Abstract
Microorganisms such as viruses, bacteria, and protozoa are the cause of many waterborne human infections. These microbes are either naturally present in aquatic environments or transferred within them by fecal sources. They remain in these environments for varying lengths of time before contaminating a new host. With the emergence of the COVID-19 pandemic, some studies have reported the presence of viral nucleic acids in stool samples from COVID-19 patients, suggesting the possibility of fecal-oral transmission. The SARS-CoV-2 RNA was thereby detected in the wastewater of symptomatic and asymptomatic people with a risk to human and environmental health. In this work, we try to discuss the different potential sources of this contamination, the forms of persistence in the environment, the techniques of partial elimination, and the possibility of creating new reservoirs.
Background: From Enteric Virus to SARS-CoV-2
Viruses are very small microorganisms, with a size ranging from 0.02 to 0.4 μm in diameter. Their replication mechanism requires a host where they inject their genome, which can be DNA or RNA, in double or single-stranded form. Each group has its own host range and tropism. 1 They can be transmitted by different routes: respiratory, via aerosols, or via the fecal-oral route. Transmission of the virus through sexual contact, with contaminated blood products, contact with infected animals (zoonotic viruses), or through vectors such as mosquitoes or ticks have also been documented. 2
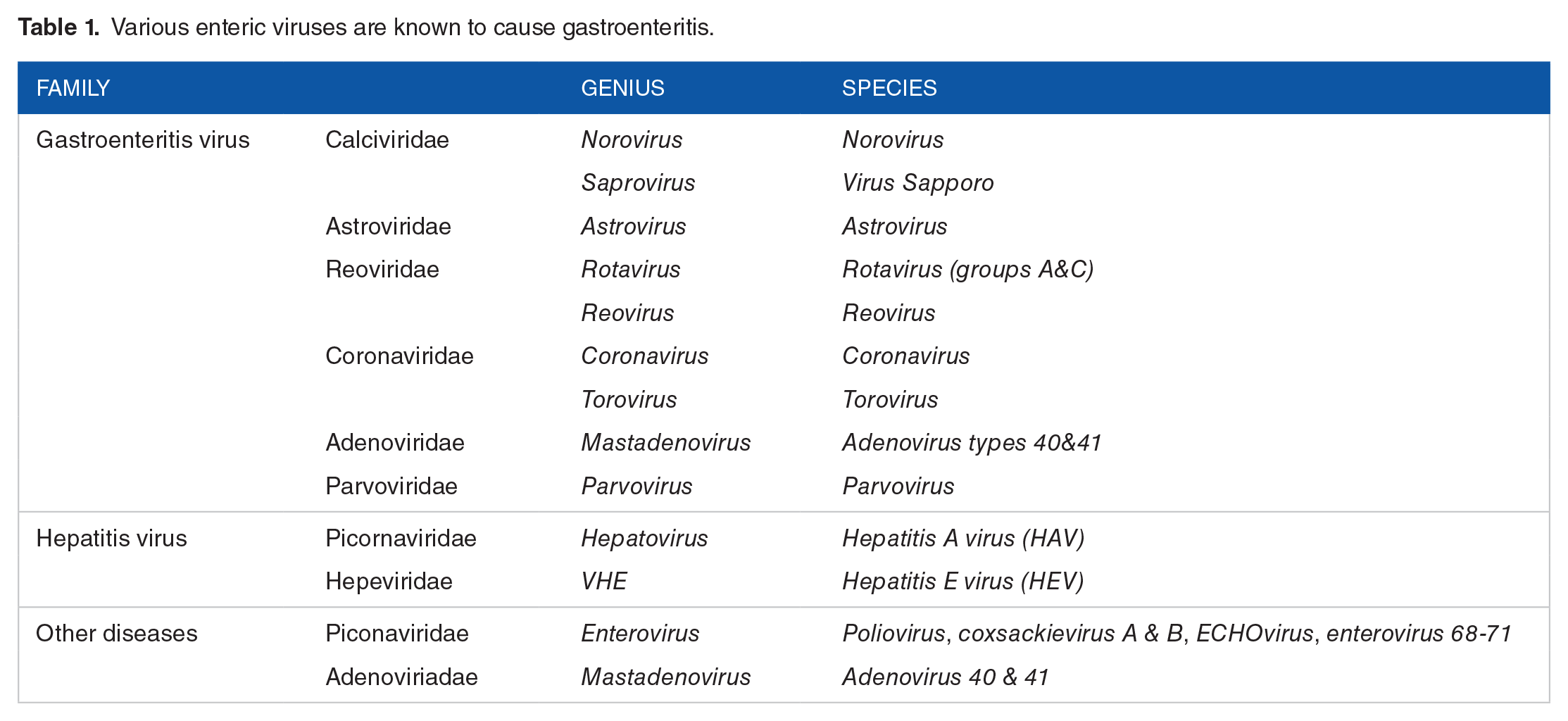
Enteric viruses are known by their direct or indirect fecal-oral mode of transmission, with the ability to reach the intestinal mucosa and multiply in enterocytes. 