Abstract
Background:
The majority of acute exacerbations of chronic obstructive pulmonary disease (AECOPD) are triggered by nonbacterial causes, yet most patients receive antibiotics. Treatment guided by procalcitonin (PCT), a sensitive biomarker of bacterial infection, safely decreases antibiotic use in many controlled trials. We evaluated PCT implementation for inpatients with AECOPD at a large academic hospital.
Methods:
All patients admitted for AECOPD during the first 6 months of PCT-guided therapy were eligible for inclusion in this retrospective cohort study. Patients with PCT performed were compared with those without PCT. The primary outcome was antibiotic days of therapy (DOT). Secondary outcomes included 30-day readmission and mortality.
Results:
Of the 238 AECOPD admissions, 73 (31%) had PCT performed. Procalcitonin-tested patients were more likely to meet systemic inflammatory response syndrome (SIRS) criteria, require intensive care unit (ICU)-level care, and have a longer length of stay (LOS) compared with those without PCT. Even after adjustment for these factors, PCT-tested patients received more inpatient DOT and there was no difference in total DOT. However, a low PCT value (<0.25 ng/mL) was associated with a 25.5% (P ⩽ .001) decrease in intravenous (IV) antibiotic DOT. Guideline-recommended follow-up testing was rare (12%). Procalcitonin measurement had no effect on 30-day readmission or mortality.
Conclusions:
In this real-world analysis of inpatients with AECOPD, PCT-guided therapy was poorly adopted by providers and was not associated with a decrease in total antibiotic DOT. However, a low PCT level was associated with a 25.5% decrease in IV antibiotic DOT, suggesting increased comfort stepping down from IV to PO therapy.
Introduction
Chronic obstructive pulmonary disease (COPD) affects 10% to 20% of Americans >40 years and is the third leading cause of death in the United States.1,2 Acute exacerbations of COPD (AECOPD), defined as acute worsening in baseline symptoms beyond normal daily variations warranting a change in therapy, are associated with significant morbidity and mortality. 3 When the exacerbation is severe enough to warrant admission, 85%-87% of patients receive antibiotics.4,5 Using antibiotics in severe AECOPD can decrease treatment failure and is consistent with the Global Initiative for Chronic Obstructive Lung Disease (GOLD) guidelines.3,6 However, the majority of AECOPD may not be impacted by antibiotics; 22% are noninfectious and only 55% of infectious etiologies are bacterial. 7
Overuse of antibacterial therapy is not without consequence. Selective pressure is a primary mechanism in creating resistant organisms. 8 Specific to respiratory illness, broad spectrum antibiotics have been linked to methicillin-resistant Staphylococcus aureus (MRSA), methicillin-resistant Streptococcus pneumoniae (MRSP), and fluoroquinolone-resistant Streptococcus pneumoniae.9-11 Furthermore, the intestinal dysbiosis from antibacterial therapy can nearly triple a patient’s risk of all-cause diarrhea and predispose them to Clostridium difficile colitis, a nosocomial pathogen responsible for nearly 30 000 deaths per year.6,12,13
Considering the unintended consequences of antimicrobial therapy and the often nonbacterial cause of AECOPD, a specific biomarker for bacterial respiratory infections would be valuable. Procalcitonin (PCT)—a precursor peptide stimulated from bacteria-specific pro-inflammatory mediators (IL1-β, tumor necrosis factor [TNF], and interleukin [IL]-6) 14 —is such a biomarker. 15 Since 2004, there have been multiple randomized trials and a Cochrane review showing that PCT-guided therapy safely reduces antibiotic exposure in acute respiratory illness (ARI).16-18 Specific to AECOPD, a decrease in antibiotic exposure was shown in a randomized trial (ProCOLD) 19 and recent meta-analysis. 20 From an economic standpoint, it has been estimated that widespread use of PCT-guided therapy in the United States in case of ARI could save US$1.6 billion annually. 21
Although PCT-guided antibiotic therapy shows promise for safely reducing antimicrobial use in AECOPD, its utility is extrapolated from predominantly randomized, controlled settings outside of North America. As hospitals increasingly introduce PCT to help antibiotic decision making, it is important to understand whether the benefits are achievable in real-world settings.
