Abstract
Gonorrhea, caused by Neisseria gonorrhoeae, is a common sexually transmitted infection and an urgent public health problem. Humans are the exclusive host, and the genital tract with heterogeneous epithelia is the primary niche of this bacterium, creating unique challenges for understanding its pathogenesis. The cervical tissue explant model that we have developed enabled us to show that the properties of the epithelial cells in the female reproductive tract are the main factors driving gonococcal adaptation. Gonococcal variants that colonize strongly and penetrate poorly, thereby causing asymptomatic infection, survive better in the cervix. Gonococci adapt to different epithelial cell types by varying their surfaces and modulating distinct epithelial cell-cell adhesion complexes through manipulation of host cell signaling. These findings provide critical new insights on the mechanisms by which N. gonorrhoeae adapts to the human mucosal surface and causes asymptomatic infection.
Gonorrhea, caused by the Gram-negative bacterium Neisseria gonorrhoeae, has been a common sexually transmitted infection and a public health issue since medieval times. N. gonorrhoeae exhibits an extraordinary ability to adapt to the human host and has developed multiple ways to evade the human immune system. Extensive efforts for the last few decades have not yet generated a protective vaccine against gonorrhea. This bacterium has developed resistance to every antibiotic 10 to 20 years after it was introduced as a therapeutic. 1 To overcome these challenges, it is critical to obtain a better understanding of the fundamental, mechanisms by which gonococci have successfully coevolved with humans for thousands of years.
Challenges for Understanding Gonococcal Pathogenesis
The major challenges for delineating the pathogenesis of N. gonorrhoeae include its exclusive human host and its constantly changing surface. Its primary niche is the human genital tract. While N. gonorrhoeae infects both men and women, female infections have much more complicated outcomes, arranging from asymptomatic colonization to severe pathology, 2 such as pelvic inflammatory disease, a major cause of infertility. The majority of female infections are asymptomatic, allowing the bacteria to spread silently and the patients to develop complications.
The female reproductive tract (FRT) presents the bacterium a long canal covered with a variety of epithelial cells. How ascending infections progress from the lower tract (the vagina and the ectocervix) to the upper tract (the endocervix, the uterus, and the Fallopian tube) is unknown. The cervix, often plugged with mucus, represents a bottleneck for sexually transmitted bacteria to ascend from the nonsterile lower tract to the sterile upper tract, where they can induce inflammation. 3 Thus, the cervix is the potential location that determines whether the asymptomatic infections transition to symptomatic. At the cellular level, the cervix is also the location where multilayered, nonpolarized squamous epithelial cells of the ectocervix (Figure 1A) and the vagina transition into single-layered, polarized columnar cells of the endocervix (Figure 1B) and the rest of the upper tract. This important epithelial cell transition occurs in a cervical region called the transformation zone (TZ). How N. gonorrhoeae deals with the cervix’s heterogeneous epithelia to establish, maintain, and advance infection was unknown.
