Abstract
Meat and vitamin B3 – nicotinamide – intake was high during hunter-gatherer times. Intake then fell and variances increased during and after the Neolithic agricultural revolution. Health, height, and IQ deteriorated. Low dietary doses are buffered by ‘welcoming’ gut symbionts and tuberculosis that can supply nicotinamide, but this co-evolved homeostatic metagenomic strategy risks dysbioses and impaired resistance to pathogens. Vitamin B3 deficiency may now be common among the poor billions on a low-meat diet. Disease transitions to non-communicable inflammatory disorders (but longer lives) may be driven by positive ‘meat transitions’. High doses of nicotinamide lead to reduced regulatory T cells and immune intolerance. Loss of no longer needed symbiotic ‘old friends’ compounds immunological over-reactivity to cause allergic and auto-immune diseases. Inhibition of nicotinamide adenine dinucleotide consumers and loss of methyl groups or production of toxins may cause cancers, metabolic toxicity, or neurodegeneration. An optimal dosage of vitamin B3 could lead to better health, but such a preventive approach needs more equitable meat distribution. Some people may require personalised doses depending on genetic make-up or, temporarily, when under stress.
Keywords
No civilisation can be sound or stable which has at its base this mass of stunted life. It must first, however, be generally believed with Sydenham, that our chronic maladies are of our own making.
Introduction
Meat transitions have been a feature of human history. Ever since the evolution of agriculture, there have been marked variances of meat intake between rich and poor nations and individuals within social classes, and therefore, oscillations of key components such as nicotinamide that when severely deficient causes pellagra.1–5 Nicotinamide and its derivatives nicotinamide adenine dinucleotide (NAD) and reduced nicotinamide adenine dinucleotide (NAD(H)) are intrinsic to mitochondrial function, and levels can be high, optimal, or low: these can involve manageable change through homeostasis or use of symbionts (or when short-lived, can be a beneficial hormetic shock). However, if nicotinamide supplies are very low, this can lead to energy decline with loss of high-energy molecules and disease often through protein modifications – as is seen with pellagra and similar to those seen with some mitochondrial and other mutations.6,7 Nicotinamide adenine dinucleotide is an important ‘food signal’ to all organisms and, often mediated by serotonin, has defining effects on development, circadian rhythms, gene regulation through chromatin remodelling, and reproductive and other behaviour.8,9 NADH as a cofactor performs more than 500 key dehydrogenase and other reactions, excluding NAD reactions that are important to metabolism, (stem-cell) development, repair, and longevity, but then is consumed – so it requires constant replenishment ultimately from dietary precursors. 10
Much has been written about modern maladaptations relative to our long hunter-gatherer days. These mismatches usually concentrate on loss of fibre or excessive sugar or fructose or gluten in recent affluent diets.5,11–23 We argue that a high risk of too little nicotinamide may be the most important difference between diets in the past 10 000 years and those in the Palaeolithic. During that long time, as we (co)evolved much of our nutrigenome, microbiome, and ‘meat cultures’, variances were usually ‘feasts or famines’ (or fatal as meat then provided most of the calories) rather than chronic shortages over lifetimes. The first Neolithic agricultural revolution and the second ‘Green revolution’ 50 years ago may have, as an unintended consequence, reduced meat and micronutrients for those in poverty. Both revolutions increased cereal availability reducing hunger but not the ‘hidden hunger’ of micronutrient deficiency in something of a ‘Faustian’ bargain. 24
Nicotinamide’s approximate concentrations in foods are shown in Table 1.25,26 Milk contains average amounts of nicotinamide but high amounts of the potent nicotinamide riboside. Culturally acquired cooking methods ‘nixtamalisation’ can improve availability of nicotinamide from the bound form niacytin: the microbiome can contribute as many bacterial symbionts can synthesise nicotinamide, though, whether they share all this with other bacteria that cannot, or, with the host is unclear in man, though, must happen in ruminants. The synthetic pathway from tryptophan is inefficient but can supply 1 mg of niacin for 60 mg of tryptophan depending on other factors including pyridoxine and iron or zinc availability and high-fat or leucine diets. Tryptophan is sourced largely from animal products boosting the importance of meat, eggs, and milk. Cereals particularly maize barely increase post-prandial levels of tryptophan, and transport across the blood-brain barrier can be reduced by other amino acids complicating effects on mood and cognition.27–32
Main sources of nicotinamide are animal products.
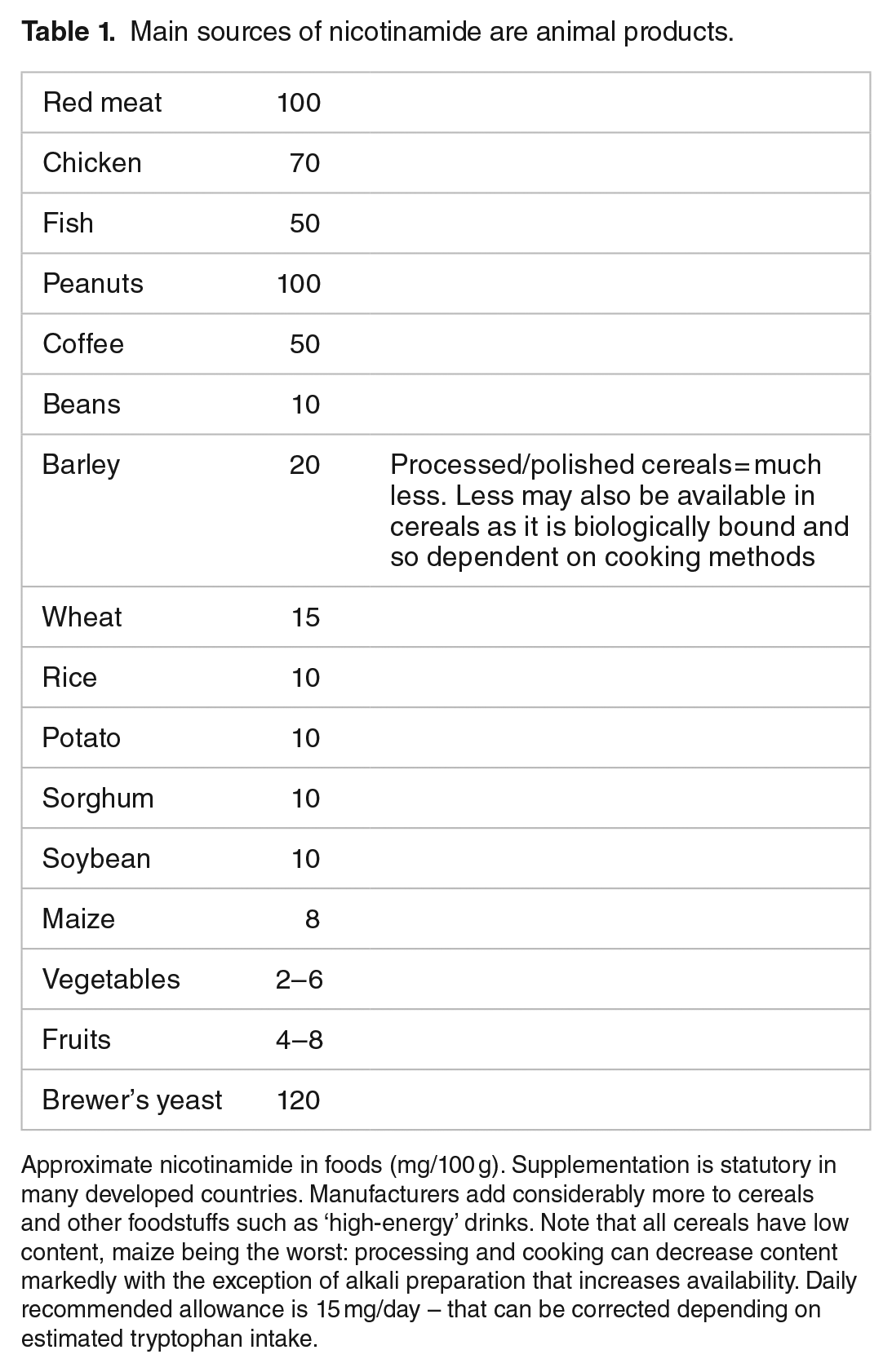
Approximate nicotinamide in foods (mg/100 g). Supplementation is statutory in many developed countries. Manufacturers add considerably more to cereals and other foodstuffs such as ‘high-energy’ drinks. Note that all cereals have low content, maize being the worst: processing and cooking can decrease content markedly with the exception of alkali preparation that increases availability. Daily recommended allowance is 15 mg/day – that can be corrected depending on estimated tryptophan intake.
‘Meatification’ of the diet doubled meat intake in the United Kingdom between 1850 and 1960 and has overall increased by 2-fold average intake from 20 to 40 kg per annum per person in 1960 to double that in 2010 (as has nicotinamide intake) and is predicted to rise to 50 kg per annum.33,34 The extremes are striking at 120 kg per annum per person as an average in many rich countries (who also obtain nicotinamide as mandated supplements and manufacturer’s additives) but 20 kg per annum per person in many poor countries with many individuals eating negligible amounts that have to affect a society’s health and industriousness.35,36
Such individuals must be at risk of the classic meat deficiency disorder particularly when on a poor monophagic cereal diet (usually maize), known as pellagra. Pellagrins can have a diagnostic rash ‘Casal necklace’ triggered by sunlight. The rash, however, is not always present or characteristic (often diagnosed as eczema) – ‘pellagra sine pellagra’. 37 Other cardinal features of pellagra such as gut infections and neurodevelopmental and neuropsychiatric syndromes and an inability to deal with stress or an absence of allergies are non-specific, so the diagnosis is bound to be missed (Figure 1). There is no easy biochemical test.

