Abstract
Diarrheagenic Escherichia coli, Campylobacter, Nontyphoidal Salmonella, and Shigella are common cause of childhood diarrhea in countries like Ethiopia, but data on their sources and coinfection profiles is limited. A cross sectional study was conducted from November 2021 to January 2023 to determine the prevalence, coinfection, and monthly occurrence rates of major diarrheagenic bacteria in diarrheic under five children and asymptomatic contacts at urban and rural settings in Ethiopia. A total of 345 stool samples were collected from; 262 diarrheic children visiting Hiwot Fana Hospital, Kersa, and Adelle Health Centers; and 83 caretakers and siblings through case based contact tracing. Samples were analyzed using standard laboratory procedures and the overall prevalence of enteric pathogens was 26.96%, with the highest isolation rate during the winter and peaks of 73.91% in February. The occurrence of the pathogens in children and tracked contacts was 27.86 and 24.09%, respectively. In our study, 8.53% coinfection and 23.66% single pathogen infection was recorded in diarrheic children. The study also showed 4.51 and 3.88% of diarrhea in children from urban and rural had attributed to bacterial coinfection, respectively. The most prevalent pathogen in diarrheic children was Diarrheagenic E. coli (10.31%), and followed by Campylobacter. On the other hand, Diarrheagenic E. coli was the second dominant bacteria following Shigella in the traced contacts, with prevalence of 8.43% and 9.64%, respectively. Based on the study site, the prevalence of Diarrheagenic E. coli and Nontyphoidal Salmonella was higher in children from urban than those from rural. However, the occurrence of each pathogen had no significant differences (P > .05) between settings. The high pathogens occurrence rate in the current study indicates the need for strong control strategies and better child carrying and treatment of diarrheal diseases at both urban and rural settings. Further studies on possible sources and factors attributing to the occurrence of enteric pathogens in children are also recommended.
Introduction
Diarrhea is usually a symptom of an infection in the intestinal tract which caused by a different biological agents including; bacterial, viral and parasites. It is the principal cause of child mortality and morbidity in the globe and annually, diarrheal disease alone kills about 525 000 children under five (UFC).1,2 Among etiological agents of diarrhea, Bacterial Foodborne Pathogens (FBPs) have a high public health concern in low- and middle-income countries (LMICs). Diarrheagenic E. coli (DEC), Shigella, Nontyphoidal Salmonella (NTS), Campylobacter spp. etc. are the most frequently identified enteric organisms causing infectious diarrhea.3-5 On the other hand, coinfection with multiple pathogens may be common among children with diarrhea and exacerbates severity of diarrheal diseases. 6
Infectious diarrhea is usually a contagious disease and causes a massive problem in many developing countries. 7 In resource limited countries like Ethiopia, where poor infrastructures and hygienic practices, people mainly acquire the infection through contaminated food or drinking-water, direct contacts; from human (person-to-person) or animals; as a result, it carries a high mortality, particularly among young children.1,8 Although the Ethiopian government and other stakeholders have implemented several intervention strategies to address childhood death due to diarrhea, Ethiopia is the second country in Africa with the highest number of diarrheal deaths. 9
Study evidence shows the incidence of childhood diarrheal infections in Ethiopia is high and it continues to pose a significant public health challenge across regions in the country 9 and bacteria, particularly Gram-negative enteric bacterial pathogens (GNEBPs) significantly contribute to infectious diarrhea in children.4,5 A study conducted in Hawassa and Debre Berhan indicated bacterial pathogens particularly Campylobacter, Shigella, and Salmonella spp. 10 and E. coli 11 are the main cause of childhood diarrhea accounting about 22.2% among the overall causative pathogens identified. The prevalence may be higher in eastern Ethiopia including Harar town and Kersa district where people have limited understanding of zoonotic disease and hand washings12,13 and practicing open defecation. A very recent data showed, in densely populated Harar town, about 12% of the residents 14 and almost half of the Kersa residents (rural settlements) do not use toilet. 15
On the other hand, enteric microorganisms normally found in all warm-blooded animals 16 and several studies reported variable prevalence of FBPs in diarrheic and non-diarrheic people of all age groups. For instance, a high prevalence of 41.38% Salmonella in all age groups 17 and 48.34% DEC in diarrheic UFC 18 were reported from Ethiopia. Therefore, asymptomatic individuals can be source attributes of enteric pathogens causing infectious diarrhea. However, majority of the studies conducted on diarrheagenic bacteria like; DEC, Salmonella, Shigella, and Campylobacter spp. in Ethiopia have reported only the pathogen prevalence in; diarrheic UFC11,19-23 or other age groups24,25 and in asymptomatic food handlers in catering establishments and street food vendors16,26-31 as a separate entities. They do not focused on tracking of direct human contacts as source attributions for diarrhea diseases. To the level of our knowledge there is also no published data reporting importance of coinfection with multiple bacteria in diarrheic UFC in urban and rural settings of eastern Ethiopia.
On the other hand, to prioritize targeted interventions of UFC diarrhea, stakeholders need to know etiologic specific important diarrheal diseases and the role of asymptomatic direct human contacts as sources of the pathogen(s). Thus, the objective of the study was to determine the prevalence, coinfection and monthly occurrence rates of DEC, NTS, Shigella, and Campylobacter spp. in diarrheic UFC and their asymptomatic direct human contacts at urban and rural settings of Harar town and Kersa district, east Ethiopia.
Material and Methods
Description of the study area
The study was conducted from November 2021 to January 2023 in Harar town and Kersa district, East Ethiopia (Figure 1). Harar is a capital of Harari regional state and also the only urban area of the region. It is one of the densely populated towns in Ethiopia. The total population of the region is estimated at 257 000 with 11% of its population aged between 0 and 4 years and 38% aged between 0 and 17 years. 13 Majority of the inhabitants of the town specialized in trading but small-scale livestock farming are also practiced by the residents. Regarding healthcare facilities, there are 7 hospitals, 8 public health centers, 32 health posts, and about 25 clinics in the region. 32 Hiwot Fana Specialized University Hospital (HFSUH) is one of the public hospitals and is a teaching hospital for Haramaya University’s College of Health and Medical Sciences. Harari has an average temperature of 19.4°C and mean monthly rainfall of 126 mm with wettest month being August. The agro-ecological of the region is 90% mid-highland between 1400 and 2200 m above mean sea level (a.m.s.l), while the remaining 10% is lowland, found below approximately 1400-m a.m.s.l. 33
Maps of the study areas.
