Abstract
Human urotensin-II (U-II) is a cyclic 11-amino-acid residue peptide with a wide range of vasoactive properties dependent on the anatomic site and the species studied. The purpose of this study was to determine the localization of human U-II in normal human kidneys and in renal carcinoma. Normal human kidneys (n=11) and eight cases of clear-cell carcinoma were immunostained with a polyclonal antibody to human U-II. In normal human kidneys, U-II was mostly present in the epithelial cells of tubules and ducts, with greater intensity in the distal convoluted tubules. Moderate U-II immunoreactivity was seen in the endothelial cells of renal capillaries, but only focal immunoreactivity was found in the endothelial cells of the glomeruli. No staining was found in the veins. All tumors expressed moderate U-II immunoreactivity in the cancer cells and vasculature. Here we demonstrate abundant expression of U-II in normal human kidneys and renal carcinoma. These findings suggest that the vasoactive and growth-mediator peptide U-II may contribute to the pathophysiology of the human renal system.
Urotensin-II (U-II) was first isolated from an extract of the urophysis of the goby, which led to the discovery of its structure (Pearson et al. 1980). Since then, several forms of U-II precursors have been identified in different species (Douglas and Ohlstein 2000). The human form of U-II is an 11-amino-acid residue peptide with potent vasoactive properties that are dependent on the anatomic site and the species studied. U-II binds to a specific high-affinity receptor and its activity is not blocked by classical receptor antagonists (Itoh et al. 1988). Ames and co-workers (1999) have recently reported that U-II was an agonist for the orphan receptor GPR14. The same group also showed that U-II, in certain vascular beds, is the most potent mammalian vasoconstrictor peptide known to date. It has also been shown that the binding between U-II and GPR14 involves calcium mobilization. Northern blotting analysis revealed that GPR14 mRNA was most abundant in the heart and the pancreas. Lower levels were detected in the kidney cortex, thalamus, superior occipital gyrus, substantia nigra, cardiac and arterial tissues, endothelial cells, and smooth muscle cells (Maguire et al. 2000). No mRNAs were found in the vena cava or venous smooth muscle cells. Maguire and co-workers (2000) located the U-II receptor in skeletal muscle, cerebral cortex, kidney cortex, left ventricle, and very little in the tissues of the atria, conducting system of the heart, and lung parenchyma. In a study by Coulouarn et al. (1998), prepro-U-II mRNA was found by dot-blot analysis in the spinal cord, kidney, spleen, small intestine, thymus, prostate, pituitary gland, and adrenal gland and, to a lesser extent, in stomach, pancreas, ovary, and liver (Coulouarn et al. 1998; Nothacker et al. 1999). However, based on another report by Coulouarn et al. (1999) renal preproU-II mRNA expression may be less evident in rodents (mouse, rat). Nevertheless, detectable preproU-II mRNA expression was found in rat kidney by RT-PCR (unpublished observations). Urotensin receptor, GPR14 or “UT” receptor, is expressed by renal smooth muscle cells at the mRNA level (Douglas and Ohlstein 2000). It is also expressed in mouse bladder smooth muscle cells (Liu et al. 1999). Indeed, U-II is a spasmogen of bladder SMCs (Yano et al. 1994). Accordingly, radioligand binding sites for [125I]-U-II have been reported in the human renal cortex (Maguire et al. 2000). Although not specifically to the kidneys, U-II immunoreactivity has also been observed in other parts of the genitourinary tract, i.e., piscine bladder (Masini et al. 1999).
A recent study has demonstrated expression of U-II and its receptor mRNAs in many types of tumors, such as T98G glioblastoma cells, IMR32 neuroblastoma cells, NB69 neuroblastoma cells, BeWo choriocarcinoma cells, SW-13 adrenocortical carcinoma cells, DLD-1 colorectal adenocarcinoma cells, and HeLa cervical cancer cells (Takahashi et al. 2001). To date, the sites of U-II production in the human kidneys remain to be elucidated. The objectives of this study were to identify the cellular sites of U-II in the human kidney and to determine whether or not U-II is also expressed in renal carcinoma.
Materials and Methods
Tissues
Normal human kidney specimens (seven male, four female) collected at the Montreal General Hospital were fixed in 10% formalin, embedded in paraffin, and processed for immunohistochemistry (IHC). Renal carcinoma (clear-cell carcinoma; n=8, six male and two female) were all collected at surgery. Eight of the normal cases studied were collected at surgery and three at autopsy.
