Abstract
Background:
Neonatal sepsis is a clinical emergency that requires sound evaluation supported by accurate laboratory analysis and timely clinical intervention for its management. This study, therefore, was conducted to identify bacteria causing neonatal sepsis and their susceptibility to the commonly prescribed antibiotic at the Neonatal Intensive Care Unit of a tertiary health care facility in the Northern Region of Ghana.
Methods:
Neonatal biodata were collected from patient folders, after which identification, isolation, and susceptibility of isolated bacteria to prescribed anti-bacterial (Kirby-Bauer disk diffusion method) were carried out on single venipuncture blood samples aseptically drawn from 275 neonates clinically diagnosed with sepsis.
Results:
275 neonates took part in the study, of which 218 (79.3%) presented with early-onset sepsis (EOS) and 57 (20.7%) with late-onset sepsis (LOS). The laboratory results confirmed a septicemia prevalence of 70.3% among neonates clinically diagnosed with sepsis. Preterm delivery (
Conclusions:
Keywords
Significance Statement
Introduction
Sepsis, a critical health care priority with a high incidence of morbidity and mortality, is the presence of bacteria in body fluids such as blood, cerebrospinal fluid, urine, pleural and peritoneal fluids. 1 In neonates, it is known as neonatal sepsis. In a technical consultation, the World Health Organization (WHO) reported that about 2.5 million neonates died of sepsis within the first 28 days after birth, with the majority of such deaths occurring within the initial 24 hours of life. 2 The burden of these neonatal deaths as a result of sepsis in regions of Africa, the Eastern Mediterranean, and Asia accounts for 73.3% of the global neonatal mortality estimated at 6.3 million. 3
Unhygienic delivery places, unsafe delivery and cord care practices, and lack of or inadequate antenatal care are some major causes of neonatal sepsis.4,5 Additionally, home deliveries that are poorly supervised or unsupervised, prematurity with its associated low birth weight and failure to practice exclusive breastfeeding have been identified as some risk factors associated with neonatal sepsis. 6 Institutionally, poor infrastructure, health systems failure, inadequate logistics, and maternal and child welfare intervention coverages among low-income earners and weak economies underpin the increase in infection rates and sepsis-related deaths. 7
Neonatal sepsis is generally classified into 2 broad groups: early-onset (EOS) and late-onset sepsis (LOS) based on the onset time.
8
Early-onset sepsis when the infection occurs within the first 7 days of life, with Group B Streptococcus (GBS),
Neonatal sepsis is a clinical emergency that requires timely intervention. However, prompt and accurate diagnosis from sound clinical evaluation and laboratory examination remains challenging in resource-limited communities. 8 A challenge inadvertently exposes neonates to other nosocomial infections and excessive antimicrobial medications during their hospital stay. This study, therefore, aimed at determining the bacteriological profile and the antibiotic susceptibility patterns of sepsis-causing bacteria isolated from neonates admitted to the Neonatal Intensive Care Unit (NICU) of the Tamale Teaching Hospital, Ghana.
Methodology
Study site
The study was conducted at the Neonatal Intensive Care Unit (NICU) of the Tamale Teaching Hospital (TTH), Northern Region, Ghana. The TTH is the only tertiary referral hospital in northern Ghana, and it also serves as a training facility for other health institutions within its catchment area. The TTH has 8 clinical departments, including the Child and Paediatric, under which the NICU is situated. The NICU, a specialized unit, is a referral facility providing advanced health care to neonates in northern Ghana and its neighbouring regions. 11
Study population
The study population comprised neonates (⩽28 days old) admitted to the NICU with symptoms compatible with septicemia according to the WHO guidelines for screening sepsis and the definition of sepsis by the International Paediatric Sepsis Conference. 12
Sampling technique and sample size determination
A purposive sampling technique was employed to select neonates diagnosed clinically as having sepsis by the attending clinician for this study. Neonates with congenital abnormalities and seropositive for the human immune deficiency virus were excluded from the study.
