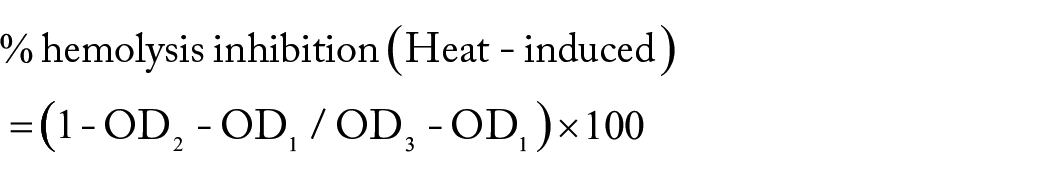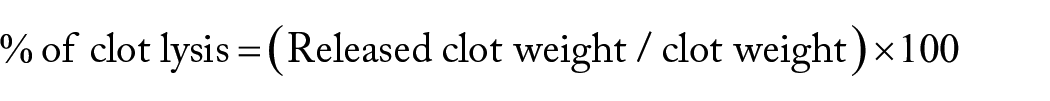Abstract
Fruit wastes are becoming popular as treasures for drug discovery in different classes of therapeutics. This research aimed to investigate the phytochemicals and potential bioactivities, such as antimicrobial, thrombolytic, anti-inflammatory, analgesic, and antidiarrheal properties of commonly available mandarin orange (Citrus reticulata Blanco) peel through experimental and computational techniques. Extensive chromatographic and 1H-NMR spectroscopic analysis was employed to isolate four purified compounds, which were characterized as tangeretin (A), nobiletin (B), limonin (C), and β-sitosterol (D). Furthermore, GC-MS/MS analysis detected over 90 compounds with a notable number of polymethoxyflavones, including nobiletin (29.04%), tangeretin (15.55%), artemetin (8.1%), 6-demethoxytangeretin (1.28%), sinensetin (0.95%), demethylnobiletin (0.14%), pebrellin (0.10%), and salvigenin (0.04%). Dichloromethane soluble fraction (DCMSF) exerted the highest antimicrobial potency Candia albicans against (20 mm zone of inhibition) in the disk diffusion assay method. The aqueous soluble fraction (AQSF) exhibited 34.71% and 48.14% inhibition in hypotonic solution-induced and heat-induced hemolysis in the membrane stabilizing assay. Similarly, the AQSF exhibited the highest anti-thrombotic property with 32.57% clot lysis. The investigated 3 doses of the methanolic extracts (100, 200, and 400 mg/kg body weight) exerted statistically significant in vivo central analgesic effects in a tail-flicking method in a time-dependent manner. Moreover, all the doses exhibited significant efficacy in inhibiting acetic acid-induced abdominal writhing and castor oil-induced diarrheal episodes in mouse models. The molecular docking studies corroborated the existing in vitro and in vivo findings by demonstrating better or comparable binding affinities toward the respective receptors and favorable pharmacokinetic properties and toxicological profiles. The present findings indicate that C. reticulata is a rich source of polymethoxyflavones, demonstrating potential efficacy against microbial infections, thrombosis, inflammation, pain, and diarrhea. Nonetheless, comprehensive phytochemical screening is imperative to identify additional bioactive compounds and evaluate their pharmacological effects against several chronic health conditions, grounded in their traditional uses and current evidence.
Keywords
Introduction
The investigation of contemporary synthetic medications is approached with caution due to their known adverse effects. 1 In contrast, conventional herbals are obtaining popularity as they are perceived as environmentally friendly, more natural, and fewer adverse effects. 2 So, despite all of the benefits of existing synthetic medications, scientists carry on to favor nature-based remedies. 3 The unique phytoconstituents of various plant parts contribute to the diverse potential of medicinal species, functional foods, or food byproducts in alleviating and treating various human disorders.3-5 In Indian culture, approximately 80 000 species of medicinal plants have been utilized as traditional medication since ancient times. 6 Plant-based medications serve as the primary resource for medical care for a substantial portion of the global population.7-9 Particularly in low-income nations, where facilities are limitted, traditional therapy and ethnic values play a significant role, making natural remedies an affordable choice for essential healthcare.10,11
Mandarin fruit (Citrus reticulata Blanco), a wide species that belongs to the family Rutaceae valued not only for its appealing flavor but also for its numerous therapeutic properties, including anticancer, antioxidant, anti-hyperlipidemic, anti-inflammatory, and antidiabetic effects.12,13 These health benefits are attributed to the rich presence of phytochemicals such as organic acids, carotenoids, sugars, amino acids, flavonoids, polyphenols, limonoids, and phenolic acids in mandarin fruit. 13 The traditional uses of mandarin fruit extend to laxative, aphrodisiac, antiemetic, astringent, and tonic properties, as well as skin-related benefits.14-16 It is also used as a stomachic and carminative.15,17,18 The pericarp and endocarp possess medicinal qualities, which are employed in the treatment and management of various conditions such as analgesia, anticholesterolemia, anti-inflammatory, antiseptic, anti-asthmatic, carminative, antiscorbutic, expectorant, antitussive, and stomachic.18,19 The immature green exocarp is utilized for alleviating, gastrointestinal distension, liver cirrhosis, liver, hypochondrium, spleen enlargement, as well as chest pains. The seed exhibits carminative and analgesic properties, making it a remedy for hernias, mastitis, lumbago, and discomfort or enlargement of the testicles. 19
In addition to its extensive biological significance, some phytochemical studies revealed that C. reticulata is a treasure trove of bioactive substances. Another report indicates that polymethoxyflavones (PMFs) are plentiful in citrus peels, particularly in mature citrus peels. These peels have been traditionally employed in Chinese medicine for numerous years to alleviate various conditions, including stomach upset, skin inflammation, cough, ringworm infections, muscle pain, and possibly lowering blood pressure.20,21
The current study utilizes Gas Chromatography-Mass Spectrometry (GC-MS) and NMR spectroscopy to identify and isolate bioactive components from methanolic extract of C. reticulata fruits peel. This research additionally explored the antimicrobial, anti-inflammatory, thrombolytic, central and peripheral analgesic, and anti-diarrheal properties of this species. The outcomes may be helpful to create a new way to discover new drugs and enhance the nutraceuticals and medicinal values of this very commonly available fruit peel waste of the C. reticulata.
Methods and Materials
Sample collection and drying
In July 2023, Mandarin oranges (Citrus reticulata Blanco) were procured from the West Dhanmondi local market in Dhaka, Bangladesh. The batch originated from the Yunnan region in southwest China. Following the purchase from the market, the procedure involved manually collecting the fresh peels and subsequently washing them with fresh water. The drying process was facilitated by prolonged exposure to open air. Strict protocols were employed to ensure that the ambient temperature remained below 30°C during the drying regimen. This precaution was taken to prevent any potential degradation and to ensure the preservation of heat-sensitive compounds. The dried fresh peels of C. reticulata are depicted in supplementary Figure S1.
Chemicals and reagents
Analytical-grade reagents and substances were employed in the study. Tween 80 and the suspending medium for the extract were procured from Merck (Germany). Glibenclamide, normal saline solution, and loperamide were supplied by BEXIMCO Pharmaceuticals Ltd., Dhaka, Bangladesh (Additional chemicals included aspirin, diclofenac sodium, and sodium thiopental.).
Extraction
Following the shadow air drying, 500 g of powdered fruit peel was soaked in 2.5 L of methanol within a sealed amber-colored vessel for 15 days with irregular jerking. The resulting solvent mixture was filtered using a clean cotton pad and filter pad. The methanol was then removed using an “EYELA Rotavapor” rotary evaporator at lower pressures and approximately 40°C to yield 58 g of gummy extract from the methanolic extract of C. reticulate peel. This represented 11.6% of the weight of the powdered fruit peel.