Kersa is one of the 20 districts of east Hararghe zone, Oromia regional state. The district is principally a rural area with 3 small towns (Kersa, Weter, and Langhe). Kersa district is found between elevation ranges of 1400 and 3200 m a.m.s.l. with a total of 463.75 km2 land coverage. The district has lowland, temperate and highland topography and experiences monthly minimum and maximum temperatures of 12.0°C and 24.2°C, with a monthly average rainfall of 65 mm (range 0-301 mm3). Kersa district has a total population of 172 626 and most of the inhabitants are farmers. The district has 6 health centers and 28 health posts that provide basic health services and no hospital at all.34,35 Natural lakes like Langey, Adelle and Haramaya lakes are found in and around Kersa district and used for multiple activities like; irrigation, fishing, swimming (bathing), washing clothes and also as drinking water for animals.
Study design and study population
A cross sectional study was conducted to determine the prevalence, coinfection, and monthly occurrence rates of major diarrheagenic bacterial FBPs in diarrheic UFC visiting health facilities (out-patient as well as in patient) and their tracked contacts (caregivers and siblings). All UFC living in Harar town and Kersa district are target population of this study, whereas UFC visiting HFSUH, Kersa and Adelle health centers for infectious diarrhea during the study period were considered as study population.
Inclusion and exclusion criteria
Diarrheic children age of 0 to 5 years who were; living in Harar town or Kersa district and visiting or admitted to the specified healthcare facilities provided that caregivers gave consents at least to collect the cases’ stool samples, were included in the study. The UFC’s caregivers and siblings (particularly the youngest individuals who were lived in the same house as the child) were also included; as long as the caregivers agreed to collect stool samples (of own and siblings). On the other hand, diarrheic UFC who were not residing in the targeted study areas for the last 2 weeks at the time of sample collection were excluded. Children with known or documented chronic medical illnesses or any type of enteropathy and those who were on antibiotic treatments during the data collection time were also excluded from the study.
Sample type and sample size determination
Childhood diarrhea caused by diarrheagenic bacteria such as; DEC, Campylobacter, NTS, and Shigella spp. is linked to many sources and transmission routes. As a result, stool samples were collected from diarrheic UFC and individuals who had direct contacts (caretakers and siblings) with the UFC cases. The prevalence of diarrheagenic bacterial pathogens varies with study areas and protocols used. Therefore, a pooled prevalence of bacterial pathogens, 6.4% in UFC, recently reported from Ethiopia
8
was used to calculate the required sample size. The expected prevalence is below 10%, thus the sample size was estimated using a desired precision of 0.03 (half of the prevalence)
36
;
Sampling techniques and procedures
Samples were collected in consultation with pediatricians and clinicians working at UFC OPD rooms, as well as laboratory technicians of HFSUH, Kersa and Adelle health centers. Before the commencement of the study, the professionals were informed about the objectives and inclusion and exclusion criteria of the study. Therefore, diarrheic UFC visiting the hospital and health centers and who fulfilled the above inclusion criteria were further checked by a pediatrician or clinician to identify whether the case was infectious or noninfectious diarrhea. Then caretakers or parents were requested to give their consents, provided that the tentative diagnosis of the case was infectious diarrhea. After getting consent, about 2 to 3 g of semi-formed or 3 mL of watery stool samples were directly collected from diarrheic UFC by caretakers with assistance from laboratory technicians. In the same way, stool samples were also collected from UFC who fulfilled the inclusion criteria but were admitted to the emergency room. About 3 g of stool samples were also collected from the child’s caregiver/mother once informed assent was obtained. Following that, face-to-face interviews were made with caretakers to obtain relevant information like the presence of similar cases or any siblings in the household, household addresses, etc. Finally, the same volume of stool samples were collected from siblings (when available) by visiting households as soon as possible, mainly within 1 to 2 days from the day of stool sample collection from the diarrheic UFC.
In this study, to link possible source attributions (contacts) to their respective UFC cases, specific sample tracking codes were used. Stool samples collected from UFC cases were first registered with a specific case ID and card (laboratory) number at the health care facilities and the tracked samples were then labeled with specific codes that were pre-determined based on the already known respective UFC case ID.
Laboratory analysis
Sample transportation, storage, and preparation
All aseptically collected stool samples were put in separate stomacher bags and kept in a cold box containing ice packs, and transported to the laboratory on the day of sample collection. On arrival at the laboratory, the samples were processed using a Standard Operating Procedure (SOP) created for the FOCAL project upon the existing protocol37,38 and analyzed at the microbiology laboratory of Haramaya University, College of Health, and Medical Sciences (HU_CHMS), Harar, Ethiopia. The samples were gently mixed using vortex and sterile plastic loop; and plated on different agar Medias aerobically (for Salmonella, Shigella spp., and E. coli) and micro-aerobically (for Campylobacter spp.). The remaining stool samples were preserved at −80°C for further meta-genomic analysis (the MG result is not included in this particular manuscript).
Isolation and Identification: for isolation and identification of Salmonella, Shigella spp. and E. coli, samples were plated in triplicate on selective media: Xylose Lysine Deoxycholate (GranuCult™ XLD) and Hektoen Enteric Agar (HE) (Oxoid CM0419B, UK) and MacConkey agar (MAC) and aerobically incubated at 37°C. After overnight incubation the 3 plates were examined for growth and morphological features of each pathogen, characteristic colonies of each pathogen were picked and subjected to gram staining. However, suspected mixed colonies of Salmonella and Shigella spp., and E. coli were sub-cultured on XLD and HE or Brilliant Green agar (BGA) (Oxoid CM0263B, UK), and MAC, respectively before gram staining. From Gram negative rod, E. coli suspected colonies on MAC, a single colony was then transferred to Levine’s eosin-methylene blue (EMB) and colonies showing green metallic sheen morphology on EMB were taken as suspected E. coli isolates.
