Abstract
Cell therapies using adipose-derived stem cells (ADSCs) have been used to treat inflammatory bowel disease (IBD) in human and dog. We previously reported the CellSaic technique, which uses a recombinant scaffold to enhance the efficacy of cell therapy. To examine whether this technique can be applied to cell therapy for colitis, we evaluated the efficacy of CellSaic in colitis mouse models. Colitis mouse models were developed by administering dextran sulfate sodium (DSS) to C57BL/6 mice for 7 days. Then CellSaic comprising human/canine ADSCs (1.2 × 106 cells) or human/canine ADSCs only (1.2 × 106 cells) were administered to the mice. The body weights were measured, and the colon length measurements and histological evaluations were conducted at 7 days after administration. After
Keywords
Introduction
Human and veterinary therapies are considered to be integrated. Synergy between veterinarians, physicians, and other scientific health and environmental professionals has been promoted in an initiative known as “One Health,” which is meant to improve the lives of all species by integrating human and veterinary medicine 1 . The “One Health” initiative aims to efficiently transform medicine, particularly the field of regenerative medicine, for all species. Moreover, the role of veterinary patients in the evolution of stem cell therapies for both human and animal patients will be explored 2 –5 .
Stem cell therapies for both human and animals have been studied for regenerative medicine, mainly adipose-derived stem cells (ADSCs) and bone marrow mesenchymal stem cells (BMSCs). There are numerous reports of cell therapies using ADSCs and BMSCs in humans 6 –8 . In veterinary regenerative medicine, autologous ADSC therapy has been commercially available since 2003 9 . Previously reported results from a blinded, controlled trial in dogs with chronic osteoarthritis of the coxofemoral joint demonstrated efficacy of a single intraarticular injection of autologous ADSC therapy 10 .
Therapies using ADSCs and BMSCs are beneficial for treating inflammatory bowel disease (IBD), a chronic relapsing disease in which pro-inflammatory immune cells and cytokines induce intestinal tissue damage and disability 11 –14 . In a clinical study of humans, more than 200 patients were treated by local injections of mesenchymal stem cells (MSCs), resulting in a complete response in more than half of the patients and overall response in approximately two-thirds of the patients 11 . In a clinical study of dogs, 11 dogs with confirmed IBD received one intravascular infusion of MSCs. Clinical remission occurred in 9 of the 11 dogs at day 42, while the 2 remaining dogs demonstrated a partial response 15 .
Wang et al. examined intravenous injection (i.v.), intraperitoneal injection (i.p.), and anal injection (a.i.) as methods for administering BMSCs. They found that i.p. leads to a better recovery from colitis and may be the optimum BMSC delivery route for treating dextran sulfate sodium (DSS)-induced colitis 16 . Chen et al. reported that interferon BMSCs, which express interferon-γ, more efficiently ameliorated DSS-induced colitis in mice, showing regained colitis-related loss of body weight, increased colon length, decreased disease activity index, and improved tissue structure of the small intestine 17 . As for other therapies using MSC for colitis, especially cell-free therapies, Legaki et al. have reported that conditioned media from MSC ameliorate DSS-induced colitis in an immunodeficient mouse model 18 . Therefore, paracrine effects by secreted molecules from MSC may have the therapeutic effects on DSS-induced mice.
In this study, we considered using a scaffold as an approach to therapy. Scaffolds are important for adherent cells because apoptosis is induced by lack of cell/scaffold attachment
19,20
. We previously reported a new cell transplantation platform, CellSaic, derived from cell- and scaffold-forming mosaic, and using a bioabsorbable biomaterial known as recombinant protein (RCP), which was developed by the FUJIFILM Corporation (Tokyo, Japan). We confirmed that cell viability in the BMSC CellSaic was higher than that in the spheroid
As described above for “One Health,” we also considered that the clinical study of CellSaic for dogs (companion animals) would enhance the development of these methods for humans. Therefore, we evaluated the efficacy of the CellSaic technique in mouse models of colitis using human or canine ADSCs. We assume this study as a step forward to prove advantages of scaffolds in treatment of disease models
Materials and Methods
Materials
Human ADSCs (hADSCs) were purchased from Lonza (PT-5006, Basel, Switzerland). The medium for hADSC was Bulletkit ADSC (PT-4505, Lonza). cADSCs were generated as previously described
22
. Briefly, adipose tissue from a TOYO beagle dog (female, 12 months old) was cut into small pieces and digested at 37°C for 1 h with 2 mg/mL collagenase (Nitta gelatin, Tokyo, Japan) in Dulbecco’s phosphate-buffered saline. The sample was centrifuged at 600 ×
Formation of hADSC/cADSC CellSaic Platforms
hADSCs were cultured at 37°C in a 5% CO2 humidified atmosphere. CellSaic platforms, containing hADSCs, were prepared by mixing hADSCs (3 × 105 cells/mL) and RCP pieces (0.25 mg/mL) in Bulletkit ADSC medium; this mixture was seeded on a 35-mm dish of EZ SPHERE 4000-903SP (AGC Techno Glass, Haibara, Japan).