3 With a diameter varying between 25 and 220 nm, they have a genome consisting of RNA (except for adenoviruses). The majority of human enteric viruses, including rotaviruses, noroviruses, and astroviruses, are characterized by a non-enveloped, naked capsid of a protein nature, 4 which presumably can tolerate gastro-intestinal fluids and enzymes. They present a great diversity and are classified according to the diseases they induce in humans: acute hepatitis, gastroenteritis, or other diseases (Table 1).5-7 Caliciviruses, rotaviruses, adenoviruses, astroviruses, and coronaviruses, are viruses that use the enteric tract as a route of entry to the human, animal, or avian host. These “enteric” viruses occur globally and share similar features. Most are RNA viruses that replicate in the cytoplasm of mature absorptive epithelial cells lining the villi of the small intestine, leading to inflammation and villus atrophy. Vomiting and diarrhea can result in dehydration and death if untreated. 8
Various enteric viruses are known to cause gastroenteritis.
Coronaviruses are enteric viruses, 3 which have been associated with gastroenteritis in humans, 9 sclerosing enterocolitis in neonates, 10 and fatal fulminant gastroenteritis in neonates, 11 but it appears that in almost 90% of cases the infection is asymptomatic. 12 Moreover, they are associated with respiratory, hepatic, and neurological diseases.13,14 Their first infection occurred in the 1960s in the context of benign upper respiratory infections. They have long been considered one of the main agents, along with rhinoviruses, of the common cold. 15 coronaviruses are also single-stranded RNA viruses, with a genome of about 30 kb, which explains their instability in bacterial plasmids. 16 Characterized by a tubular capsid with an envelope bristling with large spicules that gives the virion a crown-like appearance. 17 Their diameter is about 120 nm and their size ranges from 60 to 220 nm.10,16 Enveloped viruses are often more easily inactivated than non-enveloped viruses because the envelope is less resistant to environmental conditions and disinfectants. 