Abstract
Background
Omega-3 (n-3) polyunsaturated fatty acids (PUFAs) have modulating effects in several chronic inflammatory conditions. The aim of the present study was to test whether prior short-term dietary supplementation with n-3 (fish or seal oil) or n-6 (soy oil) PUFA rich oils would protect the development of dextran sulfate sodium (DSS)-induced colitis in rats.
Methods
Forty-eight male Wistar rats were divided into 6 groups: no intervention, sham, DSS, seal oil + DSS, fish oil + DSS and soy oil + DSS. Following 7 days of acclimatisation, 1 mL oil (seal, fish or soy) or distilled water (sham) was administered by gavage day 8 to 14. Colitis was induced by 5% DSS in drinking water from day 15 to 21. Rats were sacrificed on day 23. Histological colitis (crypt and inflammation) scores, faecal granulocyte marker protein (GMP) and quantitative fatty acid composition in red blood cells were measured.
Results
Pretreatment with fish or seal oils did not significantly influence DSS induced inflammation. In fact, all the oils tended to exacerbate the inflammation. Soy oil increased the mean crypt score (P < 0.04), but not the inflammation score or GMP. The ratio of n-6 to n-3 fatty acids (FAs) was 11 to 1 and 10 to 1 in standard diet and in red blood cells of control rats, respectively. Following administration of DSS, the ratio fell in all treatment groups (P < 0.001). The lowest ratios were seen in the groups receiving DSS + fish or seal oils (around 6 to 1).
Conclusion
Short-term pretreatment with fish or seal oils did not protect against subsequent induction of colitis by DSS in this rat model. Whether the high ratio of n-6 to n-3 FAs in the standard diet concealed effects of n-3 FA supplementation should be further investigated.
Introduction
Inflammatory bowel disease (IBD), which includes ulcerative colitis (UC) and Crohn's disease (CD), is characterised by increased mucosal production of several eicosanoids, such as prostaglandin (PG) E2, thromboxane A2, prostacyclin I2 and leukotriene (LT) B4 (1). Recently, dietary supplementation with omega-3 (n-3) polyunsaturated fatty acids (PUFAs) to IBD patients has received considerable interest because these fatty acids (FAs) have the potential to modify eicosanoid production and inflammation. However, the results of clinical trials are equivocal (2), (3).
In some prior studies, increased dietary intakes of n-3 FAs led to biochemical, histological and clinical improvements, both in patients with IBD and in experimental models of colitis (4)–(6). In one animal model, even maternal dietary n-3 FA supplementation impaired the development of colitis in the offsprings (7). However, a recent retrospective multicenter case-control study reported that total fat and fat-related nutrients, including monounsaturated FAs and PUFAs, vitamin E, and n-3 and n-6 FAs increased the risk for IBD, particularly for CD (8).
The inhibited production of metabolites of arachidonic acid, especially products of the 5-lipoxygenase pathway, LTB4, is considered the most important mechanism by which n-3 FAs influence inflammation (9). The general rule is that eicosanoids derived from n-3 PUFAs are less potent triggers of immunological and inflammatory reactions, compared to the corresponding compounds derived from n-6 PUFAs. Hence, increased dietary intake of n-3 PUFAs may shift the balance of the eicosanoid production to a putatively less inflammatory profile, such as PGE3 and LTB5. Both seal and fish oils are rich in n-3 PUFAs, but are different with respect to molecular localisation of the n-3 PUFAs on the triacylglycerol (TAG) molecule. N-3 PUFAs in seal oil are located almost exclusively at the end positions (sn-1 or sn-3 position) of the TAG molecule, whereas they are located mainly in the middle position (sn-2) of fish oil TAG (10), (11). During digestion and throughout the circulation, FAs are hydrolysed from the sn-1 and sn-3 positions by position specific pancreatic and lipoprotein lipases (12). The metabolism of structured TAGs is generally not influenced by different molecular localisation of n-3 PUFAs on TAG according to a recent review (13). However, it is not known whether effects of dietary TAGs on inflammatory and immunological reactions depend on molecular localization of n-3 PUFAs.