Pellagra has a very wide phenotype. The parallel with diseases of ageing are striking. NAD/NADH/nicotinamide imbalances may be the treatable underlying common factor to many common diseases whether from dietary deficiency or excess or varying needs from genetic mutation, toxic, anoxic, or other external stresses. NAD indicates nicotinamide adenine dinucleotide.
Hypotheses
We will argue that nobody ever systematically checked that pellagra was eliminated globally (dietary supplementation mainly happened in rich countries). Pellagra may be common and misdiagnosed masquerading as ‘environmental enteropathy’, poor cognition, eczema, or general ill health and a lack of well-being or poor homeostasis when under environmental stress with shortened lives.38,39
We also argue that environmental insults from trauma, hypoxia, toxins, stress, or mutations (such as in mitochondrial or DNA-repair genes) may require either lifetime or temporary higher doses than normally recommended (15 mg/d).
Controversially, we suggest that many people in rich countries may be on too high a dose. A hyper-vitaminosis B3 state may be common and, like pellagra, have a wide phenotype that includes the metabolic syndrome, several cancers, and some degenerative or neuro-behavioural disorders. 40
Furthermore, we propose that the transition from diseases of poverty to diseases of affluence is due to switching the dose of meat/nicotinamide too fast and too far.41,42 The most recent version of the ‘hygiene hypothesis’ concentrates on reductions in symbiotic/commensal biome diversity acquired early in development, not cleanliness during childhood and common pathogens.43–46 We will discuss how a biochemical switch away from the need to produce nicotinamide ‘in house’ from tryptophan, and the related reduced metabolic need for gut symbionts or tuberculosis (TB) on a better diet so that they are no longer ‘welcomed’ by the immune system is a more plausible explanation for the loss of microbial ‘old friends’. This switch of microbiomes reduces tolerogenic instruction to the immune system further encouraging it to over-react to otherwise irrelevant foreign proteins or self-proteins. 47
Biochemical Background
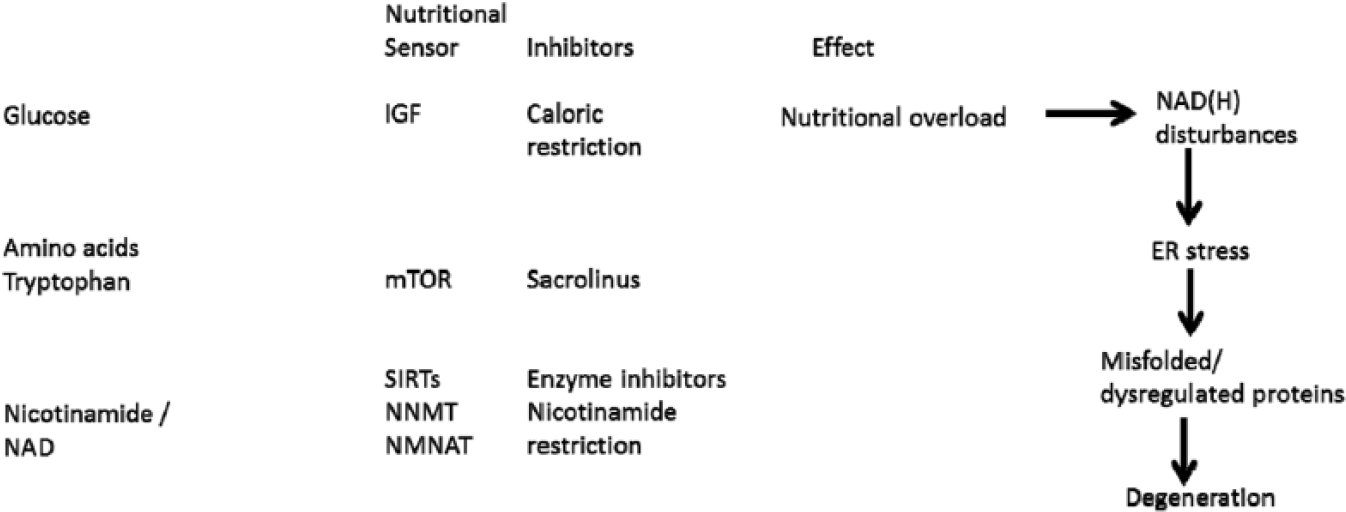
We summarise NAD metabolism in a diagrammatic form (Figures 2 to 4). Nicotinamide adenine dinucleotide has 3 related precursor vitamins – nicotinamide, nicotinic acid, and nicotinamide riboside – that have highly efficient synthetic and recycling ‘salvage’ pathways and one ‘detoxification’ pathway using nicotinamide N-methyltransferase (NNMT).48–54 There is an important backup nicotinamide/NAD ‘de novo’ synthetic pathway from the essential amino acid tryptophan. 55 This pathway is crucial to our story as it is closely linked with immunologic tolerance and intolerance.

Diet supplies nicotinamide, nicotinic acid, and nicotinamide riboside as well as the essential amino acid tryptophan. Tryptophan can be degraded to synthesise nicotinamide when there is dietary stress by the kynurenine ‘de novo’ ‘immune tolerance’ pathway. Symbionts, whether in gut or TB, are a backup source as is ‘autocarnivory’. Salvage pathways are extensive and efficient to conserve NAD/nicotinamide as NAD consumers mean that there is a continuous need for replenishment. Many are involved in repair and disease processes and ageing. Excess nicotinamide can be excreted after a methylation reaction by NNMT. NNMT, indicates nicotinamide N-methyltransferase; TB, tuberculosis.

NAD is central to metabolism and the energy supply. NAD determines cell fates during development. NAD links to the epigenome and genomic expression means close interactions with our environment that in turn supplies NAD.

Nicotinamide/NAD and tryptophan metabolism are linked by the ‘de novo’ pathway. All these pathways have been implicated in diseases of ageing and mechanisms such as proteotoxicity, and, interventions such as caloric restriction, resveratrol, parabiosis and metformin.
Nicotinamide adenine dinucleotide, the central redox co-enzyme in cellular metabolism, functions as a hydride group acceptor forming NADH with concomitant oxidation of metabolites derived from carbohydrates, amino acids, and fats. Gluconeogenesis, oxidative phosphorylation, ketogenesis, detoxification, and lipogenesis require reduced cofactors NADH and reduced nicotinamide adenine dinucleotide phosphate (NADPH). Nicotinamide adenine dinucleotide is the consumed substrate of poly(ADP-ribose) polymerases (PARPs), sirtuins (SIRTs), and cyclic adenosine diphosphate (ADP) synthetases. Nicotinamide adenine dinucleotide and NAD/NADH ratios have been described as a master controller of many physiological and repair activities and are themselves under circadian control with much cross-talk with multiple nutrient pathways, whether carbon, nitrogen, or phosphate sensing. Despite extensive recycling, cell stresses that include ageing, toxins, and over-nutrition activate NAD consumption and alongside growth requirements mean that there is a continuous need for a dietary or microbiomic source of NAD precursors.56–58
The close relationship between man and meat/milk-providing animals is not surprising. Nicotinamide adenine dinucleotide metabolism is also integral to plant primary energy acquisition from the sun on which all animals depend. Plants also use NAD-dependent oxidative bursts to ward off microbial attack. 59 Other deterrants from herbivore attack involve nicotinamide-nicotine metabolism; this is interesting as both nicotine and other stress compounds, such as salicylate and resveratrol, affect our NAD metabolism in both a medicinal and hormetic sense (Figure 5).
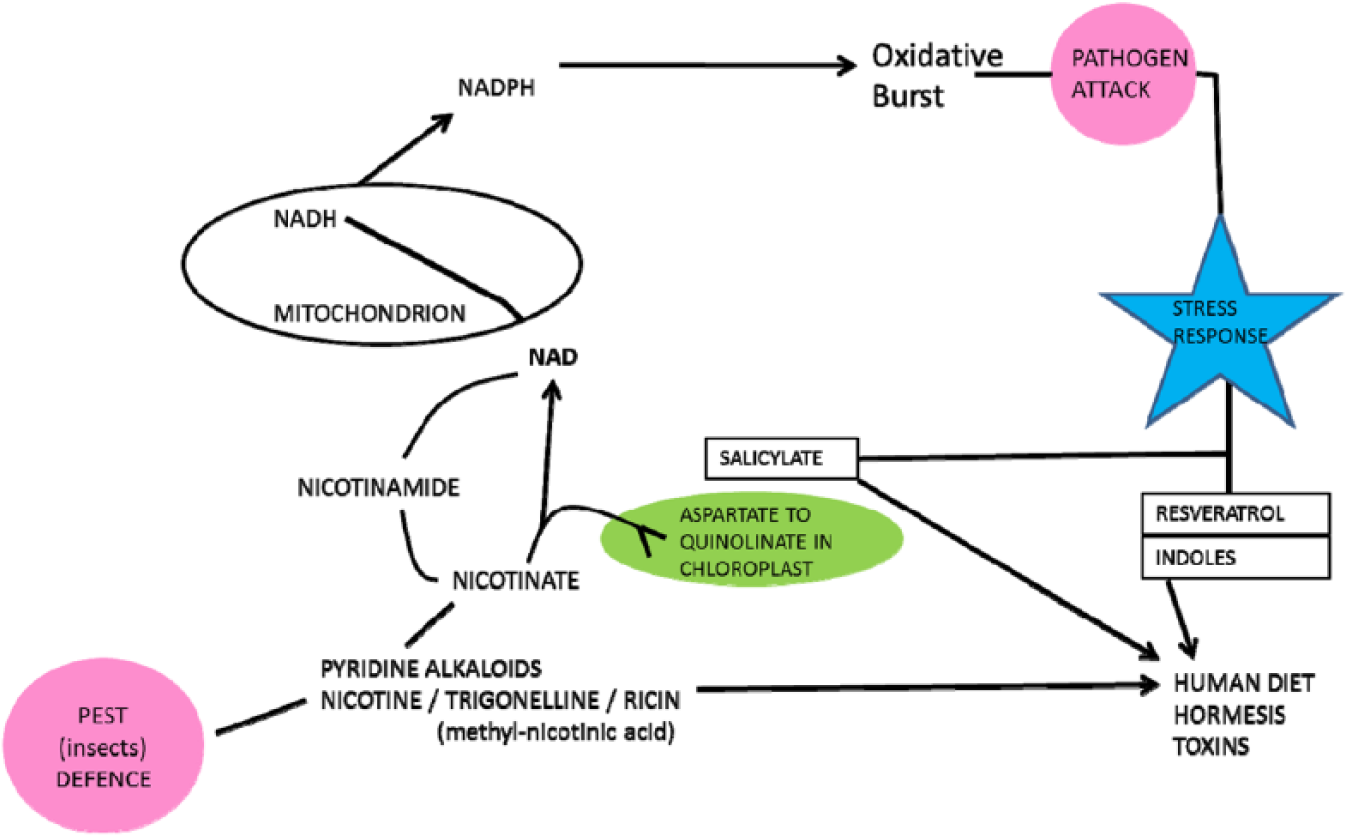
Plant and human metabolisms have active and overlapping reactions involving nicotine and nicotinamide. Stress molecules such as nicotine, salicylate, and resveratrol have medicinal and hormetic actions in man.
Some hindgut symbionts produce nicotinic acid (that they share) as does TB60–62 (Figure 6).63–65 We have argued that when meat sources are good and shortages short-lived, TB acts as a symbiont farmed for its nicotinamide. The relationship with classic pathogens is different. Many have evolved to be consumers of host NAD, and many of their toxins directly affect NAD consumer pathways66–74 (Figure 7). If the host is moderately deficient in nicotinamide/NAD in the first place, they will be less resilient – if severe, the pathogen may not have enough to survive perhaps explaining the paradoxical relationship between nutrition and infection when very poor nutrition can be protective. 75