18 This envelope consists of the glycoproteins M, E, and S (protein S is a membrane protein that organizes itself as a trimer to form spicules) and hemaglutinin-esterase HE (only for group 2 coronaviruses). The viral capsid consists of protein N (bound to viral RNA) and is helical in shape. 17 Tissue tropism and host spectrum are largely determined by the S protein, which is responsible for the attachment of the virion to the cell receptor and allows membrane fusion.17,19,20
The family Coronaviridae is divided into 2 subfamilies, the Coronavirinae and the Torovirinae. Coronaviruses, belonging to the subfamily Coronavirinae, are subdivided into 3 groups (1-3) on a serological and molecular basis, groups 1 and 2 infecting mammals and group 3 infecting birds. There are 5 human coronaviruses (HCoV): 229E, OC43 (both described since 1960), SARS-CoV (Severe Acute Respiratory Syndrome, described since the 2003 epidemic), NL63 (described in 2004), and HKU1 (described in 2005) (Figure 1).17,22-24
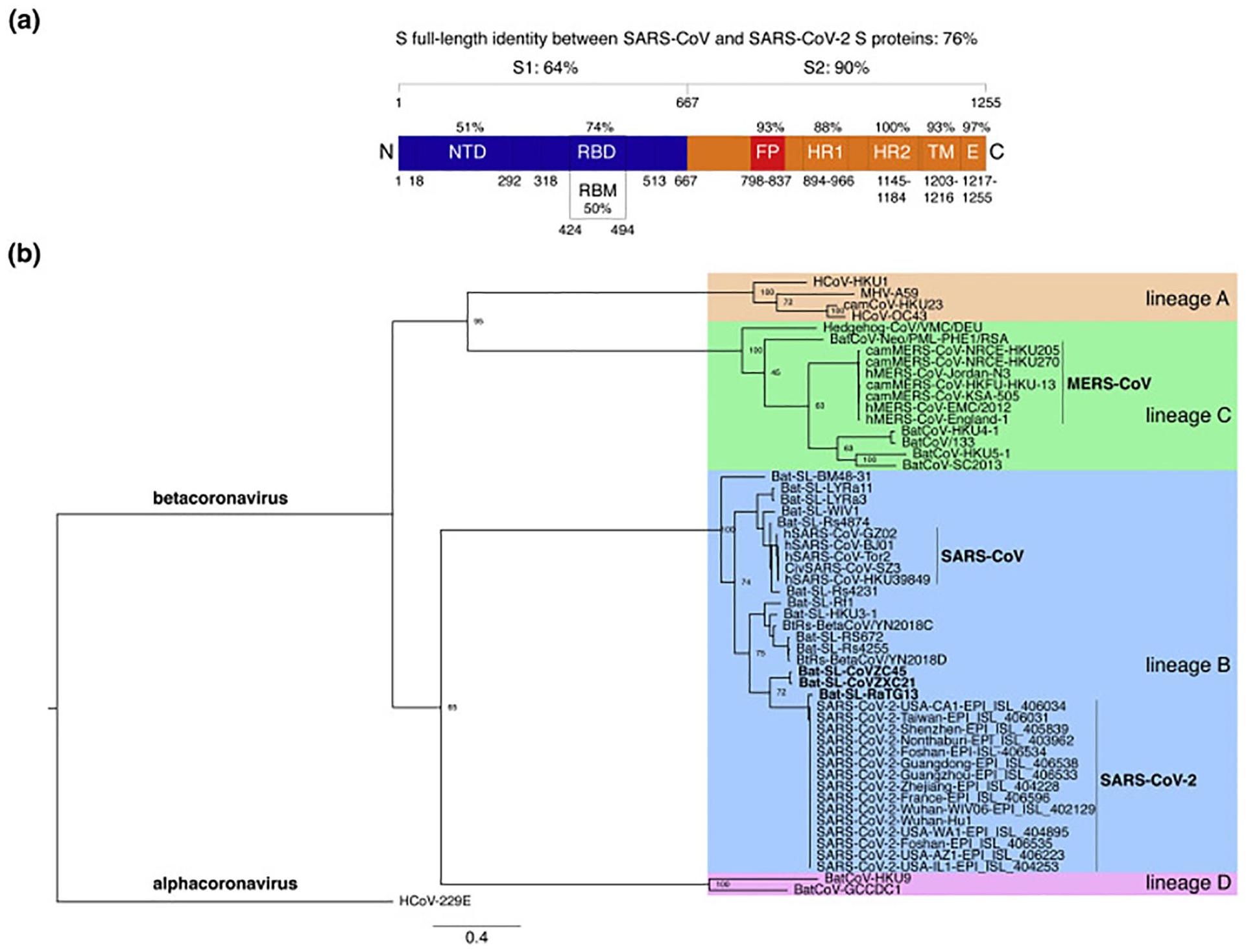
Comparative analyses of the SARS-CoV-2 S protein sequence. (a) Protein sequence identities between SARS-CoV-2 S and SARS-CoV S. (b) The S protein sequence was aligned with representatives of the 4 betacoronavirus lines. 21
Severe Acute Respiratory Syndrome Coronavirus
In order to identify this source, several studies were launched, including those of Zhang et al who had indicated an infection and replication of virions in the gastrointestinal tract,35,36 and that of Danchin et al who have indicated as well that the fecal-oral route is putatively important for the transmission of the virus. 42 The replication of the virus in the intestinal tract suggests an integration of fecal matter into wastewater,40,43 and the presence of the envelope may be related to the difficulty of isolating and detecting infectious virions in feces and sewage.
