Abstract
Sleep has a regulatory role in maintaining metabolic homeostasis and cellular functions. Inadequate sleep time and sleep disorders have become more prevalent in the modern lifestyle. Fragmentation of sleep pattern alters critical intracellular second messengers and neurotransmitters which have key functions in brain development and behavioral functions. Tryptophan metabolism has also been found to get altered in SD and it is linked to various neurodegenerative diseases. The kynurenine pathway is a major regulator of the immune response. Adequate sleep alleviates neuroinflammation and facilitates the cellular clearance of metabolic toxins produced within the brain, while sleep deprivation activates the enzymatic degradation of tryptophan via the kynurenine pathway, which results in an increased accumulation of neurotoxic metabolites. SD causes increased production and accumulation of kynurenic acid in various regions of the brain. Higher levels of kynurenic acid have been found to trigger apoptosis, leads to cognitive decline, and inhibit neurogenesis. This review aims to link the impact of sleep deprivation on tryptophan metabolism and associated complication in the brain.
Introduction
Sleep is an essential physiological process required for the survival and well-being. However, to date, the biological functions of sleep is understood fully are. Humans spend about one third of their life in sleeping. 1 Sleep plays key roles in maintaining cellular homeostasis, energy conservation, metabolic waste clearance, and regulating immune functions. 2 Quality sleep improves cognitive performance, vigilance, and psychological conditions. 3 In humans, sleep has 2 phases: non-rapid eye movement (NREM); and rapid eye movement (REM). NREM sleep consists of: stage 1 (lightest phase of sleep and lasts for 1-5 minutes); stage 2 (characterized by sleep spindles and single long delta waves); stage 3 and 4 (Slow-Wave Sleep). 4 NREM sleep is followed by REM sleep, which is distinguished by irregular eye movements, decline of muscle tone, tendency to dream and transmission of low-voltage brain waves, versus the non-REM phase. 5 By limiting neuronal oxidative damage, apoptosis, alterations in neural and cytoskeleton proteins, both NREM and REM sleep types are neuroprotective. 6 NREM disruption is found to contribute to hippocampal dependent memory decline. 7 However, because of the lifestyle modifications and night shift work schedules, the prevalence of REM sleep disruption is higher than NREM,8,9 as a result it has been linked to early ageing and in the progression of most of the neurodegenerative diseases (Figure 1).

Stages of sleep. Sleep is composed of 2 phases: Rapid Eye Movement sleep (REM), and the Non-REM sleep (NREM). The NREM phase consists of 4 stages: Stage 1, which is transition from being awake to falling asleep; Stage 2, the period of light sleep during which the eyes movements stop, Stages 3 and 4, which are also called Slow Wave Sleep (SWS) (Reproduced with permission from Bernard 10 ).
Sleep disorders have become a global and increasing health concern. Millions of people worldwide suffer from sleep deprivation, sleep apnea and insomnia on daily basis.11,12 The prevalence of insufficient sleep time and sleep deprivation (SD) has increased. 13 SD is frequently experienced because of medical conditions, sleep disorders, work pressure, social and domestic responsibilities and lifestyle (eg, shift work, prolonged work hours, stress, and social media). 14 Inadequate sleep or fragmented sleep alters the biochemical and physiological functions and has been strongly linked to neurodegenerative, 15 metabolic, 16 cardiovascular, 17 and autoimmune diseases. 18 REM SD has deleterious effects on general health. While it affects most of the organs, it has a major impact on various regions of brain like hippocampus, cortex, striatum, hypothalamus, etc and affect their functions. 15 REM SD has a major impact on brain development, cognitive functions, and long term potentiation.19,20 This has been confirmed by several studies that sleep deprivation induces apoptosis and oxidative stress in different regions of rat brain. 21 Oxidative stress is known to be associated with the development of neurodegenerative diseases as the brain is more vulnerable to reactive oxygen species (ROS) due to the high consumption of oxygen and lesser antioxidants defence. 22 While chronic sleep deprivation in young, healthy volunteers increases appetite and energy consumption, activates immune system and sympathetic tone; it decreases parasympathetic tone.23,24 Increased blood pressure, cortisol levels, and blood glucose levels are reported to be associated with chronic sleep restricted individuals.25,26 Sleep restores the brain energy during active waking 27 and clears the metabolic waste from the central nervous system by regulating glymphatic system. 