Abstract
Background:
National consensus guidelines outline recommendations for best practices in treating patients with candidemia. This study evaluated the impact of receiving care adherent to the best practice recommendations on clinical outcomes in patients with candidemia.
Methods:
This retrospective, multicenter study included patients with candidemia from 2010 to 2015 at 9 hospitals. The primary outcome was the composite of 30-day in-hospital mortality and 90-day candidemia recurrence. Outcomes were compared between those receiving and not receiving care adherent to the guideline recommendations. Inverse probability weights with regression adjustment were utilized to determine the average treatment effect of adherent care on the composite outcome
Results:
295 patients were included with 14.2% meeting criteria for the composite outcome (11.9% mortality and 2.4% recurrence). The average treatment effect of adherent care was not significant (P = .75). However, receiving appropriate initial antifungal treatment and central venous catheter removal were both associated with the composite (average treatment effect of −17.5%, P = .011 and −8.8%, P = .013, respectively). In patients with a source of infection other than the central line, central venous catheter removal was not associated with the composite (P = .95). The most common reason for failure to receive appropriate initial antifungal treatment was omission of the loading dose.
Conclusions:
Central venous catheter removal and appropriate initial antifungal treatment were associated with a lower incidence of the composite of mortality and recurrence. Additional studies are needed to determine the optimal duration of therapy following candidemia clearance.
Keywords
Introduction
Candidemia is a major healthcare burden representing the fourth most common cause of healthcare-associated bloodstream infections.1,2 Candidemia increases healthcare costs and has an estimated mortality of 30%.3–6 Appropriate management of this infection can be challenging due to complicated and comorbid patients, the increasingly common isolation of drug-resistant species, and the need to identify and remove the source of infection.1,7,8
National consensus guidelines for management of invasive candidiasis were updated in 2016 by the Infectious Diseases Society of America (IDSA).7,9 The guidelines recommend multiple best practices for managing patients with candidemia: appropriate initial antifungal treatment, repeat blood cultures to confirm bloodstream clearance, treatment duration of at least 14 days from bloodstream clearance, central venous catheter removal, and ophthalmological examination for screening of endophthalmitis. 7 Several of these recommendations are controversial and based on expert opinion. 7 Studies have examined the impact of several of these best practice recommendations on clinical outcomes; however, data are extremely limited on the impact of adherence to the recommendations collectively. 10 This study evaluated the impact of receiving care adherent to the best practice recommendations on clinical outcomes in patients with candidemia.
Methods
Study design and setting
This was a multicenter retrospective cohort study conducted at 2 health-systems located in Orlando, Florida, USA and Portland, Oregon, USA from January 2010 to June 2015. These systems comprise 9 hospitals with 2820 total beds. Hospital bed size ranges from 95 to 808 beds. These hospitals are diverse and include one major academic, 3 community-teaching, and 5 community non-teaching medical centers. While multiple protocols and guidelines are available for management of various infectious diseases, no hospital provided guidance specific to candidemia. In both health-systems, it is common practice to collect both an anaerobic and aerobic blood culture from 2 distinct sites for patients with suspicion of bloodstream infection. All aspects of treatment of candidemia were at the discretion of the primary treating practitioner. Bedside infectious diseases consultation was available to all patients upon request of the primary treating practitioner. Outcomes were compared in those that received care adherent to the guideline recommendations and those that received non-adherent care. The Legacy Health Institutional Review Board (LHS81520161) and Arnold Palmer Medical Center Institutional Review Board (831507-1) approved this study. The study was granted an exemption of informed consent by both approving boards.
Eligible patients
All patients 18 years or older with at least one blood culture positive for Candida species were screened for inclusion. Patients were excluded if they were discharged, died, had status changed to palliative care, or left against medical advice within 48 hours of blood culture collection.
Definitions
Mortality was defined as in-hospital, all-cause death within 30 days of the last dose of antifungal agent. Recurrence was defined as candidemia after initial bloodstream clearance within 90 days of the original index culture. Adherence was defined as care in accordance with the guideline recommendations (excluding ophthalmologic examination) as follows: 7 (1) proper antifungal selection initiated within 48 hours of culture positivity including appropriate loading and maintenance doses (see Appendix); (2) repeat blood cultures at least every other day until bloodstream clearance; (3) at least 14-day treatment duration after documented bloodstream clearance; and (4) removal of central venous catheters (CVC). While routine ophthalmologic examination is recommended, it may not be needed in all patients and is unlikely to affect mortality and was therefore excluded. Additionally, as early death can prevent adherence to some guideline recommendations (ophthalmologic examination, appropriate duration of therapy, and repeat blood cultures until clearance), “adherence to initial recommendations” was examined and defined as adherence to appropriate initial antifungal treatment and CVC removal (recommendations that could be achieved regardless of time to death).4,11 In patients that died during hospitalization, adherence to treatment duration was not determined except in patients that received at least 14 days of therapy after bloodstream clearance. Pitt bacteremia score was calculated as described previously. 12 Acute kidney injury was determined by the RIFLE criteria. 13 Index culture was defined as the first blood culture collected that was positive for Candida. Length of stay was not calculated in patients that died during hospitalization.