In man, incorporation of the fatty acids into cell membranes by oral administration of n-3 PUFA requires a long-term therapeutic strategy (14). Nevertheless, by duodenal administration, 10 days treatment with n-3 PUFA rich seal oil was enough to markedly alleviate joint paint in patients with IBD (15), (16). The intervention improved symptoms, fatty acid profile of red blood cells and rectal mucosa, and enhanced health related quality of life (17), but did not significantly change inflammation markers. In another double blind short-term (14-day) study using oral administration of seal oil and soy oil in psoriatic arthritis patients, we found no significant difference in symptomatic effects of the two oils (18). A major difference between the latter study in patients with psoriasis (18) and the prior studies in IBD-patients (15)–(17), was the mode of administration (oral versus duodenal).
Based on these short-term studies, we aimed to test whether prior short-term dietary supplementation with fish or seal oils (n-3 rich PUFAs), or soy oil (n-6 rich PUFAs) in addition to a background diet would influence the development of colonic inflammation following subsequent exposure to dextran sulfate sodium (DSS) in rats. To our knowledge, this is the first study where the effect of seal oil on intestinal inflammation is investigated in an animal model.
Material and Methods
Animals
Forty-eight adult male Wistar rats (Møllegaard and Blomholtgaard, Ry, Denmark), with mean weight 371 g, were singly housed in Macrolon III cages. Access to food, Beekay rodent low protein diet consisting of cereal products (88,5%), soy protein (6%), animal proteins (2,5%), soy oil (0,5%) and vitamin-, mineral- and amino acid-supplementation (2,5%) (B&K Universal AS, Nittedal, Norway), was ad libitum. The nutrient and fatty acid composition of the standard diet is given in Table 1. Tap water was given ad libitum if not otherwise stated. Animals were maintained at controlled temperature (21 ± 1 °C) with a 12 h light and 12 h dark cycle, relative humidity 55 ± 5% and 20 air changes per hour. The protocol was approved by the Norwegian Animal Research Authority.
Nutrients, energy and major fatty acids in the standard diet.
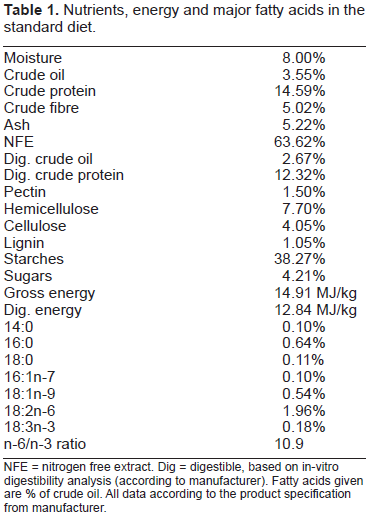
NFE = nitrogen free extract. Dig = digestible, based on in-vitro digestibility analysis (according to manufacturer). Fatty acids given are % of crude oil. All data according to the product specification from manufacturer.
Induction of Dss colitis
Acute colitis was induced by 50 g/L of DSS (MW 44 000, TdB Consultancy AB, Uppsala, Sweden) given in distilled drinking water for 7 successive days. The solution was refreshed every second day and consumption was accordingly measured.
Experimental Design
Following 7 days (day 1–7) of acclimatisation (daily handling for 5 minutes by the same individuals), the 48 animals were randomly divided into 6 groups of 8; group 1: control (no intervention), group 2: sham (1 mL distilled water once daily by gavage day 8–21), group 3: DSS, group 4: soy oil + DSS, group 5: seal oil + DSS and group 6: fish oil + DSS. In the last three groups, first, oil was given by gavage for 7 days (day 8–14), and then the rats were exposed to DSS (day 15–21) without oil gavage. Gastric gavage was performed by using a feeding tube (CH 06, 50 cm, Maersk). Animals were held in the scruff of the neck and the feeding tube was placed into the stomach (around 8 cm from the tooth row).