RNA and/or SARS-CoV Virion in Effluents: Would It Be Disseminated in the Same Way as Enteric Viruses?
Enteric viruses are rejected, and dispersed in large amounts via the fecal-oral route through human excretions, including feces (103-1010 viral particles/fecal gram) into the environment. 44 They enter the gastrointestinal tract, survive the acidity of the stomach, and initiate their infectious cycle. Viral particles are then excreted in high doses in the stool (10−7 infectious particles per gram of stool), 45 and are transferred to surface waters and environments via untreated or treated wastewater discharged from several sources. 46 The absence of a lipidic layer makes them highly resistant to physicochemical environmental conditions such as acids, temperature, dryness, pressure, disinfectants, and ultra-violet radiation. They are also able to retain their infectious character after several hours or days in seawater or on inert or biological surfaces (Figure 2).47,48
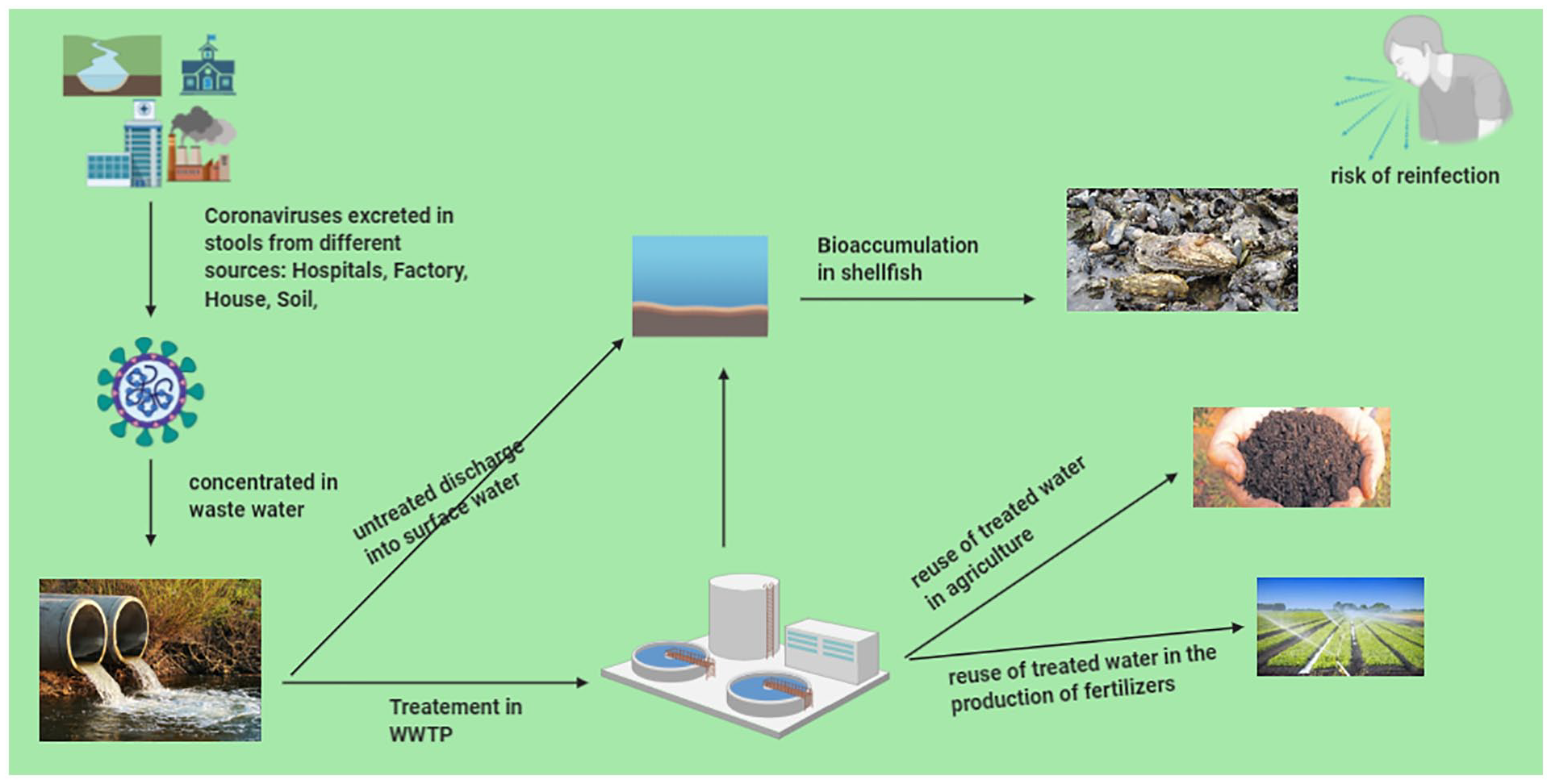
The discharge of enteric viruses into the environment through treated or untreated wastewater involves some forms of the persistence of the virus in the environment, including the formation of aerosols from activated sludge, contamination of agricultural products, bioaccumulation in aquatic resources, and so on.
Billions of liters of untreated sewage enter the coastal ocean every year, containing enteric microbes. 49 As a result of this environmental situation, water quality has deteriorated and is unfit for human consumption in some developed and less developed countries.50,51 From the environment, the fecal-oral transmission of enteric viruses occurs mainly through the consumption of contaminated food, eaten fresh or not having undergone sufficient industrial or domestic treatment, as well as through human-to-human contact. Waterborne transmission of enteric viruses can cause epidemics in countries with poor sanitary conditions.47,52 Danchin et al in their study suggested that natural water bodies contaminated by wastewater could become environmental reservoirs for SARS-CoV-2, this is probably true because the high infectivity of SARS-CoV-2 could lead to the transmission of COVID-19 in such environments. 42 During the current COVID-19 pandemic, people with poor sanitation, consuming food irrigated with wastewater and having direct contact with water resources contaminated by enteric viruses will be a danger to unexposed people. 53 It should be noted that to date no study has been carried out in this context for SARS-CoV-2, which encourages such research in countries with a weak sanitation network. 54
SARS-CoV in wastewater: Persistence or Elimination?
In aquatic environments, the evolution and persistence of native and non-native enteric microbes are governed by biotic and abiotic factors, which may in some cases favor their appearance or inhibit their growth or persistence outside the primary host.55,56 In relation to COVID-19, temperature, organic matter content, and pH have been reported as factors influencing the infectivity of SARS-CoV. 57
In general, fecal-oral transmitted viruses are persistent in the environment and are able to persist in the processes used to inactivate or control pathogens in contaminated food. 58 Most foodborne viruses are non-enveloped and therefore fairly stable outside the host, and are resistant to extreme pH (acid and alkaline), drought, radiation, and so on. 59
