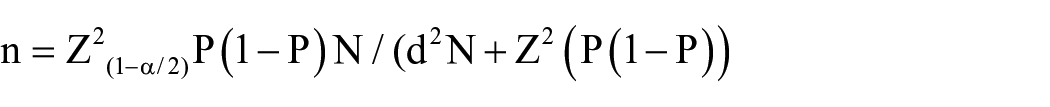Abstract
Background:
ART failure is a growing public health problem and a major threat to the progress of HIV/AIDS control. In Uganda however, little is documented on treatment outcomes and their associated factors among individuals on second line ART regimen. The rapid scale-up of ART over the past has resulted in substantial reductions in morbidity and mortality. However, as millions of people must be maintained on ART for life, individuals with ART treatment failure are increasingly encountered and the numbers are expected to rise. This could be attributed to factors such as sub-standard regimens, limited access to routine viral load monitoring, treatment interruptions, suboptimal adherence, among others. The purpose of this study was to estimate 5-year cumulative treatment failure and the associated factors among individuals on second line ART regimen Eastern Uganda.
Materials and methods:
A retrospective analysis of 541 records of HIV positive individuals, switched to second line ART regimen from January 2012 to December 2017. Inferential statistics including the Chi square test and multivariable logistic regression analysis was applied to determine associations of treatment failure against of the selected demographic, laboratory and clinical factors was performed. Associations between treatment failure and the predictors was based on a P-value of less than 5% and confidence intervals level of 95%.
Results:
We reviewed 541 records of individuals on second line ART regimen, of which 350 (64.7%) were female, 226 (41.8%) were married, and 197 (36.4%) were older than 35 years. The mean age at ART initiation was 30 years (SD = 14.8), while the mean weight at ART initiation was 47 kg (SD = 18.6), (range 4-97 kg). The overall proportion of treatment failure was 23%. The cumulative mortality risk for 5 years was 12.4% and the mortality rate was 2.5 deaths per 100 individuals per year. The odds of developing treatment failure among individuals switched to ATV/r-based regimen were 44% lower as compared to individuals who were switched to LPV/r (ORadj0.56, 95% CI 0.35-0.90, P = .016). while the odds of experiencing treatment failure among individuals that used AZT at ART initiation were 43% lower as compared to individuals that used a TDF based regimen at ART initiation (ORadj0.57, 95% CI 0.33-0.98, P = .041).
Conclusion:
The 5 year cumulative incidence of treatment failure in a cohort of 541 individuals was 23%. The type of protease inhibitor (PI) used in second line regimen and use of AZT at ART initiation were significantly associated with treatment failure. Our study also shows that the cumulative mortality risk while on second line ART regimen was 12.4% while the mortality rate was 2.5 deaths per 100 individuals per year. Given the high level of treatment failure among individuals on second line ART regimen, yet the current ART protocols limits the use of third line ART regimens to only regional referral hospitals, the Ministry of Health should strengthen the surveillance systems for identifying individuals failing on second line ART regimen even at district hospitals and lower health facilities to facilitate timely switch to optimal regimen. The Ministry of health through the Quality Improvement Division should conduct routine onsite support supervision to sites offering ART to ensure that treatment guides and other standard of care like timely switch to appropriate regimens among others are being adhered to. Knowledge gaps identified can also be addressed through onsite Continuous Medical Educations.
Background
Since the start of the Human Immunodeficiency Virus (HIV)/Acquired Immune Deficiency Syndrome (AIDS) epidemic, around 76.1 million individuals have been infected and an estimated 35 million people have died due to HIV/AIDS. At the end of 2016 an estimated 36.7 million people were living with HIV and Saharan Africa (SSA) accounts for close to 70% of the global burden of the disease. 1
Uganda’s national HIV prevalence is 6.2%, with an estimate of 1.3 million persons living with HIV; and only 84% know their HIV status. 2
The introduction and scale-up of ART has led to a reduction in HIV related mortalities up from 1.9 million AIDS-related deaths worldwide in 2005 down to 1 million people in 2016. ART scale-up has also contributed to a decline in new HIV infections by almost 11% since 2010 from 1.9 million people to 1.7 million people in 2016. 1
ART scale up of in Uganda has followed the World Health Organization (WHO) recommended public health approach. Under this approach, regimen decision is a matter of national policy guided by WHO; simple tools and approaches on when to start drug, substitute toxic drugs, switch regimen and stop drugs basing on clinical opinion and minimal laboratory investigations are developed to guide the implementation process.
