Abstract
Increased intestinal epithelial permeability has been linked to many enteric diseases because it allows easy access of microbial pathogens and toxins into the system. In poultry production, the restrictions in the use of antibiotic growth promoters have increased the chances of birds being susceptible to different enteric diseases. Thus, understanding the mechanisms which compromise intestinal function is pertinent. Based on our previous observation which showed the primary chicken enterocytes in culture undergoing dystrophic changes on treatment with phorbol myristate acetate (PMA), we surmised that this model, which appeared to mimic increased intestinal permeability, may help to understand the mechanisms of this problem. As genomic and proteomic changes are associated with many physiological and pathological problems, we were interested to find whether certain proteomic changes underlie the morphological alterations in the enterocytes induced by PMA. We exposed primary enterocyte cultures to a sub-lethal concentration of PMA, extracted the proteins, and analyzed by mass spectrometry for differentially regulated proteins. Our results showed that PMA affected several biological processes which negatively affected their energy metabolism, nuclear activities, and differentially regulated the levels of several stress proteins, chaperon, cytoskeletal, and signal transduction proteins that appear to be relevant in the cause of enterocyte dystrophy. Phorbol myristate acetate-affected signal transduction activities also raise the possibilities of their increased susceptibility to pathogens. The changes in enterocyte integrity can make intestine vulnerable to invasion by microbial pathogens and disrupt gut homeostasis.
Introduction
Increased intestinal permeability contributes to “leaky-gut,” a condition that is linked to several enteric problems because it can permeate microbial pathogens, antigens, and toxins into the system.1–3 The epithelial integrity prevents bacterial pathogens from entering blood and activate immune system, thereby maintaining a healthy mucosal immunity. In poultry production, the restrictions in the use of “antibiotic growth promoters” have increased the chances of the birds being susceptible to infections and intestinal diseases.4,5 Hence, understanding how different factors, including microbial pathogens, interact with intestine can help develop products that can improve gut health. The avian enterocyte culture, thus, has potential for screening assays and to study the mechanisms of their interaction with different dietary, microbial, and chemical factors. Previously we observed that the primary chicken enterocytes, when treated with phorbol myristate acetate (PMA), undergo dystrophic changes characterized by cell shrinkage, distended intercellular spaces, and cachectic cellular processes without significant losses of their viabilities. 6 The phorbol esters bind phospholipid membrane receptors of the cells leading to the activation of different proteins and enzymes such as NADPH oxidase, protein kinase C (PKC), and integrins, thus altering cellular adhesion, growth, differentiation, and remodeling. 7 Protein kinase C activation is one of the most studied mechanisms of phorbol esters which activate their membrane recruitment and trigger many cellular responses including inflammation, proliferation as well as cell death, and remodeling.8,9 We surmised that the dystrophic changes in the enterocytes induced by the action of PMA can be an useful experimental model to study the intestinal permeability changes associated with “leaky-gut” problem. The in vitro observation of the effect of PMA appeared consistent with evidence in the literature where the animals treated with phorbol esters or croton oil, a major source of these esters, 10 disrupt epithelial barrier of the intestine and cause inflammation and cell death.11–15 On account of the fact that proteomic changes underlie most physio-pathological transformations including morphological and functional alterations, the objective of this study was to determine the proteomic changes in the enterocytes induced by PMA that would cause enterocyte dystrophy.
Methods
Day-old male broiler Cobb 500 chicks, obtained from a local hatchery, were used to harvest intestinal villi, and the enterocytes were dissociated and cultured as described earlier. 6 The animal procedures used were per institutional guidelines. The villi were treated initially with hyaluronidase followed by trypsin/EDTA and subjected to a density-gradient centrifugation using Histopaque®-1077 (www.sigmaaldrich.com) to separate living cells and cell clusters from the debris. The cells in the interface of density-gradient medium were cultured and expanded in Dulbecco’s minimum essential medium (DMEM) containing 10% heat inactivated fetal bovine serum (FBS) IX ITS (insulin, transferrin, and selenite; www.sigmaaldrich.com) and epithelial cell growth supplement (https://www.sciencellonline.com). The enterocytes propagated as individual clusters at the beginning but reached semi-confluence when they were pooled following dissociation with Accumax (www.sigmaaldrich.com) and then replated and expanded in larger flasks. The cells in the third passage were dissociated, counted, and seeded in 12-well plates at the densities of 100 000 cells/mL and grown for 2 days before their treatments with PMA. The cell layers were rinsed twice, each with 1 mL of serum and growth-factor-free culture medium for 5 minutes to deplete exogenous proteins, and then replaced with 1 mL of the same serum-free medium. The treatment cultures were added to 1 µL (500 ng) of PMA dissolved in dimethyl sulfoxide (DMSO), whereas the control cultures received only the DMSO. All assays were done in triplicate, and the changes in the cells were examined microscopically at 24 h to ensure the anticipated morphological changes and photographed. We used replicate cultures, derived from two separate experiments, for mass spectrometric protein analysis.