Materials and Methods
Patients and study design
We conducted a retrospective analysis using electronic records at a large academic hospital in Ann Arbor, Michigan. Data were collected over the first 6 months of PCT availability (October 1, 2013-March 31, 2014). Institutional rollout of PCT testing included dissemination of institutional PCT guidelines (Figure 1) to department chairs, making the guidelines available online, and education by infectious diseases (ID) faculty at general medicine, surgery, and emergency department (ED) weekly conferences. Inpatients ⩾18 years of age with an International Classification of Diseases, Ninth Revision 22 (ICD-9) principal admission diagnosis of AECOPD were eligible and records were manually reviewed to confirm the diagnosis. Patients transferred from outside facilities were excluded. Requirement for patient consent was waived and the study was approved by the University of Michigan Health System Institutional Review Board (HUM00085581).
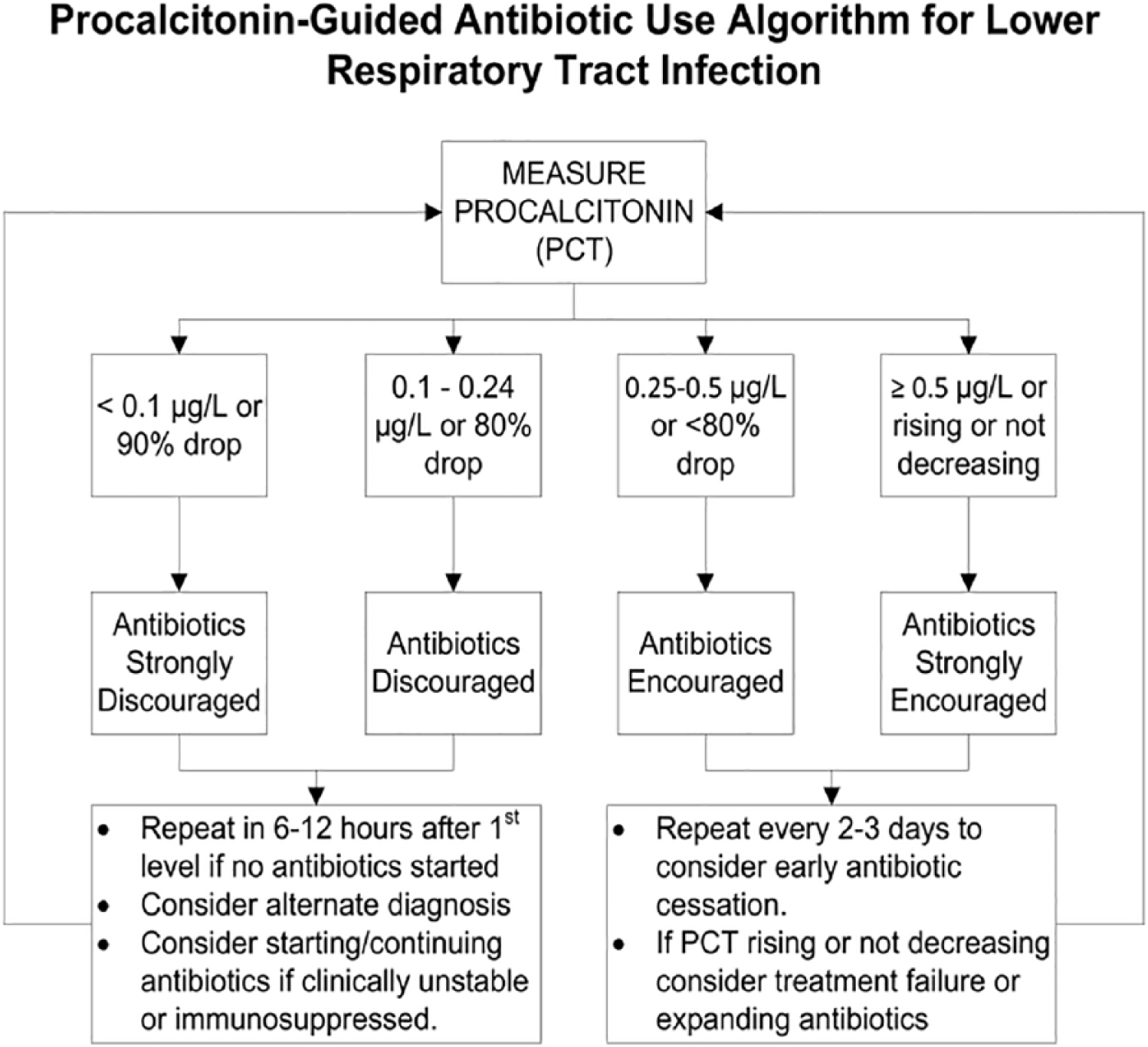
University of Michigan Health System (UMHS) procalcitonin-guided antibiotic use algorithm for lower respiratory tract infection.