This is aimed at enabling health-care workers with minimum training to deliver care to large numbers of patients. 3
Although the public health approach has enabled Uganda to reach large numbers of individuals with the ART, this approach has been associated with several programmatic limitations such as use of regimens with low genetic barrier, using less sensitive immunological and/or clinical criteria to diagnosis first line treatment failure, and lack of resistance testing. 4 In fact Uganda has one of the highest pre-treatment resistance to non-nucleoside reverse transcriptase inhibitors (NNRTIs) which stands at 15.9% compared to the WHO threshold of 10%.
This together with other obstacles to sustained ART access such as human resource constraints, frequent drug stock-outs, and weak enforcement of quality standards are likely to contribute to ART treatment failure. 5
In addition, ART is associated with life threatening adverse drug effects, drug-drug interactions, and the inconvenience of lifelong therapy, and these are known for facilitating acquisition and accumulation of drug resistance-associated mutations and may compromise the effectiveness of subsequent lines of treatment. 6
Increasing reports of multi-drug resistant virus among HIV positive individuals who have been on ART for a long period (treatment-experienced patients) are being frequently reported. Moreover, there is a possibility of newly infected ART naïve individuals being found with HIV mutants that are already resistant to commonly used antiretrovirals (ARVs). 7
These issues highlight the need for good treatment monitoring program that involves the measurement of viral loads and resistance testing. Unfortunately, routine HIV viral load monitoring is a new component in ART programs of Uganda. Until the national roll-out of viral load in 2015, ART related treatment failure was commonly identified basing on immunological and/or clinical criteria.
Resistance testing is not part of the care package during ART initiation or switching to second line, the exception is for individuals supposed to be switched to third line regimen. This implies that in an event of treatment failure, a second-line ART regimen is usually composed empirically thus making selection of a regimen with an optimal genetic barrier not feasible. 8
Responding to treatment failure in a responsible manner is key to achieving the 90-90-90 targets, particularly “the third 90.” If treatment failure increases unchecked, this target may not be reached due to limited therapeutic options and the higher costs of second- and third-line treatments, which are, respectively, 3 times and 14 times more expensive than current first-line nucleoside reverse-transcriptase inhibitor (NRTI)-based regimens. 7 ART treatment failure is also associated with poor virologic outcomes, increased mortality, and reduced durability and effectiveness of regimens. 4
In Uganda, there is paucity in data on the treatment outcomes among clients on second line ART regimen. However, a systematic review of 19 studies reporting outcomes of second-line ART in resource-limited settings indicated that the proportion of adult patients who experienced treatment failure varied widely from 8.3% to 41.2% at 24 months. 9 The proportion may even be higher in Uganda since routine viral load monitoring is not widely accessed by all ART programs in the country.
Studies that have been done within Uganda were mainly in urban settings. One study by Sigaloff et al 10 was conducted more than 5 years ago. Since then, there has been revision in the HIV treatment policy that includes the use of viral load for monitoring ART and diagnosis of treatment failure. Another recent study by Kityo et al 11 only assessed the treatment outcomes in adults on second line ART. This evidence gap regarding the outcomes of clients on second line ART in a rural setting clearly indicates a need to undertake this study. In this study, we estimated the treatment failure and the associated factors among individuals on second-line antiretroviral therapy (ART) in Eastern Uganda.
Methods
Study design and Setting
This was a retrospective cohort study conducted at Mbale Regional Referral Hospital (MRRH) in Eastern Uganda. According to the UPHIA 2017 survey, the HIV prevalence in the region is 4.8%, with one of the lowest levels of viral load suppression in the country at 52.9%.