From the suspected respective pure colonies of Salmonella, Shigella spp., and E. coli, a single colony was then transferred on to Tryptone soya agar (TSA) (Merck, Germany), and followed by biochemical confirmation tests after overnight incubation. Specific biochemical reaction of presumptive colonies for each bacterial pathogen was examined on; Triple sugar iron (TSI) and lysine iron agar (LIA), urease and IMViC (Indole (I), Methyl red (M), Voges proskauer (Vi), and Citrate (C) midia (Merck, Germany), and interpreted according to Andrews et al. 39 Besides, biochemically confirmed E. coli isolates were plated on Sorbitol MacConkey (SMAC) agar (Oxoid CM0813,UK; Lot: 3413831), and only biochemically confirmed, non Sorbitol fermenter (NSF) E. coli isolates were considered as pathogenic E. coli or DEC.
For Campylobacter spp., stool samples were plated on blood free Campylobacter selective (mCCD (Modified charcoal cefoperazone deoxycholate) agar (base) (Merck, Germany; Cat number: 1000700500) containing selective supplement (antibiotics) comprising of amphotericin B and Cefoperazone (Merck, Germany; Cat number: 1000710010) and incubated for 48 hours at 42°C in anaerobic jar 2.5 L (AG0025A, Oxiod; Cat number: 10219202) under a microaerophilic condition provided by Gas generating sachets containing 5% O2, 10% CO2, and 85% N2 (Campy-Gen 2.5 L; SSI Diagnostica, Japan; Cat number: 19438). After the incubation period, suspected colonies were subjected to gram staining and characteristic were taken on to Brain heart infusion (BHI) agar and incubated microaerophilically. 40 Finally, presumptive colonies found positive for both catalase and oxidase (Sigma, India; Cat number: 70439) tests were preserved as Campylobacter isolates.
Data quality assurance
Data quality was ensured at various activities of the study, by ensuing standard operating procedure (SOP) prepared for FOCAL project. All medias were prepared according to the manufacturer’s instructions and the test samples were incubated along with respective positive and negative control reference organisms. Quality control testing was also performed with each new lot and shipment of media and the sterility of prepared media was checked whenever media was prepared (most of the time; weekly), by incubating 2 randomly selected un-inoculated media at 37°C and 42°C for 48 hours and absence of growth following 48 hours was taken as sterile media.
Bacterial reference Strains; S. typhimurium ATCC 14028, and Sh. sonnei ATCC 25931 S. flexneri ATCC 12022 were used and compared with test culture results. E. coli ATCC 25922 and P. mirabilis ATCC 14153 were also used, sequentially as positive and negative controls for Indole and LDC (LIA) testing whereas for citrate and urease, K. pneumoniae ATCC 700603 and E. coli were used as positive and negative controls, respectively. P. aeruginosa ATCC 27853 was used in quality control for TSI and checked for no fermentation (no H2S and gas production). For Campylobacter spp. C. jejuni ATCC 33291 and C. coli ATCC 43478, and E. faecalis ATCC 29212 were used as culture positive and negative controls, respectively. The reference strains of P. aeruginosa and S. aureus ATCC 25923 were also used as a positive control for oxidase and catalase testing, respectively while the E. coli and Str. Pyogenes reference strains were used for their correspondence negative controls.
Data analysis and interpretation
The data collected during the study and the results of laboratory analysis were entered into Microsoft Excel, cross checked, coded, and analyzed by statistical methods using SPSS version 20.0 software. Descriptive statistics were used to calculate and interpret the study variables. The prevalence of enteric pathogens was estimated as the total number of positive samples divided by the total number of samples processed in the laboratory multiplied by 100. For the overall prevalence calculations, we considered a sample is positive when it was positive at least for one of the pathogen in question, and a monthly pathogen occurrence rate was estimated by considering the total number of samples tested in the laboratory in a specific month and the number of enteric bacterial isolates obtained from the respective samples in that particular month. Observed differences were considered statistically significant when P value <.05 and Pearson Chi-square (X2) test was used to compute the association between explanatory variables. Moreover, the study focused on isolation of 4 different diarrheagenic bacterial FBPs, and from none (negative) to 3 bacteria genera were recorded per sample. Therefore, in the result interpretation, the word “single infection” and “coinfection” or “cooccurrence” was used whenever; only one and multiple (2 or 3) enteric bacterial pathogens considered in the present study were, respectively, isolated from a single stool sample. Summary results were presented with tables and graphs.
Results
The study was aimed in determining prevalence, coinfection and monthly occurrence rates of major diarrheagenic bacterial FBPs in diarrheic UFC and their traced contacts in rural and urban settings of eastern Ethiopia. In the present study, the overall prevalence of diarrheagenic bacterial pathogens was found to be 26.96% (93/345), with 27.86% and 24.09% occurrence rates in diarrheic UFC and their direct human contacts (carriers), respectively (Table 1).
Overall prevalence of diarrheagenic bacterial pathogens in diarrheic UFC and their traced human contacts at Harar town and Kersa district (N = 345).