Measurements
In addition to standard demographic data, antibiotic days of therapy (DOT) and type (intravenous [IV] or oral) were recorded. Patients on chronic antibiotics (N = 6) at admission were included, but days on chronic antibiotics were excluded. Days of therapy included antibiotics initiated in the emergency department or while inpatient. If a patient was discharged with outpatient antibiotics, the anticipated course based on prescriptions was added to inpatient DOT to give total DOT. Systemic inflammatory response syndrome (SIRS) criteria were defined in concordance with the American College of Chest Physicians/Society of Critical Care Medicine (ACCP/SCCM) 1992 conference. 23 Patients were classified into GOLD classes using forced expiratory volume in 1 second (FEV1) on prior pulmonary function testing (PFT) when available. 3 Acute exacerbations of COPD within the last year and home oxygen use was determined with detailed record review. Procalcitonin measurement was at the discretion of the admitting and inpatient physicians. Procalcitonin was performed from 0.5 mL of serum using the VIDAS BRAHMS PCT assay (bioMérieux, Inc., Durham, NC, USA), a US Food and Drug Administration (FDA)-approved automated immunofluorescent assay. Procalcitonin was considered low if ⩽0.25 ng/mL. When PCT was performed, time to re-testing was compared with institutional protocol (Figure 1).
Outcomes
Our primary outcome is antibiotic DOT. Secondary outcomes include protocol re-testing compliance, length of stay (LOS), 30-day readmission, and mortality.
Statistics
All analyses were conducted in R, version 3.2.2 (R Foundation for Statistical Computing, Vienna, Austria). Variables were examined for out-of-range values and histograms were used to visualize continuous variables. Descriptive statistics included proportions for categorical variables and measures of central tendency/spread for continuous variables. These initial analyses assisted in reconstructing variables for analysis, including logarithmic transformations for DOT and LOS due to the non-normal distribution of data points. P values <.05 were considered statistically significant for all analyses. To analyze relationships between predictor variables and the primary/secondary outcomes, simple linear or logistic regression was employed. Multiple linear or logistic regression models were employed for adjusted analysis and variables were included in the final models based on significance on unadjusted analysis. Backwards elimination was then employed to result in a final candidate model. Interactions were modeled and included if significant.
Results
PCT testing and outcomes
A total of 238 patients were admitted with AECOPD during the study period: 73 (31%) had PCT testing and 165 (69%) did not (Table 1). There were no significant age or gender differences between the 2 groups. Prior PFTs were available in 160 (67%) patients with no significant differences in FEV1 or GOLD classification. On unadjusted analysis, patients with PCT testing during the admission were more likely to be admitted to the intensive care unit (ICU; odds ratio [OR]: 2.21, 95% confidence interval [CI]: 1.02-4.81, P = .042), meet SIRS criteria (OR: 1.5, 95% CI: 1.09-2.06, P = .012), and have a baseline home oxygen requirement (OR: 2.24, 95% CI: 1.28-3.92, P = .005). In total, 171 (72%) patients received antibiotics while inpatient. Patients who underwent PCT testing were more likely to receive antibiotic therapy (OR: 3.85, 95% CI: 1.79-8.3, P = .001) and receive IV antibiotics (OR: 2.7, 95% CI: 1.51-4.8, P = .001), and these associations held after adjustment for the presence of SIRS and LOS. Procalcitonin testing had no effect on discharge antibiotic prescription rates (P = .493). On average, patients with PCT testing were admitted longer (5.4 ± 3.8 vs 3.1 ± 3.3; P < .001) and had more inpatient DOT (2.8 ± 1.8 vs 2.0 ± 1.4; P < .001) than those without PCT. Despite the differences in inpatient antibiotic DOT, PCT measurement had no effect on total DOT (P = .140; Figure 2). Only 9 (12%) patients with PCT testing had guideline-recommended re-testing. There was no difference in 30-day mortality (P = .213) or readmission (P = .514) after adjusting for LOS.