MRRH is a public hospital with a bed capacity of 355 beds and runs an HIV clinic with about 9450 active clients.
All HIV positive individuals on second line regimen in MRRH were the source population for the study. Individuals who had been on second line regimen for at least 6 months and were switched to second line ART regimen between January 2012 and December 2017 were eligible.
However, clients who did not have viral load results at switch to second line regimen, those less than 6 months on second line ART and those who whose regimen was changed without documentation of treatment failure were excluded from this study.
A total of 657 clients had been switched to second line ART regimen between January 2012 and December 2017. We however, included of 541 individuals because individuals who did not have updated information on all the variables were excluded from the study.
Since we were working with a fixed sample of 657 individuals switched to second line ART in Mbale Regional Referral Hospital between January 2012 and December 2017; to demonstrate the degree of precision in estimating the parameter of interest, p, which is the proportion of the sample that experiences treatment failure. The study used the formula below for calculating the confidence interval for one proportion from a finite population.
n = Sample size
N = is the population size (541)
Z = Z statistic that corresponds to 95% level of confidence with a value of 1.96.
P = proportion of individuals who fail to achieve virologic suppression after switching to second line ART = 10% (Kityo et al, 2014)
d—Margin of error (precision error) = ±5%
Substituting into the formula,
n = 202 (minimum sample size)
Although the minimum sample size was 202, we reviewed 541 records of individuals to increase on the precision of our estimates.
A client was categorized as treatment failure if he or she had a plasma viral load of ⩾1000 copies/mL on 2 consecutive viral load measurements, the first one being done 6 months after switch to second line regimen, and the second one done 4 months after the first viral load measurement.
Socio demographic data including age, education, marital status, and sex; HIV status of other household members; tuberculosis (TB) status at ART start; WHO clinical stage at ART initiation; functional status at ART initiation; first-line ART regimen; WHO clinical stages at ART initiation; and ART regimen at switch.
Data collection, analysis, and management
Patients’ information was abstracted from the facility ART registers as well as the patients’ treatment files. The demographic variables, clients’ clinical characteristics and laboratory results were captured through a review of the patients’ charts and the facility ART register.
The collected data was analyzed using STATA 14 version. Univariate analysis was used to calculate the descriptive statistics for each of the variables. The categorical variables were summarized as proportions, while the continuous variables as means and standard deviation.
To measure the association between treatment outcome and predictors, the study utilized a Pearson’s chi-square test and logistic regression analysis. Association between treatment failure and the predictors were based on a p-value of less than 5% and the confidence interval (CI) was calculated at the 95% level.
Results and Discussions
Clinical characteristics at ART initiation
As observed in Table 1 below, majority, 181 (33.5%) of the participants were at WHO clinical stage 3, and 91 (16.8%) at WHO clinical stage 4 at ART initiation. Three hundred and forty-six participants (63.9%) did not have any signs or symptoms of TB at ART initiation. Three hundred and ninety two participants (72.4%) were working at the time of ART initiation while majority 385 (71.3%) were started on a Nevirapine based regimen at the time of ART initiation. Three hundred and ninety-four participants (72.8%) were switched to a Tenofovir (TDF) containing regimen while 343 (71.3%) were switched to Lopinavir/ritonavir (LPV/r) as protease inhibitors of choice.
Clinical characteristics of participants.
Cumulative incidence of treatment failure
Twenty three percent (123/541) of the participants experienced treatment failure in the period of January 2012 to December 2017 as shown in Figure 1 below.