Proteomic Analyses
After incubation, the cells were removed of supernatant and lysed with 0.5 mL of 4 M guanidine HCl containing 20 mM Na-acetate, pH 6.5, by freeze thaw and repeated trituration. The cell extracts were transferred to fresh tubes, centrifuged at 21 000 g for 10 minutes at 4°C, and the resulting supernatant dialyzed using 5000 MW cut-off Dispo dialyzers (www.spectrumchemical.com) against excess volumes of 25 mM ammonium bicarbonate at 4°C over a 48-hour period with 3 changes. The retentate were transferred to micro-centrifuge tubes and subjected to reduction with 10 mM dithiothreitol (DTT) at 60°C and alkylation with 20 mM iodoacetamide at room temperature for 1 hour, and then digested with 50 ng of MS grade trypsin (www.thermofisher.com) for 24 hours at 37°C.16,17 The tryptic digests were desalted using Pierce C18 spin columns per manufacturer-suggested protocol. The eluted peptides were dried and resuspended in 0.1% formic acid (FA) for liquid chromatography-mass spectrometry (LC-MS)/MS. Analyses of LC-MS/MS were done using an Agilent 1200 series micro-flow high-performance liquid chromatography (HPLC) coupled to a Bruker amaZon SL quadrupole ion trap mass spectrometer with a captive spray ionization source. Peptides were separated using a C18 capillary column (150 mm × 0.1 mm, 3.5 µm particle size, 300 Å pore size; ZORBAX SB) with 5% to 40% gradients of 0.1% FA (solvent A) and acetonitrile in 0.1% FA (solvent B), and a solvent flow rate of 1.6 µL/min over a 300-minute period each. The captive spray source was operated in a positive ion mode with a dry gas temperature of 150°C, dry nitrogen flow 3 L/min, and capillary voltage of 1500 volts. The data were acquired in the auto MS (n) mode with optimized trapping condition for the ions at m/z 1000. MS scans were performed in an enhanced scanning mode (8100 m/z/s) with collision-induced dissociation and MS/MS fragmentation scans performed automatically for top 10 precursor ions for 1 minute in the ultrascan mode (32 500 m/z/s). 18 The results were based on five replicate samples in each group from two separate experiments.
Data Analyses
Bruker DataAnalysis 4.0 software was used to pick peaks from the LC-MS/MS chromatogram using a default setting as recommended by the manufacturer to create Protein Analysis Results.xml file which were then used for Mascot database search. The parent ion- and fragment ion mass tolerances were both set at 0.6 Da with cysteine carbamidomethylation and methionine oxidation as fixed and variable modifications in Mascot search. Mascot search was carried out against Gallus proteins in UniProt database to identify the proteins in the cell extracts. The peptides from all proteins were identified with 95% confidence limit and reported based on <1% false discovery rate using at least two peptides and one unique peptide from a protein. The uncharacterized Gallus proteins were tentatively identified by their gene sequence similarities. Mascot.dat files were then exported into Scaffold Proteome Software version 4.8 (http://www.proteomesoftware.com) to identify differentially expressed proteins. 19 The quantitative differences were calculated on the basis of 95% confidence limit. The differentially regulated proteins were subjected to functional annotations by gene ontology (GO) terms of proteins using Protein Analysis Through Evolutionary Relationships software (PANTHER; http://pantherdb.org). The differentially expressed proteins were also subjected to STRING protein association network (https://string-db.org) and obtain KEGG pathway associations.
Results
Phorbol myristate acetate treatment produced cellular dystrophy, cachectic changes in the cells, and increased intercellular spaces, while the cells remained attached to the wells (Figure 1). Longer incubation up to 48 hours severely shrunk the cells’ widening intercellular spaces, often, detaching cell clusters from the substratum (not shown).