In a previous study, a fecal-oral contamination was reported in a residential complex in Hong Kong, where a large group of people with symptoms of diarrhea and oral infection were reported to be infected with SARS-CoV as a result of its spread from a poor sanitation system. It has been suggested that this SARS-CoV infection occurred through the breathing of aerosols created by flushing toilets or faulty plumbing systems. 60 The detection of SARS-CoV-2 RNA in feces and sewage,43,61 makes us think of the detection of his virions as well in wastewater and contaminated water systems (freshwater source case),62,63 as indirect routes of infection. Even if, to date, no SARS-CoV2 infectious viruses have been recovered from untreated or treated wastewater. 64
While to date, SARS-CoV-2 virions have not been detected in the environment, its RNA fragments have been detected in untreated sewage from several countries including Italy 65 ; Spain 66 ; Australia 61 ; Netherlands 43 ; United States of America67,68; France. 69 Moreover, a positive correlation between the concentrations of RNA of SARS-CoV-2 detected in wastewater and the number of clinical cases infected by this virus has been confirmed by researchers in France, 69 America,67,68 and the Netherlands. 43 And, its detection is not only limited to symptomatic but also asymptomatic people,40,70-72 because wastewater contains viruses shed by both types of people, 73 as previously demonstrated for enteric viruses, such as norovirus, hepatitis A virus, and poliovirus.74,75
The SARS-CoV-2 RNAs detected in different countries have often been localized in large cities, which makes us think of large sewage systems that contain gray water from different sources (hospitals, houses, soil leaching, rainwater runoff, and so on), this may amplify the potential for collection and dissemination, increasing the risk of transmission. 76 Without forgetting that these different sources of wastewater, both point and diffuse, contain components such as disinfectants, detergents, soaps, etc., which can minimize the concentration of viral load, 77 so there is also the dilution factor in the receiving environment, this is important because the concentration of viral load in body fluids such as saliva and sputum is potentially high. 40
Discharge of wastewater containing SARS-CoV into the environment: New risks?
In the environment, septic tank sewage leaks, pipe failures, lack of treatment at wastewater treatment plants (WWTPs), or lack of infrastructure in some countries are factors that can result in the direct release of SARS-CoV into receiving water systems (e.g. streams, rivers, ponds, estuaries, lakes, and groundwaters). In addition, treated wastewater such as secondary effluents that are discharged may also transport viruses into the environment. 69 This is true because SARS-CoV-2 RNA has been detected in a Japanese and Italian river, 65 even though there is no real detection of its virions in aquatic environments. 78
During production, irrigation of fruit and vegetable crops with water of poor sanitary quality is an important factor for viral contamination; such an observation has been documented in SARS-CoV. 79 The same is true for the use of organic fertilizers (animal dejecta, sludge not having undergone heat treatment) during traditional farming practices, which are particularly important for agricultural and vegetable production in developing countries,80,81 with a risk of food contamination that can occur throughout the agro-industrial chain, from the production of raw materials to the consumption of marketed products, through the stages of processing and development of finished products.