Fractionation
Solvent-solvent differentiation of the extract was performed using S. Morris Kupchan’s partitions (1970), as modified by VanWagenen et al. 22 Five grams of crude extract were sequentially treated with water in methanol (1:9), petroleum ether (PESF), dichloromethane (DCMSF), ethyl acetate (EASF), and water (AQSF) to generate 4 distinct fractions. Each fraction was evaporated until completely dried. The yields for the petroleum ether, dichloromethane, ethyl acetate, and water-soluble fractions were 15%, 43%, 24%, and 18%, respectively.
Phytochemical isolation and detection through nuclear magnetic resonance (NMR) technique
Secondary metabolites were isolated from the methanolic crude extract, which was then subsequently purified using chromatographic techniques. The schematic flowchart of complete phytochemical extraction, fractionation, identification, and isolation of phytoconstituents from the C. reticulata is stated in supplementary Figure S2. The size exclusion chromatography used a gradient solvent system, with lipophilic sephadex LH 20 acting as the stationary phase. The ratios of the various solvents utilized as the mobile phase were as follows: n-hexane, dichloromethane (DCM), and methanol in a proportion of 2:5:1; 10% methanol in DCM; 50% methanol in DCM; and 100% methanol (supplementary Table S1). These fractions were thoroughly screened using thin-layer chromatography (TLC) on silica gel-coated metal plates (20 cm × 20 cm) with various solvent solutions. The obtained chromatogram was visually examined under UV illumination at 254 and 366 nm to identify fluorescence quenching. Colored compounds were also recognized after spraying the plate with a 1% vanillin-sulfuric acid solution and heating at 105°C for 2 to 3 minutes. Fractions with similar TLC patterns were combined, yielding 13 sub-fractions. The phytochemicals were then separated and purified from these sub-fractions using repeated preparative thin layer chromatography (PTLC).
A total of 4 compounds (A-D; Figure 1) were isolated and characterized in the study through an extensive chromatographic and spectroscopic methods. Compound D was isolated from sub-fraction F-9 to F-12 (using a solvent mixture of 5% ethyl acetate in toluene), two compounds such as Compounds A (Rf value = 0.68) and B (Rf value = 0.46) were isolated from sub-fraction F-23 to F-28 (using 5% methanol in chloroform). In addition, compound C was isolated from sub-fraction F-30 to F-34 (using 10% methanol in chloroform; Rf value = 0.55; Tables S2 and S3).
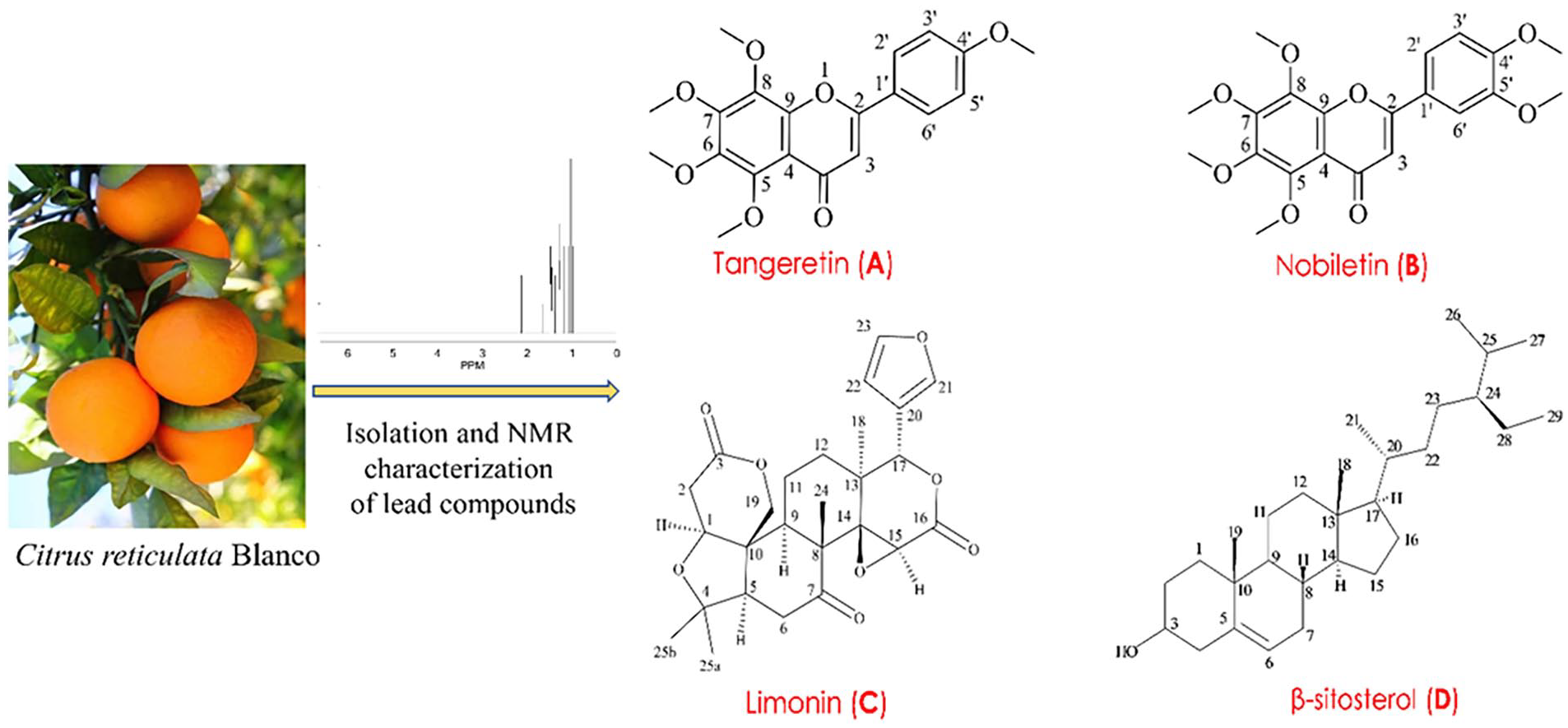
Structures of the identified and characterized compounds through the 1H-NMR and chromatographic technique from the peel extract of Citrus reticulata Blanco.
The total phytochemical isolation procedure and its schematic flowchart can be found in Supplemental Figure S2. Bruker AMX-400 NMR spectrometer was operated to generate 1H-NMR spectra while all spectra were obtained in a deuterated chloroform solvent (CDCl3). The peak values for chemical shift (δ) were standardized to tetramethylsilane (TMS) and the residual signals from the solvent.
GC-MS/MS analysis
The methanol extract of C. reticulata (MECR) was analyzed using the Shimadzu GC/MS-QP2010 ultra, which had an auto-sampler. A 5 MS/HP column (30 m, 0.25 mm, 0.25 m) was used, using pure helium as the mobile phase. The helium’s linear speed was kept at 39 cm/s, with a circulation rate of 1.12 mL/min. The oven temperature raised at a rate of 10°C/min, ranging from 110°C to 280°C, while the needle temperature was set at 250°C. Injection volume of 50 µL was used in split-less mode with a 10:1 ratio. The detector voltage remained at 0.94 kV, while the ambient temperatures for the ion source, MS transfer line, and ion source were held at 200°C and 250°C, respectively. Full-scan mass spectra were obtained at 10 000 µ/s in the (m/z) range of 85 to 500. Peaks and chemical ingredients were identified using a search of the National Institute of Standards and Technology (NIST) database.
Antimicrobial properties
A total of 13 various microbial species, encompassing 5 Gram-positive bacteria (Bacillus subtilis, Bacillus megaterium, Bacillus cereus, Staphylococcus aureus, and Sarcina lutea), 8 Gram-negative bacteria (Salmonella typhi, Escherichia coli, Shigella boydii, Salmonella paratyphi, Shigella dysenteriae, Vibrio mimicus, Vibrio parahemolyticus, and Pseudomonas aeruginosa), along with 5 fungi (Aspergillus niger, Candida albicans, Aspergillus fumigatus, Saccharomyces cerevisiae, and Aspergillus ustus), were procured in their pure culture strains from the Bangladesh Council of Scientific and Industrial Research (BCSIR), Dhaka, Bangladesh.