The minimum sample size for the study was determined using Cochran’s sample size formula 13 :

Where “n” is the sample size, “Z” is the confidence level of 95% (
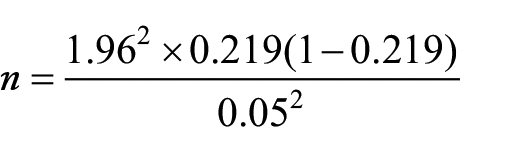

A total of 262 was obtained, of which an additional 13 were added to account for attrition.
Study design
This prospective cross-sectional study was conducted from January to July 2021 to collect neonatal biodata and laboratory investigations of neonatal blood samples. Neonatal biodata, including sex, birth weight, gestational age (weeks), duration of stay (days) at the NICU, and the axillary temperature on admission and place of delivery, were extracted from the maternal folders and admission record books. The birth weight of neonates was categorized as low birth weight (LBW) if the neonate weighed < 2.5 kg or normal birth weight (NBW) if the neonate weighed ⩾ 2.5 kg. 15 Gestational age was defined as preterm if the baby was delivered before 37 completed weeks of gestation and term if the birth occurred ⩾ 37 completed weeks as defined by the American College of Obstetricians and Gynaecologists (ACOG). 16 The axillary temperature of neonates on admission was classified as hypothermia (<36.5°C), normothermia (36.5-37.5°C) and hyperthermia (>37.5°C). 17 Place of delivery of neonates was categorized as “home” if the neonate was delivered in the house, “clinic” if delivered in a primary health centre, “secondary” if delivered at a secondary health facility and “tertiary” if the baby was delivered at a tertiary healthcare facility. Sepsis was classified as “early-onset (EOS)” if it occurred within 7 days after birth or “late-onset (LOS)” if sepsis sets in 7 or more days after birth. 9
Laboratory investigation
Single venepuncture blood samples were aseptically collected according to the protocol by Brooks 18 and dispensed into BacT/ALERT PF aerobic pediatric culture broth with a charcoal neutralizer (BioMérieux, Inc., Durham, USA). These were then incubated in the BacT/ALERT® 3D system (BioMérieux, Inc., Durham, USA) at 37°C for a maximum of 5 days, after which negative samples were discarded. 17 Positive indicated cultures were removed, and 1 ml of the broth was aseptically aspirated and streaked on Blood agar, Chocolate agar and MacConkey agar in plates for subculture. Two drops of the cultured broth were placed onto clean, dry-labeled slides for Gram staining microscopy. 17 The subculture plates were then incubated at 37°C for 18 to 24 hours, as described by Acquah et al 17 . Isolates were identified as enteric Gram-negative bacteria by the conventional standard biochemical methods and the Analytical Profile Index 20E multi-test strips (BioMérieux, Inc., Durham, USA). Gram-positive isolates were identified using the catalase, the tube coagulase enzyme and the bile esculin agar tests. 17 Susceptibility tests of isolated bacteria against commonly prescribed drugs for empiric management of suspected bloodstream infections were determined by the modified Kirby-Bauer disk diffusion method, 19 and the results of the MIC breakpoints were interpreted according to the Clinical and Laboratory Standard Institute (CLSI) criteria. 20 The following antibiotic discs (Oxoid, UK) were used for the study: amikacin (30 μg), amoxicillin (25 μg), amoxicillin/clavulanic acid (20/10 μg), ampicillin (10 μg), ceftriaxone (30 μg), cefuroxime (30 μg), chloramphenicol (30 μg), erythromycin (15 μg), gentamicin (10 μg), and tetracycline (30 μg).
Data analysis
Data collected were entered into Statistical Package for the Social Science (SPSS) version 23 (IBM Corp, USA) for analysis. Continuous variables were presented as means ± standard deviation, and categorical variables were presented as frequencies and percentages. Student t-test and Chi-square were used to test for association between variables as appropriate. A
Ethical issues
Ethical approval for the study was obtained from the Ethical Review Committee (ERC) of the Tamale Teaching Hospital, Tamale, Ghana (TTH/ERC/25/06/19/03) and consent to participate in the study was sought from all mothers/guardians by thumb-printing/signing a consent form.
A written/thumb-printed consent to participate was obtained from all mothers/caregivers of neonates aged ⩾ 18 years. For participants aged < 18 years, a written/thumb-printed consent to participate was obtained from the legally authorized representative/mother of the minor where appropriate.