The seal oil (Rieber Skinn A/S, Bergen, Norway) was a mixture of crude oil from harp seal (Phagophilus groenlandicus) and hooded seal (Cystophora cristata). The seal oil group received 0.07 g of eicosapentaenoic acid (EPA, 20:5n-3), 0.03 g of docosapentaenoic acid (DPA, 22:5n-3) and 0.07 g of docosahexaenoic acid (DHA, 22:6n-3), the fish oil group (cod liver oil, Peter Møller, Oslo, Norway) received 0.08 g of EPA, 0.01 g of DPA and 0.12 g of DHA, and the soy oil group (Mills DA, Oslo, Norway) received 0.5 g of linoleic acid (LA, 18:2n-6) and 0.06 g of α-linolenic acid (ALA, 18:3n-3) per day. The FA composition, levels of vitamin A and E and lipid peroxidation assessed by an in vitro method determining thiobarbituric acid reactive substances (TBARS) in the seal, fish and soy oils are presented in Table 2.
Major fatty acids (g/100 g), vitamins A and E and TBARS.
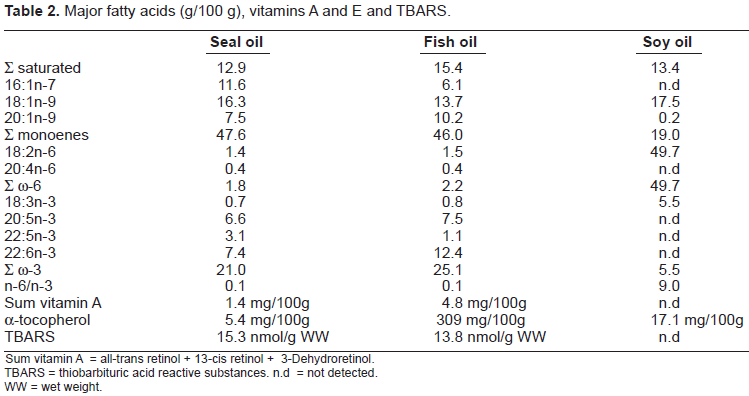
Sum vitamin A = all-trans retinol + 13-cis retinol + 3-Dehydroretinol.
TBARS = thiobarbituric acid reactive substances. n.d = not detected.
WW = wet weight.
On day 23, all rats were anaesthetised by subcutaneous injection of a combination of fentanyl citrate + fluanisone (Hypnorm®, Janssen Pharmaceutica, Beerse, Belgium) and midazolam (Dormicum®, Roche, Oslo, Norway) (Fentanyl citrate 0.079 mg/mL, fluanisone 2.5 mg/mL, midazolam 1.25 mg/mL), at a dose of 0.2 mL/100g. Thoracotomy, cardiac puncture and exsanguination were performed.
Sample Collection and analysis
Blood was drawn into a heparin-containing Vacutainer, placed on ice for 10 minutes and then centrifuged at 2000 rpm for 10 minutes at 4°C. Aliquots of red blood cells were stored at -80 °C for future analysis.
Quantitative FA composition was analysed in red blood cells (using biological sample parallels) and oils. The samples were dissolved in chloroform/methanol (2:1, w/w) and a 19:0 methyl ester was added as internal standard. The samples were filtered, saponified and methylated using 20% BF3 in methanol. FA composition of total lipids was analysed using methods described by Lie and Lambertsen (19) where the methyl esters were separated using a Trace GC 2000 gas chromatograph (“cold on column” injection, 6025 °C/min16025 °C/min190 25 °C/min 220 °C), equipped with a 50 m CP-sil 88 (Chrompack) fused silica capillary column (id: 0.32 mm). The FAs were identified by retention time using standard mixtures of methylesters (Nu-Chek, Elyian, USA), and the quantitative FA composition was calculated using an integrator (Totalchrom), connected to the Gas Liquid Chromatography (GLC)-system.