A designer symbiont. TB produces nicotinic acid but cannot recycle it to NAD so excretes it to a ‘welcoming’ host that ‘farms’ the organism at some risk of dysbiosis if the dietary dose of nicotinamide is too low. Nicotinamide is a natural and its analogues are some of the artificial anti-tuberculous agents. NAD indicates nicotinamide adenine dinucleotide; TB, tuberculosis.
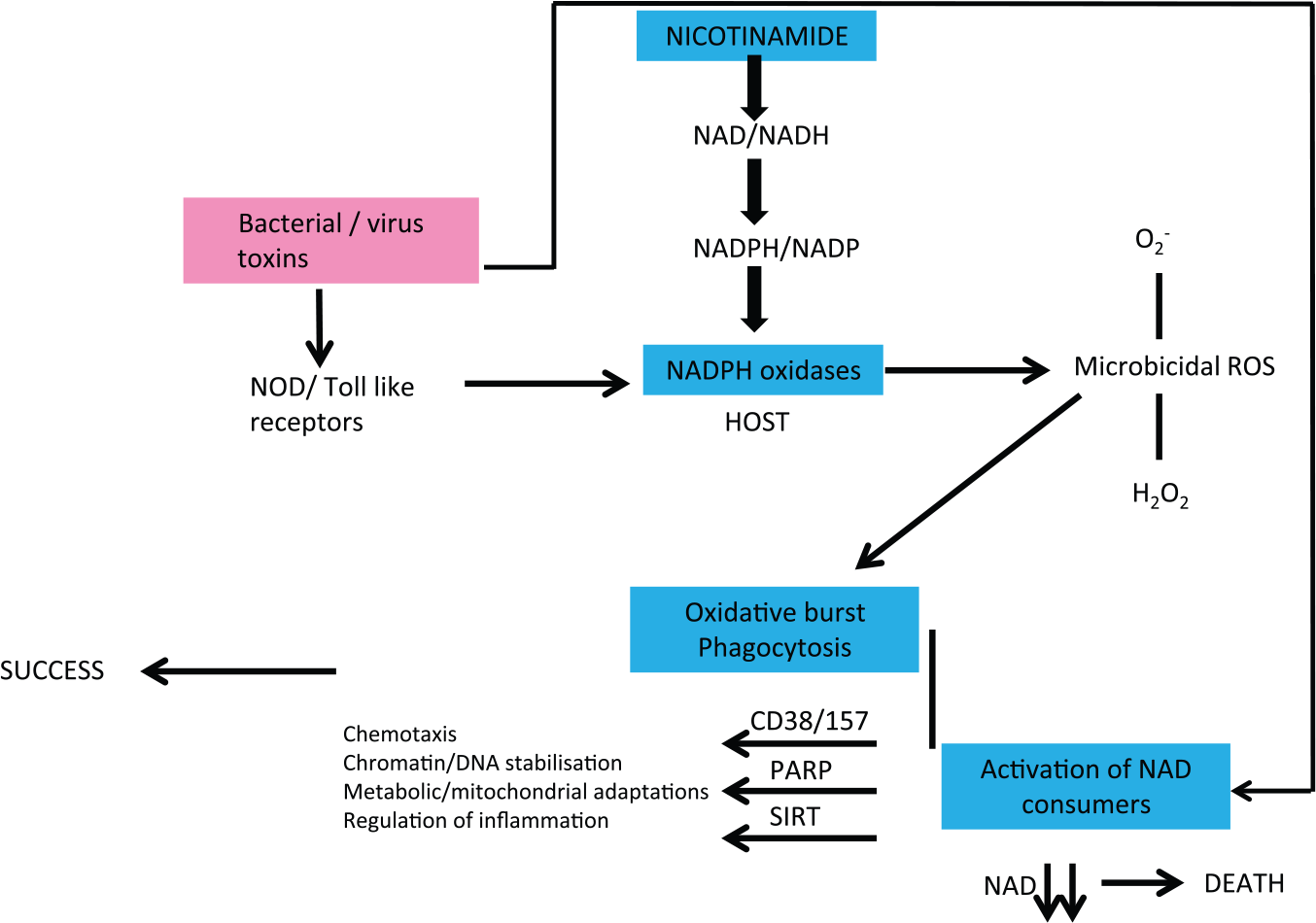
Infections and host NAD metabolism are intertwined. Many pathogens evolved to consume host NAD or their toxins lead to host NAD depletion. If the host is NAD deficient, this will exacerbate virulence and death rates. If severely deficient, there may not be enough NAD to allow the pathogen to replicate. Symbionts, by contrast, can improve host NAD levels.
Nicotinamide adenine dinucleotide pathways have been heavily implicated in ageing and many diseases from the neurological to retinal disease to cancer and can restore stem cells.73,76–103 Recent evidence fits with the very wide phenotype of disease seen with pellagra. The cellular response to NAD deficiency (and excesses) across cell lines is heterogeneous and may explain these broad phenotypes suggesting that NAD/NADH upsets might be a common underlying cause of many diseases. 104
Pathways – Over- and Under-Nutrition
Over- and under-nutrition are bad for health as demonstrated by ‘Waaler’ curves for height, weight, and mortality. 105 We propose that they share pathways that intersect with symbionts buffering poor diet and both relate to nicotinamide metabolism (Figure 8). Butyrate (and nicotinic acid) are generated by gut symbionts that include Lactobacillus or Bifidobacteria or Clostridia sp that ferment fibre and other otherwise indigestible complex carbohydrates and are in high concentration in the colon.90,106–110 Butyrate is an endogenous ligand active on the G protein–coupled GPR109A nicotinic acid receptor (expressed on macrophages, dendritic and epithelial cells, and adipocytes); this sends non-redundant signals via interleukins that affect T-cell differentiation favouring Tregs over helper T cells when the diet is poor as does nicotinamide from the diet when it is rich.90,111–118 This signal from the ‘niacin receptor’ reduces inflammation and carcinogenesis and has independently been proposed as a factor in mediating and perhaps moderating diet and disease transitions91,119
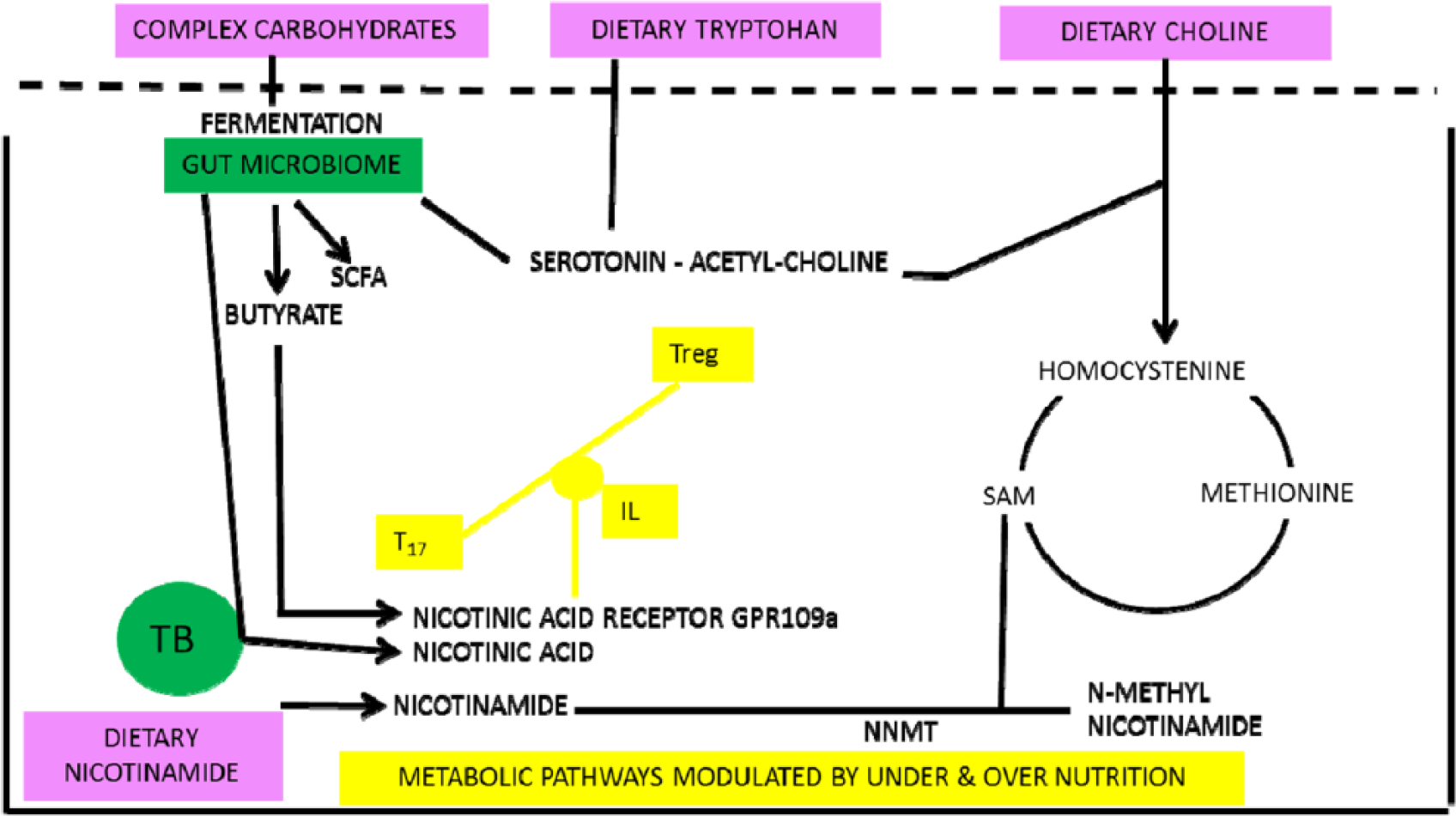
Low-meat/high-fibre diets lead to symbionts that increase nicotinamide levels or produce butyrate. Butyrate is an agonist at the nicotinic acid receptor as well as having epigenetic effects. Both butyrate and nicotinic acid will affect the T-cell balances and immunologic tolerance.
There is much supportive evidence of differences between the microbiome on rich, poor, and traditional hunter-gatherer diets and that the tryptophan pathway is involved in states of malnutrition (or rare but illustrative genetic defects that affect nutrition).120–122 Reliance on the microbiome could mean that those on a poor diet are at risk of broad-spectrum antibiotics temporarily eliminating symbionts and triggering clinical nicotinamide deficiency – pellagra (as can happen with anti-tuberculous therapy).123,124
Although many factors have been shown to alter traffic in the ‘de novo’ pathway, particularly infectious diseases, it is important to remember that its primary purpose is the production of nicotinamide/NAD from tryptophan when the diet is inadequate (Figure 9). The consequences are immune tolerance to some useful symbionts. Immune tolerance is mediated by an effect on the balance between Tregs and T17 helper cells – also the target for steroids and many modern immune therapies or artificial infection with helminths. Kynurenine-derived toxins drive auto-carnivory that also releases NAD and tryptophan at the longer term cost of organ damage. The host will be more prone to pathogens, so there will be other severe downsides for the individual.78,125–130 On a high-nicotinamide diet, the opposite may be occurring with immune intolerance to normally harmless antigens.131–135 Although induction of indoleamine 2,3-dioxygenase (IDO) has been described paradoxically (on our theory) in some of these inflammatory or cancerous conditions, there is a difference between a predisposing endo-phenotype and one that is being influenced as the disease develops: we would argue that IDO induction can be compensatory (Figure 10).136–151
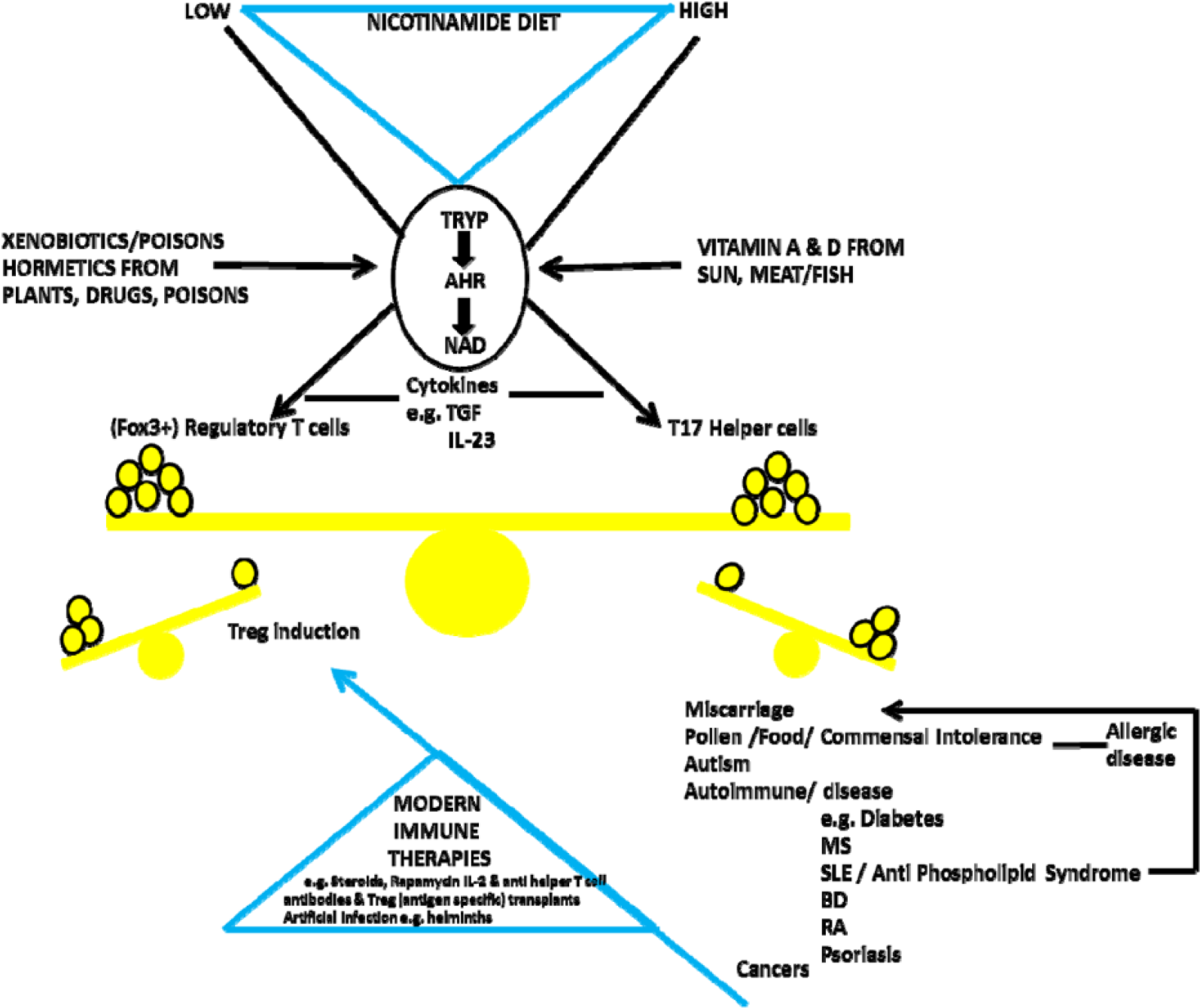
The key switch is the kynurenine ‘de novo’ pathway. When dietary supplies of nicotinamide/NAD are not sufficient, there is Treg-induced tolerance for metabolically useful symbionts but dangers to individual health from dysbioses or pathogens. However, when dietary nicotinamide is high, there is immune intolerance with too few Tregs and an excess of pro-inflammatory T17 cells and many diseases of modernity. Many immunologic therapies from steroids to recent T-cell–targeted approaches or artificial infection try to correct this imbalance. Prevention might be more effective and safer.