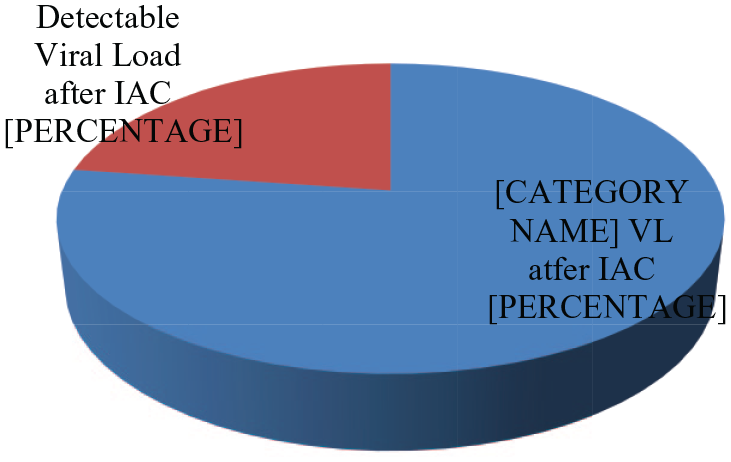
Proportion of viral load suppression.
This level of treatment failure is higher than that found in a study in Thailand on virologic and immunologic outcomes among clients on second ART line regimens which found that after 6 months of the second-line ART, 16% experienced treatment failure defined as a viral load of >400 copies/mL. At 12, 24, and 36 months of the second-line ART, 17%, 8%, and 10% experienced treatment failure respectively. 12
In South Africa, Fox et al 13 found that 23% of the patients started on second line ART experienced a treatment failure after 1 year. Treatment failure in this study however, was defined as a plasma viral load of >400 copies/mL.
In most of the second-line ART studies in Resource Limited Setting, a cut-off point of plasma viral load of >400 copies/mL was used to define virologic failure. World Health Organization (WHO) has been making new recommendations as new evidence comes in and the latest recommendation by WHO in 2016 for defining virologic failure was a viral load cut off >1000 copies/mL and that is what this used in estimating treatment failure rates. These implies that treatment failure rates among clients on second line ART regimen in MRRH could actually be higher than the 23% if a lower cut off value of >400 copies was used.
In a meta-analysis of 19 studies reporting second-line failure rates in 2035 patients across low-income and middle-income countries, the cumulative pooled proportion of adult patients failing virologically on second line ART regimen was 21.8%, 23.1%, 26.7%, and 38.0% at 6, 12, 24, and 36 months, respectively. This shows that the rates of virologic failure on second-line therapy are high in resource limited settings. Most studies (13 studies) defined virologic failure using the definition of RNA viral load >400 copies/mL but only around half of these reported 2 consecutive measures. 9
The difference in the rates of treatment failure for clients switched to second line ART regimen in this study could be attributed to the health care system challenges such as low coverage of viral services among clients on ART which is very important factor in facilitating timely detection ART failure cases. The viral load coverage in Uganda is at less than 80% unlike in the 2 different African countries where the previous studies were conducted from Ministry of Health. 2
The other possible reasons for the difference in the failure rates could be the difference in the diagnostic criteria for treatment failure. This study only used viral load results to measure treatment failure. In another study, viral load was used to assess treatment failure, in addition to other criteria such as cluster of differentiation type 4 (CD4) cell count and clinical status. CD4 cell count and clinical status however, are no longer recommended by WHO due to low sensitivity to detect treatment failure. Viral load increases before other immunologic and clinical markers, which can shorten the time to diagnosis. Due to its sensitivity, patients who have no immunological and clinical failure may already have virologic failure.
Compared to treatment failure among clients on first line in Uganda which stands at 4.1%, this is a high proportion (DHSI-2, 2018, unpublished data). Possible explanations for the observed higher failure rates with second-line therapy than with first line therapy are worse tolerability of second line regimens, acquired drug resistance during therapy, or difficulty in maintaining adherence to therapy after a longtime receiving ART, pill burden among other factors.
Analysis of treatment failure and the risk factors
Using the logistic regression model for further analysis, the results as presented in Table 2 below show that being in an age category of 26 to 35 years, WHO clinical stage 2, using Zidovudine (AZT) at ART initiation and ATV/r demonstrated a statistically significant association with treatment failure.
Predictors of treatment failure from multivariable analysis.
