Abstract
Over the past several decades, stem cell therapy for heart disease has been translated from the bench to the bedside and in clinical trials improves cardiac structure and function in both ischemic and nonischemic cardiac disease. Although the regenerative effects of stem cells in cardiac disease are mediated by both paracrine and cell-to-cell contact mechanisms, many of the downstream signaling pathways remain to be fully elucidated. This review outlines what is currently known about the main signaling pathways involved in mesenchymal stem cell and cardiac stem cell survival, proliferation, and migration and mechanisms of action to repair the damaged heart.
Introduction
Ischemic heart disease remains the leading cause of death worldwide and has remained so for the past 15 years despite advances in medical therapy. 1 After myocardial infarction (MI), cardiomyocytes begin to undergo apoptosis within hours if the vascular supply is not restored. Once necrosis and fibrosis set in, cardiac functionality declines and often heart failure results. Although medications have improved cardiac functionality following MI and other forms of cardiomyopathy, transplantation remains the only option once end-stage cardiac failure ensues. 2
Over the past several decades, stem cell therapy has evolved from the in vitro induction of cardiomyocyte contraction 3 to improvement in cardiac structure and function in both ischemic and nonischemic animal models4–9 and clinical trials.10–14 Since their discovery, a variety of stem cells including unfractionated bone marrow–derived mononuclear cells, mesenchymal stem cells (MSCs), endothelial progenitor cells, cardiac stem cells (CSCs), and induced pluripotent stem cells (iPSCs) have been tested in cardiac disease.15,16 Bone marrow–derived MSCs were among the first subpopulation discovered and tested for their regenerative capacity in cardiac disease. 17 Derived from the embryonic mesoderm, MSCs exhibit differentiation potentials into several lineages, such as cartilage, bone, adipose, muscle, and endothelial cells (ECs) under suitable conditions.18,19 Mesenchymal stem cells have been given particular attention due to their immunomodulatory and immunoevasive properties20–23 and their remarkable ability to localize to the heart and improve cardiac dysfunction.12,13,24–26
The heart was once considered a terminally differentiated organ without regenerative capacity; however, the discovery of CSCs in 2003 by Beltrami et al 27 spurred and challenged this paradigm. Subsequent research has confirmed that CSCs exist, reside in niches, and are involved in cardiomyocyte renewal throughout the duration of life.28,29 Other studies of CSCs in small and large animal models have demonstrated their expansibility in vitro and positive regenerative capacity on infarcted myocardium.5,30–34 Cardiac stem cells are multipotent and can differentiate into ECs, vascular smooth muscle cells (VSMCs), and cardiomyoctes and integrate into the host’s native myocardium.27,31,35,36 As such, they have garnered great interest as therapeutics to regenerate and restructure the damaged heart.
Mesenchymal stem cells have been studied for decades; however, we have yet to discover their exact mechanism of action. Not surprisingly, given their relatively recent discovery, we also have little information regarding the mechanisms by which CSCs act to remedy cardiac injury. Although both MSCs and CSCs can engraft into the host myocardium, they only rarely differentiate into new cardiomyocytes to replace those lost to injury. This has led to the general consensus that most stem cells exert their positive effects by secreting factors that act to stimulate endogenous repair mechanisms in a paracrine fashion. 37 However, there are underlying complex biological events, which act in unison to accomplish the task of healing a wounded heart. Therefore, it is of utmost importance to gain a better understanding of the way stem cells survive, proliferate, migrate, and interact with adjacent cells to restore damaged cardiac tissue. This review focuses on some of the main signaling pathways involved in establishing and maintaining a graft population and therapeutic efficacy of MSCs and CSCs.
Stem Cell Survival, Proliferation, and Migration
To exert their effects to repair and remodel damaged myocardium, stem cells must first home (localize) to the area of injury and migrate within the tissue and must survive and proliferate within the myocardium. Stem cell therapy is hampered by the challenges of delivering and maintaining a sizeable population of cells in the target tissue. Cell engraftment and survival efficiency depend on various factors, including the method of delivery, 38 the type of cells, cell dose, and the tissue microenvironment and comorbidities of the recipient. 39 The final population of the cells can be estimated from the following equation: graft population = (number of cell successfully delivered) × (fraction surviving) (1 + proliferation fraction)n. Proliferation fraction is defined as the proportion of cells that proliferate after transplantation and n is the number of generations of proliferation. 40 This section provides an overview of the signaling pathways involved in stem cell survival, proliferation, and migration and ways they have been manipulated to improve cell retention within the injured myocardium.
Survival
In cardiac models, most of the MSCs do not survive transplantation into the damaged myocardium. In fact, Toma et al 41 demonstrated that only 0.44% of all delivered MSCs are detectable within ischemic myocardium 4 days after transplantation in mice. Freyman et al 42 reported that only 5% to 10% of allogeneic iridium-labeled MSCs remained 2 weeks after intracoronary injection in a pig model of acute MI. Unfortunately, engraftment rates also decline over time. Hong et al 43 used quantitative polymerase chain reaction (PCR) to accurately measure CSC engraftment after intracoronary delivery in a murine model of acute MI. About 5 minutes after injection, 40% of the CSCs delivered were detectable in the myocardium. By 24 hours, only 12% of these remained, and 35 days after injection only 2.4% of the cells originally present at 5 minutes were detectable in the heart, illustrating the difficulty in maintaining a stem cell population within the target tissue. 43
Cells transplanted into diseased myocardium face several major challenges that limit their survival. First, they are exposed to hypoxia and ischemia after delivery, especially when delivered as a treatment for MI or ischemic cardiomyopathy. This hypoxic, nutrient-poor environment increases the risk of apoptosis.44–47 Moreover, the delivered cells are embedded into connective tissue in clumps, thereby hindering the diffusion of oxygen, which has a limit of ±150 µm (diameter: 7-10 cells). 40 Second, the pro-inflammatory milieu present after ischemia and reperfusion or in chronic cardiac disease increases the formation of oxygen-derived free radicals (oxidative stress). Third, the withdrawal of trophic factors, such as nutrients and growth factors, which occurs when stem cells are delivered, ischemic and inflammatory tissues can induce apoptosis via p38 pathway. 40 Finally, the lack of interaction with the extracellular matrix (ECM) and impairment in cell attachment stimulates a form of programmed cell death called anoikis. 48
The hostile environment of the damaged myocardium inhibits stem cell proliferation and induces apoptosis. Apoptosis is a highly regulated form of programmed cell death that occurs in normal cells in response to various activation mechanisms, including the intrinsic and extrinsic apoptotic pathways. Activation of the intrinsic pathway leads to the release of cytochrome C from the intermembrane space of the mitochondria. 49 This protein then binds to proteins Apaf-1 and pro-caspase 9 to form the apoptosome complex, which activates the caspase family of proteases, including caspase 3. The extrinsic pathway is activated by the ligands of the death receptors, Fas, 50 tumor necrosis factor α (TNF-α)R, 51 DR3, DR4, and DR5,52,53 which induce the activation of caspase 8 and caspase 3.50,54 The factors thought to play an important role in stem cell apoptosis include hypoxia, 55 loss of trophic factors, the immune and inflammatory response of the recipient, 56 loss of homing factors such as CXCR4, and clearance from the ischemic area via the vasculature (Figure 1).40,57,58
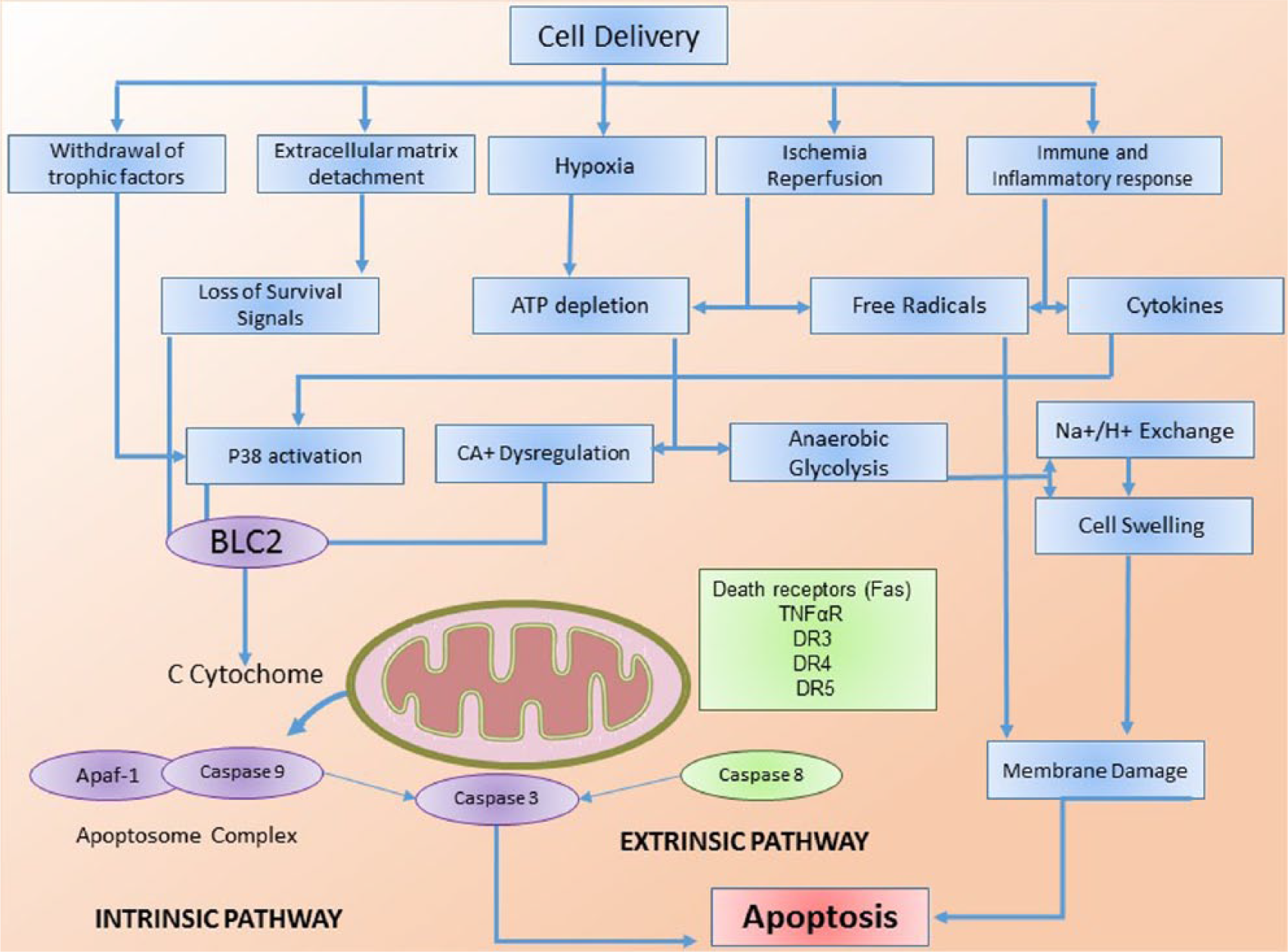
Potential mechanisms of stem cell apoptosis. The factors that play an important role for cell apoptosis after tissue delivery include hypoxia, loss of trophic factors, the immune and inflammatory responses of the recipient, 56 loss of homing factors, and wash out of the ischemic tissue.40,55,56 These activate the intrinsic or extrinsic pathways that lead to activation of caspase 3 and membrane damage of the cells.