Predisposing phenotype for inflammatory disease driven by high nicotinamide in diet leads to reduced IDO activity. The more immediate triggers to these diseases and the disease process itself may sometimes lead to the apparent paradox of induced IDO as a compensation that may exacerbate or mitigate the disease. Lack of early infections or allergens may be ultimate causes but can act later as proximate triggers. IDO indicates indoleamine 2,3-dioxygenase.
We will now look at some data from the United Kingdom, 1850 to 1950, and across the contemporary world comparing states of under- and over-nutrition and diseases and health markers that are likely to be related to nicotinamide dose, as many are components of pellagra.
Methods
All meat data were collected from ‘Eating meat: evolution, patterns, and consequences’ by Smil 152 and The Atlas of Food Who Eats What, Where, and Why by Millstone and Lang. 153 IQ and literacy data were from ‘Some British pioneers of social medicine’ by Greenwood 154 and ‘National IQS predict differences in scholoastic achievement in 67 countries’ Lynn et al. 155 Tuberculosis data were collected from McKeown 156 and the Institute for Health Metrics and Evaluation’s Global Burden of Disease (GBD) (2013). 157 The Parkinson disease (PD) data were taken from Duvoisin and Schweitzer 158 and the Institute for Health Metrics and Evaluation GBD (2013) (http://www.healthdata.org/gbd). All disease data had been corrected for age structure of populations. Cancer data were derived from ‘Mortality in England and Wales from 1848 to 1947’ by Logan 159 and the Institute for Health Metrics and Evaluation’s GBD (2013). Diabetes data were taken from The Health of Adult Britain 1841-1994 by Charlton and Murphey 160 and from the Institute for Health Metrics and Evaluation’s GBD (2013). Life expectancy data were from ‘Ecological public health: the 21st century’s big idea?’ by Rayner and Lang 161 and The Atlas of Health: Mapping the Challenges and Causes of Disease by O’Donovan. 162 Height data sets were from Galton’s midparent height revisited by Cole. 163
Statistics
Exploratory analysis was conducted on these data to identify relationships between meat consumption and other variables by conducting scatterplots. The correlation between meat consumption and other variables was analysed using the Pearson correlation coefficient. All statistics were conducted using SPSS (version 21).
Results
In summary, and in round terms in the United Kingdom between 1850 and 1900, death rates per million with both sexes combined are as follows: for TB (before any treatment) halved from 7000 to 3500, for dysentery fell by 9/10ths from 150 to 15, whereas cancer (before much effect from smoking, at least in women) increased by 2.5-fold from 700 to 1700, and diabetes increased 4-fold from 50 to 200. The incidence of Parkinson’s disease rose markedly having only been described in 1817. During this period, meat intake almost doubled. Measures of cognitive and physical health such as literacy rates increased as did height. Contemporary data comparing these conditions with average meat intake across countries support these correlations.
More specifically, the reduction in deaths from diarrhoea between 1850 and 1950 in the United Kingdom trended with the rise in meat intake (P = .085) (Figure 11).
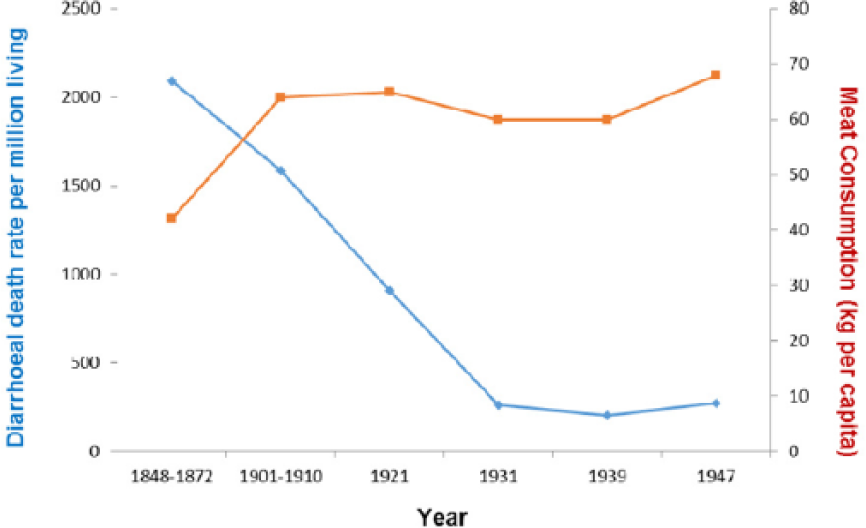
Diarrhoea plotted against meat intake in the United Kingdom, 1850-1950 (r = −0.642; P = .085).
Across the world today, deaths from diarrhoeal diseases correlate strongly with meat consumption P > .0001 (Figure 12).