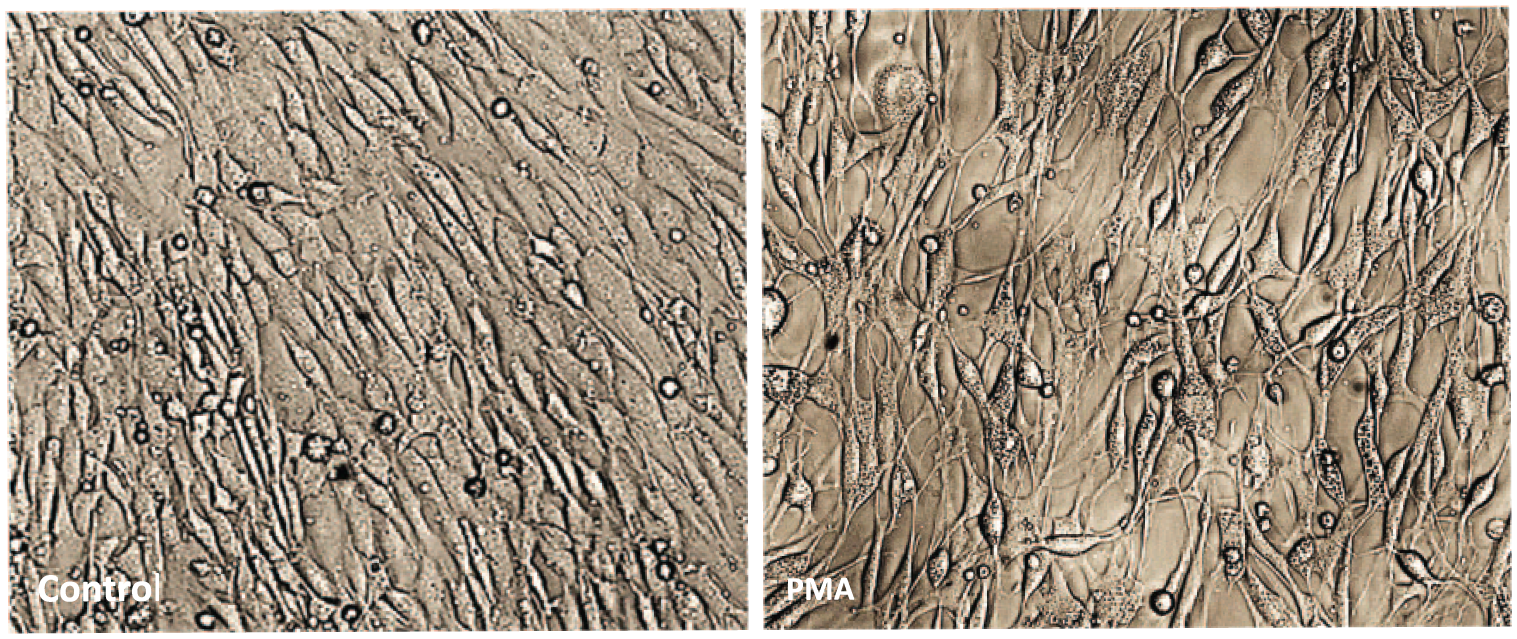
Chicken ileal enterocytes showing morphological changes induced by PMA at 24 hours following treatment (magnification 100×).
There were a total of 333 proteins consisting of 236 clusters, of which 15 clusters were uniquely present in control and 3 in PMA treated cells (Figure 2; Supplemental Table S1). Quantitative comparison showed upregulation of 24 individual (13 clusters) and downregulation of 63 proteins (51 clusters) by PMA treatment (Figure 2; Table 1).