The antibacterial efficacy of crude plant extracts and their various fractions was examined using the disk diffusion method. 23 In this process, 6 mm-diameter filter paper disks containing specified quantities of test samples (concentration of 400 µg/disk) were positioned on nutrient agar medium and uniformly inoculated with the respective microorganisms. These disks were then dried and sterilized. Antibiotics permeated the agar gel from a confined source, generating a concentration gradient. Ciprofloxacin standard antibiotic disks served as positive controls, while blank disks were utilized for the negative control. The petri dishes were refrigerated at a low temperature (4°C) for 16 to 24 hours to enhance the diffusion of test samples into adjacent media. 23 Following inversion and additional incubation at 37°C for 24 hours to promote optimal microbial growth around the disks was impeded by the test samples exhibiting antibacterial effects. This hindrance led to the formation of discernible zones of inhibition, and their diameters were measured to assess the antibacterial activity of the test sample.23,24
Anti-inflammatory effects
The crude methanolic extracts and fractions derived from the peel of C. reticulata were evaluated for in vitro anti-inflammatory activity by employing the established techniques as described by previous studies.23,25
Hypotonic solution-induced anti-inflammatory effect
To evaluate the efficiency of MECR and its related extractives in stabilizing human erythrocyte membranes, we followed known techniques from a previous research. 25 Erythrocytes were extracted from a healthy adult weighing 70 kg with fair skin and stored in a sterile container with EDTA for anticoagulation. A buffer solution containing disodium phosphate and its conjugate acid, monosodium phosphate, was created to maintain a pH of 7.4. An isotonic solution (500 mL, 154 mM) was made by dissolving 4.5045 g of NaCl in sterile distilled water, whereas a hypotonic solution (500 mL, 50 mm) was made using 1.4625 g. The erythrocyte suspension was washed 3 times with buffer and isotonic solution before being centrifuged at 3000 rpm for 10 minutes. A stock RBC suspension (0.50 mL), a 10 mM sodium phosphate buffer (4.5 mL), a hypotonic solution (50 mM NaCl), and different quantities of extractives (2.0 mg/mL) or standard acetyl salicyclic acid (ASA) (0.10 mg/mL) were used to create test samples. After a 10-minute incubation at room temperature, the solutions were centrifuged (3000 rpm for 10 minutes) and the supernatants’ absorbance (optical density (OD)) was measured at 540 nm. The proportion of inhibition of hemolysis or stabilization of membrane was estimated by employing the formula stated below:
Where, OD1 and OD2 represent the optical density of hypotonic buffered saline solution (control) and the investigated sample in hypotonic solution, respectively.
Heat-induced anti-inflammatory effect
Two sets of centrifuge tubes were created, each containing 5 mL of isotonic buffer, 1.0 mg/mL of methanolic peel extract, and different fractions. A control tube contained an equal volume of all substances except the test materials. In every well, 30 µL of erythrocyte suspension was added by inverse and gently stirring. One pair of tubes was placed in a 54°C water bath for 20 minutes, while the second set was maintained in a cold bath at 0°C to 5°C. After incubation, the supernatant was centrifuged (1300 rpm for 3 minutes), and the optical density (OD) was measured at 540 nm. The percentage inhibition of hemolysis in the investigations was estimated using the corresponding equation:
Where, OD1, OD2, and OD3 refers to the optical density of the non-heated test sample, the heated test sample, and the heated control sample, respectively.
Thrombolytic properties
In vitro thrombolytic activity was determined using the blood clot lysis technique according to the methods described by Prasad et al. 26 The positive control was a lyophilized streptokinase vial containing 1 500 000 IU while the negative control was distilled water. Healthy volunteers’ venous blood was collected in pre-weighed sterile micro-centrifuge tubes (1 mL/tube), then incubated at 37°C for 45 minutes. Following clot formation, the weight of each clot was determined after full serum extraction. Each micro-centrifuge tube with a pre-weighted clot received a 100 μL aqueous solution from different partitions, in addition to the crude extract. The tubes were incubated for 90 minutes at 37°C and kept in a close observation for clot lysis. The percentage of clot lysis was enumerated by using the following the formula:
Animal study design
The Animal Division of the International Centre for Diarrheal Diseases and Research in Bangladesh (ICDDRB) provided Swiss albino adult mice aged 4 to 5 weeks and weighed 25 to 30 g. Prior to the animal research, these mice were kept in polypropylene cages under conventional climatic settings such as a 55% relative humidity level, 25°C ambient temperature, and a 12-hour light-dark cycle. Prior to the beginning of the experiments, the animals were allotted a week to acclimate to their new environment. During this time, the animals were fed a standard laboratory diet and had unlimited access to water. Their care and handling followed the guidelines set by the Swiss Academy of Medical Sciences (SAMS) and the Swiss Academy of Sciences. The study also complied with the latest Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines. 27 The protocols for animal handling and procedures involved in the animal studies were thoroughly reviewed and approved by the Committee on Ethical Compliance in Research at the State University of Bangladesh, Dhaka, Bangladesh (approval number: 2022-01-23/SUB/A-ERC/006).
The study included 3 different doses of the methanol extract of C. reticulata (MECR) at concentrations of 100, 200, and 400 mg/kg body weight, designated as ME-100, ME-200, and ME-400, respectively. The animal subjects were divided into 5 groups, each consisting of 4 mice (n = 4). This sample size was estimated by using the “resource equation” method which was clearly explained in the previous studies.28-30 The groupings were organized as follows:
Group I (n = 4): Normal saline water [0.9% (w/v) NaCl solution BP] as negative control
Group II (n = 4): Standard drug as positive control
Group III (n = 4): 100 mg/kg body weight MECR (ME-100)
Group IV (n = 4): 200 mg/kg body weight MECR (ME-200)
Group V (n = 4): 400 mg/kg body weight MECR (ME-400)
Central analgesic activity
The analgesic effects of MECR were assessed using a heat-based approach and the tail immersion experiment, as described by previous investigation. 31 The positive control group received morphine (15 mg/mL) diluted with saline water to form the standard sample (subcutaneous, 2 mg/kg), as reported in earlier report. 32 The mice received the test drugs orally using a feeding syringe. The experiment involves submerging a mouse’s tail in water heated to 55°C. The Pain Response Time (PRT) or latency duration, which indicates how long it takes for each mouse to move its tail in hot water, was assessed at 0, 30, 60, and 90 minutes after the test samples were administered.
Peripheral analgesic activity
The peripheral analgesic effects of MECR were assessed by applying the acetic acid-induced writhing technique in mice model. 33 At the 0 hour of the experiment, the control group was administered normal saline, while the positive control group received diclofenac sodium (50 mg/kg) as standard. Subsequently, the 3 experimental groups were administered their respective doses of the designated crude extracts. After 60 minutes, all groups received an intraperitoneal injection of 1% glacial acetic acid (0.1 mL; dissolved in normal saline) to induce visceral pain. After 5 minutes of acetic acid injection, the writhing times were counted for 15 minutes. The formula for calculating the percentage of writhing inhibition was as follows:
Where, N = average amount of stomach writhing for every group.
Anti-diarrheal activity
The anti-diarrheal activity of MECR was measured by employing a castor oil-induced diarrhea model in mice, outlined in earlier studies.34,35 According to the experimental design, the positive control group received oral administration of loperamide (50 mg/kg bw). The weighted extracts, after being suspended with 1% Tween-80, were unidirectional handled and then applied to the test groups. The normal control group received only the normal saline water. To induce diarrhea, 1 mL of pure castor oil was supplied to each mouse after 1 hour. Each mouse was kept in a separate cage with blotting paper on the floor. For each test group, the floor lining of individual mouse cage was changed every hour. The number of diarrheal stools generated by each mouse was monitored hourly for up to 4 hours after castor oil delivery. The count of diarrheal feces produced by the mice was recorded at 1-hour intervals over a 4-hour observation period. The percentage inhibition of diarrheal episodes was subsequently calculated using the following equation.
Where, D = Average diarrheal episodes in every group.