Results
Out of the 914 neonates admitted to the NICU during the study period, 275 (30.1%) were due to neonatal sepsis. Two hundred forty-six (26.9%) were born preterm, 146 (16.0%) had birth asphyxia, 132 (14.4%) had neonatal jaundice, 43 (4.7%) had meconium aspiration syndrome, 28 (3.1%) had macrosomia, 24 (2.6%) had pneumonia and 20 (2.2%) had seizures.
Of the 275 neonates clinically diagnosed with sepsis and who participated in the study, 153 (55.6%) were males, with 122 (44.6%) being females. The majority, 79.3% (218/275) of these neonates who were admitted and participated in the study presented with early-onset sepsis (EOS), while 20.7% (57) presented with late-onset sepsis (LOS). Over half, 58.7% (128/218) of the neonates admitted with EOS were delivered as normal birth weight neonates, with 66.7% (38/57) of those admitted with LOS delivered as normal birth weight neonates. Low birth weight (LBW) neonates were significantly associated with EOS (
Clinical variables of neonates admitted to the NICU with suspected septicemia stratified by study variables.
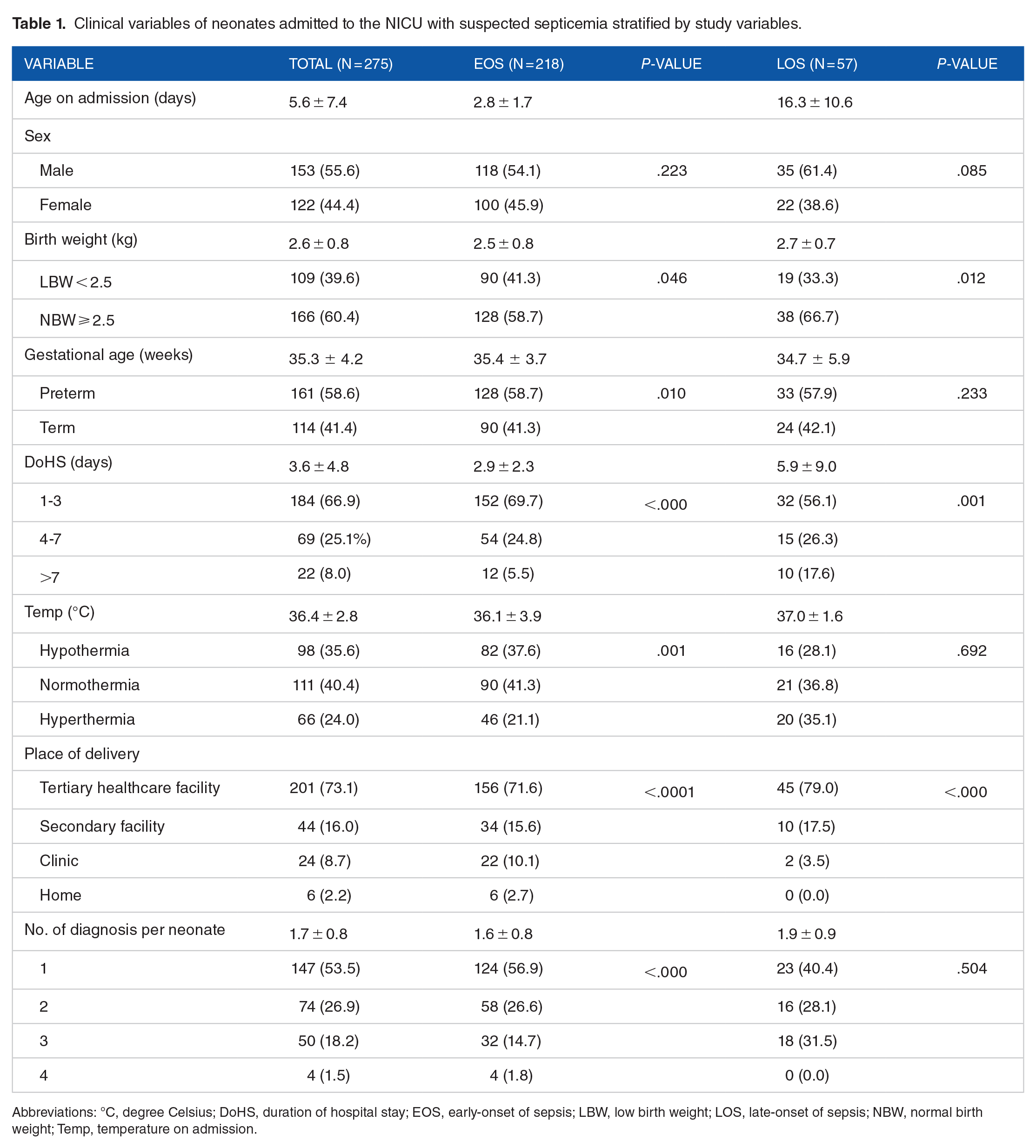
Abbreviations: °C, degree Celsius; DoHS, duration of hospital stay; EOS, early-onset of sepsis; LBW, low birth weight; LOS, late-onset of sepsis; NBW, normal birth weight; Temp, temperature on admission.