One of the primary signaling pathways that affects MSC survival is the PI3K/Akt pathway. This intracellular signaling pathway regulates apoptosis, migration, and proliferation in many cell types, including MSCs. After activation by myriad upstream regulators, PI3K activity leads to the phosphorylation and activation of Akt, which then exerts a downstream antiapoptotic effect via upregulation of antiapoptotic protein Bcl-2 and inhibition of proapoptotic Bax.59,60 The importance of Akt in regulating MSC survival is highlighted by several studies using overexpression of Akt as a strategy to improve MSC engraftment. Mangi and colleagues used retroviral transduction to induce Akt overexpression bone marrow–derived MSCs prior to transplantation in a rat MI model. They found higher myocardial retention of the transduced MSCs compared with controls. Mesenchymal stem cells overexpressing Akt reduced cardiac remodeling and infarct size, improved cardiac function, and regenerated lost myocardial volume better than controls with normal Akt expression.59,61 Similarly, Lim et al evaluated the intracoronary delivery of Akt-overexpressing MSCs in a porcine model of MI. Akt-overexpressing MSCs were more resistant to apoptosis, which resulted in improved engraftment, and these cells also decreased scar size and improved left ventricular ejection fraction better than controls. 62
Overexpression of Akt improves MSC survival largely by inhibiting apoptosis. This spurred interest in manipulation of one of the Akt’s downstream antiapoptotic targets, Bcl-2. This protein acts in the intrinsic pathway, directly inhibits proapoptotic protein Bax and has an independent action over cytochrome c. Overexpression of the Bcl-2 gene in MSCs decreases apoptosis and increased engraftment up to 6 weeks after transplantation in a rat MI model. These cells also produced greater scar size reduction, left ventricular function, and neo-angiogenesis compared with controls. 63 Overall, these data demonstrated that overexpression of Bcl-2 is an effective strategy to improve MSC survival, even in the hypoxic, post-MI myocardium. In the same pathway, glycogen synthase kinase 3β (GSK-3β) inhibition by the drug 6-bromoindirubin-3-oxim was shown to reduce apoptosis in response to TNF-α administration. 64
Upstream activators of the PI3K/Akt pathway have also been examined for their potential to improve MSC survival. Stromal-derived factor 1 (SDF-1) is a chemokine expressed in many tissues including myocardium and binds to the CXCR4 receptor present on the surface of MSCs. Stromal-derived factor 1 is essential for MSC homing to the damaged myocardium, and overexpression of the CXCR4 receptor increases MSC engraftment and migration. In addition, SDF-1 reduces MSC apoptosis via the PI3K/Akt pathway and improves survival in hypoxic conditions. Liu et al 65 found that in vitro, MSCs incubated with SDF-1 had significantly reduced rates of apoptosis and an increased Bcl-2 to Bax ratio. Stromal-derived factor 1 treatment also improved MSC migration and release of paracrine factors, both of which are vital to their therapeutic efficacy. Inhibition of both the PI3K/Akt and extracellular-regulated protein kinases 1/2 (ERK1/2) signaling pathways prevents the antiapoptotic effect of SDF-1 on MSCs under hypoxic conditions, indicating the importance of both of these pathways in SDF-1–mediated cell survival. 66
Periostin is a cell adhesion protein that binds to integrins, is expressed by MSC and fibroblasts, and is upregulated in cardiac tissue undergoing remodeling in response to injury. 67 It has been shown to increase cardiomyocyte proliferation, migration, and adhesion via the PI3K pathway and recently has been implicated in improving MSC survival in an autocrine fashion. Cho et al demonstrated that overexpression of periostin significantly increased MSC survival, and these cells improved cardiac function and scar size when transplanted into a rat model of acute MI. In addition, levels of caspase 3 were decreased in periostin-overexpressing MSCs compared with regular MSCs and the Bcl-2 to Bax ratio was increased, suggesting that periostin signaling decreases apoptosis. Furthermore, the authors found that levels of activated AKT and ERK were increased, implicating these pathways as major mediators of the antiapoptotic effects of periostin. 68