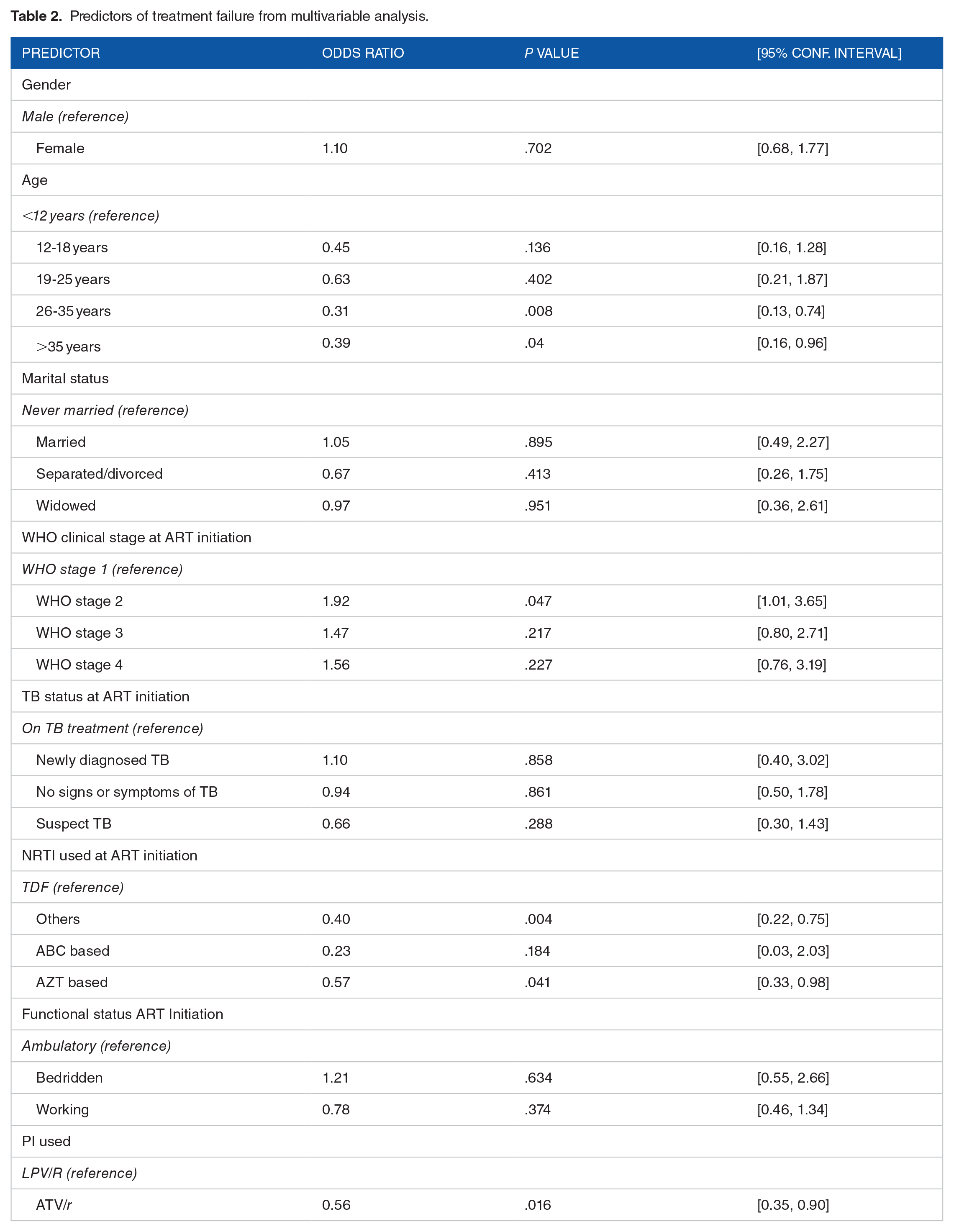
The odds for experiencing treatment failure among clients between the ages of 26 to 35 years was 69% lower as compared to clients below the age of 12 years (ORadj0.31, 95% CI 0.13-0.74, P = .008); while the odds of experiencing treatment failure among clients who are more than 35 years of age was 61% lower as compared to clients below the age of 12 years (ORadj0.39, 95% CI 0.16-0.96, P = .008). We also report that odds of experiencing treatment failure among clients with a WHO clinical stage 2 at ART initiation was 1.92 times higher as compared to clients that initiated ART with a WHO clinical stage of 1 ((ORadj1.92, 95% CI 1.01-3.36, P = .047).