Outcomes
The primary outcome was the composite of recurrence and mortality. Secondary outcomes included mortality, recurrence, hospital length of stay, and time to bloodstream clearance.
Statistical analysis
Assuming the non-adherent group would have a 35% incidence of the composite outcome, 275 patients were required to have an 80% power to detect a 50% lower incidence in the adherent group (this assumes approximately 25% of patients would receive guideline adherent care 11 ). Baseline characteristics between the non-adherent and adherent groups were compared by chi-squared, Fisher’s exact, Student’s t-test, or Wilcoxon rank-sum test as appropriate. As substantial differences in the baseline and clinical characteristics between groups was possible, we performed univariate analysis to determine variables associated with the likelihood of receiving adherent care. Variables with P < .2 were included in a backward stepwise logistic regression model with Huber-White sandwich estimators with P < .2 set for retention. Manual evaluation of interaction terms was conducted and included if found to be significant. Variables identified by this model were used to calculate propensity scores for receipt of adherent care.14,15 Similarly, backward stepwise logistic regression was employed to identify variables associated with the composite outcome with P < .05 set for retention. Inverse probability weights with regression adjustment were utilized to determine the average treatment effect of adherent care on the composite outcome. This methodology was selected as it can balance covariates between treatment groups while preserving the full data-set for analysis.16,17 Covariate balance after propensity score weighting was tested by examination of the raw and weighted differences and by an overidentification test of the full model. This same methodology was utilized for examining the individual guideline recommendations. All analyses were performed with Stata version 14.2 (Stata-Corp, College Station, TX).