In addition to overexpression and inhibition of components of the P13K/Akt pathway, other strategies to improve MSC survival have focused on their response to hypoxic conditions. Hypoxia leads to the generation of reactive oxygen species (ROS) that mediate oxidative stress and induce apoptosis in MSCs. Hemeoxygenase 1 (HO-1) is an antiapoptotic enzyme that is induced by oxidative stress and displays cytoprotective properties in hypoxic conditions. Studies in both murine and porcine acute MI models have demonstrate improved survival and cardiac regenerative efficacy of MSCs transfected with a hypoxia-inducible HO-1 plasmid.69,70
Because endogenous MSCs are physiologically accustomed to hypoxic environments, hypoxic preconditioning was examined as a means to preserve their characteristics and prevent apoptosis. Preconditioning of umbilical cord blood–derived MSCs in hypoxic media has been shown to promote resistance to apoptosis through a decrease in caspase 3 and lactate activity via by activation of the AKT pathway and subsequent inhibition of Bad, which blocks Bcl-2. 71 Liu et al demonstrated that preconditioning MSCs in 3% oxygen improved survival, adhesion, and proliferation. Hypoxic preconditioning induces expression of the CXCR4 and CXCR7 receptors and the transcription factor hypoxia–inducible factor-1α (HIF-1α), a transcription factor that is also known to upregulate CXCR4 expression. Furthermore, inhibition of PI3K/Akt signaling with wortmannin and blockade of HIF-1α reversed the beneficial effects of hypoxia preconditioning, indicating that these are key signaling molecules necessary for the beneficial effects of hypoxic preconditioning as well as survival (Figure 2). 72
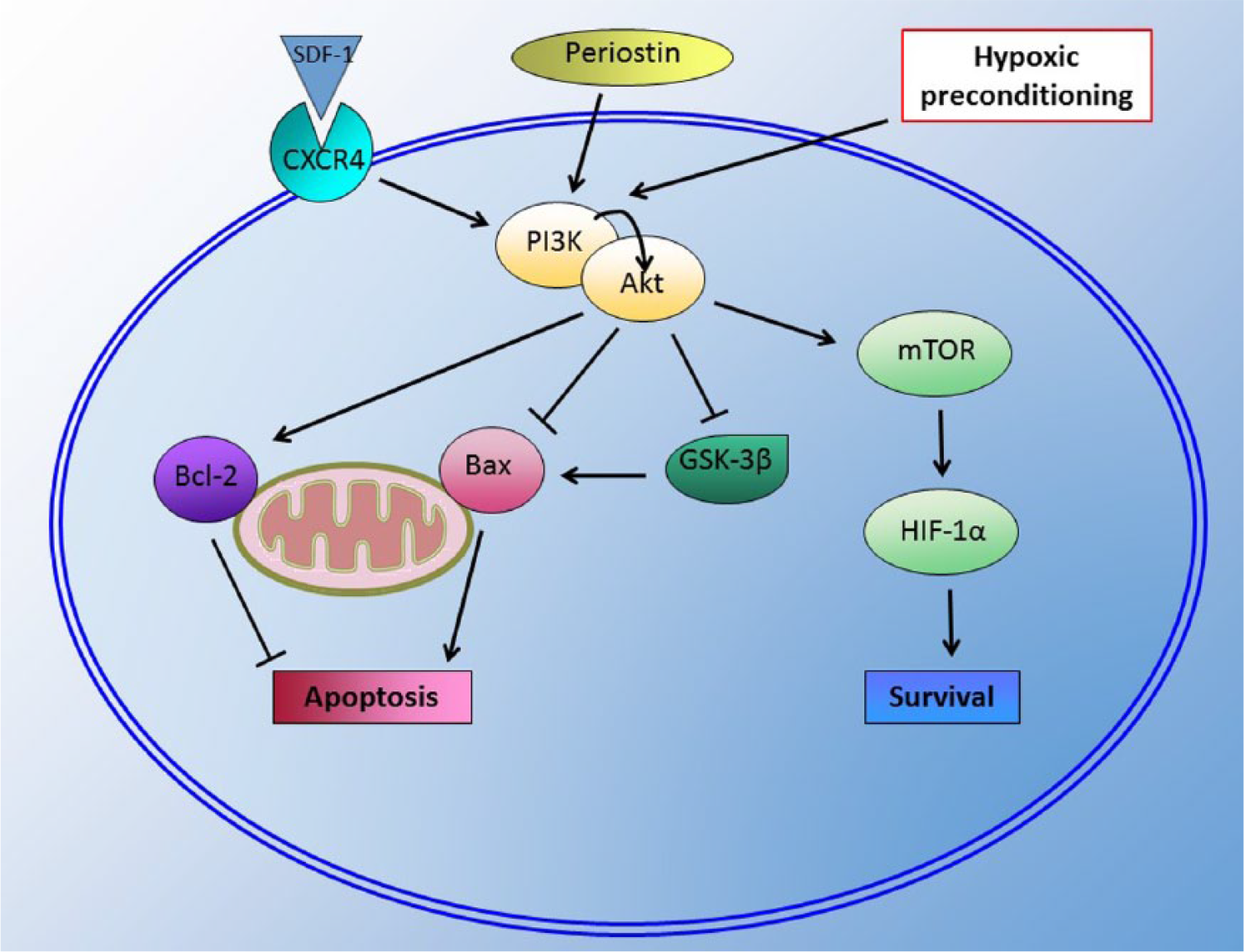
Signaling mechanisms in stem cell survival. SDF-1 binding to its receptor CXCR4, periostin signaling, and hypoxic preconditioning all activate the PI3K/Akt pathway. Active Akt inhibits proapoptotic protein Bax and stimulates antiapoptotic protein Bcl-2. Akt inhibits GSK-3β, which stimulates apoptosis via activation of Bax. Finally, Akt activity stimulates (HIF-1α) via mTOR, which leads to increased stem cell survival. HIF-1α indicates hypoxia-inducible factor-1α; SDF-1, stromal-derived factor 1; mTOR, mechanistic target of rapamycin.
The therapeutic benefits of CSCs are also hampered by cell loss, and various approaches have been investigated to improve the survival of the cells after implantation. Successful techniques include preconditioning with a HO-1 inducer 73 and nitric oxide 74 as well as overexpression of Akt and downstream kinase Pim-1.75,76 In fact, many of the prosurvival actions originally ascribed to Akt are actually mediated by Pim-1 through independent but overlapping pathways. Pim-1 is a serine/threonine kinase that inhibits apoptosis and induces proliferation in both MSCs and CSCs. This protein, such as Akt, phosphorylates and inactivates proapoptotic protein Bad and upregulates Bcl-XL and Bcl-2 to reduce apoptosis. 77 Cardiac stem cells engineered to overexpress Pim-1 have increased survival, proliferation, and ability to regenerate and repair damaged myocardium.74,78
Proliferation
Proliferation is a vital component of stem cell efficacy both because stem cells must be expanded in vitro prior to administration and because proliferation after transplantation increases the graft population. Several growth factors have been used to increase MSC proliferation in vitro. Most of these factors, including TNF-α, insulin, prostaglandin E2 (PGE2), and epidermal growth factor, stimulate proliferation in association with the PI3K/Akt pathway (Figure 3). 60
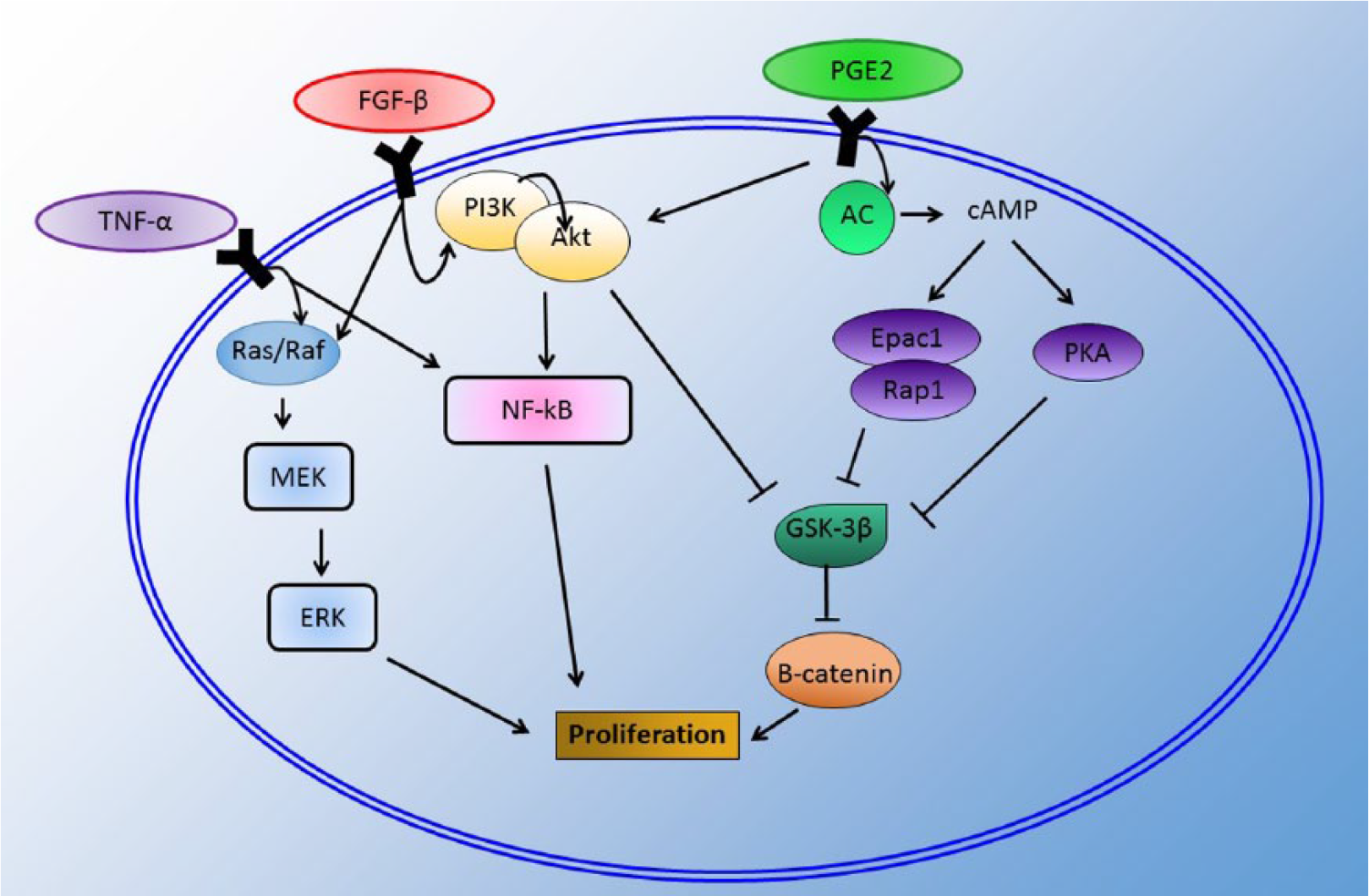
Factors that enhance the proliferation of MSCs. TNF-α stimulates proliferation via the ERK signaling pathway and direct stimulation of NF-κB. FGF-β stimulates both the ERK and PI3K/Akt pathways to increase proliferation partly by preventing GSK3-β from binding to β-catenin. Accumulation of β-catenin leads to proliferation. Binding of PGE2 to its receptor stimulated AC to produce cAMP, which activates EPAC and its downstream target RAP1 and PKA, both of which inhibit GSK3-β. AC indicates adenylyl cyclase; cAMP, cyclic adenosine monophosphate; FGF-β, fibroblast growth factor β; MSCs, mesenchymal stem cells; NF-κB, nuclear factor κB; PKA, protein kinase A; TNF-α, tumor necrosis factor α.