The odds of experiencing treatment failure among clients that used AZT at ART initiation were 43% lower as compared to individuals who used a TDF based regimen at ART initiation (ORadj0.57, 95% CI 0.33-0.98, P = .041).
The PI used at switch to second line regimen also demonstrated a significant relationship. The odds of experiencing treatment failure among clients who were switched to ATV/r-based regimens were 44% lower as compared to the patients who were switched to LPV/r based regimen (ORadj0.56, 95% CI 0.35-0.90, P = .016).
The high rates of treatment failure among HIV-positive patients who are receiving second-line ART warrant further efforts to understand predictors of virologic failure to inform interventions to improve patient outcomes.
Treatment failure was significantly associated with the type of Protease inhibitor (PI) used (P = .019) and the use of AZT as the NRTI of choice at ART initiation. Patients who underwent a change to ATV boosted with ritonavir−based regimen instead of LPV/r were 44% likely to experience treatment failure. (0.56, P = .016). ATV is generally a well-tolerated drug with less pill burden as compared to other PIs such as LPV.
We also observed that majority of the patients (47.6%) on second line regimen used AZT as part of their first line drug. The use of AZT during first line is associated with accumulation of Thymidine Analogue mutations (TAMs) that confer resistance to TDF and Abacavir (ABC) which are the choices for second line in Uganda. Eighty-six (69.9%) of the patients who experienced treatment failure were on TDF. The ART guidelines have since been revised and now TDF is preferred first line NRTI because the mutations associated with it preserve future treatment options.
Delay in switching clients has been associated with poorer outcomes among patients failing on NNRTI based first-line therapy, as opposed to those receiving protease inhibitor-based regimens. Exposure to replicating virus for relatively long periods could lead to accumulation of mutations and compromise the efficacy of second-line regimens. This study however was not able to establish this relationship because of the poor documentation of the clients who were using PI based regimens as part of their first line regimens.
We also observed that children below the age of 12 years were significantly more likely to experience treatment failure as compared to adults between 26 and 35 years. Treatment for children and adolescents presents several challenges including the complexity in ARV dosing and the need to adjust doses as the children grow. This may be difficult especially for providers who are not skilled enough with pediatric care or too busy to track the suppression status.
Treatment for children may also be affected by the type of caretakers (ie, parents vs other caretakers), schooling environment (for those in schools), and transition to adolescent. Stigma, fear of disclosure, and stress may also affect younger people more than their older counterparts. Keeping young people in care by using social workers, peer supporter groups, and training on pediatric disclosure among health workers can be one way of improving ART adherence and hence better virologic out-comes.
Our findings should be considered within the following limitations. There were cases of missing data and clients who did not have data on such important variables were excluded in the analysis. Overall there were 657 clients who had been switched to second line regimen between January 2012 and December 2017 but only 541 records were included in the analysis because of missing information. The possibility of not including all registered participants in the analysis due to missing data could have led to selection to bias.
Secondly, treatment failure was not confirmed with resistance testing, but rather the definition of treatment failure used in practice which may result in misclassification bias that can lead to an overestimate of the cases of treatment failure in the study.
Nevertheless, our results highlight the complex interplay between social and clinical factors contributing to ART adherence and subsequent risk of treatment failure among patients on second-line ART in MRRH.