Faeces were collected on day 22 and then stored at -20 °C until analysis of granulocyte marker protein (GMP), a marker of intestinal inflammation. Briefly, 1 g of faecal material was diluted in 4 mL extraction buffer and thoroughly homogenized using an Ultra Turrax (20,000 rpm) before centrifugation at 45000 g for 20 min. The upper halves of the supernatants were carefully harvested and ran on a standard one-step ELISA as described previously (20).
Tissue Preparation and Histological assessment
Colon from the colocecal junction to the anal verge was removed for histological examination. The length of the colon was recorded. The colon was rinsed with phosphate-buffered saline buffer, opened longitudinally and divided into one proximal and one distal segment, which were fixed in 10% formalin and embedded in paraffin. Eight pieces per segment were stained with haematoxylin and eosin.
Colonic crypt and inflammatory scores were determined according to a validated scoring system (21), and examined by a pathologist unaware of the experimental protocol. Crypt injury was scored as; grade 0: intact crypts, grade 1: loss of bottom one third of crypts, grade 2: loss of bottom two thirds of crypts, grade 3: loss of entire crypt with the surface epithelium remaining intact, grade 4: loss of the entire crypt and surface epithelium. The severity of inflammation was scored as; grade 0: normal, grade 1: focal inflammatory cell infiltration, grade 2: inflammatory cell infiltration, gland dropout and crypt abscess. Both scores include a measure of involvement as follows; grade 1: 1–25%, grade 2: 26–50%, grade 3: 51–75%, grade 4: 76–100%. The score is the product of either the crypt or inflammation grade by the involvement grade. The final crypt and inflammation scores are the averages of all individual scores of 16 pieces per colon.
Statistical Analysis
Data were analysed using the GraphPad Prism version 4 (GraphPad Software Inc., USA) statistical software package. Results are presented as mean ± standard error of mean. Differences between means were evaluated with one-way ANOVA and Bonferroni post-test for selected pairs of columns. P values less then 0.05 were considered statistically significant.
Results
There were no significant differences in any parameter between the control (no intervention) group and the sham group. These two groups were therefore merged into one group called controls in the presentation of results. One rat in the seal oil group bit off a piece of the distal part of the feeding tube on day 14, but the tube was visible at the upper part of larynx and was removed under isofluran anaesthesia. The experiment was completed without mortality.
Dss Intake
Total intake of water with DSS was similar in the DSS group (157 ± 11.5 mL) and the oil + DSS groups (soy oil + DSS: 146 ± 14.2 mL; seal oil + DSS: 157 ± 10.7 mL and fish oil + DSS: 152 ± 8.1 mL).
Weight Change
From the start of DSS treatment to sacrifice, weight loss was observed in all groups (DSS: –11.3 ± 4.0 g; soy oil + DSS: –7.6 ± 2.3 g; seal oil + DSS: –14.1 ± 4.2 g and fish oil + DSS: –23.4 ± 6.7 g), except in control rats (1.4 ± 1.9 g). Weight change did not significantly differ between controls and rats on DSS, or between groups receiving DSS and oils.
Colon Length
Colon length did not significantly differ neither between controls (17.6 ± 0.4 cm) and DSS group (16.8 ± 0.4 cm) nor between DSS and oil + DSS groups (soy oil + DSS: 16.3 ± 0.4 cm; seal oil + DSS: 16.1 ± 0.5 cm and fish oil + DSS: 15.4 ± 0.6 mL). However, there was a tendency to shorter colons in groups with colitis, especially in fish oil + DSS, as compared to controls.
Histology of the colon
Microscopic evaluation of colonic sections showed indications of colitis in all rats receiving DSS. Changes were most prominent in the distal colon with areas of erosions, crypt distortion and inflammatory cell infiltration as previously described (22) (Fig. 1). Soy oil significantly increased mean crypt score (p < 0.04), but not mean inflammation score, in rats with DSS-induced colitis. Such significant change was not seen in rats receiving n-3 FAs (seal or fish oil) + DSS (Fig. 2).