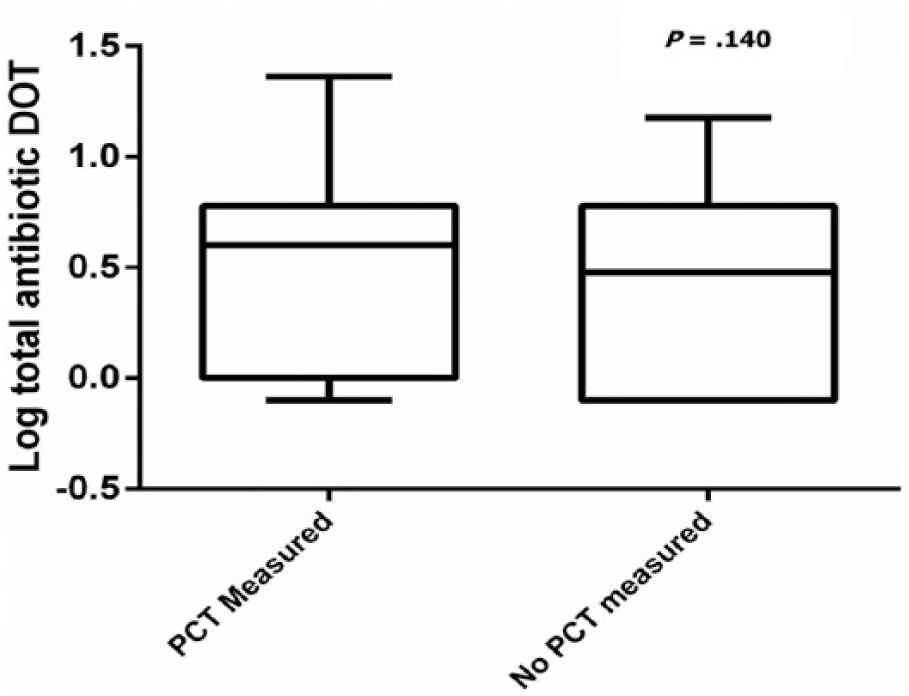
Patients with or without PCT measured versus log total antibiotic DOT. DOT indicates days of therapy; PCT, procalcitonin.
Characteristics and outcomes of patients with/without PCT measurement during their inpatient admission for AECOPD.
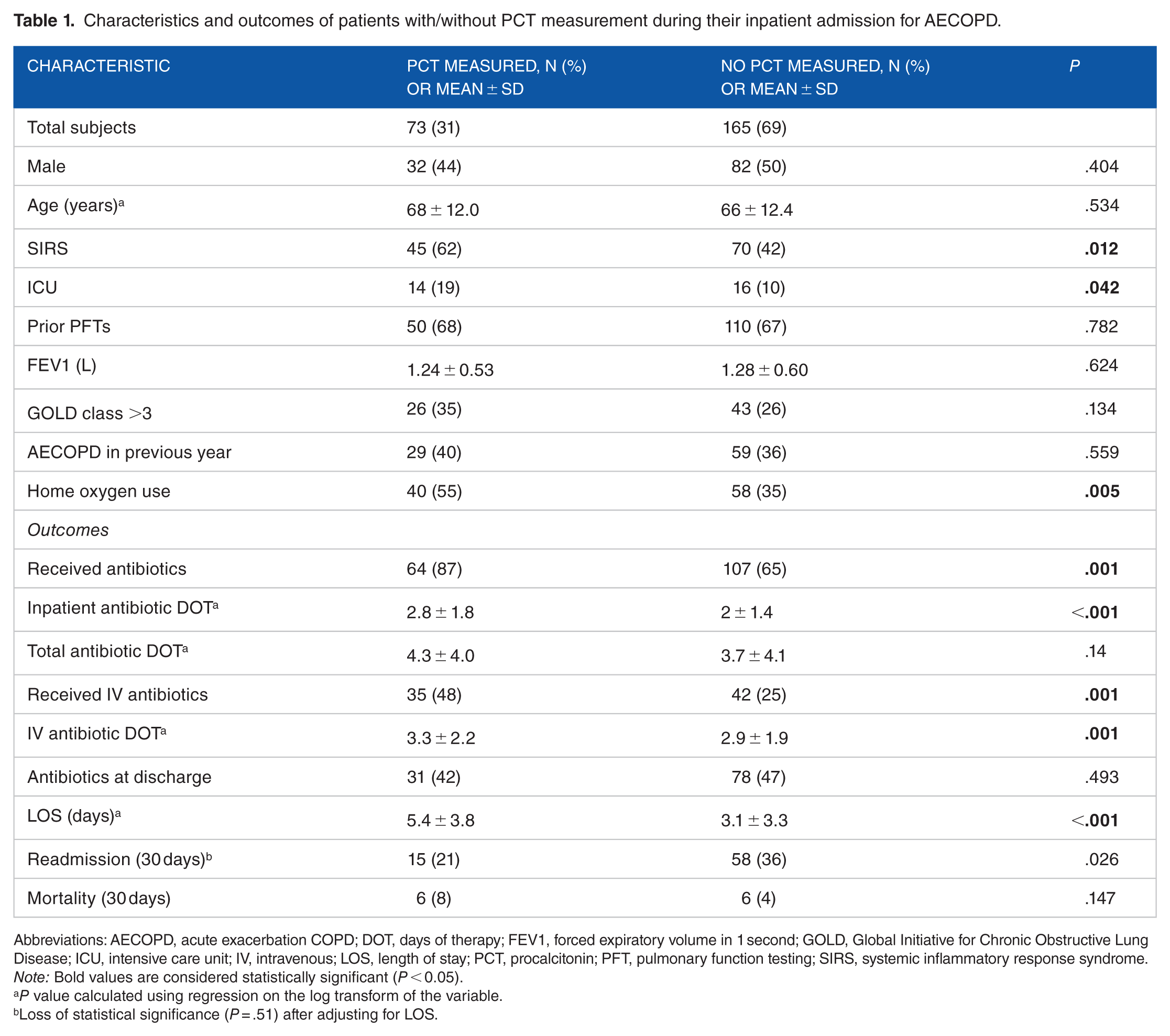
Abbreviations: AECOPD, acute exacerbation COPD; DOT, days of therapy; FEV1, forced expiratory volume in 1 second; GOLD, Global Initiative for Chronic Obstructive Lung Disease; ICU, intensive care unit; IV, intravenous; LOS, length of stay; PCT, procalcitonin; PFT, pulmonary function testing; SIRS, systemic inflammatory response syndrome.
Note: Bold values are considered statistically significant (P < 0.05).
P value calculated using regression on the log transform of the variable.
Loss of statistical significance (P = .51) after adjusting for LOS.
PCT level and outcomes
Patients with PCT testing were further divided into groups with low (⩽0.25 ng/mL) and high (>0.25 ng/mL) results; 57 (78%) of PCT results were low. All 16 patients with high PCT results received antibiotics, and 48 of 57 (84.2%) patients of the low PCT results received antibiotics. Despite high antibiotic imitation regardless of PCT level, a low PCT result was associated with a 27.2% lower (P < .001) IV antibiotic DOT (Table 2; Figure 3). This difference persisted after adjustment for SIRS criteria and LOS (Table 3). However, PCT level had no effect on inpatient DOT (P = .098) or total antibiotic DOT (P = .160). Furthermore, PCT level had no effect on use of antibiotics at discharge (P = .906), 30-day mortality (P = .747), or readmission rates (P = .237).
Selected a unadjusted analysis of IV antibiotic DOT.