Molecular docking
The isolated and purified 4 compounds (A-D) and 8 more compounds (C-79, C-83, C-84, C-86, C-87, C-88, C-90, and C-91) from the obtained compound list characterized by GC-MS/MS technique were chosen for a computational modeling study to predict their receptor binding profiles against the relevant target proteins to support the biological activities of the studied extracts and fractions. Following established procedures from several previous studies, molecular docking of these phytocompounds was conducted using BIOVIA Discovery Studio version 4.5, PyMOL 2.3, and PyRx software packages.36-39
Ligand preparation
A total of 12 phytomolecules were selected as ligands in docking investigation. These chemicals were identified as tangeretin (PubChem CID: 68077), nobiletin (PubChem CID: 72344), limonin (PubChem CID: 179651), β-sitesterol (PubChem CID: 481107734), salvigenin (PubChem CID: 161271), pentamethoxyflavone (PubChem CID: 35028119), 6-demoethoxytangeretin (PubChem CID 629964), stigmasterol (PubChem CID: 5280794), pebrellin (PubChem CID: 632255), artemetin (PubChem CID: 5320351), senensetin (PubChem CID: 145659), dimethylnobiletin (PubChem CID: 358832) from PubChem database (retrieved on May 27, 2024). The standard drugs thrombolytic agent warfarin (PubChem CID: 54678486), cyclooxygenase inhibitor diclofenac (PubChem CID: 3033), antioxidant BHT (PubChem CID: 31404), antidiarrheal molecule loperamide (PubChem CID: 3955), antibacterial agent ciprofloxacin (PubChem CID: 2764) and μ-opioid receptor inhibitor morphine (PubChem CID: 5288826) were downloaded in SDF format. Using the PubChem CIDs of the ligands, a ligand library in PDB format was created following the ligands’ sequential loading into Discovery Studio version 4.5. Using the PM6 semi-empirical approach, the accuracy of molecular interactions for every ligand was enhanced. 40
Protein preparation
To predict the potential antibacterial, thrombolytic, anti-inflammatory, analgesic, and antidiarrheal activities of the ligands, a computational docking approach was employed. Specifically, dihydrofolate reductase (DHFR; PDB ID: 4M6J), tissue plasminogen activator (PDB ID: 1A5H), cyclooxygenase 2 (COX-2; PDB ID: 1CX2), μ-opioid receptor (MOR; PDB ID: 5C1M), and kappa opioid receptor (PDB ID: 6VI4) with their corresponding active sites were selected to assess antibacterial, thrombolytic, peripheral and central analgesic, and antidiarrheal effects, respectively, based on the previously published literature.23,41-43 In order to retrieve the target proteins, protein data bank was searched. After then, the three-dimensional (3D) crystal structures were downloaded and saved in PDB format (retrieved on May 27, 2024). Then the downloaded proteins were processed by using Discovery Studio (version 4.5) and Swiss PDB viewer for cleaning and optimizing purposes, respectively. 44
Protein and ligand interaction
The PyRx software was used for molecular interaction purpose. The cleaned and optimized proteins were imported in the software and selected as macromolecules. The active amino acids were assigned, which were pre-selected according to the literature data and summarized in the Supplemental Table S4. The 3D conformers of the ligands in SDF format were uploaded to the PyRx program and minimized energy using “uff force field.” Then, all ligands were transformed into pdbqt format to determine the best hit. Then, as shown in Supplemental Table S4, the grid box was created, with the proteins’ active binding sites centered inside. The other docking process parameters were kept at their factory default values. The final docked macromolecules and ligands were exported as pdbqt format output files, and the outcomes of the docking analysis were then investigated. PyMOL software was used to combine the stored ligands’ output files and the macromolecule’s pdbqt file in PDB format for further visualization. Discovery Studio Visualizer (version 4.5) was used for visualization and 2D figure development.
Pharmacokinetics and toxicity analysis
The pharmacokinetic parameters related to absorption, distribution, metabolism, excretion (ADME), and toxicity were predicted by employing the well-known online server pkCSM (https://biosig.lab.uq.edu.au/pkcsm/prediction; accessed on May 10, 2024).
Physicochemical and drug-likeliness analysis
All the docked 12 compounds were assessed for their potential drug-like properties according to Lipinski’s rule of 5 (molar refractivity between 40 and 130, molecular weight ⩽ 500; H-bond acceptors < 10; H-bond donors < 5; and LogP ⩽ 5) using the Swiss ADME online tool (www.swissadme.ch; accessed on May 27, 2024).
Statistical analysis
The data processing and findings interpretation were done in accordance with the established protocols explained by Assel et al. 45 The in vitro experimental analysis, graphs and data processing were carried out using Microsoft Excel (version 10.0). Additionally, the treated groups were compared to the control (vehicle) group in order to do the statistical analysis for the in vivo experiments. The mean values, along with their corresponding standard errors of mean (SEM), were represented as mean ± SEM to indicate the outcomes of the in vivo assessments. The results from in vivo testing were examined and analyzed through GraphPad Prism software (version 8.0.2). A one-way ANOVA was conducted on all variables, followed by Student’s t-test. P-values less than .05 were considered statistically significant. The heatmap representing the docking score analysis and the graphs associated with the in vivo studies were generated utilizing GraphPad Prism software.
Results
Phytochemical studies
Four compounds were identified, purified, and characterized during the phytochemical isolation process through various chromatographic techniques and 1H-NMR spectroscopic analysis. These compounds were determined to be tangeretin (compound A), nobiletin (compound B), limonin (compound C), and β-sitosterol (compound D). Additionally, 91 compounds were detected and characterized using GC-MS/MS analysis. Further detailed information is provided in the following sections.
Compounds characterization by NMR technique
The 1H-NMR (400 MHz, CD3OD) and the compared literature spectroscopic data of the isolated compounds A–D from C. reticulata.

Compounds detection by GC-MS analysis
A total of 91 phytoconstituents were predominantly detected in the MECR through GC-MS technique. The spectrum obtained from the GC-MS/MS analysis is available in Figure 2. Among the identified over 90 compounds, a significant number of compounds were polymethoxyflavones. Among these, the following were quantified: nobiletin (29.04%), tangeretin (15.55%), artemetin (8.1%), 6-demethoxytangeretin (1.28%), sinensetin (0.95%), demethylnobiletin (0.14%), pebrellin (0.10%), and salvigenin (0.04%). Furthermore, some notable compounds were also detected as dihydrocarvyl acetate (4.07%), Methyl alpha D-galactopyranoside (3.37%), 3’,5’-demethoxyacetaophenone (2.55%), D-limonene (2.48%), and stigmasterol (0.35%; Table 2). Yet, some phytoconstituents were discovered as least amount such as ß-Elemene, Megastigmatrienone, Octadecanamide, Fenoterol, Salvigenin, and Bicyclon onan-2-one, 8-isopropylidene- (0.04%). Various classes of phytoconstituents such as fatty derivatives (Methyl 3-hydroxydodeca-noate, 2,6,9,11-Dodecatetraenal, 2,6,10-trimethyl-, (E,E,E)-, and Hexane,1- (isopropylidene cyclopropyl)-,) steroids (Stigmasterol), flavonoids (Demethylnobiletin, sinensetin, Pebrellin, and artemetin), and aliphatic cyclic (1,2- Cyclohexanedione, Trimethylene borate, delta-Elemene, and Furaneol) compounds were also identified in this analysis (Table 2).

GC-MS spectrum of the crude extract obtained from Citrus reticulata Blanco.
The identified and characterized phytoconstituents obtained from GC-MS/MS analysis are presented with details including the compound names, retention time (RT), percentage of area, molecular weight (g/mol), molecular formula, PubChem CID, and their chemical structures drawn using ChemDraw. Additionally, the pharmacological activities of these compounds were extracted from the literature, with the corresponding references cited in the final column.
Antimicrobial properties
Among the fractions DCMSF showed broad spectrum antimicrobial properties. From gram positive bacteria, DCMSF showed 8 mm zone of inhibition at 3 different bacteria such as B. sereus, B. subtilis, and S. aureus while the DCMSF exerted 10 mm zone of inhibition against 3 different gram-negative bacteria such as S. typhi, E. coli, and V. parahemolyticus at the antimicrobial testing assay (Table S4). DCMSF also showed potency at antifungal Sarcina lutea activity, and most effective result was found against C. albicans (20 mm zone of inhibition). However, MECE exhibits the poorest zone of inhibition only active against Sarcina lutea gram negative bacteria (15 mm; Table S4).