28 Preclinical and clinical studies have shown that SD alters glymphatic clearance and results in the deposition of toxins like amyloid β (Aβ), α-synuclein and other aberrant proteins in brain.29-34 Interestingly, chronic SD also disrupts blood brain barrier cohesion and alters its permeability. 35 The combination of increased blood-brain barrier permeability and reduced glymphatic clearance due to SD, predisposes neurodegenerative processes.35,36 Increased metabolic waste has been attributed to the impairment in brain function and development of diseases.37-40 Thus, sleep disorder or SD impairs biochemical and behavioral function in brain and evolve as risk factor for neurological disorders
Sleep Deprivation, Tryptophan, and Kynurenine Pathway
The essential amino acid tryptophan (TRP) is catabolised via 2 distinct biochemical pathways: (1) the serotonin pathway (5%) and (2) the kynurenine pathway (KP) (the majority, 95%). 41 The kynurenine pathway is a major regulator of the immune response. 42 Tryptophan is converted to kynurenine by 1 of 3 rate-limiting enzymes: indoleamine-2,3-dioxygenase-1 (IDO1), or indoleamine-2,3-di-oxygenase-2 like protein (IDO2), or tryptophan 2,3-dioxygenase (TDO). TDO is highly expressed in the liver and is activated in response to corticosteroid stress hormones, while IDO1 expression is activated by inflammatory stimuli such as Interferon gamma (IFN-γ), Tumor necrosis factor alpha (TNF-α), amyloid beta, etc. 43 Sleep is important for maintaining inflammatory homeostatic functions and its loss is linked to the alterations in the immune response. Inflammation activates the kynurenine pathway, leading to increased production of 3-hydroxykynurenine (3HK) and quinolinic acid (QUIN), major neurotoxic metabolites. Under physiological conditions, 40% of the kynurenine is produced within the brain, and 60% is taken up from the periphery through the blood brain barrier. 44 SD triggers the activation of TDO through the increase of the stress hormone, corticosterone, via hypothalamic-pituitary-adrenal (HPA) axis and IDO1 through inflammatory signalling (TNF-α, IFN-γ) leading to the catabolism of tryptophan.19,45,46 This increased production of kynurenine metabolites alters various neurotransmitter systems within the cortical and striatal regions.47-49 Kynurenine metabolites reduce dopamine levels in substantia nigra and in the striatum,48,50 acetylcholine in the cortex,51,52 and gamma aminobutyric acid (GABA) in rat striatum and cortex of rat brains.47,53 Dysregulation of kynurenine metabolism associated with SD is known to impact cognitive function and to promote the development and progression of neurodegeneration.41,54,55 In rat model of chronic sleep deprivation, tryptophan (TRP) and kynurenine (KYN) are increased in plasma.56,57 These peripheral tryptophan and kynurenine are taken up into the brain and used as extra substrate resulting in an overproduction of Kynurenic acid (KYNA) in the presynaptic neurons of hypothalamus, hippocampus, and cerebral cortex in SD rats. 55 SD alters the immune response resulting in increased levels of inflammatory mediators such as Interleukin 6 (IL-6), TNF-α, IFN-γ, and C-reactive protein (CRP).58-62 Increase in inflammatory mediators activate IDO which metabolizes tryptophan to kynurenine (Figure 2). Increase in KYN/TRP ratio infers increased expression of IDO. KYN is further metabolized to 3-hydroxykynurenine (3HK) by kynurenine monooxygenase (KMO), an enzyme also found up regulated in inflammatory conditions. 63 Further, this leads to an increased production of the excitotoxin, quinolinic acid, by macrophages and activated microglia.64-66 Imbalance between neurotoxic and neuroprotective and immune-modulator kynurenine pathway metabolites have been reported in many neurodegenerative diseases such as Alzheimer’s disease (AD), Parkinson’s disease (PD), Multiple sclerosis (MS), Amyotrophic lateral sclerosis (ALS), Huntington disease (HD).43,67-71 Increased kynurenine levels or catabolic enzyme activity are not the only mechanisms that determine the formation of KYNA in the brain. Accumulation of pro-oxidant factors after sleep deprivation also promotes non-enzymatic degradation of kynurenine to KYNA, which is a stable metabolite, and the enzymes responsible for its breakdown are absent in human.72,73 This data indicates that sleep loss is associated with higher levels of inflammation and kynurenine metabolism is increasingly linked with inflammation and neurodegeneration.