Results
A total of 475 patients were screened with 295 meeting criteria for inclusion (Figure 1). Table 1 shows the baseline clinical and demographic characteristics of the patients that received non- adherent and adherent care. Several significant differences were identified between groups including age, renal function, CVC as the likely source, days in the hospital prior to diagnosis, and infectious diseases consultation. Approximately half of the included patients were admitted to the intensive care unit with approximately one-quarter requiring mechanical ventilation at the time of index culture collection. Close to 90% of patients had a central venous catheter with 67% suspected of the line being the source of infection. Approximately half were infected with Candida albicans and an additional one-quarter infected with Candida glabrata. An echinocandin was used as first line therapy in 54% of patients with almost all other patients receiving fluconazole.
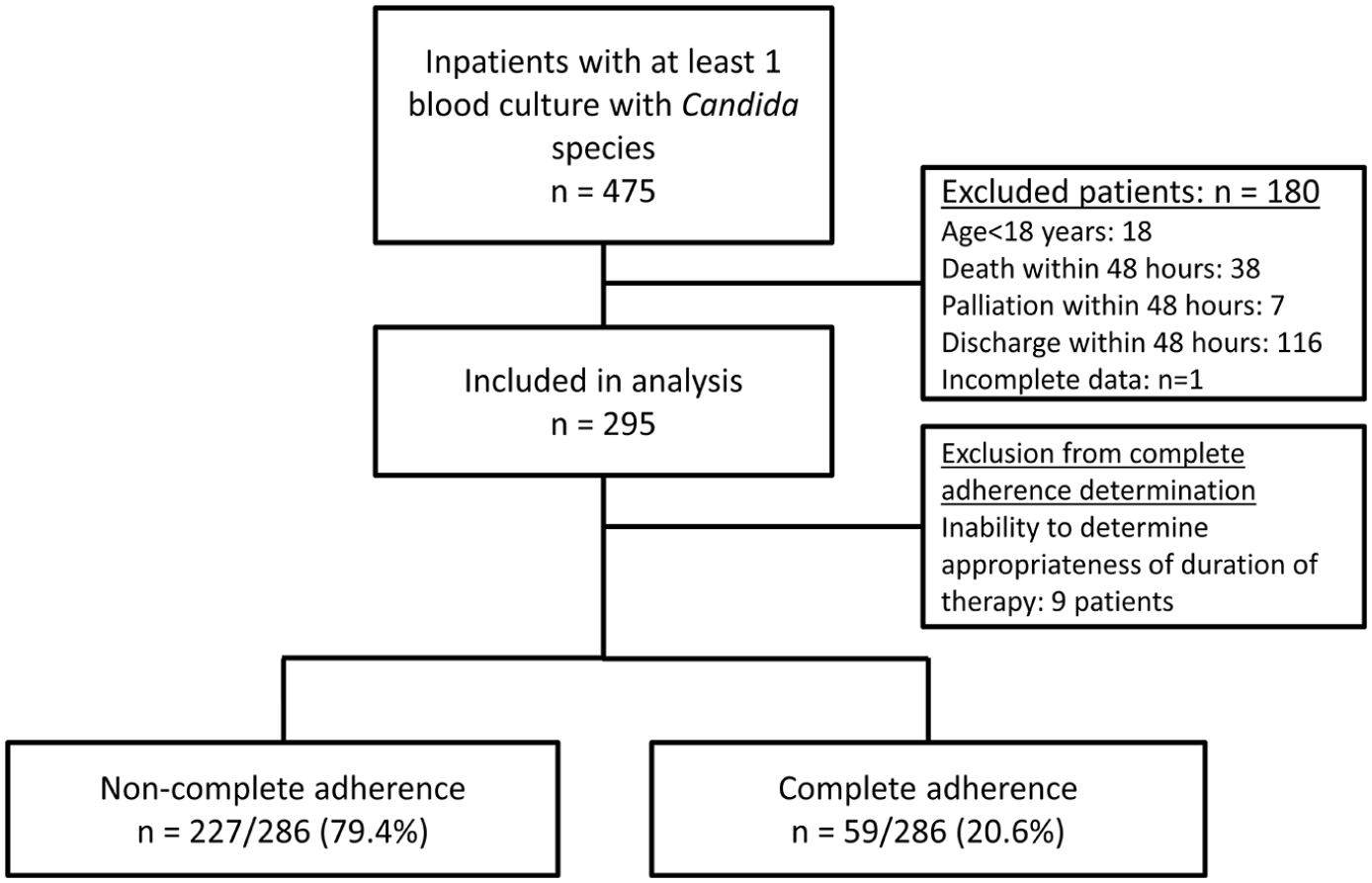
Flow diagram for patients included in the study.
Demographics and clinical characteristics in the non-adherent and adherent groups.
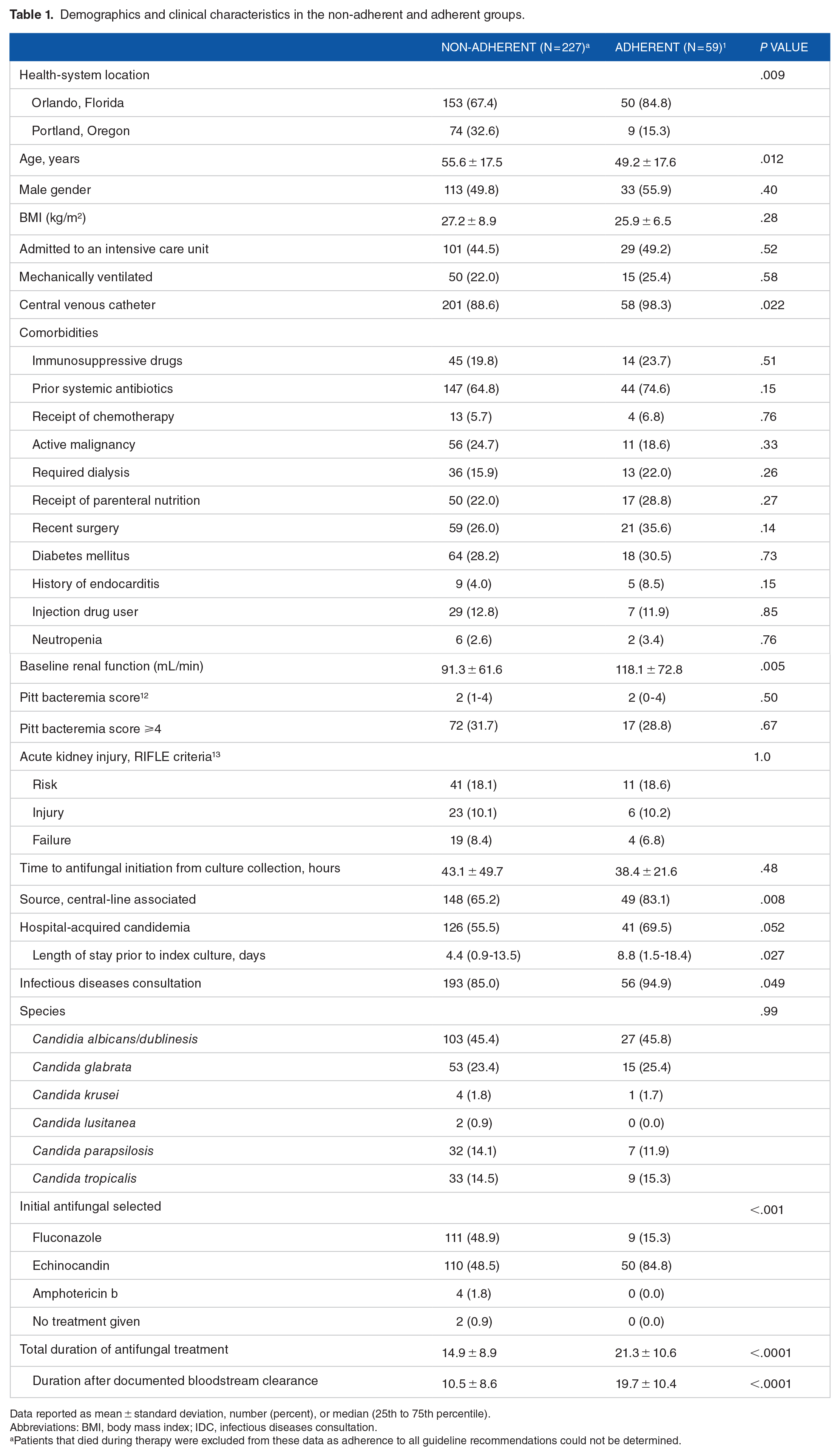
Data reported as mean ± standard deviation, number (percent), or median (25th to 75th percentile).
Abbreviations: BMI, body mass index; IDC, infectious diseases consultation.
Patients that died during therapy were excluded from these data as adherence to all guideline recommendations could not be determined.