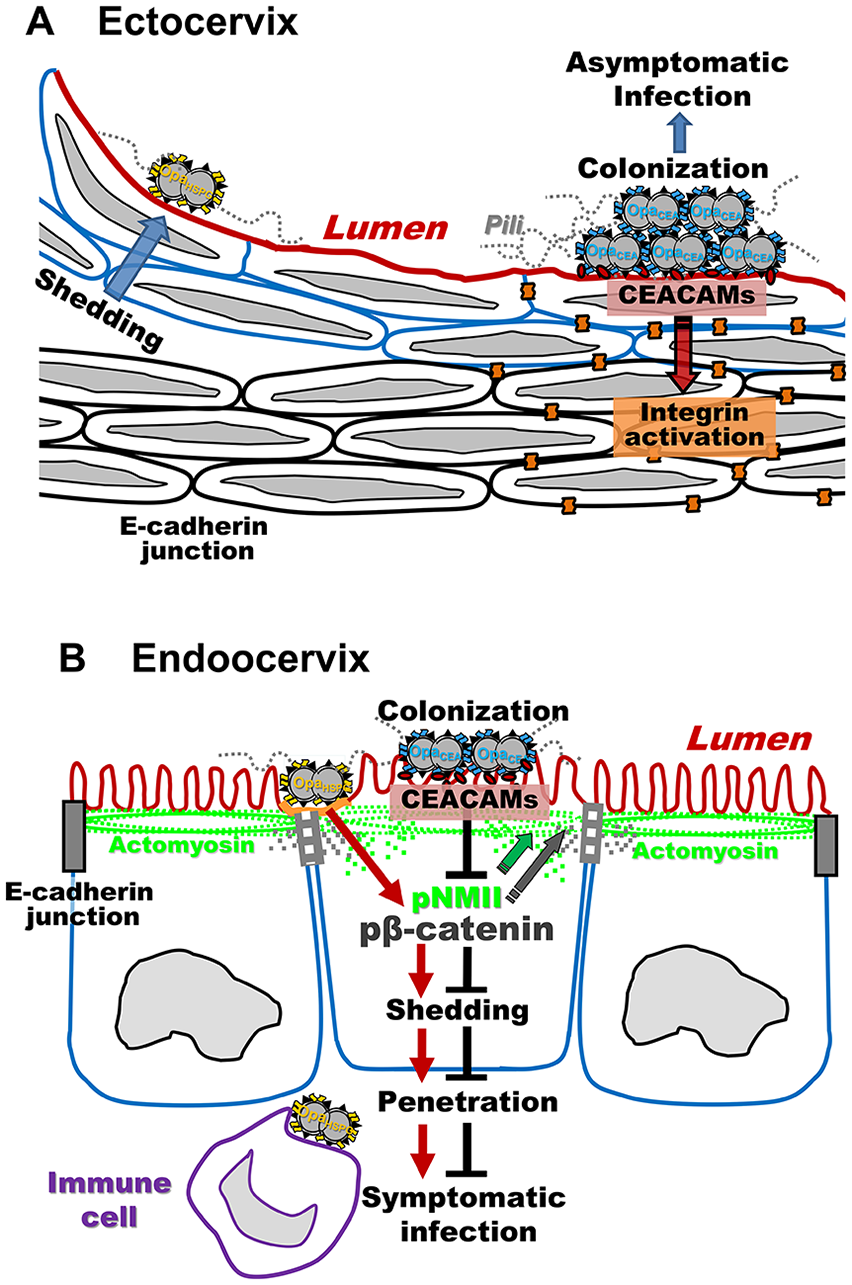
N. gonorrhoeae infects different types of cervical epithelial cells using distinct mechanisms. (A) N. gonorrhoeae colonizes but not penetrates the ectocervix, likely causing localized and asymptomatic infection. OpaCEA proteins, when expressed on the bacterial surface, directly interact with CEACAMs and induce integrin activation, which enhances colonization by inhibiting epithelial cell shedding. (B) N. gonorrhoeae colonizes and penetrates the endocervical epithelium. Penetration exposes subepithelial immune cells to the bacteria, potentially causing symptomatic infection. Interactions of gonococci with columnar epithelial cells activates both NMII and β-catenin, which lead to E-cadherin junction disassembly and epithelial shedding, allowing the bacteria penetrate. OpaCEA-CEACAM interactions inhibit bacteria-induced NMII and β-catenin activation, which restores E-cadherin cell-cell junctions and decreases epithelial cell shedding, consequently reducing gonococcal penetration and enhancing colonization.
Gonococcal Infectivity Changes with the Epithelial Cell Types
Our recent studies addressed this question using human cervical tissue explants. 4 Inspired by the successful Fallopian tube organ culture, 5 we established human cervical tissue explants as an infection model. In this system, the epithelia maintain their in vivo heterogeneous properties after in vitro culture. After inoculation, gonococci were detected on the luminal surface of all cervical regions, while subepithelial bacteria were found only in the TZ and the endocervix. 4 This distribution mimicked the in vivo pattern of gonococcal infection observed with patient biopsies. 6 The ability of the cervical tissue explant model to mimic in vivo gonococcal infections provided a previously unavailable opportunity to examine what happens when the bacteria reach the cervix. This model allowed us to show that N. gonorrhoeae modifies its infection tactics based on the type of cervical epithelial cell it interacts with. 4 When the entire luminal surface of the cervix is equally exposed to piliated and phase-variable gonococci, the bacteria prefer to colonize the ectocervix and the TZ over the endocervix. In contrast, the bacteria do not penetrate the ectocervix despite high colonization levels. Instead, the bacteria prefer to penetrate the TZ and, at much lower levels, the endocervix. 4 These data provide important evidence that epithelial cell properties are critical factors that determine the nature of the gonococcal infection. The selective penetration in the TZ and endocervix can explain both symptomatic gonococcal infection at the upper (columnar epithelial cells) and asymptomatic infection at the lower (squamous and stratified epithelial cells) FRT. Penetration exposes subepithelial immune cells to the bacteria, activating inflammatory responses (Figure 1). However, during natural infection, the vagina and the ectocervix are exposed to the bacteria first. Gonococci have to overcome local flora, mucus, and gravity to colonize the ectocervix and ascend to the TZ and endocervix. Based on this scenario, gonococcal penetration is likely to be even less frequent in vivo than in the tissue explant model. This may explain why gonococci evolved such a strong ability to colonize the ectocervix and why most female infections are localized and asymptomatic.