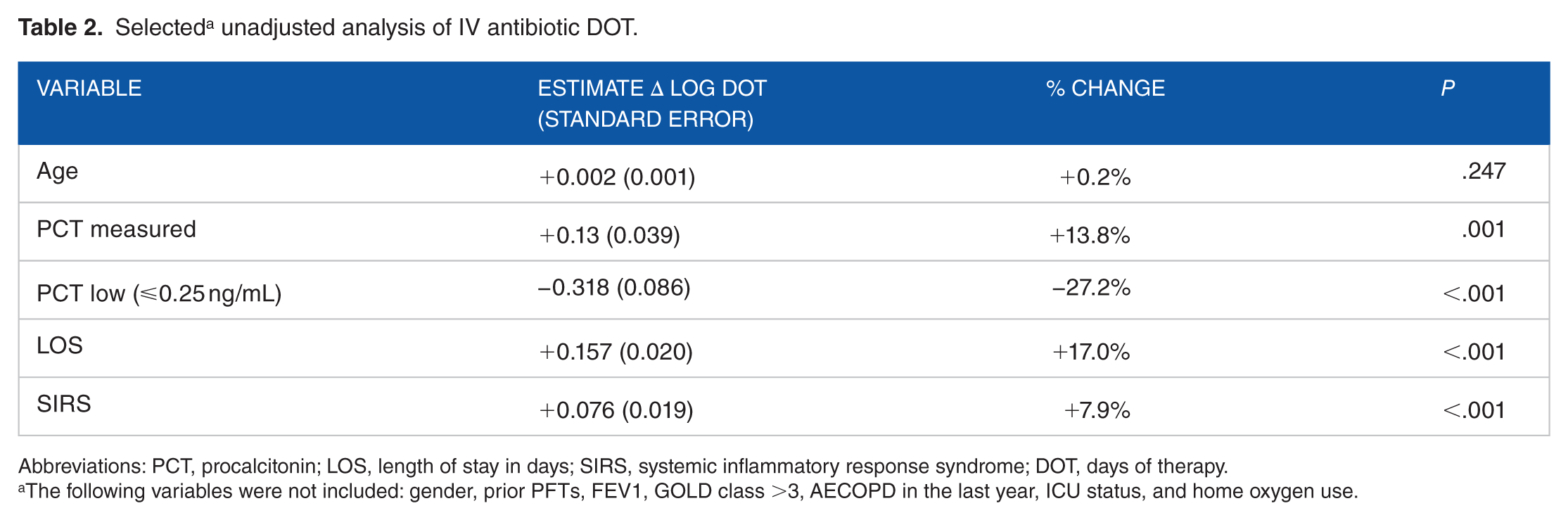
Abbreviations: PCT, procalcitonin; LOS, length of stay in days; SIRS, systemic inflammatory response syndrome; DOT, days of therapy.
The following variables were not included: gender, prior PFTs, FEV1, GOLD class >3, AECOPD in the last year, ICU status, and home oxygen use.
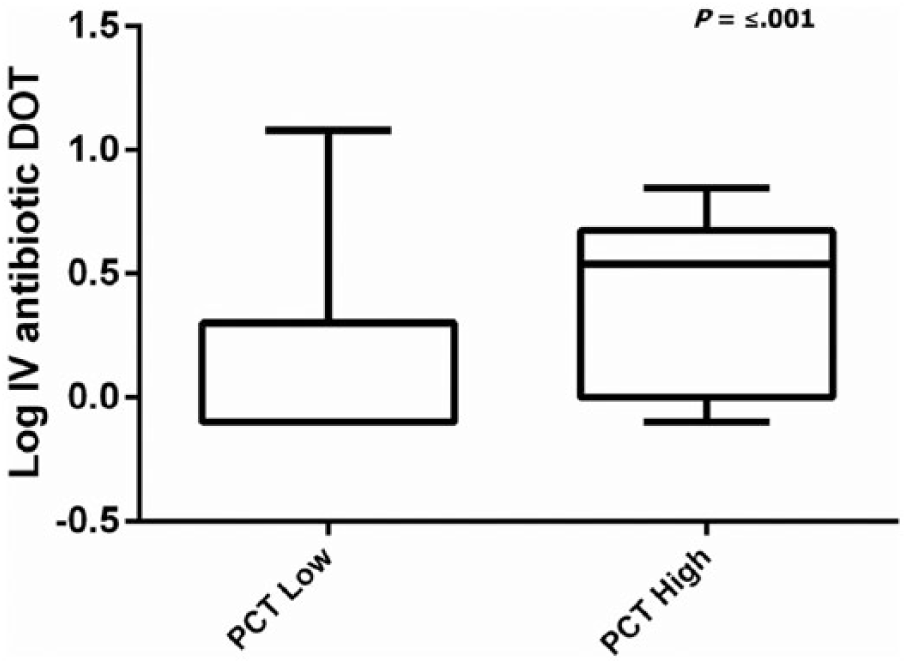
PCT level versus log total antibiotic DOT. Low PCT ⩽0.25 ng/mL. DOT indicates days of therapy; PCT, procalcitonin.