Prior to excluding patients that died within 48 hours, mortality was 21.9% and consistent with previous reports.7,18,19 In the final population, mortality was only 11.9% and median time to death was 7.5 days from index culture collection. Recurrence occurred in only 2.4% of patients with a total composite outcome occurring in 14.2%.
Association of adherence to composite guideline recommendations and clinical outcomes
Table 2 shows the overall incidence of adherence to the guideline recommendations. The composite outcome, mortality, and recurrence were similar regardless of receiving care adherent to guideline recommendations (Table 3). However, receiving adherent care was associated with a 1.7 day quicker time to clearance of candidemia but longer total hospital stay. Factors associated with adherent care were well-balanced after inverse probability weighting. Multiple factors associated with the composite outcome (Table 4) were used in the propensity-score weighted model. After inverse probability weighting with regression adjustment, the composite was similar in the non-adherent and adherent groups (Table 5). Adherence to initial recommendations (appropriate initial antifungal treatment and CVC removal) was associated with approximately 10% lower incidence of the composite outcome (Table 5) and lower mortality (average treatment effect −6.9%, P = .037).
Incidence of adherence to guideline recommendations.

Abbreviation: CVC, central venous catheter.
Defined as adherence to appropriate initial antifungal treatment, repeat blood cultures at least every other day, treatment of at least 14 days, and CVC removal; adherence to treatment duration was not able to be determined for some patients that died prior to receiving 14 days of antifungal therapy.
Included appropriate initial antifungal treatment and CVC removal.
Outcomes in the non-adherent compared to the adherent group.