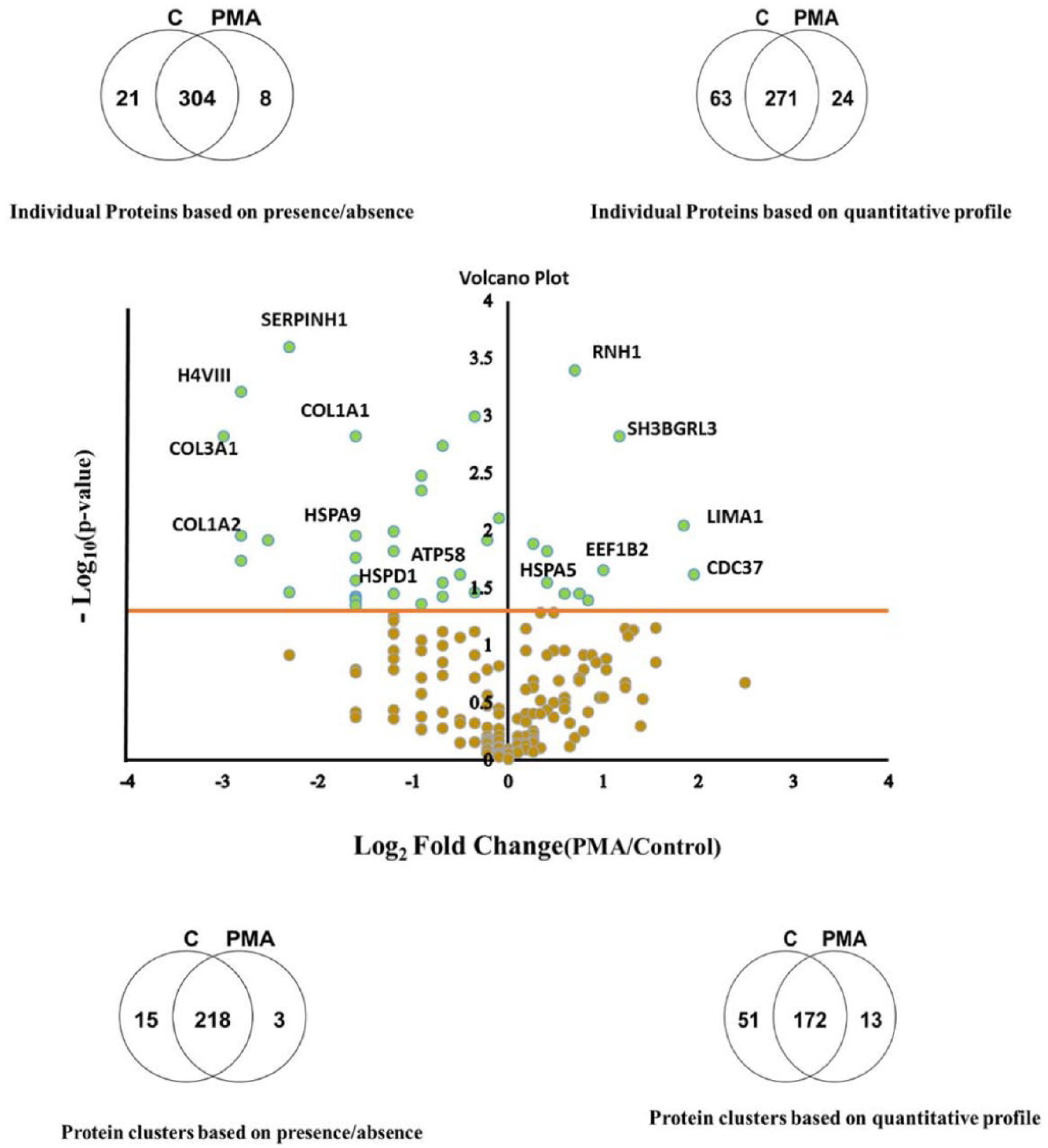
Venn diagrams showing the common and differentially expressed individual proteins and protein clusters and a volcano plot showing down- and upregulated protein clusters.
Differentially regulated proteins in the enterocytes by PMA treatment.
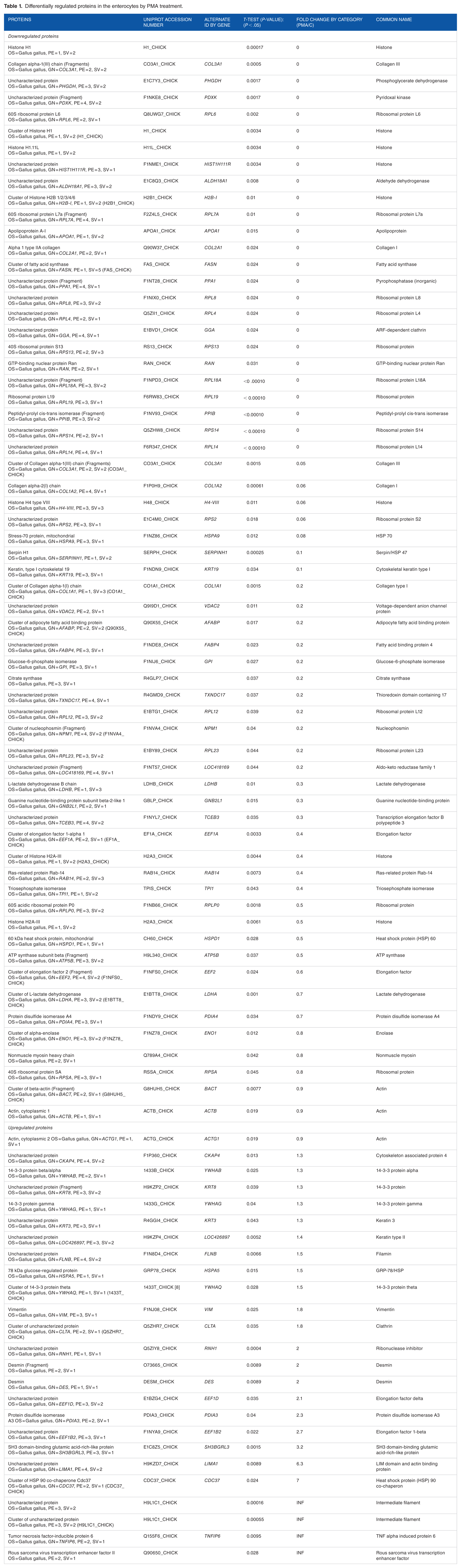
There were 10 biological processes affected by PMA, of which 8 were common to both groups although the proteins included in each of these processes were different (Table 2). Table 3 shows the KEGG pathways affected by PMA. There were 12 pathways downregulated and 3 pathways upregulated by PMA treatment. The adhesion functions of the cells were downregulated in PMA-treated cells indicated by the reduction in the levels of two collagen proteins. The downregulated proteins largely belonged to cellular and metabolic processes that included enzyme proteins associated with energy metabolisms such as alpha enolase (ENO1), ATP synthase (ATP5B), aldehyde dehydrogenase (ALDH18A1), phosphoglycerate dehydrogenase (PHGDH), triose phosphate isomerase (TPI1), lactate dehydrogenase (LDHA, LDHB), ATP synthase (ATP5B), glucose phosphate isomerase (GPI), and fatty acid metabolism and transport-related proteins (fatty acid synthase [FASN], apolipoprotein A1 [APOA1], and fatty acid binding proteins [AFABP]). Several ribosomal proteins and DNA replication and interactive proteins such as elongation factors (EEF1A, EEF2), histones (H4-VIII, H2B-I), nucleophosmin (NPM1), and cytoskeletal proteins (ACTB, ACTG1), nonmuscle myosin, and cytokeratin proteins were affected by PMA. Heat shock proteins (HSP) such as the HSPs 47(SERPINH1), 60 (HSPD1), and a mitochondrial stress protein 70 (HSPA9) were downregulated on PMA treatment, and HSP 90 co-chaperon (CDC37) and a 78 kDa glucose regulated protein (HSPA5) were upregulated (Tables 1 and 2). Other upregulated proteins in PMA-treated enterocytes included certain cytoskeletal and structural proteins such as the intermediate-sized filament proteins, vimentin, and keratins, and two or more signal transduction proteins, a clathrin light chain A and a Rous sarcoma virus transcription enhancer factor II (Table 1). Figure 3 shows the number of regulated proteins under different biological processes although all of the regulated Gallus proteins with putative biological functions (Table 1) were not accounted in the chart.
GO-annotated proteins associated with different biological processes.

KEGG pathways affected by PMA treatment.