H&E, 10X. a) Histologic appearance of the normal colonic mucosa of healthy control rat (intact crypts). b) Colonic mucosa of rat with DSS colitis showing crypt loss, inflammatory infiltration and crypt abscesses. c) Colonic mucosa of a rat which received soy oil and DSS. d) Colonic mucosa of a rat which received seal oil and DSS. e) Colonic mucosa of a rat which received fish oil and DSS (c, d and e show similar findings as in b).
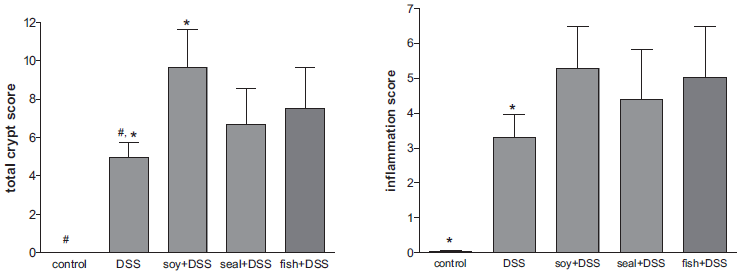
Effect of DSS and n-3 or n-6 polyunsaturated fatty acids on histological crypt and inflammation scores. Results are expressed as mean ± SEM. Symbols mean significant difference, p < 0.05.
Granulocyte Marker protein
After 7 days of DSS intake, faecal GMP levels increased significantly in rats on DSS (63.6 ± 19.5 mg/L) as compared with controls (7.8 ± 2.6 mg/L) (p < 0.01). There was no significant difference between DSS and oil + DSS groups. However, high faecal GMP levels were observed in rats receiving seal (84.8 ± 25.1 mg/L) and fish oils (88.5 ± 28.1 mg/L), but not soy oil (51.6 ± 14.9 mg/L).
Fatty Acid composition
Compared with controls, there was a significantly lower n-6 to n-3 FA ratio in red blood cells of DSS and DSS + oil groups (p < 0.001) (Table 3).
Fatty acid composition in red blood cells (mg/g) in study groups. Means (SEM).
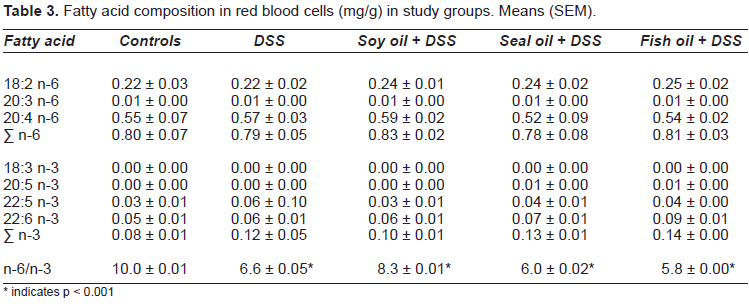
indicates p < 0.001
Discussion
In the present study short-term pretreatment with either n-3 PUFA rich seal or fish oils as a supplement to an n-6 PUFA rich (soy based) standard diet did not protect against subsequent induction of colitis by DSS in rats. Contrary to what we hypothesized, the administration of all PUFAs tended to aggravate the inflammation. The oil-induced changes were not significant except for crypt injury score in the group receiving soy oil.
Based on the results from our previous studies (15)–(18), we expected a protective effect of short-term administration of n-3 PUFAs on the development of colonic inflammation. The lack of protection against colitis might be due to the short duration of oil supplementation (7 days) and the low dose of n-3 PUFAs given as a supplement to an n-6 PUFA rich standard diet. Other studies used at least 13 days oral administration or 2 weeks rectal enema (4), (11), (3) allowing the fatty acids to be incorporated into membranes. Such incorporation into red blood cells needs several weeks (14).