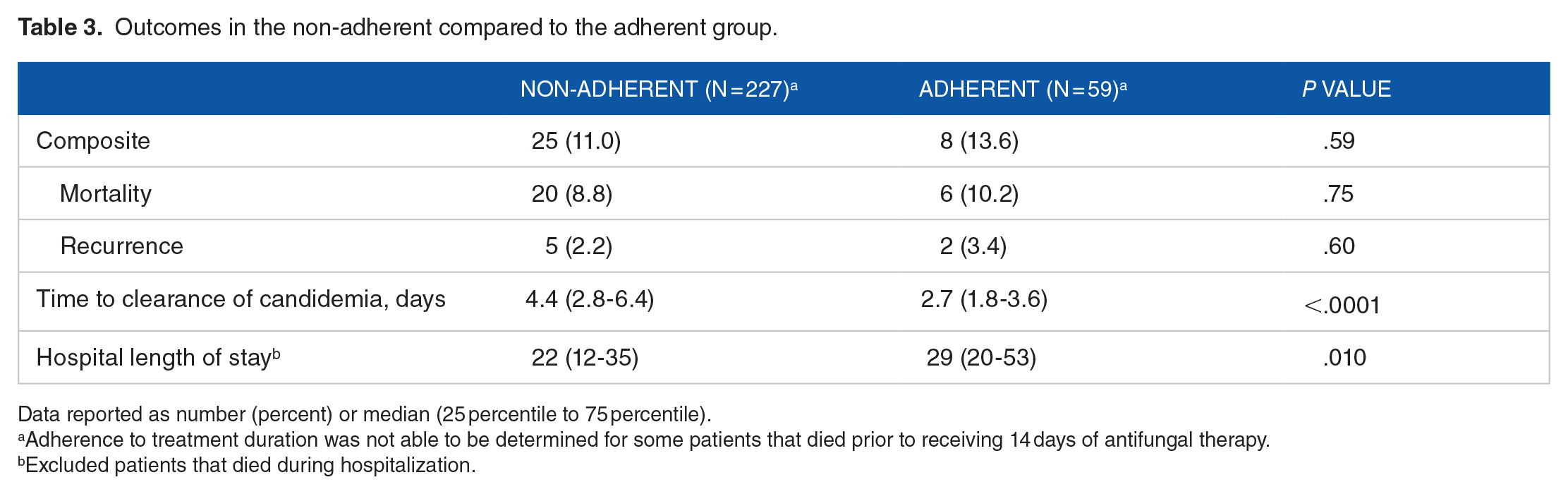
Data reported as number (percent) or median (25 percentile to 75 percentile).
Adherence to treatment duration was not able to be determined for some patients that died prior to receiving 14 days of antifungal therapy.
Excluded patients that died during hospitalization.
Multivariate logistic regression model of variables associated with the composite outcome of mortality or recurrence.
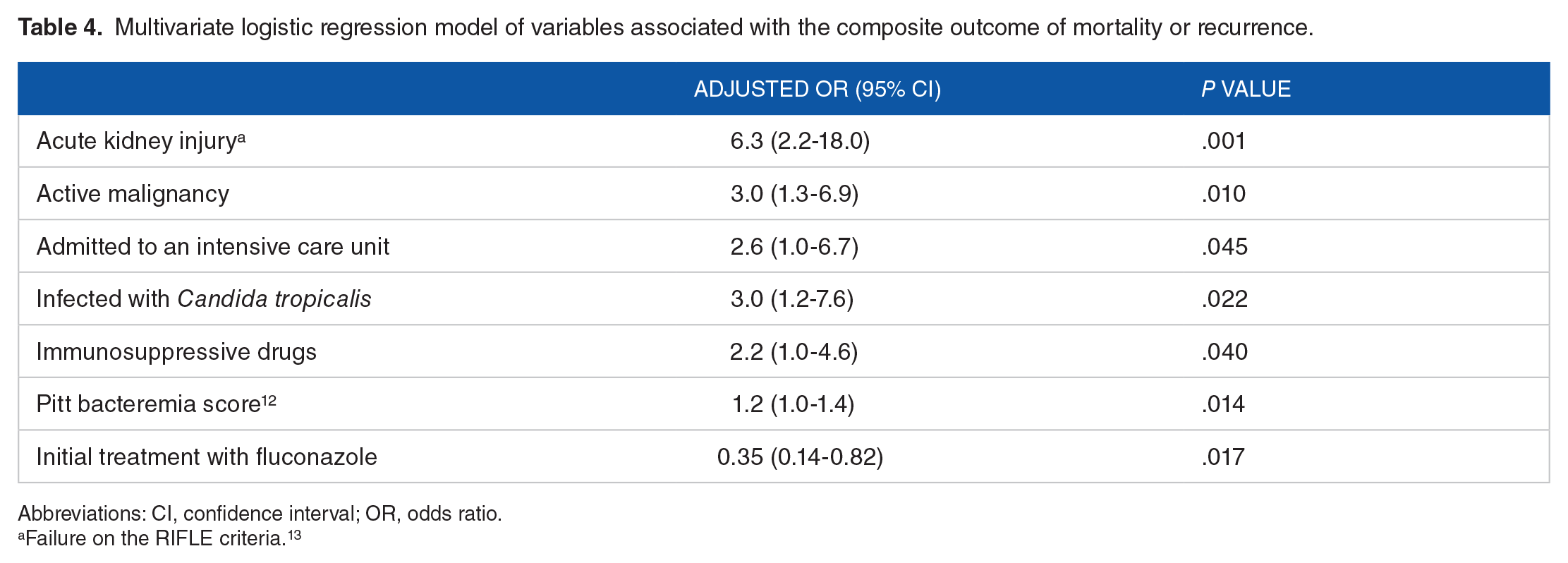
Abbreviations: CI, confidence interval; OR, odds ratio.