Prostaglandin E2’s action is transduced by its G protein–coupled receptor leading to the activation of adenylyl cyclase and cyclic adenosine monophosphate production. In MSCs, this leads to activation of protein kinase A (PKA) and Epac1 and its downstream target Rap1, which ultimately leads to phosphorylation and activation PI3K/Akt. Both PKA and Epac1 activation leads to GSK-3β phosphorylation, which increases the accumulation of β-catenin in the cytoplasm. β-catenin accumulation then activates c-Myc promoting cell proliferation. 79
Tumor necrosis factor α increases proliferation in a dose-dependent manner by the activation of ERK, JNK, and NF-κB activity (PI3K/Akt pathway) on hematopoietic bone marrow (BM) cells. 80 Similarly, fibroblast growth factor (FGF-2) has shown to stimulate MSC proliferation via activation of the PI3K/Akt and ERK1/2 pathways. 81 Conjugation of FGF-2 with nanoparticles in MSCs has been shown to increase proliferation after controlled delivery. 82 Finally, sphingosine 1-phosphate (S1P) has been studied as a factor to stimulate proliferation, differentiation, and migration. The mechanism remains to be elucidated, but it has been proposed that it is dependent on mitogen-activated protein kinase (MAPK) pathway. 83
Homing and migration
After injury, the migration of specific inflammatory mediators allows wounds to contract and granulation tissue to form. 84 Likewise, when organ-specific injury occurs, such as after an MI, endogenous MSCs and CSCs migrate to areas of myocyte injury to exert their regenerative effects. 85 Here, we discuss some of the signaling mechanisms involved in homing of MSC and CSC to sites of cardiac injury and the transduction pathways that mediate their migration.
Endogenous MSCs have been shown to home to areas of the body that have been infarcted, irradiated, or contain genetic defects.86–88 Similarly, CSCs aid in homeostasis and tissue repair via their homing effect to sites of injury.30,89 When the myocardium is injured, it releases a host of cytokines and growth factors that signal distant stem cells to migrate to the site of necrosis to aid in the repair process. 87 Clinical studies using stem cell therapy have focused on delivery methods that transplant cells near the area of injury, ie, transendocardial injections, intracoronary infusion. However, even using these delivery methods, stem cells delivered into the tissue must migrate to nearby areas of injury, and cells infused into the coronary arteries must extravagate from the vessels before migrating through the myocardium. Mesenchymal stem cells home and migrate to sites of injury via a step-by-step process similar to the migration of leukocytes. First, MSCs are activated when inflammatory cytokines in the damaged myocardium upregulate chemokines and homing receptors. When the site of injury is reached, adhesion molecules are upregulated on the MSC surface, and proteinases are secreted to allow migration through the endothelial basement membrane and into the inflammatory milieu. 90
Cell engraftment and homing to the cardiac tissue can be improved via the addition of adhesion molecules. Selectins are cell surface receptors expressed by the vascular system. These receptors bind to glycoprotein ligands, such as P-selectin glycoprotein ligand 1 (PSGL-1), which is expressed on leukocytes, and binding of selectins to ligands on leukocytes mediates tethering and rolling. 91 The process of “glycoengineering” involves attaching selectin ligands to the stem cell surface, which can then bind to selectins expressed by the endothelium in areas of injury and inflammation. This process should increase tethering and rolling of the engineered stem cells to facilitate homing and extravasation. This was tested by Lo et al 92 who added glycoproteins PSGL-1 and FUT7 to MSCs and CSCs and noted improved stem cell homing to the injured myocardium in a porcine ischemia-reperfusion model. Glycoengineering with physiological selectin ligands is thus a promising strategy to enhance stem cell engraftment. 92
Once MSCs home to the site of injury, extravasation and migration occur through cellular movement, which is dependent on the cytoskeletal changes of actin and other structural proteins. Together, these proteins act in unison to form filopodia/lamellipodia in the front of the cell and stress fibers in the rear to facilitate movement.93–95 The PI3K/Akt pathway plays a vital role in initiating these cytoskeletal changes. Although not specifically studied in MSCs, the actin-binding protein Girdin becomes phosphorylated by Akt when cells need to produce stress fibers and lamellipodia.96,97 Akt also upregulates RhoA, which then effects changes in the cytoskeleton. 60 Akt may also phosphorylate p70(S6K), a regulator of the actin filament cross-linking proteins, which has been studied in ovarian neoplasia to induce proteins, CDC42 and Rac-1, which activate PAK1, a regulator of cytoskeleton change. 98
The chemokine SDF-1 has been well studied as a means to improve homing and migration of both CSCs and MSCs (Figure 4). As described previously in this review, SDF-1 acts directly on the PI3K/Akt to stimulate cell survival and migration. In a rodent model of ischemic cardiac disease, the expression of SDF-1 messenger RNA (mRNA) was upregulated within and around the area of scar. Higher SDF-1 expression in turn leads to recruitment and increased survival of MSCs in the injured regions of myocardium via the activation of PI3K/Akt.65,100 Stromal-derived factor 1 has a complementary 7-transmembrane G-coupled receptor, CXCR4 (expressed by MSCs and CSCs), which is required for their migratory process.101,102 To test the efficacy of the SDF-1 on the migratory pattern of stem cells, one group inhibited binding of this ligand to the CXCR4 receptor with AMD3100. They reported a drastic reduction in the ability of BM-MSCs to home to injured tissue. 100
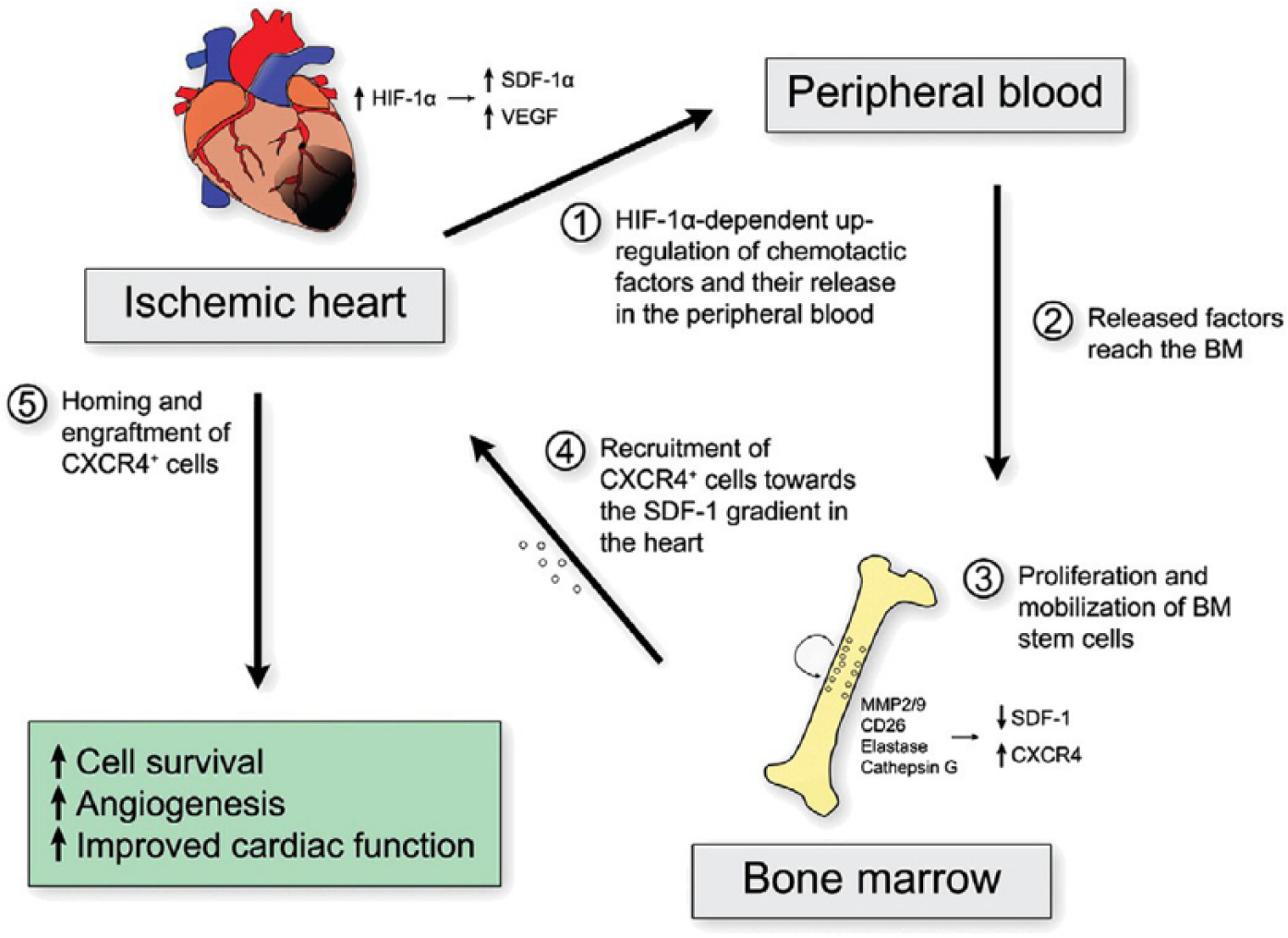
SDF-1 upregulates homing of mesenchymal stem cells to the injured regions of myocardium via the activation of PI3K/Akt. Reproduced with permission from Ghadge et al. 99 SDF-1 indicates stromal-derived factor 1.