Adjusted multivariable analysis of IV antibiotic DOT.

Abbreviations: DOT, days of therapy; IV, intravenous; LOS, length of stay in days; PCT, procalcitonin; SIRS, systemic inflammatory response syndrome.
Discussion
During the introduction of PCT-guided therapy at our large academic hospital, patients admitted with AECOPD had PCT sent infrequently (31%), and those tested had higher acuity (SIRS, ICU) requiring longer admissions. Even after adjusting for these patient factors, PCT-tested patients received more inpatient DOT and there was no difference in overall DOT when compared with patients without PCT testing. Importantly, there was a 25.5% reduction in IV DOT after adjusting for LOS and SIRS. There was no difference in 30-day readmission or mortality.
Our results are contrary to many prior randomized controlled trials and prospective studies showing PCT-guided therapy decreases antibiotic utilization in respiratory illness.15-17,19,24,25 The discordant results are likely due to differences in guideline compliance; patients in prior studies were either randomized16,17,19 or required to have PCT testing on admission with a qualifying diagnosis.15,24,25 This bypasses the reality of provider-driven decision making. When given a choice, our results show that providers sent PCT infrequently, usually on patients with more severe disease. A possible explanation for this may be a greater awareness of PCT’s role in sepsis and ICU settings26-31 or mistrust in the reported test sensitivity. It could be the medical provider’s tendency to send more tests on sicker patients. 32 Irrespective of the cause, our results suggest that providers sent PCT to confirm an already high clinical suspicion for bacterial infection rather than using it as a tool to delineate bacterial infection from nonbacterial causes.
Our findings are concordant with the recent ProACT study, an open-label randomized controlled trial of PCT-guided therapy across 14 US hospitals of 1664 patients with lower respiratory tract infection (LRTI), 32% of which had AECOPD. 33 This trial found no decrease in antibiotic DOT between the PCT measurement and usual care. Similar to our study, clinicians’ adherence to PCT guidelines was poor (49.2% for COPD diagnoses) and a significant portion of patients with low PCT still received antibiotics. Our findings, together with ProACT, highlight the challenges with implementation of a PCT-guided model and suggest that achieving a PCT-driven decrease in antibiotic DOT requires high adoption rates for initial measurement and subsequent guideline-based therapy.
Regional differences are likely playing a significant role toward our results. In AECOPD, the sentinel study showing a benefit of PCT-guided therapy (ProCOLD) was conducted in Switzerland, 19 and a meta-analysis has shown a similar benefit in 8 trials conducted outside of North America. 20 Our study examined an academic institution in the United States, with a population and provider culture more similar to studies that failed to show PCT benefit for AECOPD: the ProACT trial 33 and a recent database review utilizing patients across 500 U.S. hospitals. 34 The reason for regional differences in PCT benefit likely ties back into guideline compliance. In the largest study examining PCT use in LRTI outside of trial settings, compliance was 76% in Switzerland and 74% in France, but only 34% in the United States. 25 The provider compliance in our study—with 31% of patients having an initial PCT—is concordant to these findings. Procalcitonin compliance may be higher in European countries due to greater familiarity with PCT-guided therapy and a difference in the health care culture surrounding guidelines; for example, compared with the United States, European guidelines are more likely to have implementation tools to help providers. 35
Despite poor guideline compliance and minimal effect on total antibiotic DOT, we found a 25.5% decrease in IV antibiotic DOT if PCT result was low. This persisted after adjustment for differences in patient acuity. Although a low result is highly suggestive of nonbacterial infection and the algorithm discourages antibiotics, providers were more comfortable “stepping-down” therapy from IV to oral. The utility of this is debatable, but any decrease in broad spectrum antibiotics is welcome from a stewardship perspective and is associated with less antimicrobial resistance. 36 Furthermore, with no difference in readmission or mortality between groups, testing is unlikely to cause harm.