For cADSC CellSaic formation, hADSCs in Bulletkit ADSC medium were prepared by mixing 3 × 105 cells/mL and RCP pieces (0.0625 mg/mL). The mixture was then seeded onto an EZ SPHERE 4000-903SP. Each dish was incubated for 72 h. hADSC and cADSC CellSaics were collected into 400 µL of the respective medium and used for implantation.
Efficacy test (Fig. 1A)
Creation of the colitis model
Acute colitis was induced in C57BL/6 female mice by feeding of 3% DSS (MP Biomedicals, Santa Ana, CA, USA) dissolved in water for 7 days, followed by 7 days of regular drinking water.
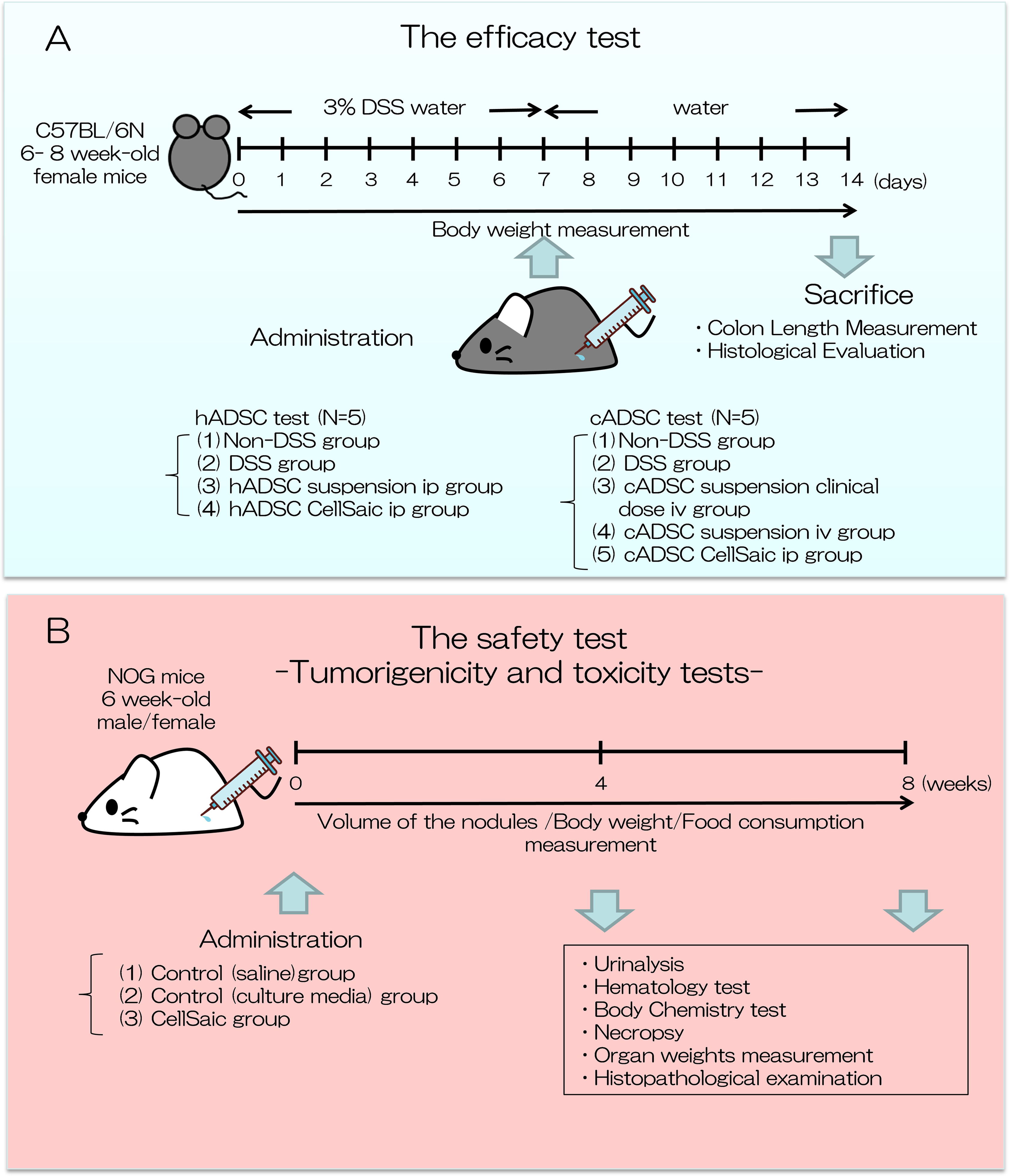
Experimental design of this study. We conducted an efficacy test and safety test of ADSC CellSaic. (A) Efficacy test: therapeutic effects of hADSC/cADSC CellSaic, evaluated in DSS-induced mouse model, compared with hADSC/cADSC suspension; Body weight was measured. Seven days after administration, mice were sacrificed, colon length was measured, and histological evaluation was conducted. (B) Safety test: Tumorigenicity and toxicity tests were conducted. cADSC CellSaic evaluated in NOG mouse. Volume of nodules, body weight, and food consumption were measured until 8 weeks. Four and 8 weeks after administration, urinalysis, hematology test, body chemistry test, necropsy organ weight measurement, and histopathological examination were conducted.