One hundred and ninety-five (195/275) out of the 275 blood samples from the study participants and cultured showed bacterial growth, giving a point prevalence of 70.3% bacteriologically confirmed septicemia among the neonates admitted to the NICU of TTH during the study period. The majority, 54.9% (107) of the culture isolates were Gram-positive cocci, 45.1% (88) being enteric Gram-negative bacteria.
Distribution of Gram-positive bacterial isolates by the onset-of sepsis.

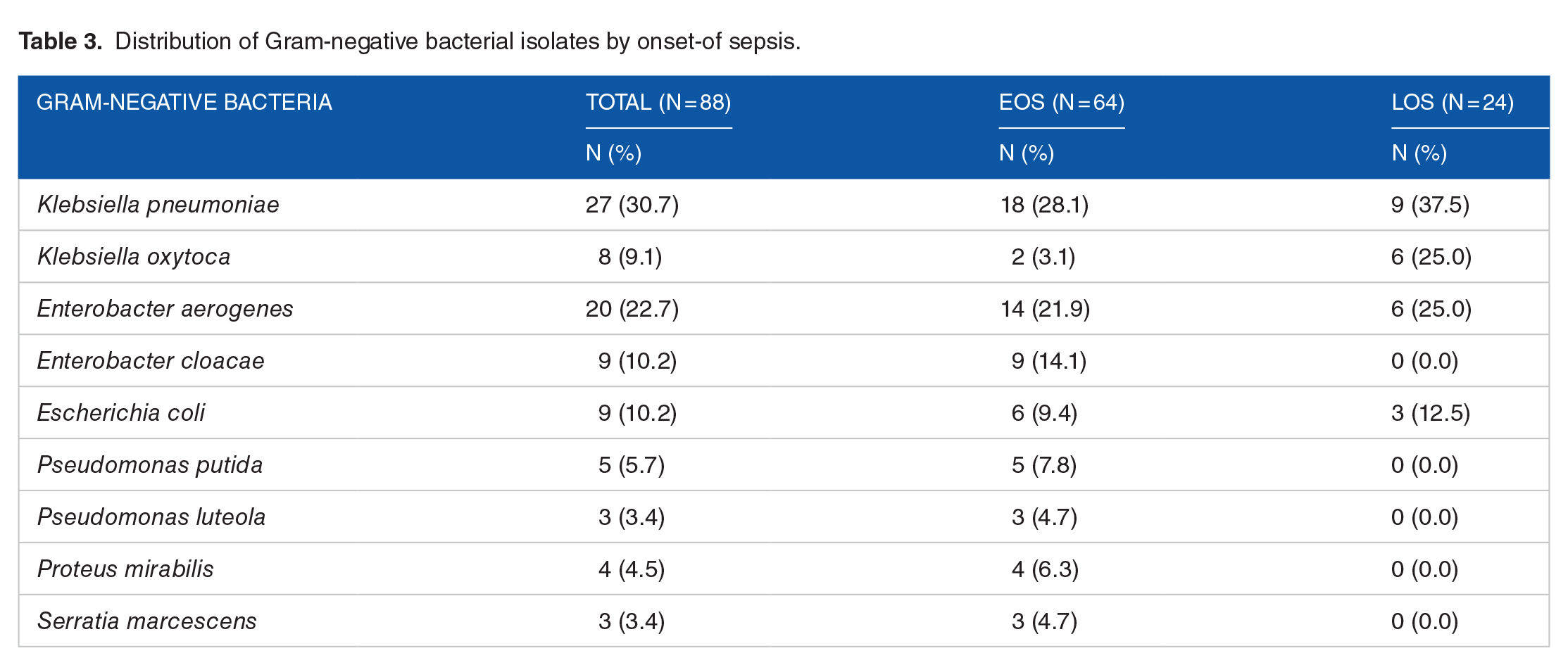
Of the 88 enteric Gram-negative bacteria species isolated, 72.7% (64) were from neonates who presented with early-onset sepsis. The most prevalent Gram-negative bacteria isolated were
Distribution of Gram-negative bacterial isolates by onset-of sepsis.