Anti-inflammatory properties
Hypotonic solution-induced hemolysis
In comparison with the standard acetyl salicylic acid at a strength of 0.10 mg/mL (61.90% inhibition against erythrocyte membrane lysis), methanolic extractives of C. reticulata peel at a concentration of 2.0 mg/mL revealed a mild protective effect (Figure 3). AQSF, in particular, displayed 34.71% effective inhibition than the other extract.

Percentage inhibition of hemolysis of different extractives of peel of C. reticulata in hypotonic solution-induced and heat-induced conditions [Here, ME, PE, DCM, EA, and AQ mean the soluble fractions of C. reticulata in methanol, pet ether, dichloromethane, ethyl acetate, aqueous conditions, respectively.].
Heat-induced hemolysis
The extractives from the peel of C. reticulata were efficient in erythrocyte lysis triggered by heat according to the outcomes. The inhibition of AQSF was around 48.14%, which is greater than that of MECR (34.39%), EASF (31.91%), PESF (30.68%), and DCMSF (24.28%), and even higher than the standard Acetyl Salicylic Acid (42.0%) under normal conditions (Figure 3).
Thrombolytic properties
Streptokinase (SK), a positive control resulting in a notable 64.55% clot lysis. In contrast, the negative control, treated with distilled water, exhibited minimal clot lysis at 5.64%. However, the AQSF of the peel of C. reticulata demonstrated the highest thrombolytic activity at 32.57%, followed by the EASF at 22.61%, DCMSF at 6.80%, MECR at 6.05%, and PESF at 4.06% (Figure 4).

Effects of methanolic crude extract and its various solvent fractions of C. reticulata peel against thrombosis [Here, ME, PE, DCM, EA, and AQ mean the soluble fractions of C. reticulata in methanol, pet ether, dichloromethane, ethyl acetate, aqueous conditions, respectively].
Central analgesic properties
Figure 5a shows the findings of the central analgesic effects of the MECR using the tail immersion technique. The pain reaction time for every sample showed dose-dependent when compared to the control medication, morphine. The 100 and 200 mg/kg body weight doses demonstrated statistically significant central analgesic effects (P < .05) at the 30-, 60-, and 90-minute observation points. In contrast, the 400 mg/kg dose showed statistically significant central analgesic effects (P < .05) only at the 30-minute observation point (Figure 5a).
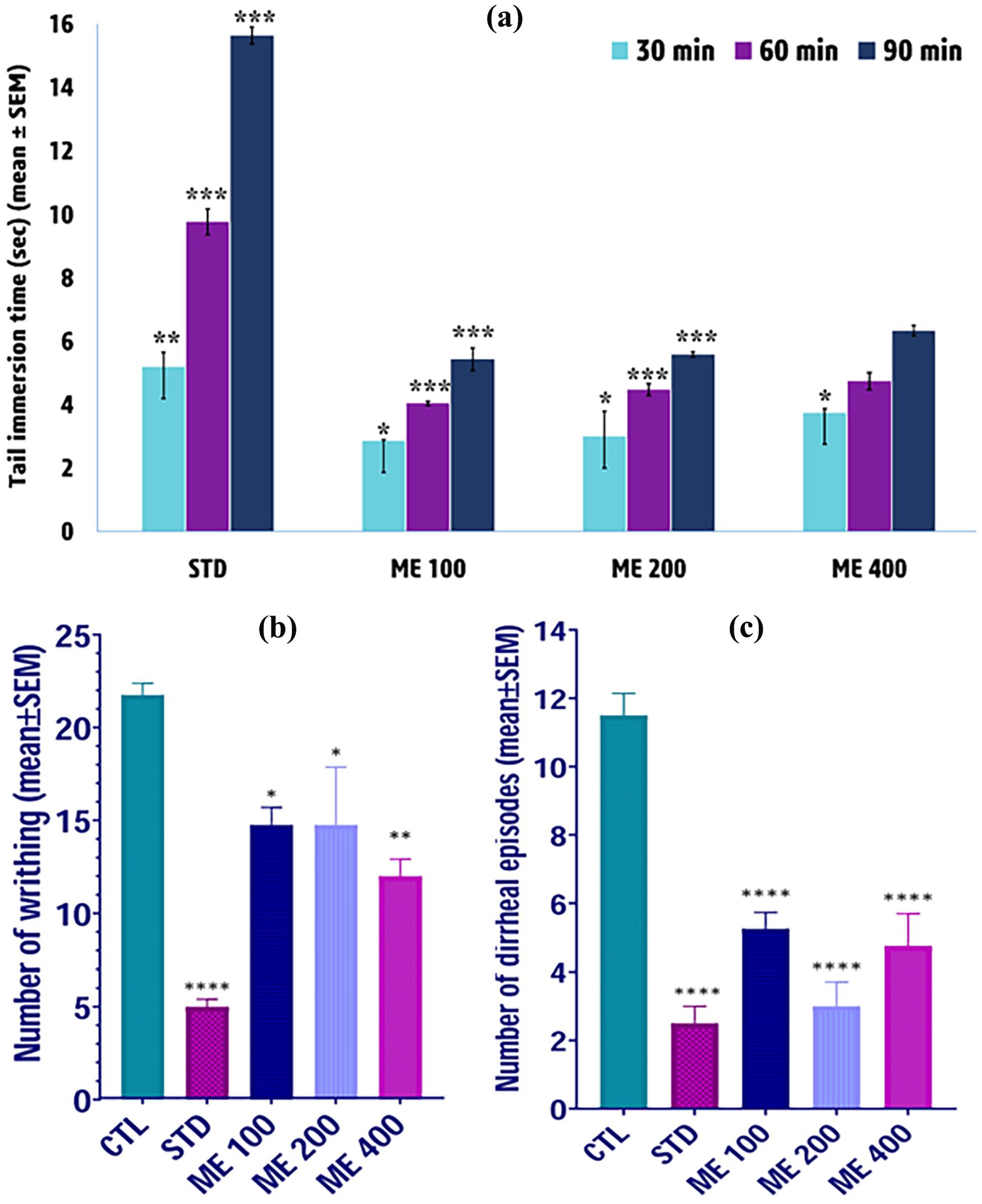
In vivo pharmacological actions of MECR: (a) effects of methanolic crude extracts of C. reticulata peel on the tail immersion time (seconds) of the mice in central analgesic test, (b) effects of the 3 doses of methanolic crude extract of C. reticulata peel on the number of writhing of mice in the peripheral analgesic experiment, (c) effects of the 3 doses of methanolic crude extract of C. reticulata peel on the number of diarrheal episodes of mice after 4 hours of administration of Castor oil in antidiarrheal experiment. The data were expressed as mean ± SEM.
Peripheral analgesic properties
The findings of the peripheral analgesic effect of methanolic crude extracts (100, 200, and 400 mg/kg body weight) in mice are shown in Figure 5b. The MECR demonstrated significant efficacy in inhibiting acetic acid-induced abdominal writhing in mice by reducing the number of writhing of mice. After 30 minutes of pain induction, the 100 mg/kg extract reduced pain by 32.18%, while the 400 mg/kg dosage inhibited writhing by 44.83% in mice, compared to the standard diclofenac sodium (77.0% inhibition; Figure S3).
Anti-diarrheal properties
Castor oil-induced diarrhea lasted up to 4 hours in the control group. The anti-diarrheal evaluation of MECR indicated a very strongly significant (P < .0001) antidiarrheal action at various dosages (100, 200, and 400 mg/kg; Figure 5c). Among the studied groups, ME 200 and ME 400 groups showed nearly 70% inhibition in diarrheal episodes at 4 hours (Figure S4), whereas the positive control had a 73.91% reduction in diarrheal episodes at 4 hours (Figure S4).