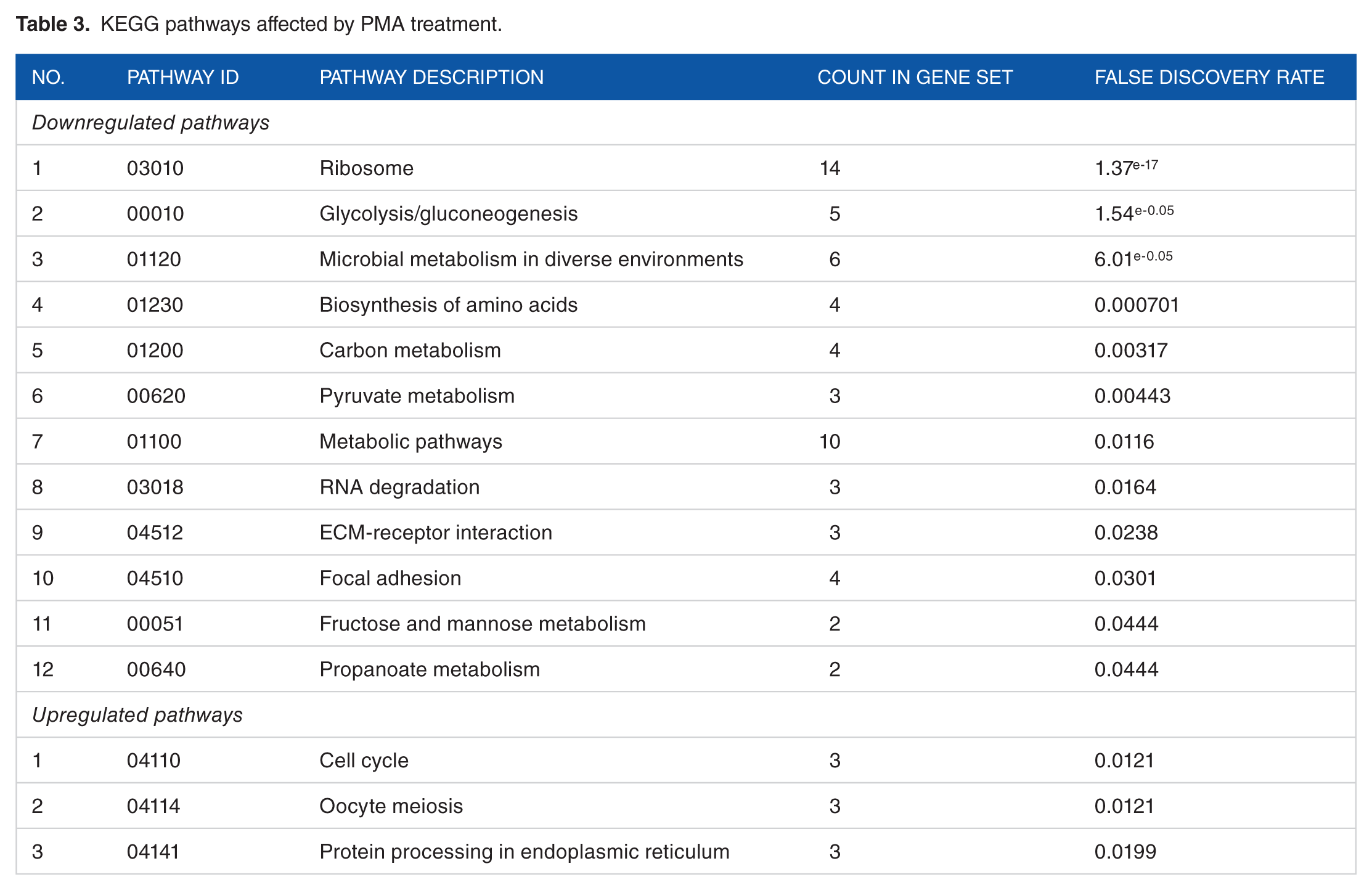
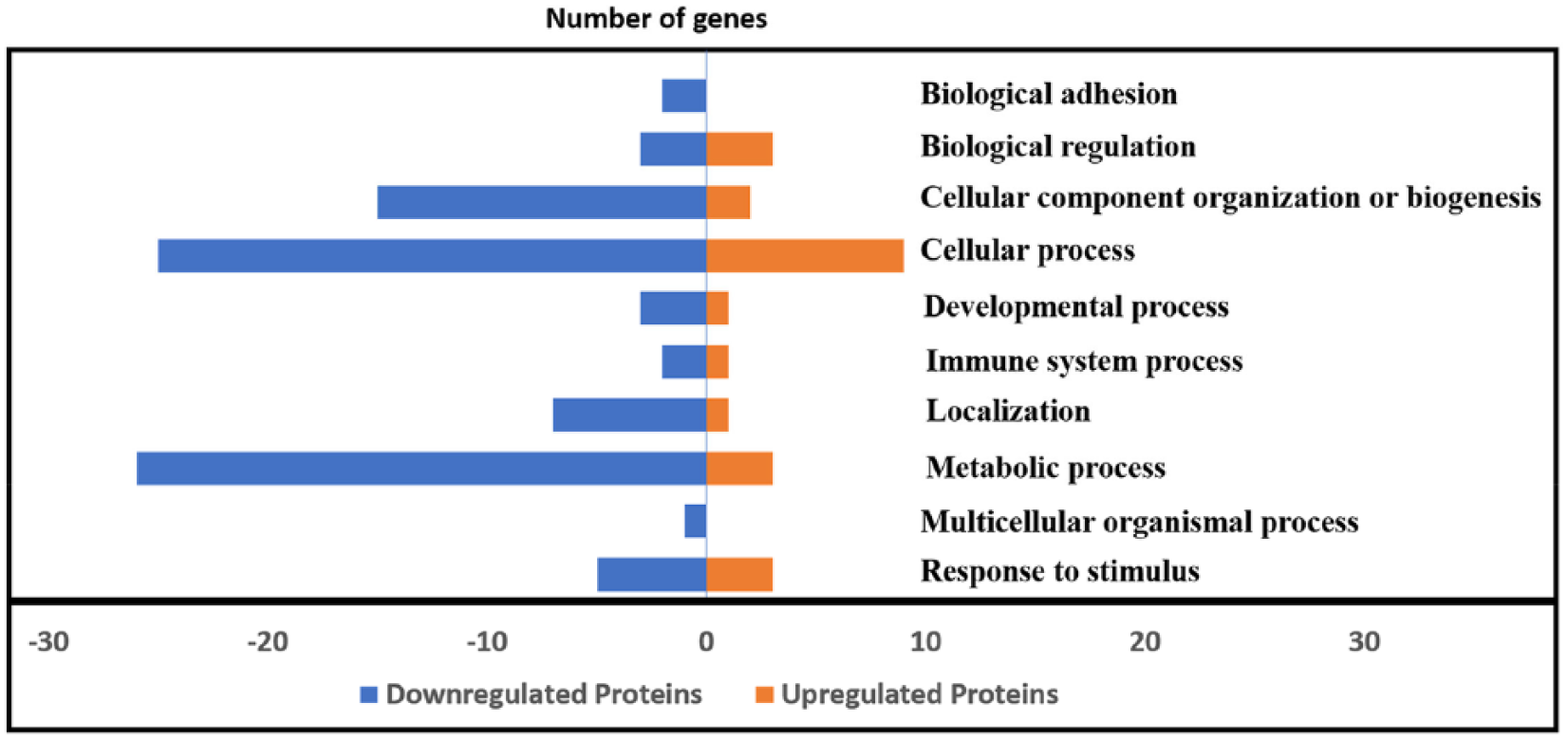
Phorbol myristate acetate-induced down- and upregulated proteins in different biological processes.
Discussion
Our results show that PMA exerts an anti-anabolic effect on enterocytes, downregulating several proteins which affect different biological processes. Many of the proteins identified were associated with energy metabolism, carbon metabolism, nuclear, signal transduction, cytoskeletal homeostasis, and cell adhesion functions. The downregulation of glycolytic and energy metabolism-associated enzymes such as ATP- and citrate-synthases, glucose 6-phosphate isomerase, and several oxido-reductase enzymes (phosphoglycerate-, aldehyde-, and lactate dehydrogenase, aldo-keto reductase, and thioredoxin domain-containing protein 17 [TXNDC17]) indicate diminished mitochondrial activities which were also further evident from the decreased levels of fat metabolism-related proteins. The fat metabolism-associated proteins such as fatty acid synthase, fatty acid binding protein (FABP), and apolipoproteins help not only in energy production but also their transportation along with other lipophilic substances such as cholesterol and triglycerides thereby playing key roles in membrane biosynthesis and signaling functions.20–23 Intestinal tissues constitutively produce FABPs which transport dietary lipids and fatty acids.24–27 Decreased levels of both FABP and ApoA1 in PMA-treated enterocytes suggest decreased lipid metabolism and their transport that can affect membrane biosynthesis and remodeling. Phorbol myristate acetate was shown to induce transcriptional suppression of ApoA4 in human hepatic and intestinal cells. 