Administration of CellSaics and suspension
At day 7 after administration of DSS water, the mice were randomized and injected with implants intraperitoneally using an 18G needle or intravenously under general isoflurane anesthesia. The Animal Care Committee of FUJIFILM Corporation approved the experimental protocol, and all experimental procedures used in animal studies were performed in accordance with international guidelines.
Groups
For the hADSC test, transplantation was performed using the same i.p. injection method to compare the efficacy of the hADSC CellSaic and hADSC suspension. The study groups were as follows. Non-DSS control group ( DSS control group ( hADSC suspension i.p. group ( hADSC CellSaic i.p. group (
For the cADSC test, the efficacy of i.p. injection of cADSC CellSaics was compared with that of i.v. injection of cADSC suspension used at a clinical dose. Clinical dose cell counting was conducted as described previously
15
. To assess the efficacy of cADSCs by the cell counting method, i.v. administration of cADSCs, at the same cell count as cADSC CellSaics, was also conducted. Non-DSS control group ( DSS control group ( cADSC suspension clinical dose i.v. group ( cADSC suspension i.v. group ( cADSC CellSaic i.p. group (
Outcome Measurements
Weight recovery
For evaluation, the body weights were recorded daily. The weight recovery percentage was calculated as (the body weight at the point) / (the initial body weight) × 100.
Colon length
Seven days after transplantation, the mice were sacrificed, and colon length measurements and histological evaluations were conducted. After the colon was removed, the colon length was measured from the caecum to the anus.
Histological evaluation of inflammatory score
To assess the inflammatory level, histological sections were prepared 7 days following implantation. Colon tissues were removed, cut into three parts (upper colon, lower colon, rectum), and fixed with 10% phosphate-buffered formalin. Hematoxylin and eosin (H&E)-stained sections were prepared for histological examination, with the center of the three parts facing the outside. The inflammatory scores of the colon sections were calculated using a scoring system. (A) Depth of ulceration, (B) area of ulceration, (C) edema, and (D) infiltration were scored individually for the samples using a graded scale of 0–4, as described in Table 1. The scores were reviewed by a pathologist and veterinarian at FUJIFILM Corp. The histological score was defined as the sum of the four parameters (A+B+C+D) of the three sections and represents the average of the effective values.
Scoring System for Histological Evaluation.

In Vitro Analysis
Cell culture level of TSG-6 analysis by ELISA
The TSG-6 analysis was performed as described previously
23
. hADSC CellSaic was prepared by mixing hADSCs (5 × 104 cells/mL) and RCP pieces (0.05 mg/mL) in DMEM; this mixture was seeded on a PrimeSurface 96U plate (Sumitomo Bakelite Co. Ltd., Tokyo, Japan) in 200 μL wells. For spheroid formation, hADSCs in DMEM were prepared using 5 × 104 cells/mL, and the cells were seeded on a PrimeSurface 96 U plate in 200 μL wells. Each plate was centrifuged using a tabletop plate centrifuge (600 ×
Safety Test
Tumorigenicity and Toxicity Tests (Fig. 1B)
The test was conducted by Nihon Bioresearch Center, Inc. (Hashima, Japan).
Population
Male and female NOG mice at 6 weeks of age (CLEA Japan, Inc., Meguro-ku, Japan) were used in the test; 36 male mice and 36 female mice were used in this test.
Groups
Mice were grouped as follows. Note that male group and female group were respectively prepared for each of the three groups. Control (saline) group ( Control (culture media) group ( CellSaic group (
Physiological saline (Otsuka Pharmaceutical Co., Ltd., Tokyo, Japan), culture medium used for cADSCs, or CellSaics (107 cells cADSC and 1.04 mg µ-pieces) was administered at a volume of 0.4 mL to the right lateral abdomen of mice under anesthesia using a 23G injection needle.