It is generally recommended that the human diet should return to a more balanced ratio of n-6 to n-3 FA (24). A ratio closer to 4 to 1 rather than the ratio of 20–15 to 1 as provided by current Western diet is supposed to be beneficial also for patients with IBD. An epidemiological study found increased intakes of n-6 FAs with a concomitant increase in the ratio of n-6 to n-3 FAs to be associated with increased occurrence of CD (25). A recent clinical study found reduced remission rates with increasing fat content when soy oil-based enteral feeds were administered to patients with active CD (26). Induction of colitis by DSS decreased the ratio of n-6 to n-3 FAs in all groups, possibly due to consumption of n-6 FAs for generation of pro-inflammatory prostaglandins and leukotriens. Even with n-3 PUFA rich seal or fish oils supplementation, the ratio remained high (around 6 to 1). Possibly due to the fact that the rats’ standard diet had a high n-6 to n-3 FA ratio (11 to 1), both baseline (control groups) and final n-6 to n-3 ratios (experimental groups) were too high to disclose potential protective effects of n-3 PUFA rich oil supplementation.
DSS in the drinking water is a widely used experimental model of colitis (27). There are few studies testing effects of fish oil in DSS-induced colitis. Camuesco et al reported beneficial effects of adding fish oil to an olive oil diet (28). In another study, the effect of fish oil was compared with Lyprinol (Lipid extract of New Zealand Green Lipped Mussel) and olive oil in mice (23), (29). The results showed no significant benefit of fish oil compared with Lyprinol or olive oil. In our study, fish oil was richest in n-3 PUFAs and provided the lowest n-6 to n-3 ratio in blood, but still the fish oil + DSS group lost most weight, had the shortest colon and the highest GMP value (not significant). The level of α-tocopherol, added as an antioxidant for protection of PUFAs against lipid peroxidation, was in fact 57 times higher in the fish oil compared with the seal oil with quite similar TBARS values. Whether the crude seal oil contained additional beneficial substances compared with refined marine oils is not known. Amending regulations on contaminants prohibit the use of crude marine oils in clinical trials.
Faecal GMP, which corresponds to calprotectin in humans, has been used to monitor gut inflammation (30), (31). GMP extracted from faeces is a non-invasive marker of gastrointestinal inflammation in rats, and is derived from neutrophils and monocytes. In our study, exposure to 5% DSS induced histological inflammation, elevated faecal GMP concentration and loss of weight similar to that reported by others (27), (31). Treatment in all oil groups tended to aggravate the colitis, however, the change was significant in the soy oil group only. The significantly increased crypt injury score with soy oil might indicate a deleterious effect of soy oil because loss of crypts is a typical and early sign of DSS induced damage (32). Notably, while rats may convert ALA to EPA and DHA rather easily, humans can not. Relatively high ALA concentrations in soy oil may therefore mask deleterious effects of a high LA diet much more in rats than in humans (33). The oils were not oxidised according to TBARS measurement, and thus probably did not cause oxidative injury. Whether the use of a gastric gavage might have stressed the rats and thus contributed to the more severe colitis in the DSS + oil groups compared with DSS group is possible since stress is known to contribute to the severity of both experimentally induced colitis and ulcerative colitis (34). Control (no gavage) and sham (with gavage day 8–21) groups was however not different, but we have no additional sham + DSS group enabling us to conclude whether the oils per se or the gavage procedure per se, contributed to the development of colitis.
In conclusion, short-term pretreatment with seal or fish oils as a supplement to a n-6 PUFA rich (soy based) standard diet did not protect against subsequent induction of colitis by DSS in this rat model. Further studies are required to elucidate the effects of different n-6 to n-3 FA ratios in the diet and tissues before subsequent induction of colitis by DSS.
Footnotes
Acknowledgement
The study was funded by The Norwegian Fishermen Association, The Royal Ministry of Fisheries and Costal Affairs, NIFES and Haukeland University Hospital. Georg Olsen and Annbjørg Bøkevoll are thanked for excellent analytical work. Oddrun Gudbrandsen and Hege Wergedahl are thanked for excellent technical work during the study. Rieber Skinn A/S supplied free samples of seal oil.