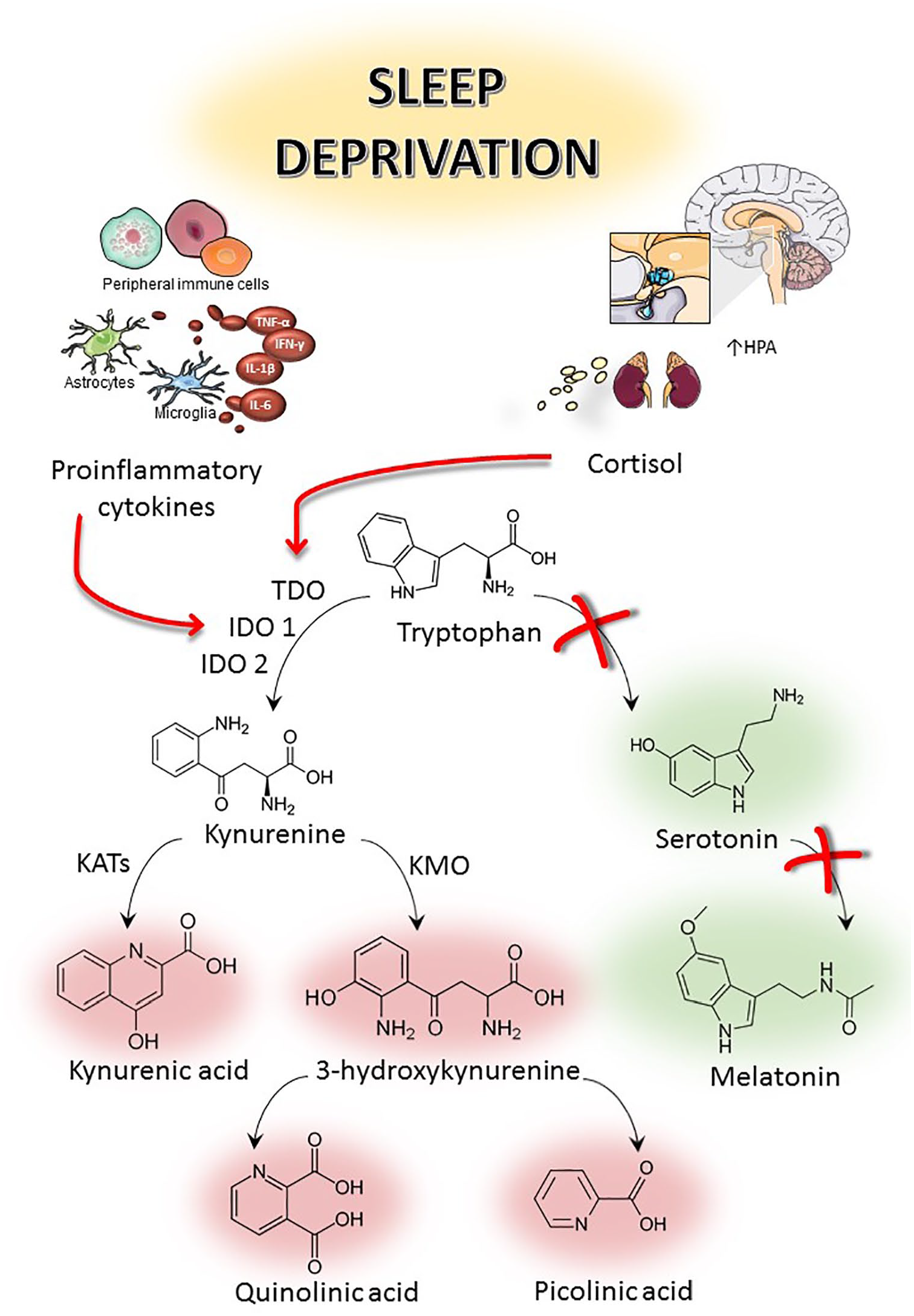
Relationship between sleep deprivation, hypothalamic-pituitary-adrenal (HPA) axis and tryptophan metabolism.