Antibody Generation
Female New Zealand White rabbits were dosed (100 μg SC followed by two booster injections at 4 and 8 weeks) with KLH-conjugated human urotensin-II (ETPD [CFWKYC] V). Antisera were subjected to protein A purification (ImmunoPure; Pierce, Rockford, IL), ammonium sulfate precipitation, and dialysis (25C in PBS overnight). The human urotensin-II polyclonal antibody (PAb) was further purified using an agarose-immobilized diaminodipropylamine column to which 2 mg human UR-II had been coupled [1 hr, 25C; EDC/DADPA immobilization, as described previously (Ames et al. 1999)]. Finally, the affinity-purified PAb was eluted from the column (0.1 M glycine, pH 2.5), pH-adjusted, and concentrated (3 mg/ml; Centricon-30; Amicon, Beverly, MA) before storage at −20C. The PAb has been tested (in an RIA format) against a range of peptide ligands, including somatostatin-14, endothelin-1, angiotensin II, CGRP, adrenomedullin, neuropeptide Y, urotensin-I, and two upstream regions of the U-II propeptide, and did not show any significant affinity (crossreactivity) for any of these ligands. SAR studies demonstrated that the conserved cyclic octapeptide core of the U-II isopeptide family is also recognized by the PAb, consistent with the observation that the PAb does cross react with U-II peptides from non-human species, including rat. The specificity of the antiserum was further confirmed by Western blotting using protein from recombinant endothelial cells expressing rat or human U-II (Figure 1).
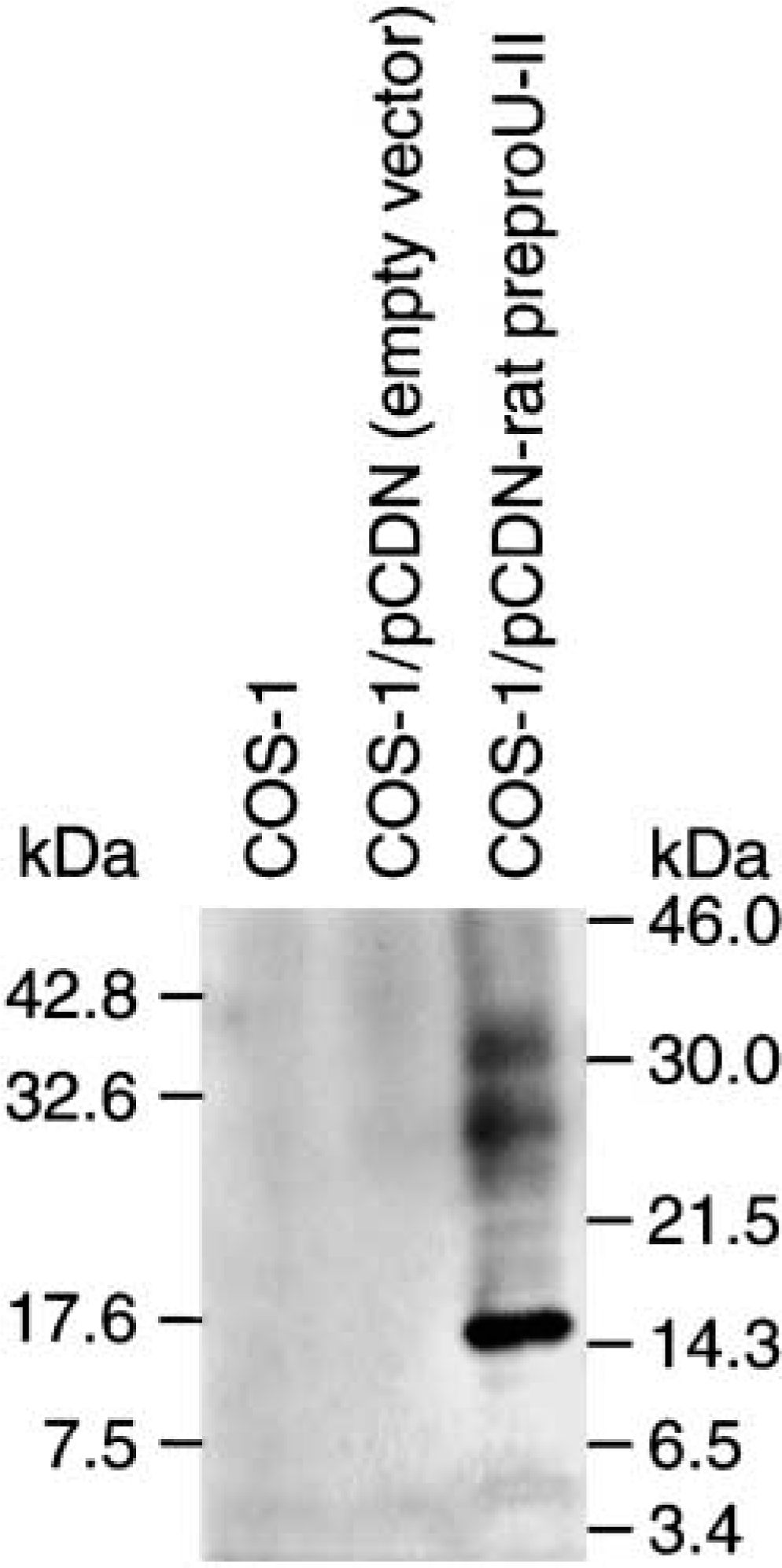
Western immunoblot using protein from recombinant endothelial cells expressing rat or human U-II, demonstrating the specificity of the U-II antiserum.
Immunohistochemistry