Antibiotic susceptibility patterns of bacterial isolates
Bacteria isolates exhibited varied susceptibility to the antibiotics tested against them.
Antibiotic susceptibility profile of Gram-positive bacterial isolates.
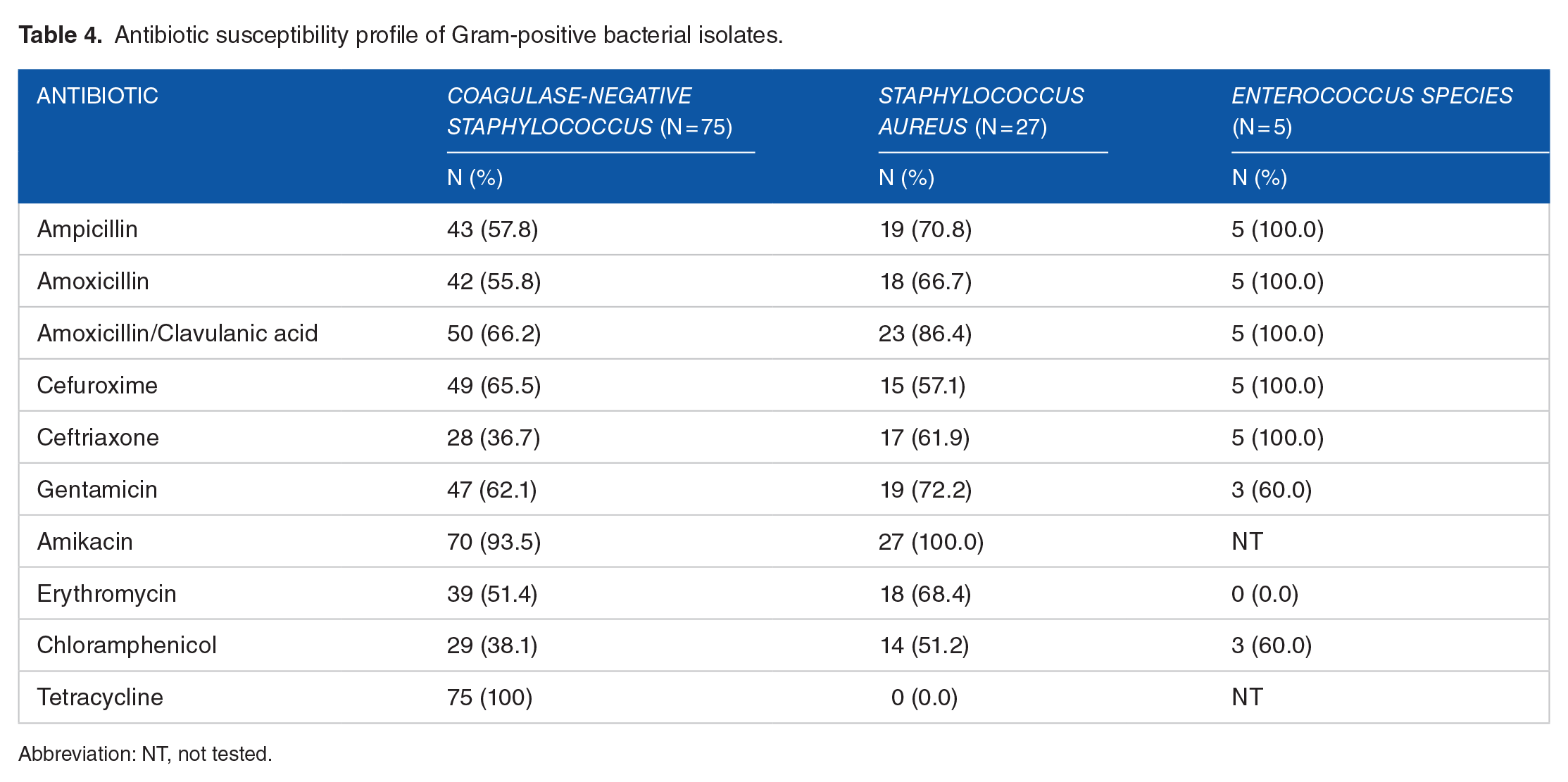
Abbreviation: NT, not tested.