Failure on the RIFLE criteria. 13
Impact of adherence to guideline recommendations on the composite of 90-day recurrence and 30-day in-hospital mortality.
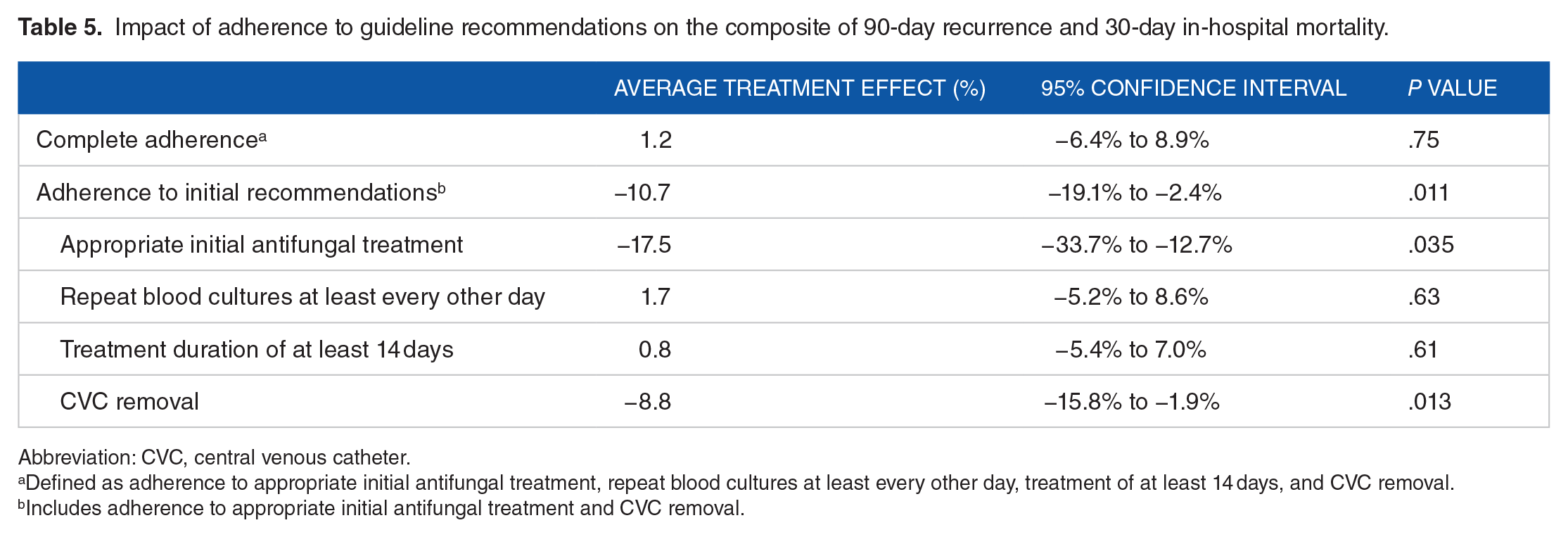
Abbreviation: CVC, central venous catheter.
Defined as adherence to appropriate initial antifungal treatment, repeat blood cultures at least every other day, treatment of at least 14 days, and CVC removal.
Includes adherence to appropriate initial antifungal treatment and CVC removal.
Association of adherence to individual guideline recommendations and clinical outcomes
Appropriate initial antifungal treatment and CVC removal were the only individual recommendations associated with a lower probability of the composite outcome after propensity-score weighting (Table 5). Of the 89 patients (30.2%) that did not receive appropriate initial antifungal treatment, 2 (0.7%) did not receive any antifungal treatment during the entire hospital course; 13 (4.4%) received their first dose of antifungal therapy more than 48 hours after culture positivity; 3 (1.0%) received caspofungin without an appropriate loading dose; 68 (23.1%) received fluconazole without a loading dose; and 3 (1.0%) received a suboptimal fluconazole maintenance dose. When limiting the analysis to patients that initially received fluconazole (n = 121), patients that received appropriate loading doses were less likely to suffer the composite outcome (adjusted odds ratio 0.27, P = .20), but this did not reach significance.