Molecular docking analysis
Molecular docking studies were performed using various computer-based methods to evaluate the bioactivities of compounds generated from Citrus reticulata peel extracts and their solvent fractions. Docking scores, as generated by PyRx, are presented in Figure 6. Supplemental Tables S6–S10 provides detailed information on the amino acids interacting with ligand atoms, including the interaction type, distance, and nature. A lower binding affinity value (kcal/mol) indicates stronger binding strength. Optimal docking predictions were identified based on projected binding affinities with a root mean square deviation (RMSD) value of zero. The following sections describe the inhibitory potential of the isolated compounds against specific enzymes and receptors.
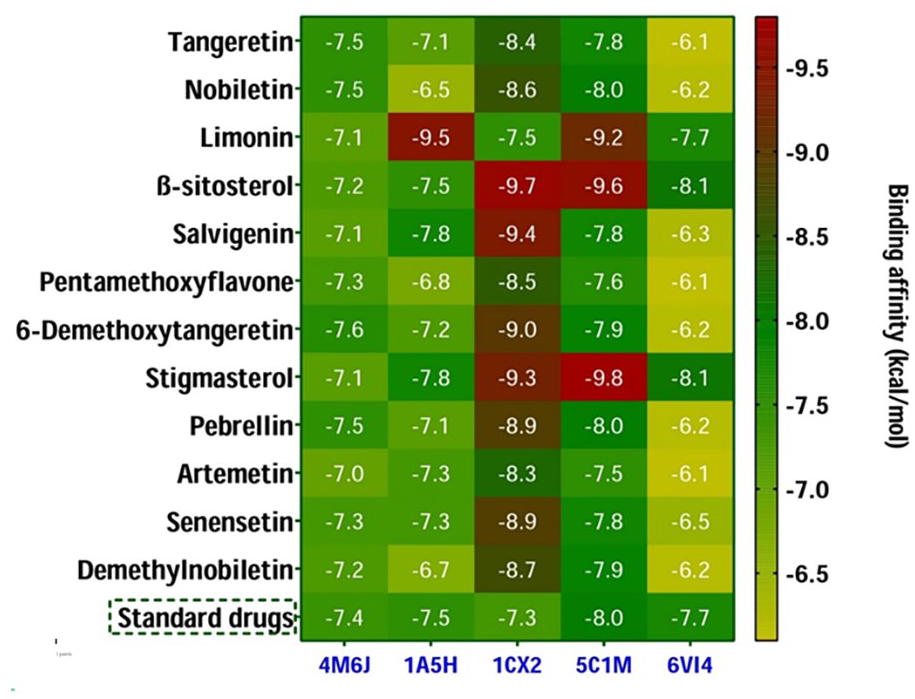
Heat map revealing the binding affinity (kcal/mol) obtained from in silico molecular docking between the isolated compounds and the target receptors. Here, the ciprofloxacin, warfarin, diclofenac, morphine, and loperamide were used as standard drugs while interactions with 4M6J, 1A5H, 1CX2, 4C1M, and 6VI4, respectively.
Inhibition of dihydrofolate reductase (DHFR)
Regarding dihydrofolate reductase (DHFR; 4M6J), all compounds exhibited similar or better binding affinities (−7.0 to −7.6 kcal/mol) when compared to the standard ciprofloxacin (−7.4 kcal/mol). Among them, C-84 had the highest binding affinity (−7.6 kcal/mol), followed by C-A, C-B, and C-87 (each at −7.5 kcal/mol) toward the DHFR protein (Figure 6). The three-dimensional (3D) and two-dimensional (2D) interactions of the best-hit 4 compounds [tangeretin (Compound
Inhibition of tissue plasminogen activator (t-PA)
In the case of molecular docking of tissue plasminogen activator (1A5H), compounds C-B, C-83, and C-91 showed lower binding affinities (−6.5, −6.8, and −6.7 kcal/mol, respectively) compared to the standard warfarin (−7.5 kcal/mol; Figure 6). However, C-C exhibited the highest binding affinity (−9.5 kcal/mol) among all docked compounds, surpassing warfarin, while the other molecules demonstrated binding affinities close to warfarin (−7.1 to 7.8 kcal/mol; Figure 6). The Supplemental Figure S6 shows the three-dimensional (3D) and two-dimensional (2D) interactions of the best-hit 4 compounds [limonin (Compound C), ß-sitosterol (Compound D), salvigenin (Compound 79), and stigmasterol (Compound 86)] while interacting with t-PA revealing anti-thrombotic properties. The interactions noted included hydrophilic (conventional hydrogen bonds and carbon hydrogen bonds) and hydrophobic interactions (alkyl and pi-alkyl; Table S7).
Inhibition of COX-2 enzyme
For cyclooxygenase 2 (1CX2), all docked molecules showed better binding affinities (−9.7 to −7.5 kcal/mol) compared to the positive control diclofenac (−7.3 kcal/mol; Figure 6). Figure 7 exhibited the three-dimensional (3D) and two-dimensional (2D) interactions of the 4 best-hit compounds [ß-sitosterol (Compound D), salvigenin (Compound 79), 6-Demethoxytangeretin (Compound 84), and stigmasterol (Compound 86)] while interacting with cyclooxygenase 2 (COX 2) revealing peripheral analgesic properties. The interactions were mostly hydrophobic, involving alkyl and pi-alkyl bonds (Table S8).

Three-dimensional (3D) and two-dimensional (2D) interactions of the 4 best-hit compounds while interacting with cyclooxygenase 2 (COX 2) revealing peripheral analgesic properties. [Here, (a-h) represent the interactions of ß-sitosterol (Compound D), salvigenin (Compound 79), 6-Demethoxytangeretin (Compound 84), and stigmasterol (Compound 86), respectively, while inhibiting the COX-2 protein.].
Inhibition of mu-opioid receptor
When investigating central analgesic properties via molecular docking analysis with the mu-opioid receptor (5C1M), compounds C, D, and 86 demonstrated more efficient binding affinities (<−9.2 kcal/mol) compared to the standard morphine (−8.0 kcal/mol). The other molecules displayed similar efficiencies (−7.6 to −8.0 kcal/mol) to the positive control (Figure 6). Figure 8 showed the three-dimensional (3D) and two-dimensional (2D) interactions of the 4 best-hit compounds [nobiletin (Compound B), limonin (Compound C), ß-sitosterol (Compound D), and stigmasterol (Compound 86)] while interacting with mu-opioid receptor revealing central analgesic properties. Most ligand-protein interactions were hydrophobic, including pi-alkyl and alkyl bonds, with some hydrogen bonds and 1 electrostatic bond also observed (Table S9).

Three-dimensional (3D) and two-dimensional (2D) interactions of the 4 best-hit compounds while interacting with mu-opioid receptor revealing central analgesic properties. [Here, (a-h) represent the interactions of nobiletin (Compound B), limonin (Compound C), ß-sitosterol (Compound D), and stigmasterol (Compound 86), respectively, while inhibiting the mu-opioid receptor.].
Inhibition of kappa opioid receptor
The compounds ß-sitosterol (C-D) and stigmasterol (C-86) exerted the highest binding affinities (−8.1 kcal/mol) toward the kappa opioid receptor than the standard molecule loperamide (−7.7 kcal/mol) revealing their promising antidiarrheal activities (Figure 6). The Supplemental Figure S7 exhibited the three-dimensional (3D) and two-dimensional (2D) interactions of the 4 best-hit compounds [nobiletin (Compound B), limonin (Compound C), ß-sitosterol (Compound D), and stigmasterol (Compound 86)] while interacting with kappa opioid receptor revealing antidiarrheal properties. The interactions were predominantly hydrophobic, such as pi-alkyl and alkyl bonds (Supplemental Table S10).