Although CSCs use the SDF-1/CXCR4 axis, they harbor a few subtle differences. Many CSCs contain a ligand known as c-kit or stem cell factor (SCF). This particular cell surface marker is critical for CSC proliferation and migration. The peri-infarct region of the left ventricle has shown to increase expression of SCF, much similar to SDF-1. 89 In vitro experiments have shown that SCF employs a 7 transmembrane–spanning G protein–coupled receptor kinase 6 (GRK6) to phosphorylate CXCR4’s downstream targets p38 MAPK and ERK1/2 which act to induce CSC migration. To test the effect of c-kit on migration, Kuang et al 103 used an antibody to block c-kit, which yielded a reduction in the phosphorylation of both CXCR4 and p-ERK1/2, negatively effecting the migration of CSCs. Furthermore, in vivo knockdown of GRK6 reduced the ability of CSCs to migrate to zone of infarction.
The SDF-1/CXCR4 axis is the most recognized pathway of MSC migration; however, a variety of disease processes, aging, and in vitro expansion of MSCs lead to downregulation of this pathway, therefore, other signaling mechanisms also play a key role in MSC migration.104–107 Hepatocyte growth factor (HGF) is one such chemokine which acts in an analogous manner and has been shown to be upregulated in a rat ischemia/reperfusion model. 108 The cognate receptor of HGF, c-met, becomes active on MSCs which in turn aids in the migratory process. 106 Another group inhibited the HGF-c-met axis with a c-met blocking agent, K-252a, and found a significant MSC inhibitory chemotactic response toward HGF. 106 However, they also noted an enhanced migratory chemotactic effect when SDF-1 and HGF act in unison. Other chemokine receptors play crucial roles in MSC migration and engraftment on infarcted myocardium. Specifically, monocyte chemoattractant protein-1 (MCP-1) also known as CCL2 and monocyte-specific chemokine 3 (MCP3) also known as CCL7 are the ligands for chemokine receptors 2 and 1 (CCR2/1), respectively. Studies have shown rapid upregulation of these chemokines following an ischemic insult, and both have been linked to enhanced homing of MSCs to murine myocardial tissue after an MI, followed by improvements cardiac structure and function.109,110
Alternative factors also stimulate CSC migration. Similar to MSC migration, c-kit+ CSCs use HGF and c-met to home from areas of viable myocardium to infarcted tissue. 32 Intramyocardial injection of HGF into rodent hearts after MI via ligation of the left anterior descending artery increased migration of CSCs to the area of infarct, stimulated new myocyte formation, and improved cardiac function. 111 A different subtype of CSC, termed side-population CSCs, expresses adenosine triphosphate (ATP)–binding cassette transporter protein (ABCG2) on their surface, which aids in proliferation and migration. Using a mouse myocardial cryoinjury model, Martin et al 112 demonstrated an upregulation of ABCG2, cell survival, and an enhanced migratory capacity of cardiac progenitor cells following myonecrosis. Conversely, an ABCG2 knockout mouse displayed blunted proliferation of CSCs and enhanced cell death. 113 Another protein residing on the cell surface, ephrin tyrosine kinase, acts on ephrin ligands under hypoxic conditions to enhance CSC migration. There are multiple subtypes of ephrins; however, only subtype A1 is able to bind to all 8 ephrinA receptors. 85 Dries et al 114 showed that ephrinA1 played a role in myocardial repair following injury. However, Goichberg et al 115 proved that the ephrinA1 ligand is able to improve c-kit+ CSC migration via its surface receptor ephrinA2. Together, these alternative markers enhance CSC proliferation and migration under hypoxic conditions.
The attraction of a chemotactic signal to its receptor allows the migratory journey to begin. Once at the site of injury, the adhesion molecules, vascular adhesion molecule (VCAM-1) and α4β1 integrin (VLA-4), secreted by ECs and MSCs, respectively, act to anchor the cells in preparation for diapedesis. 90 However, in the final step migration, stem cells must travel through the tissue and vasculature needs to be laid down to continue the voyage. The angiogenic properties of murine MSCs have been tested in a hypoxic setting. When introduced into a 1% oxygenated medium, MSCs begin to form capillary-like structures. 116 Similarly, vascular endothelial growth factor (VEGF), a regulator of blood vessel formation, stimulates c-kit+ CSC migration to myocardial injury in both in vitro and in vivo to promote vasculogenesis and myogenesis.117,118 Once the stem cells are adhered to the site of injury, proteinases are necessary to allow migration through the vessel wall. Elevated levels of metalloproteinases (MMP-2 and MMP-9) are involved in the degradation of basement membrane gelatin and collagen post-MI.100,119–121 Mesenchymal stem cells in particular express matrix metalloproteinase-2 (MMP) and the membranous-type 1 (MT1-MMP), allowing them to transmigrate across the basement membrane. 90 Similarly, CSCs exhibit an ability to upregulate MMP-2 and MMP-9 via Akt activity. 122 A recent study has also shown that subunit C1q of the classical complement pathway upregulates MMP-2 via the ERK1/2 pathway to enhance diapedesis.90,123 Given that matrix remodeling follows an infarct, it is imperative that these proteinases allow CSCs and MSCs to travel through damaged tissue to enter the site of injury and begin the reparative process.
Stem Cell Mechanisms of Action
Stem cells exert their beneficial effects by preventing and reversing deleterious cardiac remodeling that occurs after ischemic or other insults to the heart. The mechanism of action is multifaceted, involving both direct differentiation into new cardiomyocytes, endothelial and smooth muscle cells, and via stimulation of endogenous repair mechanisms. 124 This occurs through multiple signaling mechanisms including transduction pathways which lead to differentiation, paracrine effects, cell-cell contact, and exosome signaling. The following sections will focus on the better understood signaling mechanisms involving stem cell differentiation and paracrine signaling.
Differentiation
Initial interest in cell-based therapies for cardiac disease focused on stimulating stem cells to differentiate into and replace damaged cardiomyocytes. Multilineage differentiation capacity is one of the defining characteristics of MSCs.125–127 Moreover, MSCs have been shown to transdifferentiate into cardiomyocytes and vascular-like structures.128–131 It was once thought that MSCs had a tremendous regenerative potential; however, empirically MSCs only rarely transdifferentiate into cardiomyocytes after transplantation into the heart. 26 Understanding the mechanisms by which differentiation occurs is vital to stimulating and harnessing the direct regenerative capacity of MSCs. The Wnt/β-catenin, transforming growth factor β (TGF-β), MAPK, and PI3K signaling pathways are the main molecular pathways involved in MSC differentiation.
Mesenchymal stem cells are true multipotent stem cells and are in part defined by their ability to differentiate into osteoblasts, adipocytes, and chondroblasts in vitro; however, they have also been shown to transdifferentiate into other cell types as well. The chosen lineage is affected by exposure to certain bioactive factors. In the presence of VEGF, MSCs preferentially take up osteoblast lineage, and adipocyte differentiation is repressed through regulation of the transcription factors, RUNX2 and PPARγ2, as well as through interaction with nuclear envelope proteins lamin A/C. 132 Day et al 133 showed that activation of Wnt pathway results in ossification during vertebrate skeletogenesis, whereas inhibition of β-catenin promotes chondrogenesis. After exposure to insulinlike growth factor 1 (IGF-1), bone MSCs differentiate into cardiomyocyte-like cells in vitro through activation of MAPK and PI3K pathways. 134 Other factors, such as dexamethasone and TGF-β, induce MSC differentiation into chondrocytes, 135 by activating the TGF-β superfamily. 125 A neuronal phenotype is observed after the exposure of MSCs to basic fibroblast factor, dimethyl sulfoxide, β-mercaptoethanol, and butylated hydroxyanisole. 136 Adipogenic differentiation is achieved by exposing MSCs to dexamethasone, insulin, indomethacin, and 1-methyl-3-iso-butylxanthine. 127
In addition to their defining ability to differentiate into osteoblasts, chondroblasts, and adipocytes, MSCs also have been shown to transdifferentiate into cardiomyocytes, VSMCs, and ECs.26,128,129,131 Toma et al 41 reported that transplanted human MSCs can successfully differentiate into cardiomyocytes and express myocardium-specific proteins after cell transplantation into left ventricle of severe combined immunodeficiency mice.