Treated wastewater has been documented to have SARS-CoV-2 RNA, 69 thus suggesting potential risks associated with wastewater reuse for agriculture. As long as the oral-fecal transmission of SARS-CoV-2 has not been studied so far, it should be noted that such a risk has been documented with viruses of the same family. 82 Infectivity of human bovine 229E CoV was detected on lettuce leaves even after a storage time of 2 days at 4°C.82,83 In addition, washing the products does not eliminate virions. 83
Viral inactivation is highly variable and depending on the type of virus, the type of treatment, the type of matrix, the concentration of viral load, and the inactivation parameter. It is therefore not easy to determine the most resistant virus for a particular treatment in a given matrix, and there is no single treatment regime applicable to all viruses in all matrices. 84 Temperature is widely known as the main factor determining virus inactivation in the environment and is also widely applied in the food industry. 57 The inactivation of infectivity of CoV-SARS has been studied during low temperatures, for example, 14 days at 4°C and 2 days at 25°C in wastewater. 85 This allows us to think about cold and temperate seasons where the environmental survival of SARS-CoV-2 could increase, which is interesting because according to recent studies, total inactivation of SARS-CoV and SARS-CoV-2 was detected at 56°C for 30 and 90 minutes respectively.86,87
Composting is an aerobic process that facilitates and accelerates the transformation of fermentable organic matter by many microorganisms naturally present in the material to be composted. During this process, temperature changes within the compost induce changes in the composition and activity of microbial communities and promote the elimination of pathogenic microorganisms. 88 Numerous studies have focused on the dynamics of pathogenic microbes during the composting process and have shown, the growth of some pathogenic at the beginning of the thermophilic phase, or a resumption of growth of the populations during the cooling phase. 89 In some cases, compost may be re-contaminated during storage, handling, watering, or by animals. 90 The use of re-contaminated compost as fertilizer may cause contamination of plant materials with pathogenic microorganisms. 91
In composts, especially when manipulating windrows (turning), bioaerosols are generated. This transition can be considered as a form of persistence, 92 which represents a significant public health problem for humans both directly (contact) and indirectly (airborne contamination). During the first SARS-CoV outbreak, the aerosol formation was detected as a key mechanism of fecal-oral transmission, making us think of SARS-CoV-2 as the new suspect.93,94 In particular, 1 study suggested the aerosol viability of SARS-CoV-2 up to 16 hours,95,96 and that HCoV 229E remains infectious for 6 days at 25°C and longer periods at 6°C. 97 Even though no aerosol analyses of SARS-CoV-2 in sewage treatment plants have been reported, the formation of aerosols during the treatment process could be a risk for sewage treatment plant operators and facilitate dissemination, especially for sewage treatment plants in densely populated areas.98,99
Dissemination of SARS-CoV-2 in the environment
Groundwater contamination occurs from a variety of sources (natural, agricultural, industrial, residential, etc.), residential wastewater systems can be a source of various types of contaminants, including bacteria, viruses, nitrates, and organic compounds. Wells used for the disposal of domestic wastewater (septic systems, cesspools, drainage wells for exceptional rainfall-runoff, groundwater recharge wells) are particularly concerned about groundwater quality if they are located near drinking water wells. 100