A major strength of our study is our large cohort in a real-world setting, where consecutively admitted patients with AECOPD were all eligible for inclusion. Our cohort is larger than the randomized trial AECOPD arms16,17,19 and similar in size to the largest retrospective studies on PCT and antibiotics in AECOPD.37,38 To reduce confounding from facilities making antibiotic decisions without the option of sending PCT, we excluded patients transferred from outside facilities. To fully characterize the cohort, we included important COPD baseline disease factors such as PFT, home oxygen use, and recent AECOPD, which allowed determination of GOLD classification in the majority of patients.
Our study has several limitations. First, we used a retrospective cohort of AECOPD patients based off ICD-9-coded admission diagnoses. To ameliorate misclassification, we confirmed each patient’s diagnosis through extensive chart review, but it is possible that some admissions for AECOPD were not captured. In an effort to increase generalizability, we included both ICU and non-ICU patients. However, a recent trial has shown that PCT-guided antibiotic management in ICU patients admitted with AECOPD does not affect antibiotic exposure and may worsen mortality outcomes. 39 In the future, PCT testing should be limited to non-ICU inpatients with AECOPD. Much like the ProACT trial, our study was also limited by the implementation of PCT testing, algorithm availability, and provider education without active surveillance or prospective audit and feedback. Although active stewardship may increase compliance with PCT-based guidelines, we believe our design more realistically approximates testing rollout in a US health care environment where it can be difficult to obtain adequate funding for stewardship measures. 40 An additional limitation was our limited timeframe including the inpatient stay, outpatient discharge antibiotics, and 30-day outcomes; there may be benefits to PCT guidance extending beyond this time that were not realized. A final possible limitation was our inclusion of discharge azithromycin suppression of AECOPD, which could affect DOT, but may not be unnecessary as it can be effective over months to years. 41 However, the majority of prescriptions were for short-course (5-10 days) azithromycin therapy which has been shown to offer no benefit in AECOPD. 42
Current GOLD initiative and Veterans Affairs and the Department of Defense (VA/DoD) guidelines do not recommend testing PCT in AECOPD.3,43 Our study suggests that there are significant difficulties with implementing PCT-guided therapy in AECOPD, and we found no association with decreased antibiotic DOT. Nevertheless, a change in national guidelines with a subsequent increase in PCT compliance may be what is needed to have medical centers approximate the PCT benefit seen in prior AECOPD studies. Furthermore, our results suggest that current guidelines may be missing an opportunity to decrease IV antibiotic DOT when PCT returns low. Future analysis of secular trends is warranted as increased familiarity of PCT will increase use of this biomarker toward PCT-guided therapy; with this, the benefits observed in clinical trials may emerge in real-world settings of AECOPD.
Conclusions
In patients admitted with AECOPD, introduction of PCT-guided therapy was poorly adopted and was not associated with the antibiotic reducing effects seen in previous controlled trials. Our study highlights the difficulty with PCT implementation, as providers tested primarily higher acuity AECOPD patients requiring a longer LOS. Even after adjusting for these patient factors, PCT testing did not associate with a decrease in overall antibiotic DOT. However, patients with a low PCT received 25.5% less IV antibiotic DOT, suggesting comfort with stepping-down therapy and a possible benefit from an antimicrobial standpoint.
Footnotes
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr Rao was supported by grants from the Claude D. Pepper Older Americans Independence Center (grant number AG-024824) and the Michigan Institute for Clinical and Health Research (grant number 2UL1TR000433). Dr Flanders receives grant support from Blue Cross Blue Shield of Michigan and AHRQ and royalties from Wiley publishing. The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Authors’ Note
The abstract of this paper was presented at the Infectious Disease Society of America national meeting “IDWeek,” October 8-12, 2014, in Philadelphia, PA.
Author Contributions
Data Availability
All data contributing to this manuscript are available by request to the corresponding author.