Several biochemical substances can induce MSC differentiation into cardiomyocytes in vitro. Only 24 hours after exposure, 5-Azacytidine (5-Aza), a chemical analogue of cytidine, can trigger MSCs differentiation into myotubule-like cells, and in 7 to 10 days, these cells express cardiac-specific proteins. Jaquet et al 137 demonstrated that human MSCs incubated with 5-Aza begin to express smooth muscle actin within 2 weeks. The signaling pathways involved in differentiation of MSCs after 5-Aza treatment include the following: MEK/ERK, MEF-2c, and STAT3 activation. 138 Pharmacological inhibition of MEK reverses the phosphorylation of these proteins in a dose-dependent manner and downregulates the expression of MyoD and cTnt (both markers of cardiomyocyte-like cells). 138
Although 5-Aza is the most commonly used chemical inducer of MSC cardiomyogenesis, its efficiency is low due to its toxic effects on cardiomyocytes. Bone morphogenetic protein-2 (BMP-2) plays a fundamental role–directed differentiation of CSCs and the development of embryonic heart. 139 Hou et al 140 found that BMP-2 in combination with 5-AZA induces differentiation of bone marrow–derived MSCs in vitro and ameliorates apoptosis caused by 5-AZA. Other studies showed that angiotensin-II (Ang-II) has a potential to stimulate proliferation of VSMCs and fibroblasts. Moreover, it can promote MSCs to differentiate into cardiomyocyte-like cells in vitro through potential ERK-dependent activation of the autocrine TGF-β1-Smad2 cross talk pathway.141,142
Coculture of MSCs with other cell types is another strategy to induce differentiation. Bone marrow–derived MSCs cocultured with ventricular myocytes differentiate into cardiomyocytes that express sarcomeric α-actinin, desmin, cardiac troponins, ryanodine receptor, and SERCA2. 143 He et al 144 demonstrated that coculture of rat bone marrow mesenchymal stromal cells (BMSCs) with the neonatal cardiomyocytes enhanced myogenic conversion in a dose-dependent manner. Moreover, BMSCs contracted synchronously with the neonatal cardiomyocytes and even exhibited action potentials. Plotnikov et al 145 showed similar results when they cocultured human MSCs with rat cardiomyocytes and proposed that a potential mechanism of this transdifferentiation is mitochondrial transfer from one cell type another. In a study, where mouse MSCs were cocultured with rat ventricular myocytes in a ratio of 1:40, MSCs became actin positive and formed gap junctions with the endogenous myocytes. In contrast, MSCs separated from myocytes by a semipermeable membrane were negative for α-actinin, suggesting that MSC transdifferentiation also requires direct cell-cell interaction. 146 Although it has been shown that MSCs differentiate directly into cardiomyocytes,26,34 the majority remain as MSCs and generate cell to cell interactions with host cardiomyocytes, stimulating endogenous cardiomyocyte turnover and CSC differentiation.26,34,147
In addition to stimulating myocyte turnover and progenitor cell recruitment, 148 endogenous CSCs also promote repair through direct remuscularization 27 and these effects are enhanced by MSCs. Although some findings suggest that resident CSCs contribute only minimally to cardiac regeneration, 149 others have demonstrated that this is a variable process which is significantly enhanced after cell-cell contact with MSCs.34,147 In a recent study, Hatzistergos et al 150 showed that MSCs stimulate CSC proliferation, migration, and differentiation into new cardiomyocytes through regulation of c-kit and SDF-1/CXCR4 signaling pathways. Taken together, these studies suggest that cardiac microenvironment, factors secreted by myocytes, and cell-cell contact take part in stem cell differentiation into cardiomyocytes.
Cardiac stem cells expressing the surface marker c-kit+ are self-renewing, clonogenic, and possess the ability to differentiate into cardiomyocytes, smooth muscle, and ECs. After 7 to 10 days of induction of CSC differentiation in vitro, most of the cells exhibited signs of biochemical differentiation into myogenic (29%-40%), smooth muscle cell (20%-26%), or EC (18%-23%) lineage. 27 Unfortunately, the phenotype of the in vitro–differentiated CSC was morphologically and functionally immature, as indicated by a lack of structural organization and contractile activity. 27 In vivo, a study in rodents demonstrated that 20 days following transplantation into the infarct border zone, BrdU-labeled c-kit+ cells had colonized the infarcted area and appeared to have matured as indicated by the expression of cardiac myosin heavy chain, α-sarcomeric actin, N-cadherin, and connexin 43-positive intercalated discs. 27 Moreover, this was accompanied by an increase in the density of capillaries and arterioles in cardiac tissue after CSC implantation. Differentiation of CSCs involves activation of Wnt/β-catenin signaling pathway. Bergmann et al 151 demonstrated that the inhibition of β-catenin triggers CSC differentiation to contractile cells. β-Catenin depletion enhances resident CSC differentiation to the cardiac lineage and it is associated with upregulation of cardiac lineage markers Tbx5 and GATA4. 152
The Notch pathway is involved in the regulation of tissue homeostasis and cardiomyogenesis at multiple stages, from the commitment of mesoderm to the maturation of myocytes. 153 In the developing heart, Notch is involved in ventricular trabeculation, cardiomyocyte proliferation and differentiation, and valve formation. 154 Together with the target gene HES-1, the Notch pathway is activated in cardiomyocytes at the border zone of the scar. 155 Accumulated evidence suggests that resident CSC recruitment and differentiation are mediated by Notch-mediated induction of reparative mechanisms in the damaged heart.155–158 Urbanek et al 159 showed that pharmacological inhibition of Notch reduces the number of cycling c-kit+ CSCs and compromises their cardiac commitment. Moreover, inhibition of Notch signaling leads to severe dilated cardiomyopathy in newborn mice secondary to the reduced differentiation rate of cardiac progenitor cells. 159
Paracrine effects
Both MSCs and CSCs are known to produce myriad soluble factors which act in a paracrine fashion to stimulate endogenous repair mechanisms and improve cardiac function. Given that stem cell engraftment and survival are fairly low and differentiation even rarer, most of the therapeutic capacity of stem cells is mediated by their paracrine effects. Through paracrine effects, MSCs aid in repairing the heart by stimulating angiogenesis, reducing fibrosis, immunomodulation, and improving cardiomyocyte and resident CSC survival. The mechanisms and paracrine signals involved in these processes will be discussed in the following section.
Angiogenesis
Mesenchymal stem cells induce angiogenesis by direct differentiation into ECs and smooth muscle cells, as well as by secreting paracrine factors such as VEGF and basic fibroblast growth factor (bFGF). Considering the low differentiation of MSCs in vivo, paracrine signaling is the most influential factor of neoangiogenesis 160 and VEGF regulates this process predominantly by binding to VEGFR-2/FLK1 of ECs to induce arterial morphogenesis. 161 Mesenchymal stem cells function as pericytes, which wrap around the ECs of capillaries and venules and function with ECs to produce a common basement membrane. They promote angiogenesis in response to angiogenic signals or chemokines released by hypoxic, inflammatory, or tumor cells. 161 Vascular endothelial growth factor regulates both vascular EC migration, proliferation, and permeability and functions as an antiapoptotic factor for newly formed blood vessels. 162 Basic fibroblast growth factor is also a well-known angiogenic factor that activates receptors (FGF receptors) on ECs or indirectly stimulate angiogenesis by inducing the factor release from other cell types. For instance, in the heart, FGF-mediated signaling fuels vessel growth by stimulating the release of hedgehog, angiopoietin-2, and VEGF-B. Low levels of FGF are required for the maintenance of vascular integrity. 163 Mesenchymal stem cells with overexpression of connexin 43 have been shown to improve myocardial protection in ischemic hearts by increasing levels of both VEGF and bFGF. 164
Extracellular matrix and antifibrotic effects
Mesenchymal stem cells are recognized for their antifibrotic effects and ability to reduce scar and fibrosis; interestingly, this is not achieved via direct modulation of the ECM but rather due to interplay with fibroblasts. 165 Fibroblasts are the main regulators of the ECM. Ohnishi et al 166 reported that MSC-conditioned medium upregulates antiproliferation-related genes, such as elastin, myocardin, and DNA damage–inducible transcript 3, which result in the suppression of cardiac fibroblast proliferation. Mias et al 167 reported that MSCs promote the secretion of the ECM-degrading enzyme MMPs and decrease their endogenous inhibitors (tissue inhibitors of metalloproteinases) via paracrine signaling of cardiac fibroblasts. Meanwhile, Li et al argue that paracrine signaling alone cannot explain the antifibrotic properties of MSCs. The authors demonstrate the importance of intercellular communication between adipose-derived MSCs and fibroblasts in an in vitro coculture model and show that MSCs significantly inhibit viability and α-smooth muscle actin expression of fibroblasts when compared with MSC-conditioned medium. They also show that HGF secreted by MSCs contributes to reduced fibrosis in multiple organs. 168 Little is known of the antifibrotic effects of resident CSCs.