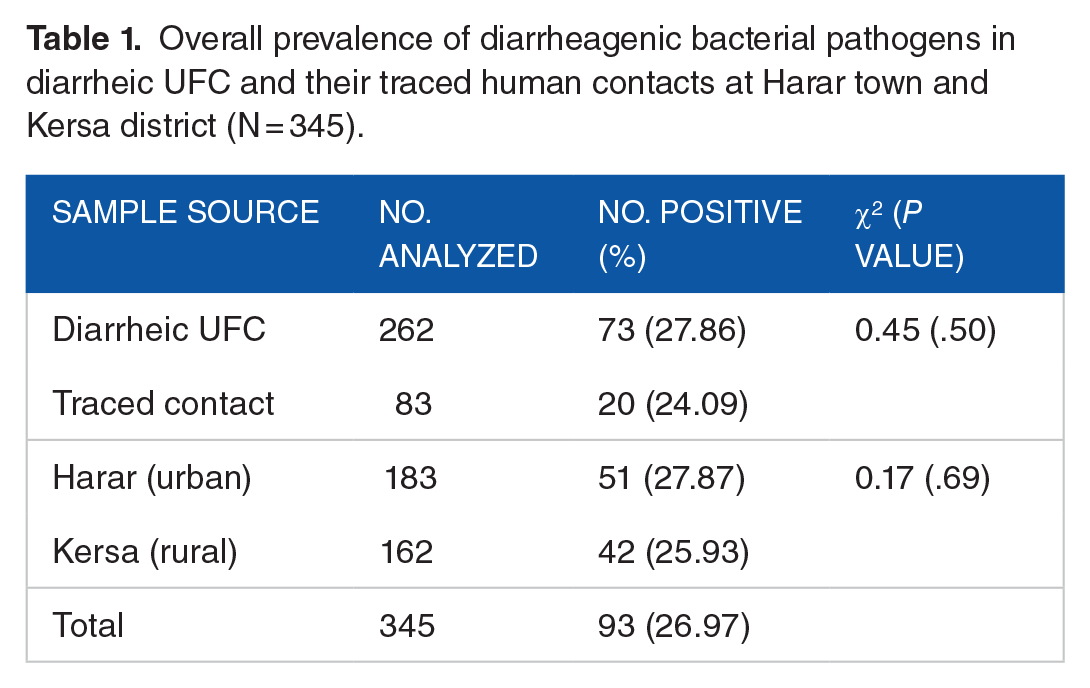
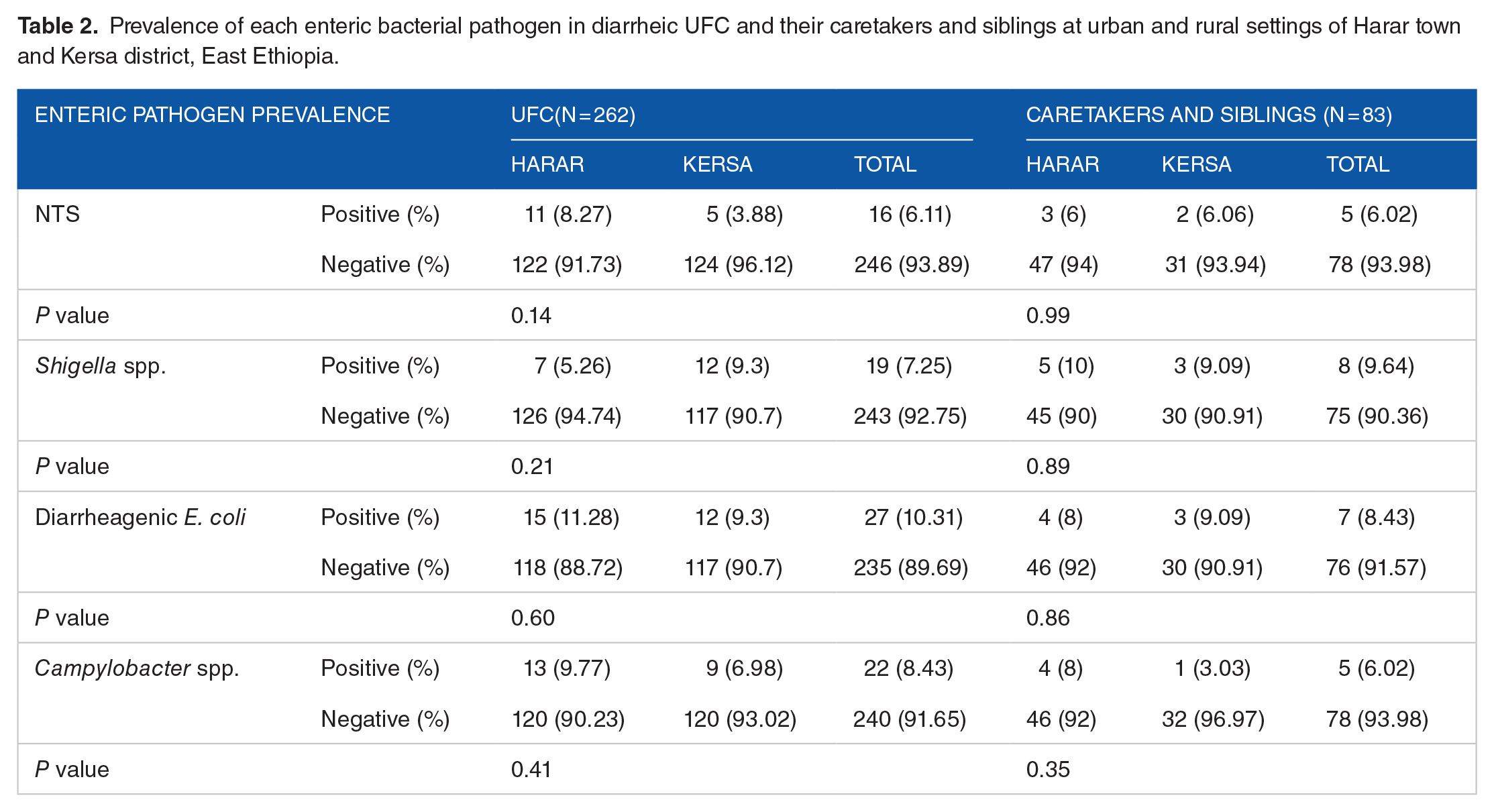
The prevalence of NTS, Shigella, DEC, and Campylobacter spp. in diarrheic UFC was; 6.11%, 7.25%, 10.31%, and 8.4%, respectively, and its correspondence in asymptomatic contacts (carriers) was, sequentially; 6.02%, 9.64%, 8.43%, and 6.02%. Diarrheagenic E. coli was found the most prevalent pathogen in diarrheic UFC while Shigella was the dominant bacteria in their direct human contacts (caretakers and siblings). Unlike the other enteric pathogens, the isolation rate of NTS in Kersa (6.06%) and that of Shigella spp. in Harar (10%) was higher in asymptomatic caretaker or siblings than in their respective diarrheic UFC cases. However, occurrence of each pathogen in both sample sources had no significant differences (P > .05) between the urban and rural settings (Table 2).