The morphological characteristics of viruses confer them high mobility in groundwater, 101 which is possible because of their external peak glycoproteins, which give them interaction with flow paths and fractures. 102 The SARS-CoV-2 known by its size ~100 nm and its long survival time in water and surfaces, 77 indicate that the virus can migrate into the subsoil and contaminate aquifers. However, the presence of envelope, extracellular enzymes in bacteria such as hydrolases and proteases are able to inactivate SARS-CoV-2.103,104
While enveloped viruses in the environment are likely to be eliminated more than non-enveloped viruses,103,104 studies targeting the detection of RNA in sludge had documented the presence of CoV genes in 80% of untreated sewage sludge samples, of which HKU1 CoV was the most dominant. 61 The lack of data in this context for SARS-CoV, 105 calls molecular techniques such as Virome metagenomics 106 and Transcriptomics, 107 studies, because the low concentration of viral load will be an obstacle for its detection. In some countries with partial or non-existent sewerage systems, there are still people who practice open defecation, 108 which can amplify the viral load locally, especially in the presence of rainfall.
In addition to groundwater contamination, the release of wastewater into surface water leads to the bioaccumulation of certain microbes in fisheries resources. 49 Bathing and consumption of raw or undercooked seafood from coastal waters polluted by enteric viruses leads to more than 120 million cases of gastrointestinal (GI) diseases and 50 million cases of respiratory diseases. 109
Bivalve molluscs (oysters, mussels, clams. . .) are one of the fishery resources that filter, through their gills, large quantities of water (100-650 L/hour/kg of mussels), thus accumulating enteric viruses in their digestive gland.110,111 Unlike bacterial contaminants, the adhesion of viruses to the cells of the digestive mucosa involves specific bonds that strongly limit the effectiveness of the depurative periods that precede marketing. 112
In a study conducted by Gabrieli et al, 2007, 137 bivalves were collected for environmental and market monitoring; all samples were analyzed by RT-PCR assay. Bacteriological enumerations meeting European Union criteria for molluscs were performed in 69.5% of all samples, while overall positive values for the presence of enteric viruses were obtained: 25.5%, 18.2%, 8.0%, and 2.1% for rotaviruses, astroviruses, enteroviruses, and noroviruses, respectively. Mussels appeared to be the most contaminated bivalves, with 64.8% of samples positive, 55.7% and 22.7% respectively for clams and oysters, while in bivalves collected for human consumption 50.7% were positive for enteric viruses, compared to 56.4% of samples collected for the classification of growing areas. The overall positive sample was 54.0%. 111
It is true that the COVID-19 pandemic originated from a live animal and seafood market in Wuhan, China. 26 But it should be noted also that SARS-CoV-2 belongs to the family Coronaviridae and the genus Betacoronavirus – which have only been reported to infect mammals.17,22-24 At present, there is no evidence to suggest that SARS-CoV-2 can infect aquatic food animals (e.g. fish, crustaceans, molluscs, amphibians) and therefore these animals do not play an epidemiological role in the spread of COVID-19 to humans. 113
Aquatic food-producing animals and their products, like any other surface, can potentially be contaminated with SARS-CoV-2, particularly when handled by persons infected with the virus. Nevertheless, with proper food handling and sanitation, the likelihood of contamination of aquatic animals or their products with SARS-CoV-2 should be negligible.