Our novel finding that N. gonorrhoeae differentially infects epithelial cells with different properties reconciles conflict data accumulated over the years, placing them in an in vivo perspective. Such data were generated in vitro using different model systems, where the properties of the epithelial cells varied with their sources and culture conditions. The problem of conflicting in vitro observations further underscores the importance of establishing infection models that mimic the in vivo properties of infection sites. The unique advantage of the tissue explant model provides opportunities for studying infections and coinfections of other sexually transmitted pathogens. Furthermore, research using tissue explant models can be instrumental for tissue engineering, potentially providing consistent, in vivo-mimicking, and manipulatable infection models for sexually transmitted pathogen research.
Mechanisms by Which N. gonorrhoeae Infect Different Types of Epithelial Cells
While searching for the mechanism by which N. gonorrhoeae changes infectivity according to the type of cervical epithelial cells, we discovered that the bacteria differentially modulate cell-cell junctions. While epithelial cells in the three cervical regions all form E-cadherin-based cell-cell junctions with neighboring cells, gonococci only induce the disassembly of the E-cadherin junction in the TZ and the endocervix (where gonococci can penetrate) but not in the ectocervix (where gonococci cannot penetrate). 4 This finding provides strong supportive evidence for our previous observations showing that gonococci penetrate epithelia by inducing disassembly of cell-cell junctions.7,8 We further revealed two mechanisms underlying gonococci-induced epithelial junction disassembly (Figure 1B). The first mechanism is inducing β-catenin phosphorylation exclusively in the TZ and the endocervix. 4 β-catenin is responsible for linking E-cadherin junctions to cell signaling and the cytoskeleton, and phosphorylation disassociates β-catenin from the junction complex, causing junction disassembly. 9 The second mechanism is inducing Ca2+-dependent activation of nonmuscle myosin II (NMII) and reorganizing activated NMII from epithelial cell-cell junctions to gonococcal adherent sites, resulting in disassembly of the actomyosin ring supporting the junction complex. 10 How gonococci trigger β-catenin and NMII phosphorylation is not completely understood. However, the bacteria can trigger host cell signaling, such as epidermal growth factor receptor (EGFR) activation and other signaling pathways. 11 The failure of gonococci to target β-catenin and NMII in the ectocervix is likely due to a unique absence of E-cadherin and NMII expression in ectocervical epithelial cells, particularly in the luminal layers that are directly contacted by colonizing bacteria. Furthermore, there are additional differences between epithelial cells at different cervical regions, such as expression levels of gonococcal host receptors, signaling regulation, and cytoskeleton organization. All of these can potentially influence gonococcal infection.