In patients with a CVC as the suspected source of infection, removal was associated with a lower incidence of the composite outcome (12.6% vs 30.4%, P = .023) which persisted after propensity-score weighting (removal associated with average treatment effect of −18.8%, P < .001). Patients suffering the composite outcome had a slower time to catheter removal than those that did not (median 7.7 vs 2.4 days, P = .042). Of the 65 patients with a CVC with a suspected source other than the CVC, the composite occurred with a similar frequency in those with removal and retention (15.6% vs 15.0%, P = .95).
While 123 patients (41.7%) did not have blood cultures taken every other day until clearance, most patients had documented negative blood cultures during the hospital admission (86.8%). In-hospital mortality was not the main factor for failing to document negative blood cultures as those suffering mortality only accounted for 18% of these failures.
Of the 134 patients that did not receive an appropriate duration (see Table 2), 27% received 7 days or less, 41% received 7 to 13 days, and 28% received 14 or more days of total antifungal treatment; however, the latter group only received an average of 8.8 days of antifungal treatment after bloodstream clearance and therefore most did not qualify as having an appropriate duration of treatment.
Few patients had an ophthalmologic examination (Table 2). In those receiving care without an infectious diseases consultation, only 7.9% received this examination; however, even in patients with an infectious diseases consultation, only 26.5% had this examination.
Discussion
This study failed to identify a benefit to care adherent to the 2016 IDSA guidelines. However, we did identify a benefit when care was adherent to initial recommendations (appropriate initial antifungal treatment and CVC removal), which supports the guideline recommendations. We attempted to control for bias by calculating propensity scores for receipt of adherent care and modeling with inverse probability weights with regression adjustment. To our knowledge, this study is the largest evaluating guideline adherence and association with clinical outcomes.
Several studies have evaluated the impact of adherence from previous iterations of the guidelines. In 2004, Patel and colleagues performed a similar study by evaluating adherence to the guidelines published in the year 2000 based on 3 criteria: (1) selection of an appropriate initial antifungal; (2) CVC removal; and (3) antifungal treatment for all patients. 4 They found a mortality of 32% and overall adherence of 76%; the latter was associated with lower mortality after controlling for severity of illness (P = .003). Similarly, we identified a benefit in mortality and the composite outcome in patients that received care adherent to initial recommendations (see Table 5). In 2017, Ashong and colleagues evaluated guideline adherence to the 2009 IDSA guidelines for management of candidiasis in 94 patients with candidemia at a single center. 11 They found only 17% adherence to appropriate duration and initial antifungal treatment; however, those that were adherent were much less likely to die at 6 weeks (0% vs 42.3%, P = .001; overall mortality 33%). It is unclear how the authors classified duration of therapy in those that died prior to day 14 and if that may have affected the identified difference in mortality. As this study had few patients, the authors did not attempt to adjust for potential confounding.
In 2020, Cardozo et al 10 published an analysis examining the impact of a practice improvement implemented (2016-2018) in 11 hospitals in Spain. Infectious diseases specialists identified patients with candidemia via prospective microbiology screening and made recommendations based on a bundle that included the guideline recommendations. Patients with candidemia during the practice improvement period were much less likely to suffer early and overall mortality as compared to historical controls (2010-2011). This study provides evidence to support implementation of similar best practice bundles. However, the study did not compare patients with and without guideline-adherent care and conclusions about individual recommendations cannot be made. In addition, it is difficult to determine whether additional changes in management between the pre- (2010-2011) and post-implementation (2016-2018) periods affected the outcomes.
Similar to the finding in our study, previous studies have identified that earlier administration of appropriate antifungal therapy is associated with lower mortality.4,7,11,20 This conforms to the general understanding in treatment of infectious diseases. 21 In accordance to the guidelines, we classified fluconazole as an appropriate initial choice in non-neutropenic patients if they received an appropriate loading dose. Recently, a prospective randomized controlled trial found that initial therapy with isavuconazole (a triazole) failed to meet the definition of non-inferiority compared to caspofungin (with step-down to oral voriconazole therapy in close to 40% of patients). 22 This recent study, in combination with other previous reports,23-25 suggests that echinocandins are likely the favored initial antifungal therapy regardless of patient factors or prevalence of triazole-resistance. Our study identified a lower incidence of the composite outcome in those receiving fluconazole as initial therapy (see Table 4). This may represent incomplete control for severity of illness by other covariates instead of an actual benefit of empiric fluconazole; 22 a previous retrospective cohort study also reached this conclusion. 26 Our study illustrates that fluconazole is still frequently utilized as a first-line agent, but often with inappropriate initial dosing (59.5% of patients treated with fluconazole in our study did not receive the loading dose). Lack of a loading dose may further affect outcome when fluconazole is chosen for empiric therapy. Although lack of a fluconazole loading dose itself was not statistically associated with the composite outcome in this study, it did account for 76% of the patients who did not receive appropriate initial antifungal therapy. Due to difficulty in appropriate dosing and suggested greater risk of poor clinical outcome,23,24 we believe echinocandins should be preferentially used for initial treatment of known candidemia, with step-down to oral triazoles considered in stable patients once susceptibility is known. When triazoles are used for empiric or definitive therapy, the dosing should be optimized.