Conclusions and Recommendations
In this retrospective cohort analysis, the 5-years’ cumulative incidence of treatment failure among clients on second line ART regimen was 23%. The type of PI used in second line regimen and use of AZT as the NTRI of choice in the first line ART regimen were found to have a significant association with treatment failure.
Given the high level of treatment failure among individuals on second line ART regimen, yet the current ART protocols limits the use of third line ART regimens to only regional referral hospitals, the Ministry of Health should strengthen the surveillance systems for identifying individuals failing on second line ART regimen even at district hospitals and lower health facilities to facilitate timely switch to optimal regimen.
The Ministry of health through the Quality Improvement Division can conduct routine onsite support supervision to ensure adherence to treatment guides and other standard of care for example, intensive adherence counselling to individuals with high viral load, timely switch to appropriate regimens among others quality of care standard. This will also facilitate the process ensuring that all individuals on ART access a timely viral load and the results are correctly used for making decision in regard to patient care.
Following evidence from study that revealed high levels of pre-treatment drug resistance (PDR) estimated at 15.9% to NNRTIs, exceeding the threshold of 10.0% set by WHO for first line ARVs, the MoH of Uganda with guidance from WHO made a recommendation to substitute Nevirapine and Efavirenz with Dolutegravir among adults and LPV/R among children weighing less than 30 kg; and these changes were included in the national treatment guidelines in 2019. This process is referred to as ART optimization. The Ministry should therefore track the optimization process to ensure complete phase-out of the legacy regimens (Nevirapine (NVP) and Efavirenz (EFV)) since evidence shows that Dolutegravir (DTG) achieves viral Load Suppression better than the 2 NNRTIs.
Although the National HIV care and treatment guidelines recommend enhanced adherence counseling and support for patients whose first VL result is beyond the threshold, it is not clear whether the health care system is giving enough attention to these patients and adhering to the viral load algorithm in the management of patients with high very viral loads. Another study could be conducted to evaluate the compliance of health workers to the treatment protocols in the management of clients with high viral loads.
Study limitations
There were several limitations to the study.
There were cases of missing data especially on a number of parameters. For instance the variables of weight, clinical staging, Tuberculosis status, and functional status among others we often incomplete. Individuals who did not have data on such important variables were excluded in the analysis. Overall there were 657 individuals who had been switched to second line regimen between January 2012 and December 2017 but only 541 records were included in the analysis because of missing information. The possibility of not including all registered Patients in the analysis due to missing data may have created selection bias.
Finally, treatment failure was not confirmed with resistance testing, but rather the definition of treatment failure used in practice which may result in misclassification bias that can lead to an overestimate of the cases of treatment failure in the study.
Nevertheless, our results highlight the complex interplay between social and clinical factors contributing to ART adherence and subsequent risk of treatment failure among individuals on second-line ART.
Footnotes
Acknowledgements
We would like to acknowledge data collection team and the staff of Mbale Regional Referral Hospital-ART clinic for the assistance during the data collection process.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author received some research funds from NORAHED (SURVIVAL PLUS Project) and RUFORUM program.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MS, JBT, and JPMM: Conception of research protocol, study design, literature review, data extraction, data analysis, interpretation, and drafting the manuscript. DA contributed on data analysis, manuscript review, and edition and validation.
Ethics Approval and Consent to Participate
The study was approved by the Busitema University Faculty of Health Sciences Higher Degrees and Research Committee as well as the Mbale Regional Referral Hospital Research and Ethics Committee (Ref No. MRRH-REC-IN-COM 025/2018). Permission to conduct the study was further obtained from the Medical Director Mbale Regional Referral Hospital. Waiver of informed consent was attained because the study involved secondary review of existing medical records that were initially collected for purposes of patient care however privacy and confidentiality of personal information was observed. The tool used for data collection provided for numbers not names which ensured that no individual personal data was exposed and the date collected was stored securely to avoid any other people not involved in research from access. The research presented no risk of harm to subjects.
Availability of Data and Materials
The data that support the findings was from Mbale Regional Referral Hospital. The datasets used can be made available on reasonable request from the corresponding author.