Paraffin sections of 5-μm thickness were immunostained by the avidin-biotin-peroxidase complex (ABC) method. Sections were deparaffinized and rehydrated in alcohol. After washing in PBS, the sections were placed in a Triton X-100 solution (0.2% in PBS) for 30 min at room temperature (RT). After washing in PBS, the sections were placed in a hydrogen peroxide solution (5% in PBS) for 30 min at RT. They were then washed in PBS and incubated for 30 min at RT in 10% normal goat serum (Vector Labs; Burlingame, CA). The sections were incubated at 4C overnight (18 hr) in U-II polyclonal antibody (1:75 in PBS) in 0.1% BSA. After washing again in PBS, they were then incubated for 45 min at RT in biotin-conjugated goat anti-rabbit IgG (Vector). After washing in PBS, the slides were incubated at RT for 45 min with the avidin-biotin complex (Vector). Sites of U-II immunoreactivity were developed in 3,3-diaminobenzidine (DAB) solution and hydrogen peroxide. After washing in PBS, sections were counterstained with hematoxylin. Negative controls were immunostained with Ag/Ab mixture or with the non-immune serum in place of the primary antiserum.
Results
In normal human kidney there was abundant expression of human U-II, mainly in the tubule epithelial cells. The cytoplasm of epithelial cells of distal convoluted tubules and collecting tubules exhibited the strongest U-II expression (Figures 2A and 2C). The cells of the proximal convoluted tubules exhibited weaker U-II immunoreactivity compared to those of the distal convoluted tubules (Figure 2A). The collecting ducts were weakly immunostained for U-II (Figure 2C). The Henle loops showed weak immunoreactivity for U-II. In the glomeruli, only a small number of cells expressed strong U-II immunoreactivity (Figure 2B). There was abundant U-II immunoreactivity in the endothelial cells of renal capillaries. Endothelial cells and smooth muscle cells of renal arteries showed moderate U-II immunoreactivity (Figure 2E). No immunostaining was found in the endothelial cells of veins.