ADMET, physicochemical, and drug-likeliness analysis
Poor ADMET profiles are well-known barrier to design effective therapeutic molecule. One of the major challenges in drug development during clinical investigation is addressing these pharmacokinetic characteristics. To overcome these challenges, in silico techniques have been employed to evaluate ADMET properties and predict the drug-likeness of isolated and identified 12 compounds (4 isolated compounds and 8 selected phytocompounds which were detected by GC-MS/MS technique) as potential therapeutic candidates.98-100 The results of these ADMET and drug-likeliness analyses are summarized in Tables 3 and 4.
Summary of the parameters regarding the pharmacokinetic and toxicological properties of the lead identified compounds from the peel of C reticulata.

Summary of the parameters regarding the physicochemical properties and drug-likeliness qualities.

Abbreviations: TPSA, topological polar surface area.
Most of the molecules exhibited similar water solubility (3.89 log mol/L), with the exceptions of C-D, C-86, and C-87, which had solubility values of 6.77, 6.68, and 3.47 log mol/L, respectively (Table 3). Nearly all compounds demonstrated Caco-2 permeability, except for C-87. The phytocompounds also showed comparable human intestinal absorption and skin permeability properties. While C-79, C-87, C-88, and C-91 were not substrates for P-glycoprotein, approximately all other compounds, except for C-87, were able to inhibit P-glycoprotein-I (Table 3). In terms of distribution, most compounds were unbound in a small fraction, except for C-D and C-86, which were fully bounded (Table 3). Compounds C-B and C-87 exhibited higher blood-brain barrier (BBB) permeability compared to other molecules. Additionally, all compounds, except for C-D and C-87 (−1.705 and −1.65, respectively), showed strong central nervous system (CNS) permeability (Table 3). Regarding drug metabolism, all compounds could act as substrates for CYP2D6 and CYP3A4, except for 87 (Table 3). Compounds C, D, and 86 did not show inhibitory action against any types of CYP450 in the docking study, and no compounds were able to inhibit CYP2D6 (Table 3). For renal clearance, most compounds exhibited total clearance (expressed as log mL/kg/min) ranging from 0.422 to 0.789, except for C-C (0.088). All the compounds acted as non-substrates for renal OCT2 (organic cation transporter 2), excluding C-79 (Table 3). The compounds displayed no AMES toxicity, hERG I inhibition, hERG II inhibition, hepatotoxicity, or skin sensitization (Table 3). According to the OECD 423 model, 101 the obtained LD50 values indicated no acute oral toxicity, classifying the substances as class VI (“non-toxic”) under the Oral Toxicity Classification. Furthermore, nearly all compounds adhered to Lipinski’s rule of 5, with a bioavailability score of 0.55 in the drug-likeness analysis. However, compounds D and 86 had an octanol-water partition coefficient log P higher than Lipinski’s recommendation (log P ⩾ 5; Table 4).
Discussion
Fruit plants and vegetables are considered a significant reservoir of bioactive phytochemicals, illustrating various pharmacological properties. Substantial quantities of secondary metabolites are inherent within fruit and vegetable waste or byproducts, thereby the presence of phenolic molecules, dietary fibers, and other biologically active constituents through extraction and isolation techniques can be identified. Scientific research has elucidated the prevalence of phytochemicals and essential nutrients within various components of fruits and vegetables, including peels, seeds, and pulp. 102 The systematic isolation and characterization of these phytochemicals represent a well-recognized approach for identifying bioactive agents derived from fruit waste. Within the phytochemical analysis, several chromatographic methodologies, encompassing column chromatography (CC), thin-layer chromatography (TLC), gas chromatography (GC), and preparative thin-layer chromatography (PTLC), are employed in the purification and refinement of bioactive compounds from botanical extracts. In our study, we conducted a chemical profiling of Citrus reticulata fruit peel utilizing methanol as the solvent, employing gel permeation chromatography, TLC screening, and subsequent PTLC analysis. In our study, NMR approaches was applied to confirm the subsequent isolated phytochemical structures in the investigation. Furthermore, GC-MS technique was employed to comprehensively identify additional phytomolecules from the methanolic extract of C. reticulata peel.
The present research demonstrated the isolation of 4 compounds from C. reticulata, including tangeretin (compound A), nobiletin (compound B), limonin (compound C), and ß-sitosterol (compound D), as depicted in Figure 1. These phytochemicals were also detected from the same species reported in previous studies.103-105 Nobiletin and tangeretin were separated from the peel extract of C. reticulata and reported by Kaushal et al 106 and Mizuno et al, 107 respectively. Additionally, tangeretin and limonin were extracted from the same botanical component and documented by Yaqoob et al. 108 Nevertheless, while ß-sitosterol was previously identified and documented in the stem bark extract, 105 as per our searching experience, this current study represents the first report of its isolation from the C. reticulata peel extract. Furthermore, GC-MS approaches detected and characterized over 90 bioactive compounds, notably, D-limonene (antioxidant, antidiabetic, anticancer), emylcamate (anti-cancer, antioxidant), furaneol (antimicrobial), thymine (antibiotics), ethanamine, N-ethyl-nitroso (anti-cancer), ribitol (anti-microbial), thymol (anti-inflammatory, antiseptic), phytol (cytotoxicity, antioxidant, antimicrobial), ß-elemene (antibacterial), humulene (antifungal, anticancer), trehalose (anti-inflammatory), bis (2-ethylhexyl) phthalate (antimicrobial, cytotoxicity), fenoterol (antimicrobial), salvigenin (anti-inflammatory). In addition, many fatty derivatives such as cis,cis,cis – 7,10,13-hexadecatrienal, cis-11-eicosenamide, 9,12-octadecadienoic acid (Z,Z)-,2,3- dihydroxypropyl ester, and hexadecanoic acid 2-hydroxy1- (hydroxymethyl) ethyl ester were also identified in our phytochemical analysis, which are mainly promising for their antioxidant properties (Table 2).
The assessment of the antibacterial efficacy of the methanolic extract of C. reticulata and its assorted fractions was conducted with the aim of identifying novel candidates for antimicrobial agents. This initiative fueled from the escalating issue of resistance exhibited by numerous strains of bacteria against conventional antibiotics, thereby emphasizing the imperative for alternative therapeutics. Additionally, contemporary antibiotic treatments often entail heightened adverse effects, underscoring the urgency to explore alternative avenues for combating bacterial infections. 109 Microorganisms resistant to antimicrobial agents have been identified as the primary causative agents in an estimated 700 000 fatalities annually worldwide. This alarming figure underscores the urgent need for concerted efforts, including discovering novel antibiotics to address antimicrobial resistance. 110
The present investigation revealed antibacterial efficacy across various extractives of C. reticulata, with notable antimicrobial activity observed specifically within the dichloromethane-soluble fraction. These findings underscore the potential of natural sources such as C. reticulata in pursuing novel antimicrobial agents amid the escalating global challenge of antimicrobial resistance. 111 In addition, the isolated compounds such as tangeretin and nobiletin showed potential antimicrobial activity at several previous studies. 112 Moreover, a previous study of the fruit peel showed that the presence of tangeretin and nobiletin exhibited potent antimicrobial activity. 113 Additionally, the isolated compounds demonstrated notable binding affinities toward the dihydrofolate reductase (DHFR) enzyme, with values ranging from −7.0 to −7.6 kcal/mol. These affinities compare favorably to the standard drug ciprofloxacin (−7.4 kcal/mol). In addition, several compounds such as santolina epoxide, 3-(2-piperidinyl) sulfate, ß-elemene, α-farnesene, dodecanoic acid, megastigmatrienone, 2,3-anhydro-d-mannosan and isovaleric acid, eicosyl ester were identified through the GC-MS analysis which have anti-microbial properties (Table 2). Further investigation is needed to establish the exact mechanism of actions of these phytochemicals from the C. reticulata fruits peel toward microbial infections.
