6 in older adults. Another potential mechanism is the association between TRP and oxidative stress and neurotoxicity. Some TRP metabolites (eg, KYN) increase oxidative stress, 28 and associate with neurodegenerative disorders. 29 Furthermore, KYN and KYNA are associated with physical activity, 30 aging, 26 and muscle function. 31 Recently, several studies linked the conversion of KYN to KYNA with increased physical activity.32,33 Another randomized controlled trial showed that intensive exercise is associated with increase conversion of KYN into KYNA as well. 34 These changes in KYNA levels may have multiple biological consequences, which are unlikely to affect the central nervous system as these metabolites barely cross or penetrate the blood-brain barrier. 35 Our current study observed that sarcopenic and frail patients have a lower KYNA content in their plasma compared to controls without any association with physical function or inflammation. In contrast, KYNA/QUIN ratio was associated with a better function.
QUIN is known for its excitotoxicity via its activation of N-methyl-D-aspartate receptor (NMDA) receptors, 36 and other neurotoxic mechanisms 37 thus, suggesting a possible role of QUIN in oxidative stress and neurotoxicity. In this study, we observed higher levels of IL-6, worse physical function, and a higher prevalence of frailty in subjects with increased QUIN levels. Whether these findings are explained by the biological mechanisms listed above could be investigated in future clinical trials.
To assess the biological role of the KYN pathway, ratios between TRP metabolites are frequently employed. In this case, KYNA/KYN and both KYNA/QUIN and KYNA/PIC are ratios used to estimate the balance between the neuroprotective and the neurotoxic metabolites.38,39 These ratios showed to be associated with inflammation. 40 Although we did not find any association with IL-6 levels, all the calculated ratios were associated with a lower likelihood of frailty and sarcopenia, with some being associated with better physical performance (KYNA/QUIN, KYNA/KYN, and KYNA/PIC).
The study has several limitations, including small sample size and a lack of precise mechanisms showing how the identified TRP metabolites affect muscle function and physical performance. In addition, the cross-sectional study design may have implications about the longitudinal relationship between serum levels of these metabolites and the clinical and physical measures that we measured. Although sensitive methods of separation, such as high-pressure liquid chromatography coupled with GC/MS have been used to detect TRP metabolites independent of their biological source, it should be noted that the circulating levels of TRP metabolites can reflect production (or changes in production) in discrete amounts in tissues and organs. Consequently, a current challenge for the field is interpreting the complexity of differences in TRP expression in different cells and tissues and under different circumstances and relating this back to biological function; this could explain these discrepancies between studies. Nevertheless, our study has several strengths. This is the first study to explore most of the major TRP metabolites in a population that included both pre-frail and frail individuals where a very comprehensive functional assessment was performed.
In conclusion, we have identified novel associations between TRP metabolites and physical function and frailty (summarized in Figure 2). Importantly, our data are consistent with the results from in vitro and animal experiments showing the role of TRP and its products in musculoskeletal health,22,28,41 and provide clinical evidence that serum TRP metabolites demonstrate strong biological plausibility to become robust biomarkers for frailty and musculoskeletal diseases in the future. However, a larger sample assessed longitudinally will be required to determine the predictive value of these metabolites to adverse outcomes. In addition, defining the underlying mechanisms may permit the development of new diagnostic and therapeutic strategies using specific KP metabolites.

The biological effect of tryptophan metabolites on older persons.
Supplemental Material
sj-docx-1-try-10.1177_11786469211069951 – Supplemental material for Association Between Tryptophan Metabolites, Physical Performance, and Frailty in Older Persons
Supplemental material, sj-docx-1-try-10.1177_11786469211069951 for Association Between Tryptophan Metabolites, Physical Performance, and Frailty in Older Persons by Ahmed Al Saedi, Sharron Chow, Sara Vogrin, Gilles J Guillemin and Gustavo Duque in International Journal of Tryptophan Research
Supplemental Material
sj-docx-2-try-10.1177_11786469211069951 – Supplemental material for Association Between Tryptophan Metabolites, Physical Performance, and Frailty in Older Persons
Supplemental material, sj-docx-2-try-10.1177_11786469211069951 for Association Between Tryptophan Metabolites, Physical Performance, and Frailty in Older Persons by Ahmed Al Saedi, Sharron Chow, Sara Vogrin, Gilles J Guillemin and Gustavo Duque in International Journal of Tryptophan Research
Footnotes
Author Contributions
Study design: AAS and GD. Study conduct and experiments: AAS, SC, GJG, GD. Data collection: AAS, SC, and GD. Data analysis: AAS, SC, SV, GJG and GD. Data interpretation: AAS, SV, GJG and GD. Drafting manuscript: AAS, SV and GD. Revising manuscript content: AAS, SC, SV, GJG and GD. Approving final version of manuscript: AAS, SC, SV, GJG and GD. SV and GD take responsibility for the integrity of the data analysis.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a seed grant from the Australian Institute for Musculoskeletal Science (AIMSS).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