The enteric Gram-negative bacteria showed varying susceptibility to the antimicrobials administered to neonates with suspected bloodstream infection at the Unit.
Antibiotic susceptibility profile of Gram-negative bacterial isolates.
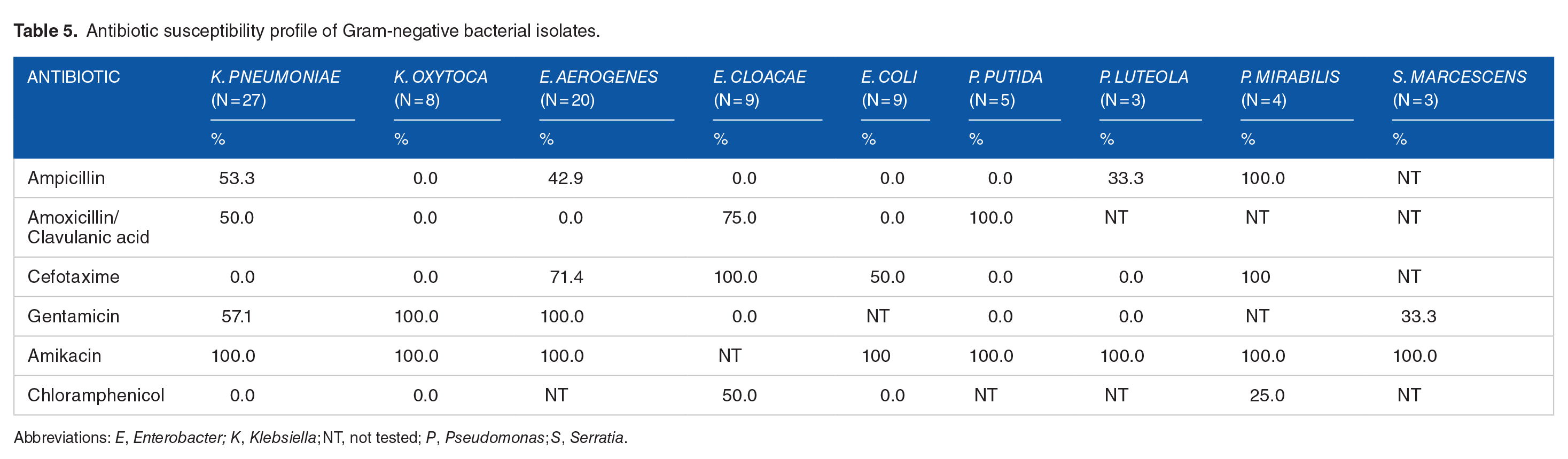
Abbreviations:
Except for
Discussion
Neonatal sepsis is a clinical emergency that requires sound clinical evaluation and laboratory analysis for accurate and timely intervention. However, waiting for blood culture and susceptibility testing results will demand that treatment be delayed for at least 48 hours. Consequently, early empirical antibiotic treatment is initiated immediately after a blood specimen is drawn for laboratory analysis. Appropriate early empirical treatment shortens the disease course, prevents serious complications, and reduces mortality. 6 Generally, the choice of drugs for empirical therapy is based on previous experiences, clinical observation, and knowledge of the current susceptibility profile of common pathogens in the region. In resource-limited countries with a paucity of information on the established drug resistance profile of known pathogens, clinicians use broad-spectrum antibiotics, which may lead to the overuse of these drugs and a subsequent increase in adverse drug effects, costs and antimicrobial resistance. 21 This study was conducted to identify common risk factors, causative bacterial agents and their susceptibility to common antibiotics used at the NICU of the tertiary healthcare facility to guide empirical therapy. Our study is the first prospective study on the bacteriological and antibiotic-sensitive patterns at the Unit, with the only previous study being retrospective. 17 Therefore, this study provides the facility with valuable local reference data on which empirical treatment of sepsis can be based.