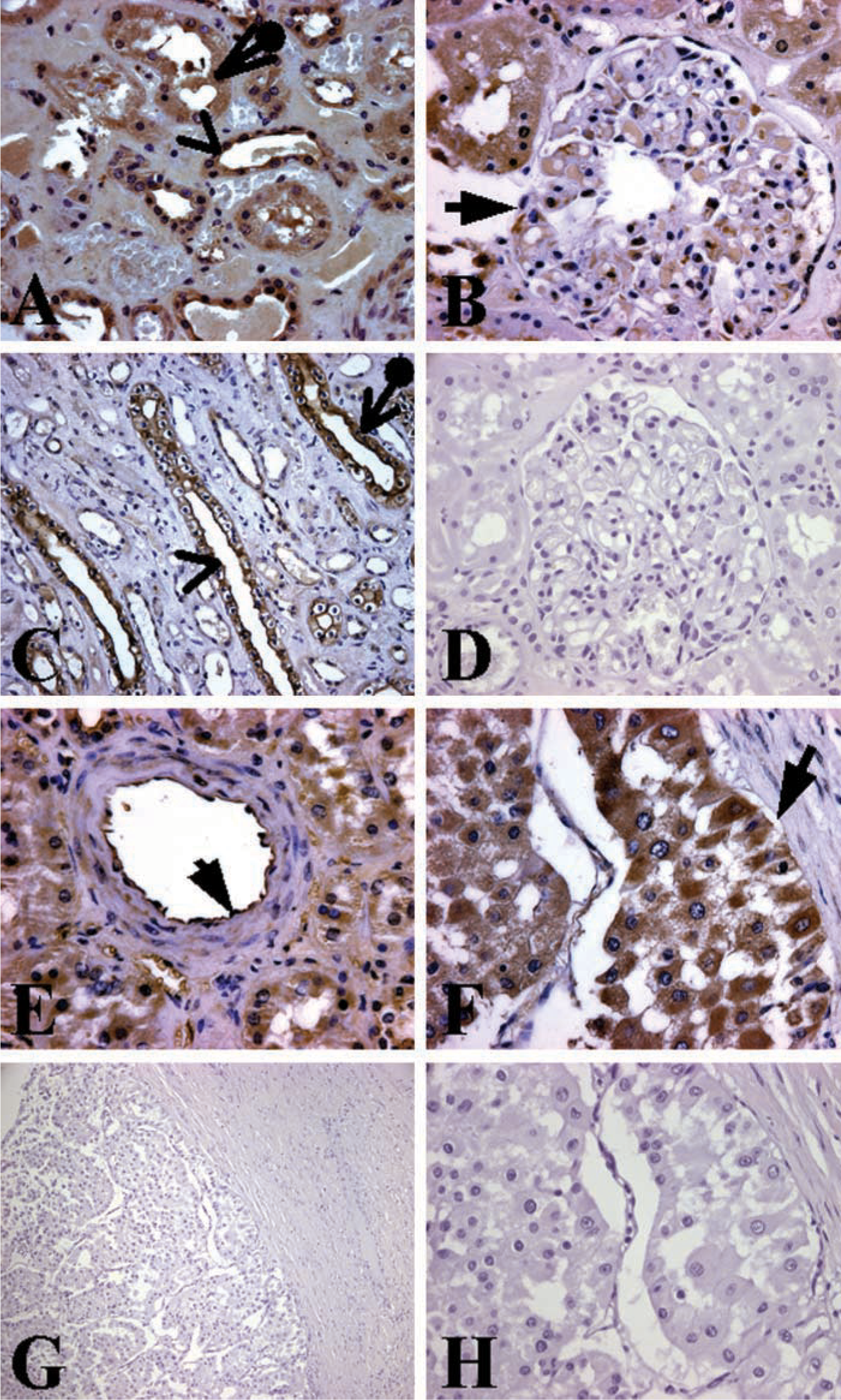
Localization of U-II immunoreactivity in the human kidney.
In the cases with renal clear-cell carcinoma, we observed moderate levels of U-II immunoreactivity in the cancer cells (Figure 2F). The intensity of U-II immunoreactivity in the tubule epithelial cells of normal and cancer tissues appeared comparable. The vasculature of the tumors expressed moderate U-II immunoreactivity. We also observed U-II immunoreactivity in the inflammatory cells within and surrounding the tumor. In contrast, very little immunostaining was found in the inflammatory cells of the tissue free of tumor.
Negative control experiments showed no immunostaining, confirming the specificity of the antisera and techniques used in this study (Figures 2D, 2G, and 2H).
Discussion
This is the first report demonstrating the presence of U-II by IHC in the kidneys and in clear-cell renal adenocarcinoma. Our results showed abundant expression of U-II in normal human kidney. The epithelial cells of distal convoluted tubules exhibited the most intense U-II expression, followed by collecting tubules, collecting ducts, and proximal convoluted tubules. U-II was also evident in all vascular endothelial cells except those of veins. In the renal cell carcinomas, there was abundant U-II immunoreactivity in both cancer cells and normal epithelial cells. These results demonstrate the presence of U-II protein in normal human kidneys and in renal carcinoma, suggesting that the presence of the peptide may contribute to the pathophysiology of human kidneys.