28 Song et al 29 reported the negative effects of phorbol on membrane remodeling based on their studies with human intestinal epithelial cells. Both nuclear and nucleolar functions of enterocytes were also suppressed by PMA which were evidenced by the decrease in the levels of several ribosomal proteins, histone, elongation factor, and nucleophosmin proteins that are involved in transcription, translational activities, ribosome biogenesis, and degradation. Phorbol myristate acetate also affected both protein maturation and degradation functions indicated by differential changes in HSPs and other chaperon proteins such as peptidyl prolyl cis-trans isomerase (PPIB), thioredoxin domain containing 17, and protein disulfide isomerase (PDIA) family of proteins. 30 The chaperon proteins, particularly the HSPs, are stress proteins that are involved in a variety of functions which include peptide translocation, folding, stabilization, and the degradation of misfolded proteins.31–33 The HSPs protect the cells against apoptosis and have been implicated in some gastrointestinal diseases.34–36 HSPs appeared to be differentially regulated by PMA. The HSPs 90, GRP-78, and PDIA3 were upregulated by PMA, the HSPs 47 and 60, and PPIB, whereas PDIA4 were downregulated on PMA treatment. The differential regulations of these stress proteins in the enterocytes are not understood but reciprocal regulations of different stress proteins such as GRP 78 and HSP 70 have been observed in other cells. 37 In certain neurodegenerative diseases, the HSPs 90 and 70 have been shown to have opposing roles on the stability of their respective interacting proteins and their proteosomal degradations. 38 HSPs help maintain intestinal integrity and provide defense against epithelial permeability and their decreased expressions have been reported in conditions that increase gut permeability.39,40 HSP 47 (SERPINH1), which decreased in PMA-treated enterocytes, has been implicated in the etiology of intestinal fibrosis in experimentally induced colitis. 41 Because fibrosis is a condition that relates to the overproduction and deposition of collagens, 42 a decreased expression of HSP 47 in PMA-treated enterocytes, along with the decrease in collagen content, observed in this study seems consistent.