Diarrhoea plotted against meat intake in the contemporary world across nations (r = −0.508; P < .0001).
Literacy rates in the United Kingdom between 1850 and 1900 correlated with the rise in meat consumption P > .001 (Figure 13).
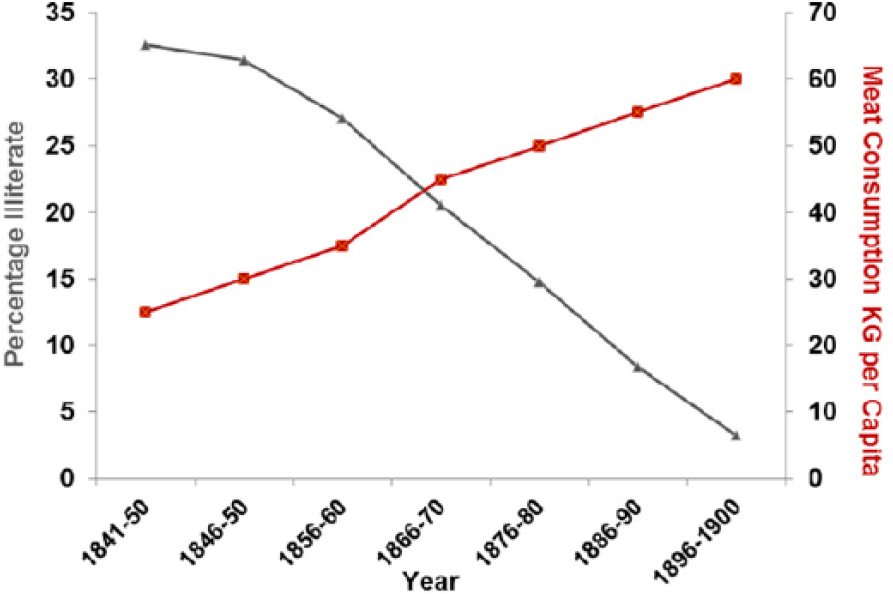
Literacy rates plotted against meat consumption in the United Kingdom, 1850-1900 (r = −0.988; P < .001).
In the contemporary world across nations, literacy rates correlate strongly with meat consumption P > .001 (Figure 14).

Literacy rates plotted against meat consumption in the contemporary world across nations (r = 0.531; P < .001).
Measured IQ correlates with meat consumption currently across countries P > .001 (Figure 15).

IQ plotted against meat consumption in the contemporary world across nations (r = 0.538; P < .001).
Between 1870 and 1970, boys height increased and correlates with meat intake (P > .001) as does height across nations currently (P > .001) (Figures 16 and 17).
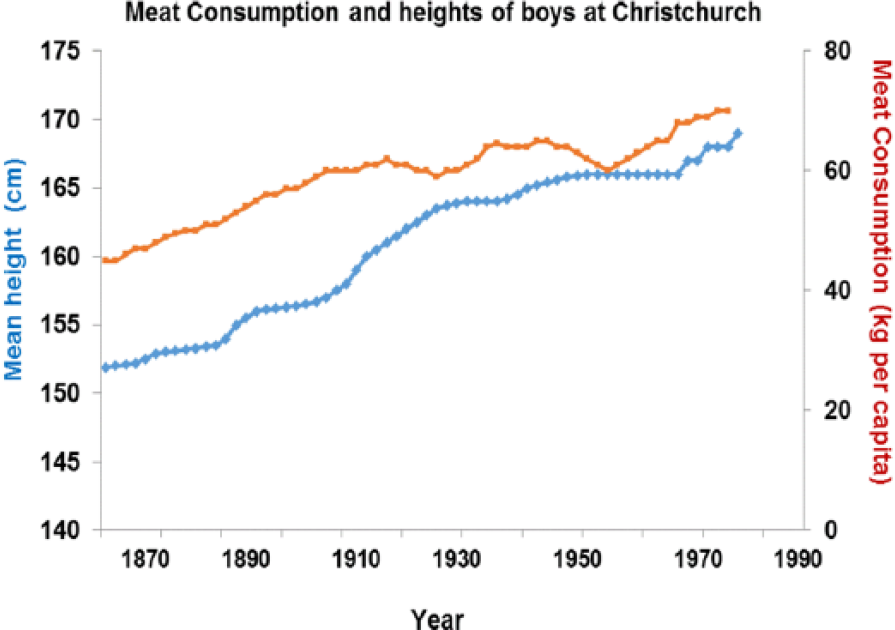
Increased height correlates strongly with higher meat intake (r = 0.934; P < .001).