The kidneys are very vascular organs and play an important role in regulating arterial blood pressure. They do so by controlling water reabsorption and the glomerular filtration rate (GFR). The renin-angiotensin complex is well known for its major role in water reabsorption and the control of cardiovascular peripheral resistance. Regulation of GFR is obtained by three main mechanisms: myogenic response, tubuloglomerular feedback, and reflex control of GFR. The tubuloglomerular feedback consists of a chemical message sent by the distal tubules to nearby afferent arterioles when the flow increases in the tubules. The chemicals sent produce a vasoconstriction in the arteriole, reducing GFR. It is uncertain which chemicals act in this feedback, but a paracrine vasoactive hormone is a likely candidate. Because U-II is a vasoactive peptide and is abundantly expressed in the tubules and the vasculature, it is reasonable to suggest that U-II plays a role in this mechanism by a paracrine or an endocrine action.
There is also a reflex control, which occurs when blood pressure falls, causing vasoconstriction and decreasing GFR. This decrease in GFR augments blood volume and systemic blood pressure. This process is regulated by different hormones such as angiotensin-II, a vasoconstrictor, and prostaglandins, which act as vasodilators. U-II is known for its very strong vasoactive effects and may be one of the hormones responsible for this reflex. This is supported by the recent findings of increased U-II levels in patients with renal failure (Totsune et al. 2001). Furthermore, angiotensin-II and U-II have already been coupled in a few processes. In research on the central nervous system of Aplysia californica, Gonzalez et al. (1995) found that urotensin-I (U-I), U-II, and angiotensin-like peptides co-existed within the same cells of the F-cluster of the cerebral ganglia. Another study showed co-existence of adrenocorticotrophic hormone, angiotensin II, and the urophysial peptides U-I and U-II in the process responsible for cortisol secretion by interrenal preparations of seawater and freshwater-adapted trout (Arnold-Reed and Balment 1994).
U-II also modulates transepithelial [Na+]/[Cl-] ion transport in fish (skin epithelia, operculum, intestine, bladder), and it is therefore proposed that U-II might possess an osmoregulatory function in mammals (Loretz et al. 1985; Conlon et al. 1996,1997). The proposal that U-II plays a physiological role in osmoregulation is supported by the observation that exposing fish (flounder) to an osmotic stress (freshwater vs saltwater environment) elevates circulating U-II-like immunoreactivity (coincident with alterations in plasma osmolarity and [Na+]) (Winter et al. 1999). U-II may possess osmoregulatory functions in aquatic animals, although little is known to date about similar effects in mammals. U-II is an extremely potent constrictor of renal blood vessels from primates with a -log (EC50) of 9.6, an order of magnitude more potent than endothelin-1 (Ames et al. 1999). However, this is a species-dependent phenomenon and is not seen in rat, canine, or porcine isolated renal arteries (Ames et al. 1999; Douglas et al. 2000). In vivo, systemic administration of U-II to the anesthetized primate results in profound renal vasoconstriction (Ames et al. 1999). In contrast to the primate, systemic U-II administration to the conscious rat is associated with renal hyporemia secondary to its effects on arterial blood pressure (Gardiner et al. 2001).
Recent reports have shown a mitogenic role for U-II through induction of smooth muscle cell proliferation (Sauzeau et al. 2001; Watanabe et al. 2001). In addition, U-II has been shown to induce collagen deposition by fibroblasts (Tzanidis et al. 2000). It also causes cardiomyocyte hypertrophy on overexpression of its UT receptors (Tzanidis et al. 2001). U-II may also induce release of nitric oxide, which is an antagonist (MacLean et al. 2000). Moreover, a recent study has demonstrated that U-II and its receptor mRNAs were found in many types of tumor cell lines (Takahashi et al. 2001). Therefore, the finding of moderate expression of U-II in renal clear-cell carcinoma shown in our present study suggests a mitogenic role for the peptide in renal carcinoma.
In summary, we report abundant expression of the human U-II peptide in the kidney tubules and vasculature and in renal carcinoma. These morphological findings suggest a possible role for this vasoactive and mitogenic peptide in the pathophysiology of the human renal system. Further studies are needed to address the role played by this peptide in this system and the mechanism of its regulation in health and disease.
Footnotes
Acknowledgements
Supported by the Canadian Institute of Health Research and the Fond de la Recherche en Sante du Quebec.
We wish to thank Ghada Hassan, Fazila Chiouali, and Zhaohui Ao for their help during the course of the study.