The majority of neonates admitted to the NICU being diagnosed with neonatal sepsis in this study was consistent with assertions by the World Health Organization that bloodstream infections are the number one cause of death in neonates globally. 2 Sepsis has been the most common cause and responsible for about 30% to 50% of all neonatal deaths annually, as elucidated by Shobowale et al. 22 Clinical signs of sepsis are usually non-specific, and neonatologists should always have a high index of suspicion for prompt diagnosis. As a result, it is queried as one of the main conditions affecting neonates admitted to intensive care facilities worldwide. 23
The preponderance of male neonates admitted to Neonatal Intensive Care Units reported earlier by Aku et al in a similar health setting was corroborated in this study. 10 The male child in a West African traditional set-up is viewed as a treasure and continuity of the family’s lineage compared to a girl child. 24 The birth of such neonates is greeted with fanfare and overexposure to all sorts of environmental conditions that contribute to their exposure to infectious agents and their subsequent increase in their admission rates at the NICU. In addition, the neonatal male gender is known to be associated with a higher risk of infectious, cardiovascular, pulmonary and neurological morbidities relative to female neonates and, as a result, has a higher likelihood of being admitted to the intensive care facility. 25
The association of gestational age with the incidence of early-onset bloodstream infections noted in this study was congruent with the finding by Wynn. 23 Low gestational age has been strongly linked with the neonatal immune apparatus’s poor development, maturation and responses and a reduction in pathogen-specific antibodies acquired from the mother. 1
Neonates who are delivered outside a health facility have been reported to be at a higher risk of developing sepsis compared with those delivered at a health facility, a fact confirmed in this study and emphasized by Shobowale et al 22 findings. This is not surprising as most extramural deliveries, especially home deliveries, are carried out in a septic environment with various predisposing factors, including unclean and unsafe settings and inadequate human resources, thereby increasing the likelihood of infections. In addition, the innumerable high-risk septic factors during this phase of an infant’s life increase the likelihood of contracting bloodstream infections. 7
Comparatively, the mean duration of stay on admission in this study (3.6 ± 4.8 days) was lower than the earlier mean duration of stay of 7.6 ± 5.7 days on admission reported by Vaniya et al. 26 However, other authors have reported a mean duration of stay as high as 22 days. 27 Neonatal sepsis is considered a clinical emergency, with its management having an implication for the neonate’s lifelong survival. Due to this, much attention is given to these neonates to ensure they are in a better state of health before discharge.
Bulkowstein et al 28 findings in a study comparing early-onset and late-onset sepsis noted that a longer stay at the NICU was associated with EOS compared to those admitted with LOS could not be confirmed in this study. However, this study’s findings indicated a relatively longer duration of stay for LOS (5.9 ± 9.0 days) compared to those with EOS (2.9 ± 2.3 days). Most neonates who presented with EOS were delivered at the study facility, leading to early admission and initiation of empirical treatment. However, it is unclear how the place of delivery affected this early presentation for treatment.
The current study revealed a prevalence rate of 70.9% of blood culture positivity which is comparatively higher than the rate of 25.9% reported by Acquah et al 17 in Ghana, 43.5% by Peterside et al 29 in Nigeria and 44.7% by Shitaye et al 30 in Ethiopia. On the contrary, the prevalence was lower than the 77.9% estimated by Getabelew et al 6 among neonates in some selected NICUs in Ethiopia. Contrary to the current study’s findings, Getabelew et al (5) observed a comparatively high prevalence of confirmed sepsis among neonates reporting early-onset sepsis. Most neonates with culture-positive blood samples were preterm neonates with immature systems. These variations in blood culture positivity prevalence can be attributed to the difference in the sampling techniques and clinical criteria for determining neonatal sepsis in the various studies.