Increased height correlates strongly with higher meat intake (r = 0.635; P < .001).
Between 1850 and 1970, also between 1850 and 1950 (before any drug treatment), TB rates fell dramatically and this correlates with increased meat intake (P > .001). The decline in TB did not happen at the same time even within Europe and was also delayed in other industrial nations such as Japan where improvements in diet particularly of meat were also delayed. 164 Reverses are well described under famine conditions165,166 (Figure 18).
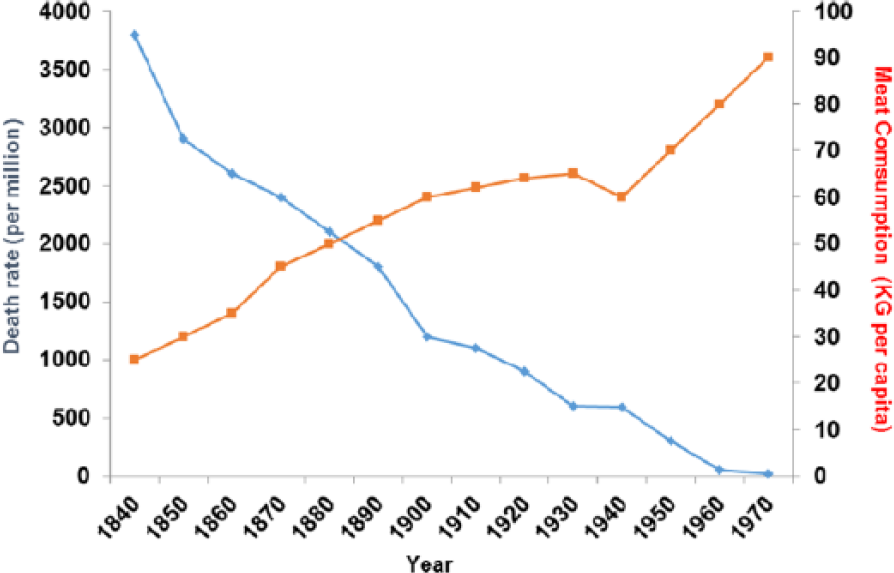
Tuberculosis plotted against meat consumption in the United Kingdom, 1850-1920, before there was any drug therapy (r = −0.958; P < .001).
This correlation is true across countries today where meat consumption correlates strongly with deaths from TB (P > .0001) (Figure 19).
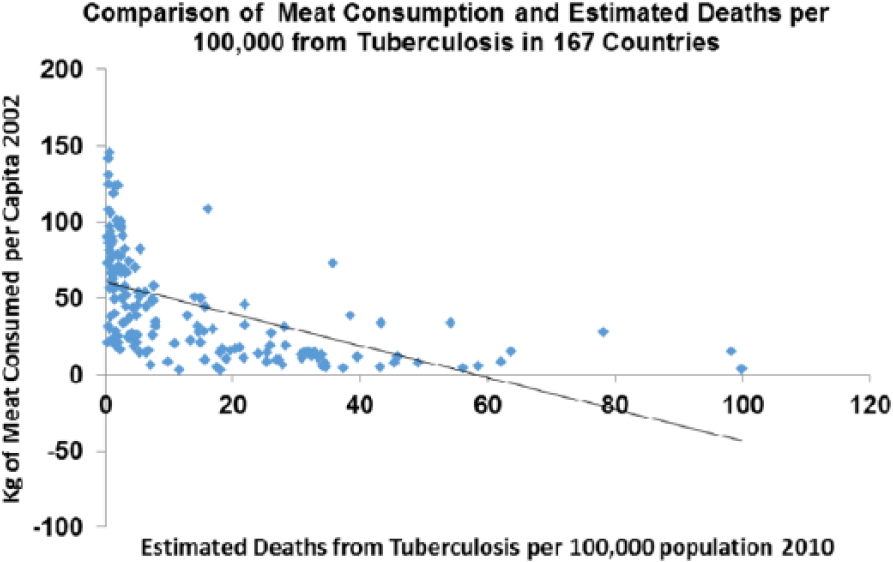
Tuberculosis plotted against meat consumption in the contemporary world across nations (r = −0.545; P < .0001).
Diabetes deaths rose between 1850 and 1950 and correlate with meat intake (P > .05) as they do across the world today (P > .001) (Figures 20 and 21).

Diabetes plotted against meat consumption in the United Kingdom, 1850-1950 (r = 0.756; P < .05).

Diabetes plotted against meat consumption in the contemporary world across nations (r = 0.247; P < .001).
Cancer rates were increasing in both sexes in the United Kingdom from 1850 to 1900 before there was much effect from smoking (and none in women) and correlate with meat intake (P > .0001) as they do across the contemporary world (P > .0001) (Figures 22 and 23).
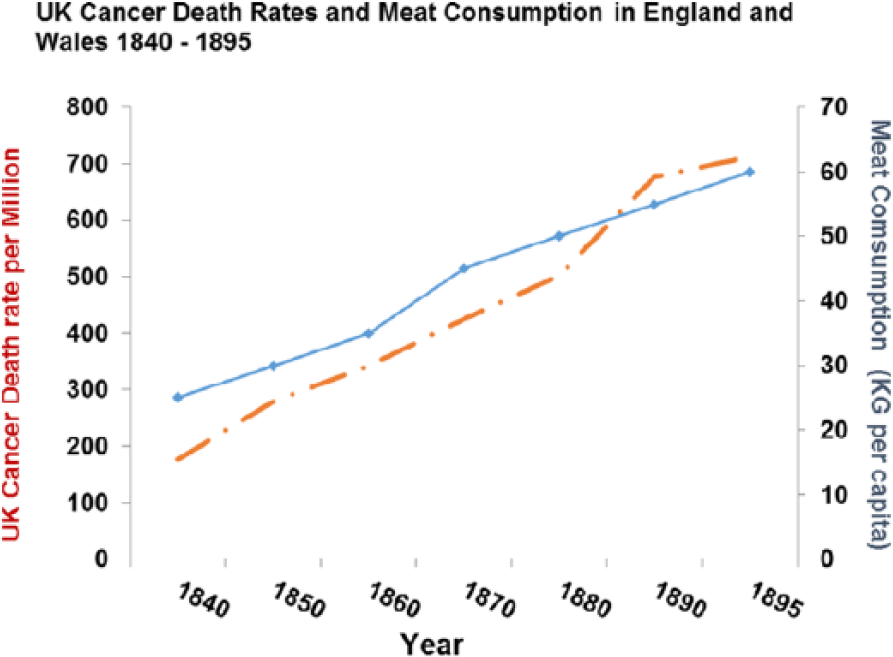
Cancer death rates plotted against meat consumption 1850-1950 (r = 0.981409; P < .0001).

Cancer death rates plotted against meat consumption in the contemporary world across nations (r = 0.667; P < .0001).
Rates for the Parkinson disease were increasing between 1850 and 1960 (P > .001) and correlate with meat intake as they do across the world today (P > .0001) (Figures 24 and 25).

Parkinson disease plotted against meat consumption 1850-1900 (r = 0.842; P < .001).

Parkinson disease plotted against meat consumption in the contemporary world across nations (r = 0.842; P < .0001).
The Allergy Epidemic, 1870-Till Now
The recent rise and exact timing of the rise in hay fever and allergic/auto-immune disease are less well-documented numerically – although the facts are not in dispute and average meat intake has also doubled again between 1960 and now, so if they were strong correlations would be obtained. 34 Apart from a very few references in ancient literature, allergy first in the form of hay fever was first described in the mid-19th century exactly at the time the TB epidemic was abating in both the United Kingdom and in the United States.167–170 Observers at the time described these conditions and noted that they were on the increase, to begin with among the fashionable wealthy. Allergic disease had been unknown in previous generations (much as they are still unknown in poor African villages today). Increases are being recorded in countries that are getting richer or have had the economic benefits of European re-unification.171–180 Proximate triggers such as grass and ragweed pollen were noted early on, but they do not contain new antigens even if some became more prevalent. Rather paradoxically avoiding the allergen can make matters worse, as recently demonstrated for peanut allergy, so treating the proximate allergen as the actual or preventable cause is problematic. 181
The ultimate cause of the epidemic has been linked by others to the decline of TB using evidence from BCG vaccination and inverse associations with tuberculin responses or other infections.182–185 Current maps of the incidence of infections such as helminths and TB are diametrically opposed to incidences of allergic and auto-immune diseases 186 (Figure 26). Reduction in helminth infection, as with TB, is also associated with relief from poverty and poor diet with limited overall impact of antibiotics at least at a population level: as animal parasites, their interaction with host nicotinamide status and biochemistry could be either positive or negative but has not been studied.187–191

Incidence of helminth infestation and tuberculosis is diametrically opposed with incidence of autoimmune disorders now. In 1850, this map would have looked more homogeneous.
A reasonable proposition as to timing of the allergy epidemic in the northwest is shown 192 (Figure 27). Not only numbers but also diversity of auto-immune disease continues to radiate. 193 Links with NAD metabolism through CD38 and allergen/pollen NADPH oxidases have been made; the latter links in to oxidative stress and T-cell imbalances that could cause allergic disease but only if Tregs were unbalanced in the first place, prior to exposure to the immediate extrinsic signal.194–200 High tryptophan levels and low IDO activity have also been recorded in allergic disease whether asthma or food allergies.201–203 In addition, the modern outbreak of depression appears, despite cross-cultural issues to be a feature of diseases of affluence, and may influence many of the others and can clearly be related to disturbed tryptophan and serotonin pathways.204,205 The commonest cause of infertility – polycystic ovary syndrome – is also far commoner in developed economies and has been related to the metabolic syndrome and auto-immune disease; this is relevant to the argument we make in our companion article about increasing infertility in high-meat economies. 206