Thrombosis stands as a leading contributor to morbidity and mortality across a diverse range of vascular dysfunctions. 114 By the influence of activated thrombin, fibrinogen undergoes conversion to fibrin, leading to the formation of a thrombus or blood clot. Individuals with occluded veins or arteries receive treatment with fibrinolytic medications, eliminating thrombi by activating tissue plasminogen activator (t-PA) and manifesting a thrombolytic effect. 115 Diminishing platelet aggregation serves as a protective measure against specific conditions, such as atherosclerosis. 116 In this present investigation, the EASF and AQSF of C. reticulata displayed moderate thrombolytic activity (22.61% and 32.57% clot lysis, respectively). Furthermore, a prior study suggests that the identified compound nobiletin could be a promising therapeutic agent for preventing or treating thromboembolic illnesses. 117 Another compound, tangeretin, blocks agonist-induced human platelet activation in a concentration-dependent manner. It hinders agonist-induced integrin αIIbβ3 inside-out and outside-in signaling, intracellular calcium mobilization, and granule secretion. 118 Furthermore, nearly all the selected compounds from C. reticulata for molecular docking exhibited binding affinities toward human tissue plasminogen activator that surpassed those of the standard thrombolytic drug warfarin (−7.5 kcal/mol). The findings from this investigation may contribute to the find out the thrombolytic properties of the fruit peel of C. reticulata.119,120
Substances capable of stabilizing the erythrocyte membrane may also stabilize the lysosomal membrane, thereby exhibiting anti-inflammatory properties by influencing the activity and release of cell mediators due to their structural similarities.25,121 In this context, the AQSF of C. reticulata showcased notable anti-inflammatory efficacy by averting erythrocyte hemolysis in conditions induced by both hypotonic solution and heat. GC-MS analysis revealed the presence of several anti-inflammatory compounds in MECR, including α-farnesene, dl-citrulline, trehalose, ethyl cholate, octadecanamide, salvigenin, vitamin E, sinensetin, and demethylnobiletin. Furthermore, a compound like limonin exhibited potent inhibition of IL-1β-Induced Inflammation, mitigating osteoarthritis inflammation by activating Nrf2. 122 Additionally, all the docking compounds demonstrated the enhanced binding affinities for the cyclooxygenase protein (COX 2), which supports the outcomes of the experimental studies. Plausibly, these molecules might be accountable for the anti-inflammatory activity of C. reticulata. Additional investigations should be conducted to isolate and identify the specific molecules responsible for the membrane-stabilizing properties.
Exploration for pain-alleviating substances is emphasized from natural reservoirs as substitutes for synthetic medications due to their lower adverse effects. 123 The current investigation revealed the analgesic potential of the methanol extract from the peel of C. reticulata using the writhing and tail immersion methods in mice. In our experimental setting, the plant extract mitigated acetic acid-induced pain responses in mice more effectively than heat-induced pain reactions. The findings propose that phytochemicals within the fruit peel extracts of C. reticulata may possess the capability to diminish pain by impeding prostaglandin synthesis.124,125 Some of the isolated and detected compounds of this phytochemical investigation showed high binding affinity than the conventional analgesic medicine morphine and diclofenac. One prior study indicated that the isolated compound β-sitosterol exhibited analgesic effects in hot plate and acetic acid-induced assessments by either inhibiting central opioid receptors or facilitating the release of endogenous opioid peptides, and inhibiting the production of prostaglandins and bradykinins. 126 Furthermore, the GC-MS technique detected certain analgesic compounds such as gamma-Terpinene, Linalool, and Benzofuran, 2,3-dihydro, suggesting the fruit peel of C. reticulata possesses analgesic attributes.
Since time immemorial, medicinal flora has been utilized to address various gastrointestinal (GI) disorders, such as diarrhea. Nonetheless, the safety and effectiveness profiles of the majority of these botanicals have not been scrutinized. As a result, this investigation was done to assess the efficacy of the peel extract of C. reticulata fruits as a potential remedy for diarrhea. Ricinoleic acid, the primary element in castor oil, is known to provoke irritation of the intestinal wall, eliciting peristaltic motion and ultimately leading to diarrhea. 127 The MECR of C. reticulata exhibited significant properties to counteract diarrhea. Even the smaller dosage (100 mg/kg bw) displayed a significant antidiarrheal effect. This exploration illustrated that the anti-diarrheal capabilities of the MECR could be ascribed to the bioactive phytochemicals accountable for the anti-diarrheal effect. 128 Molecular docking displayed that 3 compounds C, D, and 86 had higher binding affinities than the standard drug loperamide. β-Sitosterol, an isolated secondary metabolite from C. reticulata, might assume a significant function in diminishing peristalsis in the GI system, consequently averting GI motility.127,128 While the GC-MS method did not reveal any potent antidiarrheal phytomolecules, according to the literature pharmacological activities records, antioxidant compounds may be instrumental in mitigating intestinal motility. 129 These antioxidant agents could alleviate the impact of castor oil-induced intestinal inflammation while several antioxidant compounds were identified through the GC-MS approach. Furthermore, 2 flavonoid molecules, tangeretin, and nobiletin, possess potent antioxidant attributes.130,131
Limitations and future research
The current study presents certain limitations and gaps. The purified compounds were not pharmacologically investigated. Instead, the study reported their actions computationally to support the pharmacological properties of the MECR and its various solvent fractions. Nevertheless, the study will serve as a foundation for future research focused on the extensive phytochemical isolation of additional polymethoxyflavones and other flavonoids and their pharmacological evaluations. Moreover, it is recommended that the most abundant purified polymethoxyflavones be assessed for their potential effectiveness against chronic disorders, including infections, thrombosis, inflammation, pain, and diarrhea, based on the present evidence. Additionally, the MECR has not been subjected to acute, subacute, or chronic toxicity evaluations in this report, which should be addressed in future studies.
Conclusion
The present study successfully identified and isolated four purified compounds: tangeretin, nobiletin, limonin, and ß-sitosterol, utilizing column chromatography, TLC, and NMR spectroscopic techniques. Additionally, the study detected over 90 phytoconstituents in the fruit peel extract of Citrus reticulata through GC-MS/MS analysis. The study also evaluated the pharmacological properties of the peel extract of C. reticulata, focusing on antibacterial, thrombolytic, anti-inflammatory, analgesic, and antidiarrheal activities. Promising in vitro antimicrobial and anti-inflammatory properties were exhibited by the methanolic fruit peel extract and its various solvent fractions. Statistically significant in vivo peripheral analgesic and antidiarrheal effects were observed with the crude extract at the tested doses (100, 200, and 400 mg/kg bw), where the outcomes were further supported by in silico investigations. In addition, the reported promising compounds demonstrated favorable pharmacokinetics and safety profiles, which are essential for the development of new drugs. The presence of various polymethoxyflavones in this plant underlines the numerous therapeutic benefits of C. reticulata fruit peels. However, additional research is needed to fully understand the precise mechanisms and discover the bioactive compounds that govern the actions of C. reticulata through a combined experimental and computational approach.
Supplemental Material
sj-pdf-1-nmi-10.1177_11786388251327668 – Supplemental material for Phytochemical Isolation and Antimicrobial, Thrombolytic, Anti-inflammatory, Analgesic, and Antidiarrheal Activities from the Shell of Commonly Available Citrus reticulata Blanco: Multifaceted Role of Polymethoxyflavones
Supplemental material, sj-pdf-1-nmi-10.1177_11786388251327668 for Phytochemical Isolation and Antimicrobial, Thrombolytic, Anti-inflammatory, Analgesic, and Antidiarrheal Activities from the Shell of Commonly Available Citrus reticulata Blanco: Multifaceted Role of Polymethoxyflavones by Md. Jamal Hossain, Md. Abdus Samadd, Mst. Nusrat Zahan Urmi, Mst. Farzana Yeasmin Reshmi, Md. Shohel Hossen and Mohammad A. Rashid in Nutrition and Metabolic Insights
Footnotes
Acknowledgements
We extend our gratitude to the Department of Pharmacy at the State University of Bangladesh for the laboratory facilities and logistical support essential for conducting this research.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics
The protocols for animal handling and procedures involved in the animal studies were thoroughly reviewed and approved by the Animal Ethics Committee at the State University of Bangladesh, Dhaka, Bangladesh (approval number: 2022-01-23/SUB/A-ERC/006).
Informed Consent
Not required.
Data Availability Statement
Data included in article/supp. material/referenced in article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.