Contrary to the current study’s finding, Pokhrel et al
31
reported a preponderance of enteric Gram-negative bacteria isolates among a similar cohort in Nepal. However, the spectrum of Gram-negative bacterial isolates identified in this current study is consistent with findings by Acquah et al
17
in Ghana.
The implication of the CoNS and
Of the aminoglycosides indicated in the first-line treatment for neonatal sepsis, amikacin showed 100.0% sensitivity compared to gentamicin. Gentamicin is less expensive and often the drug of choice, exposing it to frequent use and subsequent development of resistance by some of these enteric Gram-negative bacteria. The complete susceptibility of all the isolated bacteria to amikacin for which its usage in the empirical treatment of sepsis in neonates is strongly recommended is congruent with earlier findings by Jyothi et al 7 and Zamarano et al. 36 Daily single-dose PK-PD parameters of administered amikacin at 15 mg/kg in neonates point to the drug’s efficacy and safety at minimum inhibition corresponding to what was tested in this study.37,38 However, Smits et al 39 suggested the consideration of neonatal covariates of current weight, postnatal age, and perinatal asphyxia in dosing of the drug.
The susceptibility rate of Gram-positive cocci isolates to ampicillin ranged from 57.8% to 100.0%, which was at variance with findings by Aku et al 10 in which all the Gram-positive cocci they isolated and tested against ampicillin were resistant. Lone et al 40 assertion that the Gram-positives were generally more sensitive to ampicillin relative to the Gram-negatives was confirmed in this study.
As noted in this study, the implication of
Earlier findings by Acquah et al
17
showed that
Limitation
This study was a single-centered-based cross-sectional study; therefore, the results obtained do not represent the general population, and with the relatively small sample size, it might either lack the scientific precision or external validity needed to assist widespread changes in practice. Hence, large-scale, multi-center prospective studies are needed to validate our findings. Methicillin-resistant
Conclusions
Low gestational age, low birth weight, and place of delivery were the main risk factors associated with bloodstream infections among neonates. The predominant Gram-positive cocci and enteric Gram-negative bacteria isolates were
Recommendations
The high sensitivity of Amikacin toward both Gram-positive and negative bacteria isolated makes it a strong candidate for consideration in the empirical treatment of neonatal sepsis. To study their sensitivity profile, routine surveillance of common sepsis-causing agents within the NICU should be established.
Footnotes
Acknowledgements
We acknowledge the valuable contribution of the Neonatal Intensive Care Unit staff at Tamale Teaching Hospital.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author’s Contribution
KOA and EKV conceived and designed the study; KOA and SEA drafted the manuscript; SEA, LQ, and AA performed the data analysis; SEA, AA, KBM, and LQ critically reviewed the manuscript; MFS, AS, and EFB collected the data, did the laboratory analysis and contributed to the study design. All authors read and approved the final manuscript.
Abbreviations
CLSI - Clinical and Laboratory Standard Institute
CoNS
EOS - Early-onset sepsis
GBS - Group B Streptococcus
LBW - Low birth weight
LOS - Late-onset sepsis
NICU - Neonatal Intensive Care Unit
NBW - Normal birth weight
Availability of Data
All data generated or analyzed during this study are included in this published article [and its supplementary information files]. However, the dataset and/or analysis for this work are available from the authors on a reasonable request.
Consent for Publication
Not applicable.
Ethics Approval and Consent to Participate.
Ethical approval for the study was obtained from the Ethical Review Committee (ERC) of the Tamale Teaching Hospital, Tamale, Ghana (TTHERC/25/06/19/03). A written/thumb-printed consent to participate was obtained from all mothers/caregivers of neonates aged ⩾18 years. For participants aged <18 years, written/thumb-printed consent to participate was appropriately obtained from the legally authorized representative/mother of the minor.