Prevalence of each enteric bacterial pathogen in diarrheic UFC and their caretakers and siblings at urban and rural settings of Harar town and Kersa district, East Ethiopia.
In the present study, 84 and 25 diarrheagenic bacterial isolates were correspondingly obtained from 262 diarrheic UFC and 83 traced human contacts recruited in the study. The analysis pattern indicated fluctuations in monthly occurrence rate of the pathogens at the study areas. The highest pathogen isolation rate, 84.62% in diarrheic UFC and 75.0% in their caretakers’ stools, was found in February and August, respectively as shown in Figure 2B.
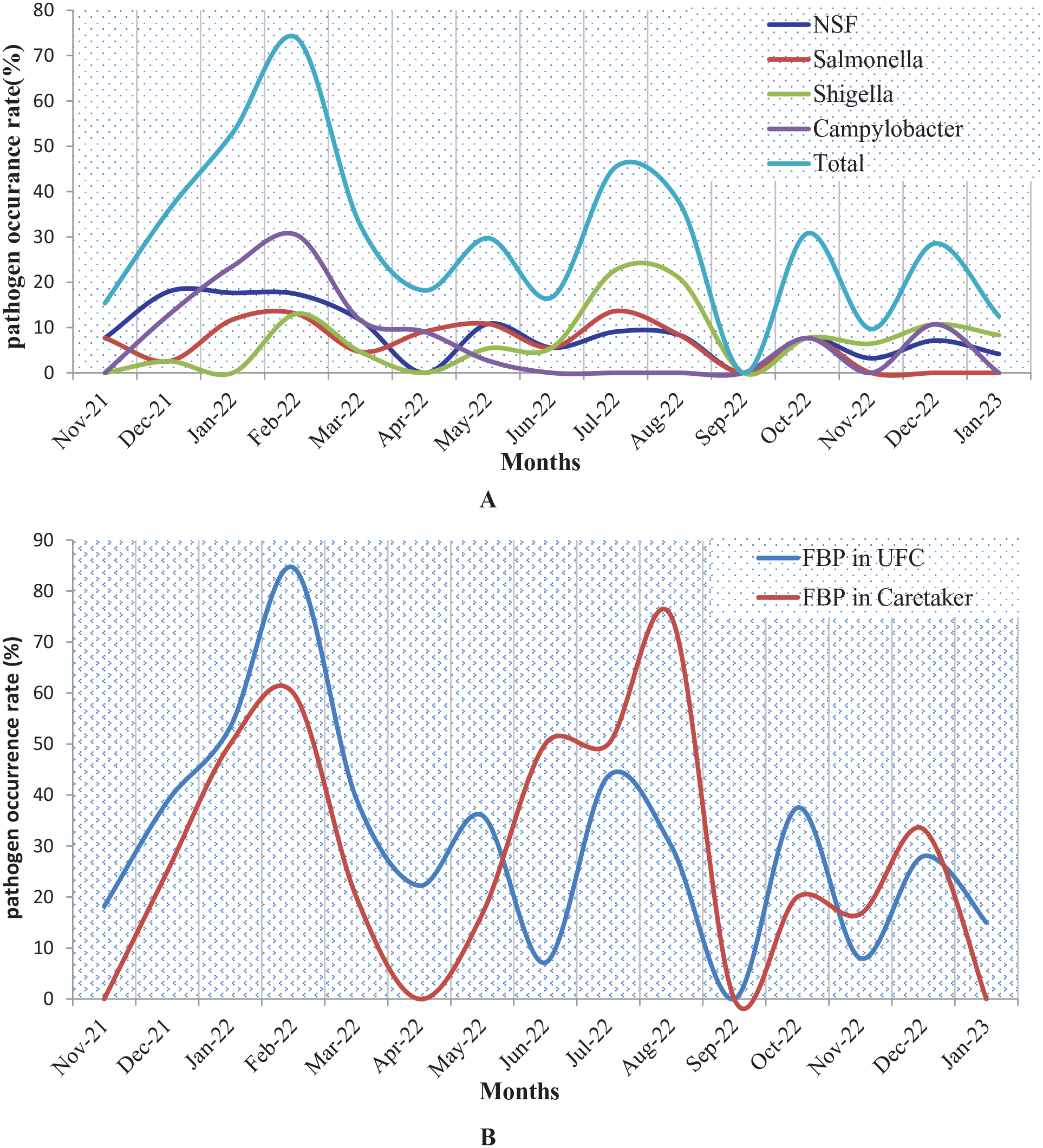
Pathogen specific (A) – and overall FBP (B) – monthly occurrence patterns in diarrheic UFC and their caretaker or siblings at the study areas (November 2021-January 2023).
The 15 months result also revealed, the occurrence rates of diarrheagenic pathogens was linearly increasing during the 2021/22 winter season with peaks of 73.91% in February 2022, and falls during the next, winter 2022/23. Unlike the other pathogens, the isolation rate of Campylobacter spp. was zero (0) between June and September. Exceptionally, only 2 eligible cases of UFC were diagnosed for infectious diarrhea in September 2022 and none of the pathogens was isolated from both cases (Figure 2A).