The rapid and sequential rise of allergic and auto-immune disease in the United Kingdom is shown. During this period, meat intake on average doubled again, so if accurate figures were available for the new diseases, strong correlations would be apparent. Incidence and severity may be stabilising for some such as asthma (as is meat intake).
Questions
We now ask a series of questions to further explore and try and convince readers that the dose of vitamin B3 is a significant health issue and that deficiencies and perhaps excesses are commoner than is usually supposed.
Question 1a: was pellagra conquered or is it still endemic in some poor meat areas?
The short answer is that nobody has checked systematically at a clinical or biochemical level. The few studies done in high-risk populations such as refugees in war-torn Africa suggest that it may be common but rarely diagnosed.207–211 Pellagra may be treated inadvertently in alcoholics while restoring thiamine levels with multivitamins for Wernicke encephalopathy.212–215 Indeed, it may genuinely be difficult to separate nicotinamide deficiency from deficits of other micronutrients as seen with tropical neuropathies; even when there are putative (cyanogenic or amino acid) toxins involved, nicotinamide deficiency could play a part in degenerative diseases when on poor monophagic plant diets such as those described on Guam as well as Lathyrism and Konzo.216–219 Some of these conditions including pellagra, like scurvy in the past, can get considered to be ‘badges of dishonour’ (even though captains Columbus and Cook were probably affected) or wilful self-neglect rather than due to chronic food shortages obfuscating calls to action. 220
Pellagra happens to those on a very low-meat/milk and maize diet, so many in Africa and Asia must be at risk. The rash is photosensitive, so may not be present as often in those with pigmented skin – earlier epidemics were mainly in poor whites. Diarrhoea and poor mental development are endemic. Kwashiorkor, when first described, was felt by some to be a form of pellagra, but the argument was initially lost to the proponents of calorie or protein deficiency.221–224 The renamed ‘environmental enteropathy’ with gut dysbioses, cognitive impairment, and later ‘epigenetic’ metabolic syndromes may have a closer relationship with a ‘new version’ of pellagra than is generally appreciated.225,226 Pellagra may be masquerading as general ill health and shortened lives with lower than expected IQ and exacerbate both TB and human immunodeficiency virus.227–229
Question 1b: was pellagra sine pellagra conquered or is it still a common cause of poor intellectual development or premature ageing including dementia?
Before the biochemistry and treatment of pellagra were sorted out in the 1940s, it was pointed out that lack of meat in diet had profound effects on health and height, and the clinical manifestations went outside the classic pellagra phenotype.230,231 This view was criticised by vegetarian groups (including Gandhi). We now realise that there is a world of difference between an economically driven poor monophagic vegetarian diet and a voluntary vegetarian diet of good quality that often includes some animal products and supplements – so the latter has few dangers and some advantages. 232
During the American and earlier European epidemics of pellagra, often in families with other members diagnosed, there were many with poor physical and mental development. Indeed, a high proportion of both blacks and ‘white trash’ in the Southern states may have been affected. The average IQ of Confederate recruits was in the ‘moron’ category and that was not true for the richer Union recruits. The correlations we now see between IQ/literacy and meat intake may represent nicotinamide deficiency. The dramatic changes in IQ known as the ‘Flynn effect’ as countries become more prosperous may reflect increased nicotinamide intake and a better brain in the first place more capable of learning from a better educational system and less prone to dementia.233–235 Indeed, there is already considerable evidence that nicotinamide could prevent or benefit Alzheimer’s disease and has a role in serious neuropsychiatric disease such as schizophrenia and post-traumatic dementia.236–248 Increased longevity could also represent improving nicotinamide dosage. This is supported by pellagra causing premature ageing and dementia and evidence that NAD levels fall with age given that NAD consumers, whether SIRTs or PARPs, are largely responsible for ageing and repair mechanisms 249 (Figure 28).

A summary of how poor diet can interact with the microbiome and with internal stresses such as mutations and with external stresses. These can all contribute to a single NAD endo-phenotype with multiple clinical phenotypes. NAD indicates nicotinamide adenine dinucleotide.
Question 2: is there a strict recommended dose of nicotinamide or do some people with some genetic mutations require a personalised dosage and do some others temporarily need a boost in their dosage when under stress?
The recommended daily dose of nicotinamide of around 15 mg/day was a reasonable informed ‘guesstimate’ designed to avoid or treat pellagra. However, there are examples of increased needs under some mutational circumstances, such as those causing muscular dystrophy or optic atrophy.250–252 The same may be true of DNA-repair or metabolic defects such as those causing ataxia-telangiectasia, Friedreich ataxia, defects in glutamine synthetase, and some cancers.253–256 Other diseases such as Huntington’s are known to have a disturbed ‘de novo’ pathway and an intervention with nicotinamide could work.257,258
It has been known for a long time that the dose needs to be increased in patients with the carcinoid or Hartnup syndrome who shunt tryptophan to increased serotonin synthesis or have a tryptophan transport defect.259,260 If there is genotoxic stress such as that from chemicals or drugs, sunlight, or even emotional stress, it would be expected that the extra NAD consumption would be easier to support if the dietary dosage was high. There is emerging evidence that nicotinamide dosage can be (neuro)protective under a wide range of environmental insults whether traumatic, anoxic, or toxic suggesting that prophylactic boosting of the dosage or treating soon after the insult may reduce cell damage in a number of organs, including the brain.96,104,236,261–290 Many diseases where the environmental trigger is not known may have an NAD-deficient endo-phenotype. This is suggested by the extraordinarily varied phenotype of pellagra where a wide range of dementias and neuropsychiatric conditions was described including mimics of Creutzfeldt-Jakob disease, PD, motor neuron disease, multiple sclerosis (MS), and cerebellar syndromes alongside migraine and epilepsy. Evidence for such a deficit is already strong for some, such as prion diseases, where there is phenotypic overlap with pellagra.291,292
Question 3: was the change in dosage of nicotinamide the transition factor converting diseases of poverty to diseases of affluence? Did this lead to the loss of ‘old friends’ such as TB allowing a poorly educated immune system to become overactive?
Nicotinamide adenine dinucleotide status is probably crucial to the poor resistance to pathogens seen in poor countries. Many pathogens or their toxins target the host’s NAD system by disrupting NAD consumer pathways, or by secreting NAD glycohydrolase, break down the host’s NAD levels. 293 We have also argued that TB and some gut symbionts originally co-evolved to supply nicotinic acid. Such symbionts become dysbiotic and behave as pathogens when the diet is low in meat for protracted periods of time, or if driven to mutate using antibiotics. Nicotinamide was the first anti-tuberculous antibiotic discovered. The largely spontaneous disappearance of TB as countries and diets become richer becomes less of a mystery and is less likely to be related to better hygiene. There are, indeed, good examples of improving sanitation improving water-borne infection such as cholera but having little impact on other infections, until diet and particularly meat intake is addressed. 294 In one famous example, improved sanitation caused an increase in mortality as the increased rents led to a decline in food quality. 295 A famous other example was rebuilding barracks to no effect on TB rates until the meat intake was increased, and of course, TB becomes rampant during many famines. 296
It is notable that TB sanatoria were not closed but were transformed into sanatoria for allergies that first became a problem for the rich on rich diets.15,297–303 A ‘nicotinamide switch’ towards the evolutionary norm of a high-meat diet could be responsible for the switch from TB and other chronic infections, now co-evolved to affect the development of the immune system, to a poorly educated overactive immune system responding to normally innocuous antigens.81,304–310 The biochemical nature of this switch was outlined earlier (Figure 9). The speed at which the switch is turned could be important, with the highest risk within a lifetime or a generation or two for disease of affluence – thereafter, immune adaptations and a different starting point of the microbiome may occur compatible with little evidence that hunter-gatherers had auto-immune/allergic disease (trauma leading to shorter lifespans may have spared them later onset diseases).
The spread and origin of the obesity epidemic is interesting regarding this as modern maps of the epidemic and obesity, diabetes, and stroke ‘belts’ are strikingly similar to maps of pellagra a century ago suggesting that the switch can manifest over several generation raising future discussion over ‘thrifty’ (and in other contexts ‘non-thrifty’) nicotinamide-related genotypes and phenotypes and developmental plasticity having effects on later disease compatible with the known involvement of nicotinamide, butyrate, and methyl groups in epigenetic modifications 311–316 (Figure 29).