Similar to other findings,7,24 our study identified a benefit of CVC removal. In addition, we identified that a quicker time to removal was associated with a lower incidence of the composite outcome. This finding may be confounded by other factors including severity of illness. Conversely, we identified no benefit with removal when the source was that other than the central line. Other studies have questioned the benefit of CVC removal, and some have questioned specifically removal in patients without CVC as the source.27-29 While the guidelines continue to support early CVC removal in all non-neutropenic patients regardless of source, 7 a definitive answer is unlikely to be reached until performance of a randomized controlled trial examining catheter management including early versus late removal and removal versus retained catheter. Evaluating the role of CVC removal for sources other than the CVC may be especially important, as our study failed to show a benefit in this subgroup. Due to the low number of neutropenic patients in our study, we were not able to explore CVC removal in this subpopulation.
There is little evidence to suggest that collecting follow-up blood cultures to document bloodstream clearance is related to clinical outcomes. 7 Duration of therapy for uncomplicated infection is based on practice in randomized controlled trials and high-quality prospective studies evaluating the impact of duration of therapy on clinical outcomes are lacking. 7 Routine ophthalmologic examination in patients with candidemia is controversial and there is little evidence linking examination to clinical outcomes. 7 We failed to identify a link between follow-up blood cultures, duration of therapy, or routine ophthalmologic examination and clinical outcomes. Routine ophthalmologic examination should likely continue to be performed based on the prevalence of ophthalmologic involvement despite lack of symptoms in patients with candidemia. 30 Despite lack of confirmatory data, follow-up blood cultures and adequate duration of therapy should continue to be recommended until prospective studies suggest otherwise.
Other studies have consistently identified a protective effect of infectious diseases consultation (IDC) on mortality in patients with candidemia.18,19,31 While patients in our study with an IDC had a higher incidence of adherent care (Table 2), we did not identify lower mortality in patients with an IDC (data not shown, P = .56). This may be related to the low number of patients without an IDC in our cohort. As there can be a substantial delay between identification of candidemia and an IDC (delay in primary practitioner request), antimicrobial stewardship programs that perform prospective audit with feedback may be ideally placed to optimize appropriate initial antifungal treatment. As we identified a lower incidence of the composite outcome in those that had appropriate initial antifungal treatment, we believe antifungal selection and dosing to be major targets for antimicrobial stewardship programs.
Substantial limitations are present in this study. First, this was a retrospective study and is subject to bias. Variables associated with clinical outcomes may be causative, consequences of the outcome, or merely associated. 28 Substantial differences between the non-adherent and adherent groups were present. We attempted to control for these differences via multivariate modeling and propensity score weighting; however, additional bias may still be present. Second, few patients received care adherent to all guideline recommendations; this small number of adherent patients may have been insufficient to identify a clinical benefit to adherence. Third, our study included few patients with concurrent neutropenia and the results cannot be extrapolated to this unique population.
Conclusions
In this multicenter study, we found a clinical benefit in patients that received appropriate initial antifungal treatment and underwent CVC removal. We suggest that antimicrobial stewardship programs may be ideally placed to improve the percentage of patients receiving appropriate initial treatment. While we did not find a clinical benefit to ophthalmologic examination or patients that had repeat blood cultures until bloodstream clearance, we believe these practices should continue until further evidence suggests otherwise. In addition, prospective randomized studies are needed to identify the ideal duration of therapy in patients with candidemia.
Footnotes
Appendix: Appropriate initial treatment
The following were considered appropriate initial treatment if received within 48 hours of culture positivity:
Acknowledgements
The authors would like to acknowledge their respective health-systems for allowing this research to be completed.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
All authors designed the study; SJJ, SC, and RBT collected the data; RBT and SJJ analyzed the data; SC, SJJ, and RBT were principally responsible for drafting the paper; all authors gave final approval of the manuscript.