In the current context of trade liberalization and globalization, these productions represent a significant risk that must be taken into consideration. 114 An infected individual, whether or not he or she develops clinical symptoms, will excrete enteric viruses in his or her feces for relatively long periods of time (from a few days to several weeks depending on the virus). 115 In 94 norovirus epidemics recently studied in the United States, the involvement of a handler at 1 level of the production chain was demonstrated in 48% of cases. 116
Also, article 14 of the European regulation EC n°178/2002 indicates that “no food shall be placed on the market if it is unsafe” and that “food is considered unsafe if it is: (a) injurious to health; (b) unfit for human consumption.” In its recital 2, Regulation EC No 2073/2005 specifies that “food must not contain micro-organisms or their toxins or metabolites in quantities which present an unacceptable risk to human health.” Enteric viruses are therefore perfectly within the regulatory framework of these texts which are the basis of the Hygiene Package in force since 2006.
SARS-CoV: Host changing and adaptive capacity
Coronaviruses are both highly adaptive and capable of infecting different human tissues. They transfer easily between new host species and adapt to a variety of ecological conditions through the accumulation of point mutations and homologous recombination. 117 The research carried out to identify the animal reservoirs of SARS-CoV has highlighted the strong evolutionary potential of these viruses, their very broad host spectrum, and their high genetic diversity.118-120
It is certainly true that to date there have been no SARS-CoV-2 virions in the environment, 78 and this may be related to the lack of environmental monitoring. But, the phenomena of its bioaccumulation in bivalves and its dispersion in water environments, makes us think about the possible interactions and genetic exchanges between the different viruses present in wastewater. 121
It is true that the rate of mutation in SARS-CoV-2 is not yet clear,122,123 but, the rate of intra-host variants observed and shared between different individuals suggests adaptive evolution of the virus in patients. This high level of diversity could potentially affect the antigenicity, virulence, and infectivity of the virus, making its elimination more difficult during a reinfection. 124
In addition, for the detection of SARS-CoV-2 RNA in wastewater, viruses are considered infectious when they can penetrate the cell membrane and express at least 1 viral gene or replicate their intact genome, as well as if the viral capsid is undamaged and retains its ability to bind to the cell receptor,125,126 therefore, the persistence of “naked” nucleic acid (DNA/RNA) in the environment suggests a possible horizontal gene transfer between non-indigenous and indigenous communities by the transformation. These processes of horizontal gene transfer may participate in the diffusion of genes and virulence.55,127
Conclusion
Zoonotic infections are a separate category for human health but unfortunately, to date, few studies have been published on the use of environmental surveillance to monitor the contamination of water systems by SARS-CoV.
In both developed and less developed countries, wastewater treatment is an incomplete process that leads to the accumulation of enteric microbes in the receiving environment. It is essential to control and safeguard the quality of wastewater containing enteric viruses and their products. The health risks of COVID-19 through waterborne transmission may be greater than originally thought, and wastewater should be further investigated as a potential pathway for COVID-19 transmission.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contribution
All authors contributed conception and design of the study YS wrote different sections of the manuscript. FC and FB contributed to manuscript revision. All authors contributed to manuscript revision, read and approved the submitted version.