Sleep Deprivation and Kynurenic Acid
Kynurenic acid is mainly produced by astrocytes within the central nervous system. 74 Nanomolar levels of KYNA measured in the brain and cerebrospinal fluid of patients with schizophrenia.75,76 Animal studies have also shown an increased level of KYNA producing cognitive deficits, 77 impairing auditory sensory gating 78 and reduction in the dopaminergic and glutamatergic neurotransmission.48,79 KYNA has been shown to prevent the excitotoxicity induced neurodegeneration when administered at lower concentrations exogenously. 71 However, the central nervous system (CNS) concentration required to produce neuroprotection is still not clear. 80 Dysfunctional KMO activity may directly increase KYNA level and this is recorded in the cerebrospinal fluid (CSF) 81 and post-mortem brains of patients with schizophrenia and bipolar disorder.76,82 The impact of a change in KYNA levels on cognitive function has been extensively studied as it blocks N-methyl-d-aspartate (NMDA) and affects human cortical development, 83 desensitizes α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors 84 and inhibits α7 nicotinic acetylcholine (α7nACh) receptors. 85 Increased KYNA levels in the brain are associated with increased cognitive deficits, whereas decreased KYNA levels, have been reported to improve learning and memory because they impair both glutamatergic and cholinergic transmission.86,87 An increase in the levels of KYNA in the presynaptic area inhibits extra synaptic NMDA receptors, nicotinic acetylcholine receptors and AMPA receptor-mediated currents.44,84,88 This results in cognitive decline and long term potentiation in rodents. 89 At nanomolar concentrations, KYNA disrupts hippocampus-based memory in rodents.86,87 A decrease in the brain KYNA by pharmacological interventions, ameliorated the cognitive functions in rats. 87 KYNA alters acetylcholine and glutamate which are critically involved in learning and memory, sleep dependent synaptic plasticity, and regulation of circadian rhythm.90-93 Also, increased KYNA level restrains dopamine release into the synaptic cleft. 48 Elevated levels of KYNA in the brainstem, cortex, and hippocampus have been found to impair contextual memory and disrupt the sleep wake cycle in rodents. 94 Exogenous administration of KYNA was shown to impair rat performance in the behavioral paradigms like open-field, and Morris water-maze tests.86,95 Decline in glutamate levels have been associated with age related cognitive decline. 96 Recently it was found that sleep deprivation impacts the hippocampal and cortical levels of KYNA in sex dependent manner. 19 Increased levels of KYNA in hippocampus was found in male rats after sleep deprivation results in cognitive decline whereas female SD rats didn’t show any impairment in memory.19,94 This indicates that sex hormones also play an important role in the metabolism of tryptophan. Further, 96 hours SD in rats showed to suppresses neurogenesis in dentate gyrus region of hippocampus, which has been attributed to the accumulation of KYNA in the hippocampus.97,98 These evidences strengthen the correlation between increased levels of KYNA and sleep deprivation which might promote the progression of neurodegenerative diseases.
Sleep Deprivation and Serotonin
Tryptophan is the unique precursor for the biosynthesis of serotonin and melatonin which both play key regulating roles on the sleep-wake cycle. Melatonin and its analogs have been used for the treatment of various sleep disorders for many years.99-101 Serotonergic system is susceptible to SD. For example, SD for 1 night exerts an immediate but short-lived antidepressant effect which may be due to a transient increase in dopamine and serotonin metabolites.102,103 Levels of extracellular serotonin are highest during waking, lower in slow-wave sleep and lowest in REM sleep in all regions of brain including the hippocampus and cortex.103,104 This effect is, however, short lasting. SD stimulates the suprachiasmatic nucleus (SCN) and stimulates the release of serotonin in hamster. 105 Toru et al reported an increase in the levels of 5-hydroxyindoleacetic acid (5-HIAA), the precursor of serotonin in the dorsal raphe nucleus and thalamus of 24 hour SD rats. 106 Also, total SD increases the mean firing rate of serotonergic neurons in the dorsal raphe nucleus in cats. 107 Chronic sleep restriction desensitizes Serotonin1A receptors, 108 inactivation of Serotonin1A receptors alters synaptic plasticity and impairs learning and memory. 109 In contrast, Bjorvatn et al, reported that 8 hours SD does not increase the levels of extracellular serotonin in hippocampus or cortex. 110
Sleep Deprivation and Melatonin
Melatonin is the main hormone secreted by pineal gland having a critical role in circadian rhythm. 111 Melatonin is synthesized from serotonin via acetylation in the presence of enzyme N-acetyl transferase (NAT). The activity of NAT at pineal gland is dependent on the noradrenergic input received from nervi conarii. Increase in the activity of nervi conarii initiated by the suprachiasmatic nucleus of the hypothalamus stimulates the synthesis of melatonin during night time. 112 Day time melatonin levels are low. Optimal levels of melatonin is a powerful free-radical scavenger and combats oxidative stress. 113 Melatonin improves sleep efficacy 114 and enhances long term memory. 115 Decrease in melatonin have been related to insomnia and sleep disorders in elderly people. 116 Total sleep deprivation for 72 hours in mice decreases plasma melatonin level which results in increased expression of inflammatory cytokines and reduced antioxidants. 117 Similarly, a decrease in melatonin production was observed in patients suffering from insomnia. 116 Chronic decrease in melatonin was found in night time shift workers. 118 In a clinical study, it was found that melatonin excretion increases in healthy males subjected to sleep deprivation by exposing them to white light. 119 Exogenous administration of melatonin inhibits inflammatory mediators and increase the reduced glutathione levels in locus coeruleus nucleus in sleep deprived mice, 120 reduces oxidative stress and improves memory consolidation in SD rats. 121 Melatonin has also been found to be efficacious for sleep disorders in children with autism. 122 Oral administration of melatonin has also shown to improve the sleep quality and increase the antioxidant capacity in patients with fibromyalgia. 123 These data indicate the involvement of sleep deprivation in the metabolism of the melatonin which results in activation of inflammatory mediators and memory consolidation.