US map illustrates the obesity epidemic and a diabetic belt. The distribution is almost exactly the same as maps of pellagra a century ago. Has the change from very low levels of nicotinamide to high levels been the crucial inter- and intra-generational change rather than calories or allergens?
Question 4: over and above a rash of immune diseases has the dose of nicotinamide become so high that there is an unrecognised hyper-vitaminosis B3 syndrome?
Nicotinamide is widely viewed as having little serious toxicity at least in the short term.317,318 We suggested nicotinamide toxicity as a causative factor for the Parkinson’s disease, the metabolic syndrome, and some cancers based on direct or indirect measure of high levels of NNMT.53,83,319–333 Too low a dose and nicotinamide can be protective for cancers and Parkinson’s, so we are proposing a double-edged dosage effect. 333 As an inducible enzyme, a logical culprit for NNMT overexpression is the dose of nicotinamide – even if other factors such as caloric restriction, stress, and exercise play their part.334,335 Background genetic variation in levels may reflect exposure in earlier generations – certainly, this is true of species as the enzyme is not expressed in herbivores. 336 Our original hypothesis for PD was that N-methylnicotinamide resembled the dopaminergic toxin MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) and so may be toxic at high levels even though beneficial at lower levels. Part of the jigsaw may be that the nicotinamide dose upsets the microbiome that in turn contributes to the proteotoxicity and affects the nicotinic acid receptor. 337 Other metabolites may be nephrotoxic. 338
Several other plausible mechanisms for nicotinamide toxicity exist whether from excessive inhibition of NAD consumers such as SIRTs or PARPs or consumption of valuable methyl groups depleting the methylome and the epigenome (Figure 30). Recent evidence shows that nicotinamide promotes adipogenesis probably via SIRT inhibition and adipogenic proteins (eg, peroxisome proliferator–activated receptor gamma and FABP4 [fatty acid–binding protein 4]) and is associated with neonatal adiposity – with surprisingly little need for excessive calories or fats.339–342

The presence of a detoxification pathway suggests that nicotinamide can be toxic. A balance may have been required as NAD consumers evolved to be an important control mechanism that both needs a supply of NAD from nicotinamide and whose enzyme activity is affected by nicotinamide.
Question 5: how hard would it be to moderate the dose of nicotinamide worldwide or personalise dosages?
As dietary manipulations go reducing, the dosage would be easy as supplements could be reduced in rich countries with no behavioural change necessary. Reducing meat intake may also be fairly painless, even if nicotinamide like nicotine has some addictive qualities, as it is now practised by the ‘healthy wealthy’.
Increasing the dose to those really at risk would also be relatively easy and not very expensive if talking about supplements or biofortification of crops. More meat, however, is not as easy as it is expensive but may be necessary if nicotinamide is not the only factor necessary or is actually dangerous on its own as it is a drain on methyl groups. Redistributing meat from those eating too much to those eating too little would improve the health of both the groups.
Conclusions
Hypo- (subclinical/misdiagnosed pellagra) and hyper-vitaminosis B3 (nicotinamide overload) may both be far commoner than has been supposed and have equally wide phenotypes transcending many conventional clinically convenient disease categories. The transition from populations of low to high intake may explain the extraordinary shifts within single generations from Rowntree’s ‘stunted life’ with chronic diarrhoeal infections and TB and disproportionally high death rates from pathogens – to non-communicable diseases. Non-communicable diseases include multiple and expanding allergic, inflammatory, and auto-immune diseases along with the metabolic syndrome, many cancers and neuropsychiatric disease, but in the context of longer lives. Geography per se has little to do with these phenomena as they track much more closely with poverty and affluence and therefore meat intake.343–345 Even the term ‘tropical diseases’ is a misnomer as many of these infections were common in Victorian England and the pellagra-prone southeastern American states a century ago.
Much current effort in rich countries is for personalised precision and genetic medicine usually for people who already have contracted often rare diseases. Understanding these disease transitions may be less eye-catching but offers more potential for low-tech population-based preventive approaches. Personalised NAD-related medicine can be nutritional, rather than glitzy genetic manipulation, as several genetic diseases may be due to genes that originally evolved at times of nicotinamide luxury or thrift.
Predicted tidal waves of dementia may be preventable (too little nicotinamide) as may some neuropsychiatric disease, diabetes and the metabolic syndrome, some cancers, and Parkinson’s disease (too much). 346 Nicotinamide overload may not work alone but with other hyper-vitaminosis or deficiency states. One example might be high nicotinamide but low vitamin D in MS compatible with the known epidemiology and genetics implicating little sun but a lot of meat and known involvement of the ‘de novo’ pathway.347–349 It will have been partly iatrogenic if increasing the dose of nicotinamide with supplements also affects caloric hunger and obesity levels and other diseases where NNMT induction has been demonstrated.350,351
Phenotypic diversity may be explained by individual variation whether genetic or from previous environmental exposure and in differential sensitivity of cell types to NAD upsets. Homeostatic responses that fail or have a longer term price whether auto-carnivory, use of symbionts, or inflammatory or cancerous tissue that over-express NNMT or NAD consumer enzymes or induction of IDO, may also be responsible for clinical heterogeneity. New phenotypes may be occurring. Predicted Armageddon as antibiotics rapidly become ineffective from multi-drug resistance may be due to not dealing with the fundamental nutritional cause. Antibiotics are a strategy that encourages mutations and emergent pathogens. Better NAD status will reduce the need for broad-spectrum antibiotics by reducing virulence. Reliance on symbionts (including TB) also means that antibiotic use could trigger pellagra that will probably not be recognised or treated.352–356 When symbionts are relied on too heavily, ‘blooming’ in the gut can lead to dysbiosis and pathogen evolution relevant to ‘environmental enteropathy’ that may be ‘new version’ pellagra88,347,357 (Figure 31).

An optimal dose of nicotinamide is suggested with trouble at the extremes. Transgenerational effects may be marked. Dysbioses that begin under these circumstances could put the affluent at risk. Within generation effects may be mismatches between early and late life exposure with poor nicotinamide in early life predisposing to the metabolic syndrome later if the dose increases. TB indicates tuberculosis; PD, Parkinson disease.
All our questions could be answered definitively. The variances in environmental exposure could be analysed in more detail epidemiologically and not just using meat intake as a surrogate. Clinical assessments sensitive to the possibility of pellagra in at-risk groups could be made. Joint clinical and biochemical assessments are the likely way forward closely followed by interventional studies.358,359 Biochemical measures have been available for a long time measuring tryptophan levels, or urinary N-methylnicotinamide excretion or NNMT levels are enough to confirm pellagra (or nicotinamide overload) but have rarely been used in the field. Metabolomics could make this more practical. Direct measurement of NAD/NADH or NAD/NADP (nicotinamide adenine dinucleotide phosphate) (known as the ‘niacin number’) ratios could be further developed. Laboratory studies could explore nicotinamide overload further as it has been assumed that NNMT or high N-methylnicotinamide levels are a marker or reaction rather than directly involved in causation. 360 The long lifetime nature of this toxicity with doses only 2 to 4 times the upper recommended range would need to be recognised in the experimental design (Figure 32).
Nicotinamide overdosage is unlikely to be working alone. It may act in concert with other excess dietary factors known to be involved with ageing and pathological pathways and may take many years to express toxicity.
Moderating the dose of nicotinamide to begin with would be easier than most dietary manipulations as supplementation is happening where it is least needed in rich countries. 361 Supplementation should be targeted at some 2 billion who are already known to be micronutrient deficient (iron, zinc, iodine, vitamin A, and folate) including the one-fifth of children who are physically or cognitively stunted. The cost has been shown to be low relative to the benefits (16-fold) either at an individual level where many are robbed of future earnings or by population – malnutrition loses 10% of gross domestic product in many parts of Africa and Asia.362–367 Meat subsidies or vouchers or conditional cash transfer systems as part of a meat ‘entitlement’ reducing meat insecurity would make sense as nicotinamide is unlikely to be the only fortification factor involved. Adding nicotinamide to diet without methyl groups could be problematic as they would be lost as excesses of nicotinamide are excreted.
Systems need to be developed that nourish rather than feed, and sometimes this means more meat not less and should be driven by evidence not ideology. 361 At first glance, there is a big environmental price to pay for more equitable meat intake. Access is as much of an issue as lack of available meat, so redistribution would mitigate many ecological effects. When combined with the demographic argument we made in the accompanying article, this may all have to be addressed objectively – unnecessary transgenerational disease or high population density is the greater ecological danger to future generations. The diet instinctively followed by the ‘healthy-wealthy’ may be a win-win diet improving health and human capital and be environmentally sustainable (Figure 33). 368 In addition, the wealthy who make more effort to exercise in safe environments and virtual hunting as sport, and consequently control weight, may be overcoming not so much evolutionary tendencies to gain weight preparing for famines (thrifty genotypes and phenotypes) but to overcome a fear of exercise as it was previously linked to danger from predators (Figure 34).369–371

The ‘healthy-wealthy’ have instinctively worked this out. Can lessons learnt influence policy in developing nations?

Finally ancient meat gluts lead to an adaptive strategy saving lives from periods of safety not hunting and later stabilising weight. This is now maladaptive unless there are alternative strategies to increase exercise levels. Guarding against thrift may not have been as important as starvation may not have been as common as previously suspected.
Learning from history could help developing countries avoid the trap of the Western diet with lack of exercise and meat intake ‘overshoots’. There is little sign of such wisdom currently as ‘the double burden’ in developing countries reflects a break down in the Engel law whereby poor-to-moderate wealth transitions classically increased the absolute (but not the proportional) amount of income spent on food increasing meat intake. 372 This economic transition is now more likely to lead to increased ‘empty calories’ from ultra-processed foods such as sugar-sweetened nicotinamide-enhanced drinks driving the apparent paradox of diseases of poverty and the metabolic syndrome co-existing in developing countries and sometimes in the same individual.
Footnotes
Acknowledgements
A.C.W. would like to thank Mike Hammond and the Richardson family for their support.
Peer review:
Five peer reviewers contributed to the peer review report. Reviewers’ reports totalled 288 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is funded by QEHB Charities.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
ACW and LJH analysed the data, agree with manuscript results and conclusions, made critical revisions and approved final version. ACW wrote the first draft of the manuscript. LJH contributed to the writing of the manuscript. All authors reviewed and approved the final manuscript.