Phorbol myristate acetate also induced differential changes in several cytoskeletal and structural proteins which affect extracellular matrices (ECMs)-receptor interactions and focal adhesion of cells. These included actin, nonmuscle myosin, and keratin 19, all of which were downregulated whereas some others, particularly the intermediate filament-associated proteins, and desmin, and vimentin were upregulated along with keratins 3 and 8. The relationship and the differential regulation of different cytoskeletal proteins are not understood, but it can be related to their metabolic dynamics such as their turnover rates. The dysregulations of cytoskeletal proteins and the adhesion of the cells to their ECM are known to affect their morphology and survival. Activation of PKC affect cell polarization also causes their morphological changes through mechanisms regulating actin cytoskeleton and other stress fibers.43–45 Similarly, the collagens which are an important component of ECM play significant role in cell-matrix interactions and influence cell attachment and their spreading, and contribute to their phenotypes. 46 Both type I and type III collagens were significantly downregulated in PMA-treated enterocytes. Although there are no data of enterocytes producing collagens, there are many reports of different epithelial cells producing collagens.47–50 In addition, the PMA-treated enterocytes showed a downregulation in protein named RPSA which is a receptor for laminin present in ECM that facilitates cell adhesion. 51 Taken together, it appears that the changes in cytoskeletal proteins along with the decrease in their collagen production likely affect their spreading and dystrophic changes in the enterocytes.
Phorbol myristate acetate also affected signaling function proteins indicated by the reductions in the level of a mitochondrial, voltage-dependent, anion-selective channel protein 2 that is responsible for ATP channeling and a GTP binding RAS-related nuclear proteins which contribute to the nuclear activities of the cells. Similarly, there were other signal transduction proteins that were upregulated in the enterocytes on PMA treatment. These were three 14-3-3 proteins—beta/alpha, theta, and gamma—which bind to different kinases, phosphatases, and transmembrane receptors. Heightened expressions of these proteins have been linked to different pathological conditions. 52 The other upregulated proteins were a Rous sarcoma virus transcription enhancer factor II and a clathrin light chain A protein, both of which can increase the cell’s susceptibility to microbial invasions.
In conclusion, our results show that PMA downregulates mitochondrial function, impairs energy metabolism and nuclear functions of the enterocytes such as their transcriptional and translational activities, and disrupts cytoskeletal homeostasis and focal adhesion which most likely contribute to cellular dystrophy, cachexia, and distension of intercellular spaces. Extrapolating it to an in vivo situation, such changes in enterocytes can inevitably increase intestinal permeability and cause epithelial damage resulting in enteropathy. The PMA-induced changes in enterocytes may help understand the “leaky-gut” problem which is linked to several avian intestinal diseases.
Supplemental Material
PMA_Supplementary_Table_1_xyz12969d4659385 – Supplemental material for Phorbol 12-Myristate 13-Acetate-Induced Changes in Chicken Enterocytes
Supplemental material, PMA_Supplementary_Table_1_xyz12969d4659385 for Phorbol 12-Myristate 13-Acetate-Induced Changes in Chicken Enterocytes by Narayan C Rath, Anamika Gupta, Rohana Liyanage and Jackson O Lay Jr in Proteomics Insights
Footnotes
Acknowledgements
The authors thank Sonia Tsai and Scott Zornes for technical assistance.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
NCR and RL conceived and designed the experiment; AG, RL, and NCR carried out the experiment; and NCR and RL wrote the manuscript along with JOL.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