Sleep Deprivation and Hypothalamic-Pituitary-Adrenal Axis
Hypothalamic-pituitary-adrenal axis is responsible for regulation of the physiological response to stress.124,125 Stress stimuli induce the release of corticotropin-releasing hormone (CRH) from the paraventricular nucleus region of the hypothalamus. CRH triggers the release of adrenocorticotropic hormone (ACTH) from the pituitary gland. ACTH stimulates the adrenal cortex to release glucocorticoids (cortisol in humans, corticosterone in animals).126,127 Sleep plays a key role in modulating stress reactivity of the HPA axis. 128 Preclinical and clinical studies have confirmed that the potential mechanism by which SD modulates the HPA axis is by increasing the levels of glucocorticoids.129-132 An increase in the levels of cortisol have been found in children with lower sleep efficiency133,134 and in healthy SD adults. 130 Similarly, higher levels of glucocorticoids have been found in sleep deprived rodents and healthy young females.135-137 Higher levels of glucocorticoids in humans 138 and animals 139 during SD have an impact on KP metabolism. 46 Increased corticosterone increases KP metabolism and leads to mass production of neurotoxic metabolites KYNA, 3HK and QUIN in brain and periphery.140,141 Stress influences KP metabolism by activation of the key enzyme TDO. 142 Stress in pregnancy not only affects the development of the fetal brain but also increase the risk of offspring developing psychiatric disorders such as schizophrenia and depression 143 which may be attributed to increase in the levels of KYNA. 75 This sleep deprivation activates HPA axis and increased levels of the stress hormones play a crucial role in neurodegeneration by increasing the neurotoxic metabolites.
Conclusion
Sleep regulates the immune and endocrine systems, helps to encounter deleterious stimuli, improves central nervous system functioning and reduces the metabolic wastes. It has an essential role in learning and memory and synaptic plasticity formation. Sleep deprivation precipitates neurodegenerative diseases such as AD, PD, HD, and motor neuron diseases. Tryptophan is metabolized by majorly via kynurenine pathway. However inadequate sleep or sleep deprivation increases the expression of key enzymes such as TDO and IDO resulting in increased levels of KP metabolites such as KYNA, 3HK, and quinolinic acid which participate in the progression of neurodegenerative diseases. Exogenous administration of melatonin reduces the neuroinflammation and improves memory consolidation. Also, IDO1, and TDO enzymes can be potential targets in correcting sleep disorders and sleep deprivation induced neurodegeneration.
Footnotes
Acknowledgements
AB is supported by the IBRO-APRC Exchange Fellowships program, Macquarie University and Senior Research Fellowship from Indian Council of Medical Research (ICMR), Govt of India, New Delhi. GJG is supported by the National Health Medical Research Council (NHMRC), the Australian Research Council (ARC), the Hanbury Foundation, the Mason Foundation, and Macquarie University. This paper has been professionally edited by Red Fern Communications, Sydney Australia.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Note
Authors’ Contributions
AB wrote the first draft. ASP prepared the figures. VT, SBC and GJG revised the manuscript. All authors read the final version and approve it.