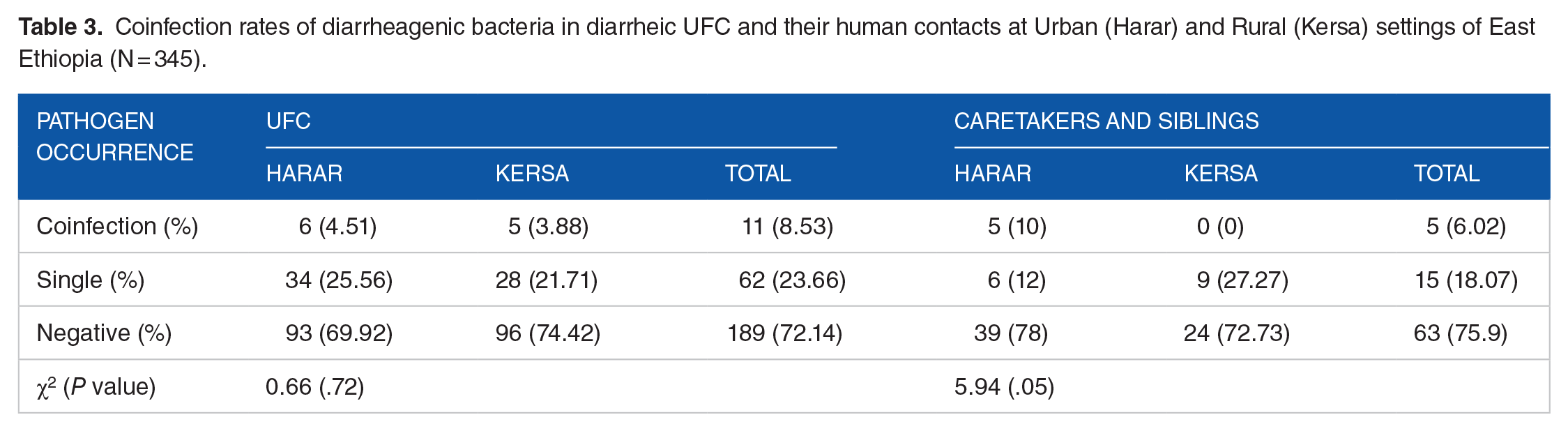
Our study also indicated that 4.51 and 3.88% of diarrhea in UFC in Harar and Kersa had attributed to bacterial coinfection, respectively. On the other hand, in both diarrheic UFC and their caretaker, the exposure to multiple pathogen infection was lower than exposure to a single bacterial infection. Regarding study site specific pathogen prevalence, significantly higher pathogen co-occurrence rate (P = .05) was found in asymptomatic human contact samples collected from Harar town than those from Kersa district. Conversely, the probability of getting coinfection in diarrheic UFC had no statistical differences between Urban and Rural settings of the study area (Table 3).
Coinfection rates of diarrheagenic bacteria in diarrheic UFC and their human contacts at Urban (Harar) and Rural (Kersa) settings of East Ethiopia (N = 345).
Discussions
Caretakers, mainly mothers who have direct contacts with children can be sources of diarrheagenic pathogens like Salmonella, Shigella spp., DEC, Campylobacter spp. etc. which cause diarrheal diseases in their children. In the present study, major bacterial pathogens causing diarrhea in UFC were identified, and their potential human source attribute samples were tracked through case based contact tracing. The overall of prevalence of bacterial FBPs, 26.96%, found in the current study is high and to the level of our knowledge; this is the first study report that estimates specific diarrheagenic pathogens in diarrheic children by linking to their possible human contacts as a source attributes of diarrheal diseases. Moreover, the current overall prevalence finding is much higher than a meta-analysis based pooled prevalence of FBP in human and environmental samples previously reported from Ethiopia. 8 Other studies conducted in Ethiopia also reported only 15.81% 5 and 18.1%, 41 enteric bacterial prevalence in diarrheic UFC.
The isolation rate of the pathogens in diarrheic UFC visiting healthcare facilities and their asymptomatic human contacts was 27.86% and 24.09%, respectively (Table 1). The result is comparable with the findings of study conducted in Manitoba. 42 However, Balew and Kibret 3 reported 13.1%, enteric bacterial prevalence in diarrheic UFC from Bahir Dar, Ethiopia. Attributing to this finding, studies conducted in Hawassa and Debre Berhan reported bacterial pathogens particularly Campylobacter, Shigella, and Salmonella spp. 10 and E. coli 11 as the principal cause of childhood diarrhea accounting about 22.2% among the overall causative pathogens identified. Likewise, Ahs et al. 43 mentioned that diarrheagenic E. coli, Salmonella, and Shigella spp. are important enteric pathogens following rotavirus in LMIC. The higher prevalence could also due to the fact that infectious diarrheic cases had been exposed to at least one pathogen that probably associated with the development of diarrhea, for which the diarrheic UFC was visiting or admitted to healthcare facilities.8,44 In fact the stool samples analyzed in this particular study were collected in collaboration with pediatricians and clinicians after case diagnosis at the local healthcare facilities that is, stool samples were collected only from diarrheic UFC provided that the tentative diagnosis of the cases were infectious diarrhea and the cases were not on antibiotic treatment. It is also factual that bacteria, particularly GNEBPs are the most common cause of infectious diarrhea in children.4,5 Conversely, a much higher prevalence of enteric pathogens was previously reported from Ethiopia, 18 Bangladesh, 45 and Guinea-Bissau. 46 The prevalence variation could be associated with differences in intrinsic and extrinsic risk factors.
On the other hand, the higher prevalence of enteric pathogens in human contact samples as in diarrheic UFC might demonstrates that a higher probability of pathogen transmission in households because of poor hygienic practices. Because all stool samples for the current study were collected from mothers or siblings who had direct contact with diarrheic UFC. Moreover, asymptomatic individuals can equally be sources of pathogens as diarrheic patients because enteric microorganisms found in all warm-blooded animals.16,47
On the other hand, the result showed prevalence of diarrheagenic pathogens had no significant difference between the rural (Kersa district) and urban (Harar town) settings of the study areas (Table 1). The burden of diarrheagenic FBPs such as DEC, Salmonella serovars, Shigella spp., etc. is high in resource-limited countries.5,48 Thus, our study might suggests the need for strong control strategies of the pathogens in urban equally as in rural settings in countries like Ethiopia.