Immunomodulation
The immunomodulatory properties of MSCs are attractive for a variety of clinical applications, such as hemopoietin engraftment and graft-vs-host disease (GVHD). In cardiac disease, immunomodulation by MSCs acts to reduce inflammation following MI and aids in cardiac regeneration by shifting immune system from away from a pro-inflammatory state. 169 This occurs by virtue of paracrine effects by several factors that are secreted from MSCs including TGF-β, HGF, interleukin (IL)-10, leukocyte inhibitory factor, human leukocyte antigen G, indoleamine 2,3-dioxygenase, hemeoxygenase (HO), galectins, and tumor necrosis factor α–stimulated gene 6 (Figure 5). 170 Mesenchymal stem cells produce this broad range of immunomodulatory paracrine factors to modulate key cells that constitute the immune system, including T and B lymphocytes, dendritic cells (DCs), natural killer cells, and monocytes or macrophage, and develop immunotolerant phenotypes such as regulatory DCs 171 and T cells.172,173
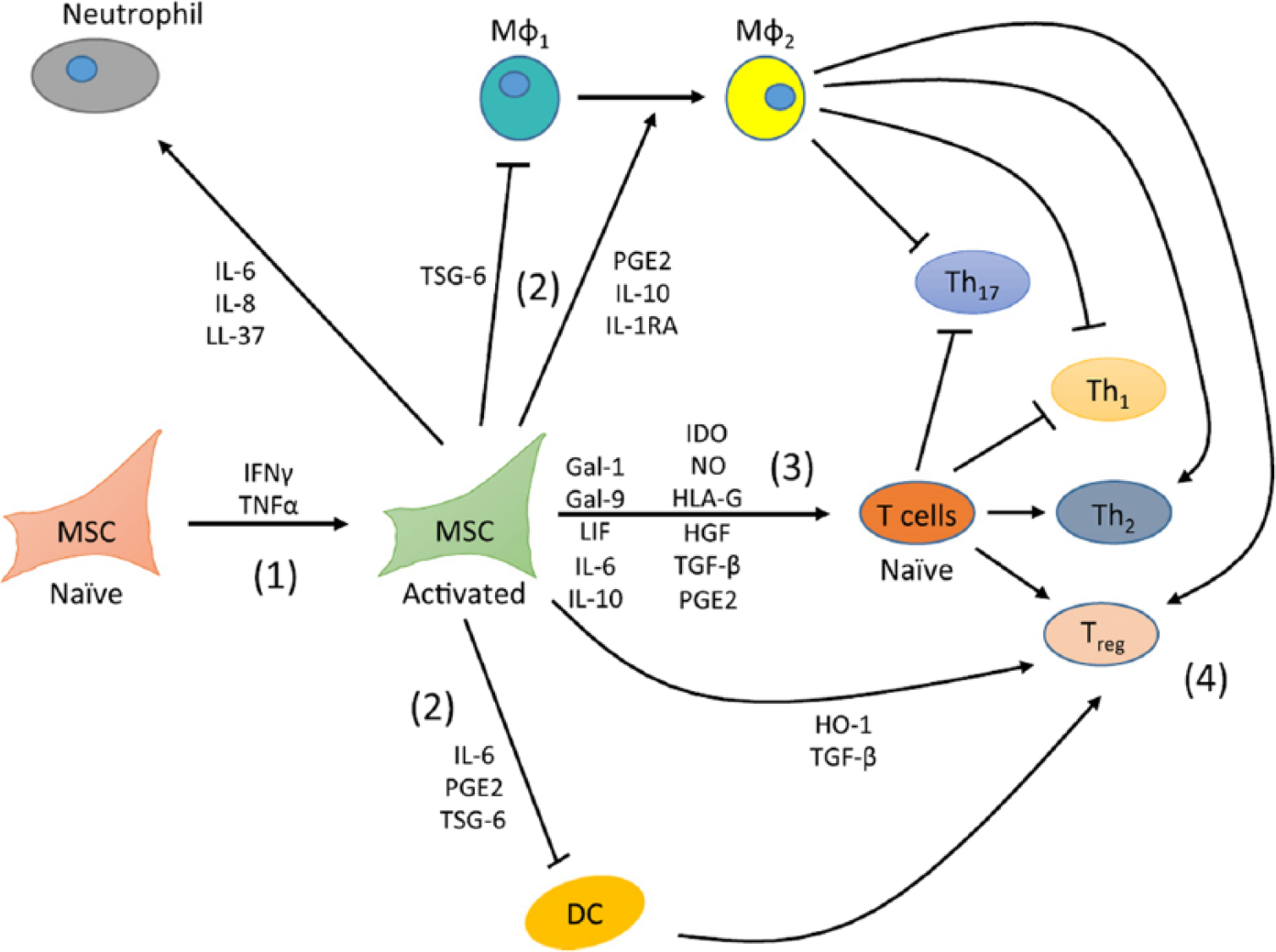
The immunomodulatory properties of mesenchymal stem cells (MSCs). Paracrine effects of several factors that are secreted from MSCs, including transforming growth factor β (TGF-β), hepatocyte growth factor (HGF), interleukin 10, leukocyte inhibitory factor, human leukocyte antigen G, indoleamine 2,3-dioxygenase, hemeoxygenase, galectins, and tumor necrosis factor α–stimulated gene 6. Reproduced with permission from Fontaine et al. 170
Transforming growth factor β increases CD4+ CD25+ FoxP3+ regulatory T lymphocytes (Tregs) in the presence of MSCs. 174 This is associated with the suppression of pro-inflammatory TH1 response which is characterized by TH1 that cells produce IFN-γ, IL-2, and tumor necrosis factor (TNF)-β and evoke cell-mediated immunity and phagocyte-dependent inflammation. 175 Moreover, Nemeth et al observed that TGF-β from MSCs was beneficial in the TH2-dominant system, which is activated in allergic responses. TH2 cells produce numerous ILs, including IL-4, IL-5, IL-6, IL-9, IL-10, and IL-13, and evoke antibody responses including those of the IgE class and eosinophil accumulation. The authors showed that these TH2 cytokines were decreased in a rat model of ragweed-induced asthma treated with MSCs. 176 Using STAT6-deficient mouse MSCs in the same ragweed-induced asthma showed no elevation of TGF-β, and given that IL-4 receptor is known to activate the STAT6 signaling pathway, 177 it is thought that STAT6 activation via IL-4 receptor induces TGF-β production in MSCs. 176
As indicated earlier, HGF is a cellular growth factor that is produced by MSCs that play a major in angiogenesis. However, HGF treatment ameliorates GVHD, 178 and MSC with HGF gene knockout increases lung injury via lipopolysaccharides (LPS), 179 suggesting that HGF bears immunomodulatory properties as well. Furthermore, MSCs overexpressing HGF in a mouse MI model have shown to enhance cell survival and paracrine effects. 180 The regulation of HGF has not been fully elucidated and may vary due to the microenvironment. 181
Prostaglandins are lipid autacoids derived from arachidonic acid and are generated from arachidonate by the action of cyclooxygenase. The arachidonic acid cascade consists of many of these lipids that are involved as second messengers in cell signaling pathways, and prostaglandins may function in both the promotion and suppression of inflammation. 182 Specifically, PGE2 has been recognized as having anti-inflammatory effects, and its expression is upregulated in MSC-mediated immunomodulation. 183 Its key role is to mediate immunoregulatory function of immune cell subtypes and is important in the interaction between MSCs and DCs. First, direct MSC-DC contact and activation of Notch signaling pathway enhance switching of DCs toward an immunosuppressive subtype. 171 Second, MSCs suppress CD8+ T-cell–mediated activation by suppressing natural killer group 2, member D protein receptor (NKG2D) expression and secreting PGE2 together with indoleamine 2 (IDO 2), and TGF-β. 184 Third, MSCs potentially modulate the inflammatory response to the bacterial injury by altering the macrophage M1 phenotype toward the M2 phenotype, which are considered a tolerogenic phenotype, thereby decreasing macrophage TNF-α production, but enhancing IL-10 secretion. 185 Finally, PGE2 secreted by MSCs decreases macrophage activation by interacting with prostaglandin EP2 and EP4 receptors on macrophages and leading to their release of IL-10. 185
Several other secreted factors also play important roles in MSC immunomodulation. Hemeoxygenase is a stress-inducible enzyme involved in heme catabolism and consists of 3 isoforms. Hemeoxygenase 1 produced by human mesenchymal stem cells specifically induce differentiation of CD4-naïve T cells into TGF-β positive–producing TH3 favoring a tolerogenic immune response and is indicated as a key contributor for MSC-mediated suppression of alloactivated T cells. 186 Tumor necrosis factor α–stimulated gene 6 (TSG-6) is a multifunctional protein involved in inflammation, ECM formation, cell migration, and development through cell-cell and cell-matrix interaction. 170 In an acute myocardial infarction model in mice, TSG-6 expression by MSCs was found to be upregulated 27-fold to 47-fold higher than baseline. 187 Inflammatory signals (TNF-α, IL-1β, or LPS) or environmental stress such as aggregation or hypoxia rapidly activate MSCs to secrete TSG-6, 188 demonstrating their vital role under stressed conditions.