Cervical Epithelial Cells Select Gonococcal Variants
N. gonorrhoeae can change their surfaces by phase variation, such as turning on/off the expression of type IV pili and any of 11 opacity-associated proteins (Opa). The host receptors of Opa are best defined among all gonococcal surface molecules. Ten of the 11 Opa isoforms in the MS11 strain bind to carcinoembryonic antigen-related cell adhesion molecules (CEACAMs, OpaCEA), but only 1 binds to heparin sulfate proteoglycan (HSPG). Gonococcal surface variation has been implicated in gonococcal adaptation to various mucosal surfaces of the human genital tract. The roles of gonococcal surface molecules in the infection have long been the focus of the field. 12 However, the results, particularly regarding Opa, have varied depending on the infection model used. The cervical tissue explant model provides an excellent opportunity to clarify this area. Consistent with all previous studies using multiple infection models, pili are essential for gonococcal colonization in all of the cervical regions regardless of the epithelial cell types. 4 This finding suggests that despite their heterogeneity, all cervical epithelial cells express sufficient levels of host receptors for pili that mediate bacterial binding and colonization. In contrast, Opa proteins are not essential. Instead, they differentially regulate the infection in different cervical regions. The expression of an OpaCEA, representing 10 of the 11 Opa proteins of MS11, enhances bacterial colonization in the ectocervix and the endocervix while inhibiting gonococcal penetration in the endocervix. 4 Surprisingly, OpaHSPG expression has no significant impact on the infection in all three cervical regions. A lack of CEACAM surface expression on TZ epithelial cells explains the unresponsiveness of gonococcal infection to OpaCEA expression. 4 However, the reason for the unresponsiveness of the infection to OpaHSPG is unclear, but it could be because the cervical surfaces lack the type of HSPG that OpaHSPG can bind and/or trigger regulatory effects. The similar infective behavior of phase-variable and OpaCEA-expressing gonococcal strains confirms that most Opa proteins expressed on the phase-variable MS11 strain bind CEACAMs.4,7 The high level of CEACAMs on the luminal surface of the ectocervix and the enhancement effect of OpaCEA on gonococcal colonization potentially drive the selection of CEACAM-binding variants as the dominating Opa proteins expressed on the bacteria. OpaCEA proteins enable gonococci to colonize the cervix effectively while limiting their penetration in cervical tissues, which triggers immune responses detrimental to bacterial survival.
Mechanisms by Which Gonococcal Surface Molecules Modulate the Infectivity
Epithelial cell shedding is a host self-protective mechanism, which removes bacteria associated with host cells. Muenzner et al. 13 have shown that OpaCEA enhances gonococcal colonization in the lower FRT of humanized mice and in nonpolarized epithelial cells in culture by inhibiting epithelial shedding through activation of integrin β1. We observed the similar effects of OpaCEA in the ectocervix and the endocervix. However, OpaCEA targets different cell-cell adhesion machineries in these two types of cervical epithelial cells. 4 In the human ectocervix, Opa-CEACAM interaction activates integrin β1 but not β-catenin, which reduces epithelial shedding without affecting E-cadherin-based cell-cell junctions (Figure 1A). In contrast, in the endocervix, Opa-CEACAM interaction inhibits β-catenin phosphorylation, which restores E-cadherin junctions and reduces epithelial cell shedding, consequently inhibiting gonococcal penetration and enhancing colonization (Figure 1B). 4 These findings reveal the impact of different cell-cell adhesion machineries in multilayered ectocervical (integrin-based) and monolayered endocervical epithelial cells (E-cadherin-based) on OpaCEA-mediated regulation of gonococcal infection.
Our results further shed new light on the potential signaling mechanism by which OpaCEA regulates gonococcal infection. The OpaCEA effects are mediated by direct interaction of Opa with CEACAMs and CEACAM downstream signaling, likely the activation of SH2-containing tyrosine phosphatase 1/2 (SHP1/2) by the immunoreceptor tyrosine-based inhibitory motif in the cytoplasmic domain of CEACAM1. 4 Besides SHP1/2, CEACAMs have been shown to interact with multiple signaling and cytoskeleton pathways, 14 and their contributions to gonococcal infection in the human cervix remain to be explored. It will also be of great interest to investigate the role of other CEACAM isoforms coexpressed with CEACAM1 on cervical epithelial cells, such as glycosylphosphatidylinositol-anchored CEACAM5 15 in gonococcal infection and OpaCEA-mediated regulation.
Summary
Our recent studies based on the cervical tissue explant model have revealed new insights into the puzzle of gonococcal asymptomatic and symptomatic infection in the FRT. The results have provided evidence and a mechanistic basis for the notion that the clinical outcomes of gonococcal infection in the FRT are determined by multileveled interplays between randomly changing surface molecules of the bacteria and the heterogeneous mucosal surfaces of the FRT. While the variation of gonococcal surface molecules is random and constant, the mucosal environment of the FRT drives the selection of gonococci that express pili and CEACAM-binding Opa proteins. These surface molecules are essential for gonococci to establish and maintain colonization while inhibiting gonococcal penetration, reducing their chance to encounter subepithelial immune cells.
Footnotes
Acknowledgements
We thank Dr. Norma W Andrews for critically reading and editing the manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Allergy and Infectious Diseases to W.S. and D.C.S (grant nos. RO1 AI123340 and RO1 AI141894). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
W.S. wrote and prepared and Q.Y., L.C.W., and D.C.S. edited the text and the figures.