In the present study, prevalence of 3.88% NTS in Kersa, and 11.28% DEC in Harar was the lost and highest prevalence in diarrheic UFC, respectively. Its correspondence for caretakers was 3.03% Campylobacter and 10% Shigella spp. Likewise, inconstant prevalence of diarrheagenic bacterial FBPs have been reported from different parts of Ethiopia in human stool samples ranging from 0%29,49-51 to 41.38% Salmonella in all age groups 17 and 48.34% DEC in diarrheic UFC. 18
The prevalence of 6.11% NTS, 7.25% Shigella, 10.31% DEC, and 8.4% Campylobacter spp. in diarrheic UFC was partially in agreement with the findings reported for Salmonella,52-55 Shigella spp.,10,20,55 DEC, 56 and Campylobacter spp. 57 from similar studies conducted in different regions of Ethiopia. However, the results are lower than the prevalence of 12.57% 19 and 12.17% 20 Salmonella; 13.28%, 58 10.45%, 21 9.09%, 22 and 9.09% 59 Shigella spp.; 24.11%, 22 24.11%, 59 and 23.83% 11 DEC; and 16.74%, 23 12.66%, 10 and 15.44% 60 Campylobacter spp., previously reported from Ethiopia. Nevertheless, the current prevalence is higher than the earlier reports of; Salmonella and Shigella spp.,61,62 DEC, 55 and Campylobacter spp. 63 from similar studies.
Differently, higher prevalence of Shigella spp. was found in diarrheic UFC coming from rural (Kersa district) than in those children from urban area (Harar). Shigella bacteria can be acquired from contaminated food or by drinking or swimming unsafe water. 64 People living in Kersa has an access to swim natural lakes found in and around Kersa district which could enhance the chance of acquiring shigellosis in Kersa than in Harar.
Prevalence variation among individual studies and bacterial genera is expected in such studies. The possible reasons for prevalence variation are differences in exposure to various risk factors among UFC like; poor drinking water, life style including societal culture. For instance, in Hararghe culture, caretaker mainly mother goes everywhere she wants including farming site, marketplaces, community meeting etc. with her child by carrying child at her back and such practices could play a major role in exposing children to infections. Prevalence variation might also be associated with differences in geographic location, season of the study, the study protocol etc.65,66 Non-uniform protocols for sampling and pathogen identification might have also contributed to heterogeneity; in our case, stools were collected only from children with infectious diarrhea. Several epidemiological studies on infectious diseases have also demonstrated the importance of regional differences in disease risk and burden.67-69
Regarding occurrences of diarrheagenic pathogens in people who had direct contacts with UFC cases, uniquely, the overall (9.64%) and at Harar site asymptomatic carriage rate of Shigella spp. (10%) was higher than the isolation rates from their respective diarrheic children cases. Similarly, the asymptomatic NTS infection rate in Kersa (6.06%) was higher than its isolation rates from the diarrheic UFC (Table 2). Hence, the findings depicted that a chance of acquiring NTS and shigellosis from apparently healthy individuals like caretakers was higher in rural and urban settings of the study areas, respectively. Unlike Shigella spp., the overall isolation rate of NTS, DEC and Campylobacter spp. was higher in diarrheic UFC than in their corresponding caretakers and siblings. Supporting the current findings, higher prevalence of enteric pathogens in symptomatic cases than in asymptomatic carriers was previously reported.8,44 The higher rate of notified cases in younger age group could be due to increased severity or susceptibility to other secondary infection and increased likelihood to seek medical care. 65
In contrary to the current Shigella spp. prevalence, in developing countries where substandard hygiene and poor water quality persists children under 5 years of age are most at risk for developing shigellosis.70,71 Likewise, different previous studies reported 0%; DEC,51,72Shigella spp,26,28,72 Salmonella,50,73 Campylobacter spp. 73 in asymptomatic individuals from Ethiopia. Nevertheless, the prevalence of Shigella spp. is consistent with the report of Nasrin et al 69 who reported overall average incidence rate of 10.78% asymptomatic Shigella infection recently in 7 low-resource settings countries including Bangladesh, Brazil, India, Nepal, Peru, South Africa, and Tanzania. Other study also reported, 11% and 15% asymptomatic shigellosis from lowland and highland sites of Papua New Guinea, respectively; though the corresponding Salmonella infection reported from both sites is 0%. 74
On the other hand, a higher incidence of asymptomatic shigellosis, 17.8% was also reported from Tanzania 75 and much higher prevalence, 80.68% reported from Antananarivo, Madagascar in control groups of asymptomatic carrier children. 76 This could be due to the fact that Shigella infection symptoms usually begin 1 to 2 days after infection and a mild case usually clears up on its own within a week in patients whom provided with rest and fluids. 64 It might also attributed to a scientific justification that shigellosis can occurs as asymptomatic infection to mild diarrhea although S. dysenteriae type 1 causes the most severe life-threatening dysentery. 74 Presence of natural lakes at our current study area might also attribute to higher shigella prevalence in asymptomatic individuals because adults have higher chance of swimming lakes and acquiring shigellosis. However, the prevalence of NTS in caretakers (7.5%), in the present study is comparable to the earlier report of study conducted on asymptomatic food handlers in Ethiopia.77,78
Attributing to our study, Getie et al 30 also reported GNEBPs like Shigella spp., enterohemorrhagic, E. coli (EHEC), and S. enterica as important pathogens among asymptomatic food handlers. The higher prevalence of enteric bacterial FBPs in caretakers might also due to the small sample size considered in this particular study. Differences in access to safe drinking water and methodology (study participants, study design) might be the possible reasons for the variation in the prevalence. 65 On the other hand, Abdulahi 27 recently reported only 2.9% Shigella spp. and 2.4% Salmonella prevalence in apparently healthy food handlers. The variation in magnitude of diarrheagenic pathogens among the studies might be attributed to the difference in exposure to different risk factors among households such as; educational level, access to safe drinking water, personal hygiene, and awareness on hygienic food preparation and handling.29,79 Methods used for sample diagnosis, study site and other intrinsic factors could have also contributed to variation in enteric pathogens prevalence.