Inactivation of oxidative stress
Similar to the inflammatory immune response, oxidative stress plays a major role in the pathogenesis of coronary heart disease and heart failure. It is characterized by deregulated production of ROS and reactive nitrogen species and the inability of the antioxidant system to neutralize them. These reactive molecules play a central role in damaging and remodeling myocardium after injury. Sources of ROS in the myocardium include fibroblasts (via nicotinamide adenine dinucleotide phosphate oxidases), infiltrating inflammatory cells and the damaged cardiomyocytes themselves. 189 The increase in ROS within the ischemic tissue is a result of 2 factors: (1) a decrease in the normal antioxidant defense mechanism including mitochondrial superoxide dismutase and reduced glutathione and (2) an increase in production as a result of elevated production of free radicals and toxic oxygen metabolites. 190 Excess ROS stimulates proapoptotic signaling pathways and induces the production of pro-inflammatory cytokines and MMPs. Oxidative stress also causes EC dysfunction, upregulation of adhesion molecules, and increase in EC permeability and migration of neutrophils.191,192
In conditions of oxidative stress, the accumulation of free radicals can damage various biomolecules, including DNA, protein, and lipids. High ROS levels in stem cells cause damage and dysfunction, but a low basal level of ROS is necessary for maintaining cellular proliferation, differentiation, and survival.193,194 Multiple studies using different disease models195–198 demonstrated that MSC could exert an antioxidative effect leading to a decrease in severity of organ injury. 199 This effect is primarily regulated through secretion of soluble factors, which prevent ROS accumulation through enhanced scavenging and antioxidant upregulation. 200 Human MSCs have a high resistance to oxidative stress–induced death due to low levels of intracellular reactive species and high levels of total glutathione, the major cellular antioxidant. 201 Human MSCs lose their capacity to manage oxidative stress when they are depleted of glutathione. 201 However, other studies suggest that MSCs have low antioxidant activity and are sensitive to oxidative stress compared with more differentiated cell types.202,203
Under ischemic injury, elevated ROS production may influence differentiation of endogenous or implanted stem cells at the infarct site. Stem cells have both adaptive and maladaptive responses to oxidative stress. For example, short ROS increase may induce embryonic stem cell differentiation toward the cardiomyocyte phenotype, whereas prolonged exposure to H2O2 inhibits differentiation. 204 Another study demonstrated that H2O2 or glucose oxidase–mediated oxidative stress promotes cardiogenic differentiation of MSCs through upregulation of Notch1 signaling. 205 Li et al 206 reported that ex vivo hypoxia in BMSCs stimulates the synthesis of vascular EC growth factor mRNA, resulting in differentiation of BMSCs into ECs in ischemic hind limb.
Apoptosis plays a central role in cardiac ischemia-reperfusion injury, and inhibition of cardiomyocyte apoptosis during reperfusion is an important strategy for therapeutic intervention.207–209 The role of prosurvival protein kinases, such as Akt and Erk1/2, which induce antiapoptotic effects, has been established in a variety of cardiovascular events.210,211 Activation of Akt signaling has been shown to minimize cardiomyocyte apoptosis in vivo after ischemia-reperfusion injury,212,213 and Akt activation has been shown to preserve the function of hypoxic cardiomyocytes at levels comparable with those of normoxic controls. 213 Increased AKT activity is one of the mechanism of cardiomyocyte protection exerted by bone marrow stem cells under ischemia. 214
Tsubokawa et al 215 transplanted modified MSCs that transiently overexpressed HO-1 and found that they have a better antioxidative and antiapoptotic properties than unmodified MSCs. Under oxidative stress, VEGF production from modified MSC was dramatically reduced by the PI3K/Akt pathway inhibitors. 215 In contrast, the overexpression of Akt can induce production of VEGF 216 that also has a direct effect on the survival of cardiomyocytes by activating the downstream Akt cascade system. 217
In addition to various growth factors and cytokines, MSCs produce exosomes, small extracellular vesicles that contain RNA and proteins which can affect various biological functions. It has been shown that MSC-derived exosomes reduce infarct size and decrease oxidative stress and cell death after myocardial ischemia/reperfusion injury.218,219 In vitro and in vivo studies have shown that MSC-released exosomes can decrease oxidative stress–induced injury by reversing the depletion of key enzymes in ROS metabolism and the accumulation of toxic products from the electron transport chain.218–220 Mesenchymal stem cell–derived exosomes have enzymatically active CD73, the major enzyme responsible for the formation of extracellular adenosine from released adenine nucleotides. 221 Exosomes activate adenosine receptors and induce adenosine-induced phosphorylation of Erk1/2 and Akt in H9C2 cardiomyocytes. 222 Moreover, intact MSC-derived exosomes directly target cardiac cells and reduce infarct size and adverse remodeling in vivo. The mechanisms involved include increased ATP production, decreased oxidative stress, and induced PI3K/Akt signaling pathway. 219
All together, the data indicate that MSCs, through cell-to-cell contacts and production of growth factors, cytokines, and exosomes, create an environment that suppresses ROS production by neutrophils, inhibit generation of effector T cells, and increase the number of Tregs.
Limitations of Cell-Based Therapies
As with any new therapy, there are multiple limitations to stem cell therapies for cardiac regeneration. Over the past 30 years, there have been great strides in demonstrating the efficacy of a number of stem cell therapies in cardiac repair as well as great insight into some of the key signaling pathways involved. Unfortunately, despite years of preclinical research, the exact mechanism of action of stem cells is unknown, making it difficult to develop means of improving their efficacy. In addition, there are many challenges in translating these therapies to the clinical setting. Clinical trials are the ultimate test of stem cell efficacy, as animal models do not respond in the same way as patients. Although there have been many stem cells evaluated in both the preclinical and clinical settings, there have been few head-to-head comparisons of different cell types, stem cell source, and timing of delivery. It is very difficult to reproduce the exact environment in which stem cells function in vivo, making it challenging to determine the mechanism of action of stem cell efficacy that is seen in clinical trials. Specifically, although cell modification may work in vivo, it is unclear whether pretreatment or genetic modification will improve the efficacy of cell therapies in clinical trials. Although many clinical trials are still ongoing, there remain many unanswered questions as to the best cell type, the benefit of autologous vs allogeneic stem cells, the timing, dose, and method of delivery, and whether pretreatment of cells is beneficial.
Conclusions and Future Directions
Stem cell therapy has evolved from in vitro studies to improvement in cardiac structure and function in both ischemic/nonischemic animal models and clinical trials. Regenerative effects are explained in large part by paracrine and cell-to-cell contact mechanisms. Most of the studies demonstrate that the engraftment and survival of stem cells are limited, which mitigates the reparative and regenerative capacity of stem cells. Elucidating these mechanisms of action has provided insights into developing genetic strategies to enhance the regenerative potential of stem cells, including promoting cell survival, migration, and transdifferentiation. Moreover, enhancing the capacity for immunomodulation, angiogenesis, and oxidative stress reduction are fundamental to maximize the regenerative properties of stem cell therapy.
Future directions of research into stem cell therapies for cardiac disease will likely focus on cell-cell contact mechanisms that enhance endogenous repair mechanisms, mitochondrial exchange between stem cells, and cardiomyocytes and “cocktails” that help induce stem cells to commit to specific cell lineages such as cardiomyocytes. In fact, the use of a cardiopoietic cocktail has been used to create MSCs that are lineage specified toward a cardiac phenotype. Unfortunately, these cells did not significantly improve cardiac structure, function, or quality-of-life parameters in the recent CHART-1 (Congestive Heart Failure Cardiopoietic Regenerative Therapy) clinical trial, 223 highlighting the need for further exploration into which factors make increase stem cell’s therapeutic potency in vivo.
The next phase of research will likely focus on methods to enhance the host tissue environment, further testing of genetic modifications and pretreatments to improve cell survival and engraftment, and clinical testing of the ideal timing of cell administration. Given the modest effects of cell types tested so far, it may be that injection of a single, unmodified cell type is not enough to reverse the process of cardiac remodeling. Future studies involve combinations of stem cells, which work in concert to repair tissue and avoid destruction by the host immune system, and these have been tested in animal models.5,6 The application of iPSCs, adult cells that have been manipulated to become stem cells, may provide more options for differentiation. Embedding cells within biomaterials and matrices is also novel method being evaluated which may hold promise in replacing damaged myocardium, and clinical trial in Japan is currently underway to test surgical engraftment of sheets of myocardium derived from autologous iPSCs. 224
As the signaling pathways involved in stem cell survival and mechanism of action are further elucidated, strategies to improve their therapeutic efficacy will arise. As we await for answers from clinical trials, further breakthroughs in stem cell signaling will provide new avenues to pursue.
Footnotes
Peer review:
Six peer reviewers contributed to the peer review report. Reviewers reports’ totaled 1651 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by NIH grants, UM1 HL113460 and R01 HL084275, the Starr Foundation, and the Soffer Family Foundation.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
ACR, VF, BAT, and MN wrote the first draft of the manuscript. ACR, VF, BAT, MN, MB, and IHS contributed to the writing of the manuscript and agree with manuscript results and conclusions. ACR and IHS jointly developed the structure and arguments for the paper. MB and IHS made critical revisions. All authors reviewed and approved the final manuscript.