Coinfection by multiple groups of enteric pathogens is demonstrated to be the norm in diarrheal cases and asymptomatic controls. 80 Likewise, in the present study, 2 or 3 enteric bacterial pathogens (co-infection, co-occurrence) were found in 8.53% diarrheic UFC and 6.02% of their human contacts. The enteric bacterial coinfection rates is higher than the reports of Cho et al, 6 and lower than the finding of Potgieter et al 81 who recently reported, 1.43% and 29% from Gyeongju, Korea, and South Africa, respectively. In agreement to the present study, Andersson et al 82 and Zhang et al 80 also reported enteric bacterial coinfection in diarrheic and nondiarrheic individuals with higher occurrence rates in diarrheal cases. Children in LICs might be exposed simultaneously to multiple enteric pathogens due to poor sanitary conditions in the environment, and poor quality of food and drinking water contributing to the widespread of microorganisms.80,81,83,84
The study result also showed the overall occurrence rate of the diarrheagenic FBPs was linearly increasing from December 2021 to February 2022 (winter) and June to August (summer) with peaks of 73.91% and 45.45%, in February and July, respectively (Figure 2A). Pediatric diarrheal disease is caused by a wide variety of pathogens and various studies have found that some pathogens are seasonal, peaking at different times of the year. 85 Frequently, the seasonal periodicity of diarrheal disease is attributed to weather, which could drive incidence by diverse mechanisms.86,87 In our case, there was a monthly variation in number of cases identified with infectious diarrhea and fluctuation in the monthly occurrence rate of the pathogens could be due to such inconsistences.
In agreement with earlier reports, 88 the isolation rates of NTS and Shigella spp. was higher in the summer. The occurrence rate of pathogens in asymptomatic human contacts was higher during the summer season with peaks of 75.0% recorded in August, the wettest month of Harari region (Figure 2B). Bacterial pathogens are known to peak in summer months 89 and during the summer flooding can contaminate natural lakes found in and around Kersa district which could also enhance the chance of getting pathogens, for instance, shigellosis, as the surrounding community uses the lakes for multiple purposes including swimming.
Nevertheless, the overall occurrence of enteric pathogens as well as their occurrence in diarrheic UFC was found higher during the winter. Similarly, DEC and Campylobacter spp. isolation rate was higher during the winter season (Figure 2A and B). This might be due to water storage during the dry period which increases the risk of water contamination as the same water sources are used for animals and irrigation purposes in many villages found in and around the study areas. Likewise, during the winter season, water is less accessible for personal hygiene, which increases the risk of potential diseases.1,81 Other possible explanation could be due to food shortage during the dry (winter) season. Higher proportion of foodstuffs is shipped from other regions and contamination of food during transportation could contribute to higher prevalence of diarrheagenic FBPs during the winter.
Limitations of the study
Even though it is the first research based study report that estimates specific diarrheagenic FBPs in diarrheic children and their possible human contacts in Ethiopia, we believe that the study has some limitations. Most people at the present study areas were not taking children with mild to moderate infection to health facilities and hence, health facility based study may affect the overall occurrence of diarrheagenic FBPs analysis at the targeted area. In the case of Campylobacter spp., samples were incubated at 42°C to minimize competing enteric organisms though Campylobacter spp. grow best at 37°C to 42°C. Furthermore, our study detected the pathogens using culture and biochemical tests.
Conclusion and Recommendations
Infectious diarrheal diseases caused by FBPs have a high public-health and economic impacts particularly in developing countries where proportion of UFC is greater. In the current study, the overall prevalence of enteric pathogens was 26.96%, with 27.86% and 24.09% occurrence rates in diarrheic children and their human contacts (carriers), respectively. The prevalence of NTS, Shigella, DEC and Campylobacter spp. in diarrheic UFC was; 6.11%, 7.25%, 10.31%, and 8.4%, respectively, and its correspondence in asymptomatic caretakers was, sequentially; 6.02%, 9.64%, 8.43%, and 6.02%. Unlike in the diarrheic children, significantly higher pathogen co-occurrence rate (P = 0.05) was found in asymptomatic contact samples collected from Harar town than those from Kersa district. However, the occurrence of each pathogen had no significant differences between the urban and rural settings; and therefore, the study demonstrates that in countries like Ethiopia, enteric pathogens are equally important in urban and rural settings.
The high prevalence of diarrheagenic pathogens in both diarrheic UFC and their contacts (caretakers) in our study also indicates the need for developing strong disease control strategies and enhancing diagnostic capacity to accurately identify disease causing agents for better case management and treatment of diarrheal diseases. Caretakers should also be neat and wear clean cloths, and wash their hands before carrying their children, and minimize unwanted contacts made with their children. Further studies on the possible sources and factors attributing to the occurrence and co-infection with multiple enteric pathogens in children are also recommended.
Footnotes
Acknowledgements
The authors like to thank FOCAL research project funders, and Haramaya University School of Medical Laboratory for their cooperation during laboratory analysis. We are also great full to pediatricians, clinicians and laboratory technicians of Hiwot Fana hospital, Kersa, and Adelle health centers for their cooperation during sample collection. We would also thank FOCAL research project local data collector teams.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study is part of the FOCAL (Foodborne disease epidemiology, surveillance and control in African LMIC). FOCAL is a multi-partner multicountry research project co-funded by the Bill & Melinda Gates Foundation and the Foreign, Commonwealth & Development Office (FCDO) of the United Kingdom Government [grant agreement investment ID OPP1195617].
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
In general, all authors participated in designing the study methodology, proposal writing, as well training, and supervising data collectors. They also contributed to the final writing of the manuscript by giving comments on the draft version of the manuscript. DB also conducted laboratory analysis, data entry, analysis, and interpretation of the results and drafted the manuscript.
Availability of Data and Materials
The data used in this study was presented in the document and complete data including supplementary file (if any) may be accessible by requesting, the corresponding author.
Consent for Publication
All authors consented for the publication of this study.
Ethics Approval and Consent to Participate
The study protocol involves human subjects and ethical approval was obtained from Institutional Health Research Ethics Review Committee (IHRERC) of Haramaya University; and National Research Ethics Committee, Ethiopia. The study was also granted ethical approval from Ministry of Education (MOSHE) with a letter written on Feb. 11/2020 (Ref Number: MoSHE/RD/14.1/9849/20). The caregivers were informed about the study and asked to give their informed consent to participate in the study. The caregivers were also asked for their willingness to collect stool samples in collaboration with pediatricians or clinicians from UFC visiting HFSUH and Kersa and Adelle health canters for infectious diarrhea cases. They were also asked to declare their voluntary consent to let us visit households and agreed to provide a feces sample of own and their child’s youngest sibling too. Information of the study subjects and that of the parents or child’s caregivers of study subjects were kept confidential.