Outcome measurements
After administration, the sizes of subcutaneous nodules were measured over time using a digital caliper (CD-15, Mitsutoyo Corporation, Kasugai, Japan) until 56 days after administration. Volume of nodules was calculated using the following formula: (long diameter × short diameter2)/2. The means and standard deviations of subcutaneous volume of nodules were calculated for each group. Other details related to general toxicity study are described in the Supplemental Materials and Methods.
Statistical Analyses
The results are presented as the means ± standard deviation. Colon length and histological score were analyzed using the
Results
hADSC CellSaic Recovers Body Weight and Colon Length in DSS-Induced Mice
We examined the anti-inflammatory effects of hADSC CellSaic using a DSS-induced model of colitis. Oral administration of 3% DSS for 7 days induced acute colitis in C57BL/6 mice, which was confirmed by the reduction of body weight (Fig. 2A). In the DSS group that did not receive cell injection, body weight decreased to 86% at day 11 and recovered to only 93% at day 14. In the hADSC suspension i.p. group, following administration of hADSCs alone at day 7, body weight decreased to 84% at day 11 but recovered to 97% at day 14; this group showed little improvement in body weight recovery. In the hADSC CellSaic i.p. group, body weight increased from day 8 to 11, recovering to 94% at day 11 and 101% at day 14, reaching 105% of the non-DSS group. The hADSC CellSaic i.p. group showed significant differences in body weight on days 13 and 14 (

hADSC CellSaic attenuates recovery of body weight and colon length. (A) Percentage of body weight changes. Non-DSS group [no treatment with DSS,
hADSC CellSaic Decreases Ulceration and Inflammatory Cells
We evaluated pathological sections to determine the depth and area of ulceration, edema, and infiltration of inflammatory cells. Pathological specimens are shown in Figs. 3A and 3B. There was no ulceration or infiltration in the non-DSS group. In the DSS group and the hADSC suspension group, ulceration and infiltration were more severe than in the hADSC CellSaic group. Scoring results are presented in Fig. 3C. Mean total scores were 0 in the non-DSS group, 13.8 in the DSS group, 15.8 in the hADSC suspension i.p. group, and 6.6 in the hADSC CellSaic i.p. group, confirming the narrower area of ulceration, smaller areas of edema, and reduced infiltration of inflammatory cells in the hADSC CellSaic i.p. group. Histological examination showed that hADSC CellSaic attenuates inflammation and disruption of the crypt architecture.

ADSC CellSaic decreased inflammatory cells and repaired ulceration in histological analysis. Histopathological comparison of colitis in lower colon at day 14. There was no ulceration and no infiltration in the non-DSS group. In the DSS group and hADSC suspension group, ulceration and infiltration are more severe than in the hADSC CellSaic group. The arrows indicate ulceration and infiltration. Scale Bar is 100 µm. (A) Histopathological comparison of colitis in the rectum at day 14. There was no edema in the non-DSS group. In the DSS group and hADSC suspension group, edema was more severe than in the hADSC CellSaic group. The arrows indicate edema. Scale bar is 100 µm. (B) Histological evaluation of the colon at day 14. The value of 75% – median (blue box), median – the value of 25% (red box), the value of average (green cross). Non-DSS group [no treatment with DSS,
cADSC CellSaic Promotes Improvement of DSS-Induced Colitis
A test similar to that used to assess the efficacy of hADSCs was performed using cADSCs. Our results indicate that the body weights in the DSS group, the cADSC suspension i.v. group, and the cADSC suspension clinical dose i.v. group were 86–88% at days 8 and 11, and 96–98% at day 14, respectively (Fig. 4A). In addition, the body weights in the cADSC CellSaic i.p. group were 89% at day 8, 96% at day 11, and 101% at day 14, indicating a recovery in body weight. The cADSC CellSaic i.p. group showed significant differences in body weight on day 11 compared with the DSS group (

cADSC CellSaic also attenuates recovery of body weight and colon length. (A) Percentage of body weight changes. Non-DSS group [no treatment with DSS,
hADSC CellSaic secretes more TSG-6 than does hADSC only in vitro
Secretion of the anti-inflammatory protein TSG-6 was tested under the culture condition of hADSC CellSaic and hADSC spheroid. The TSG-6 secretion levels after 48 h of incubation were 6072 pg for the hADSC CellSaic and 1946 pg for the hADSC sphenoid in culture medium containing TNFα (Fig. 5). These results indicate that more TSG-6 was secreted in the presence of hADSC CellSaic. The hADSC CellSaic showed significant differences from the hADSC spheroid (
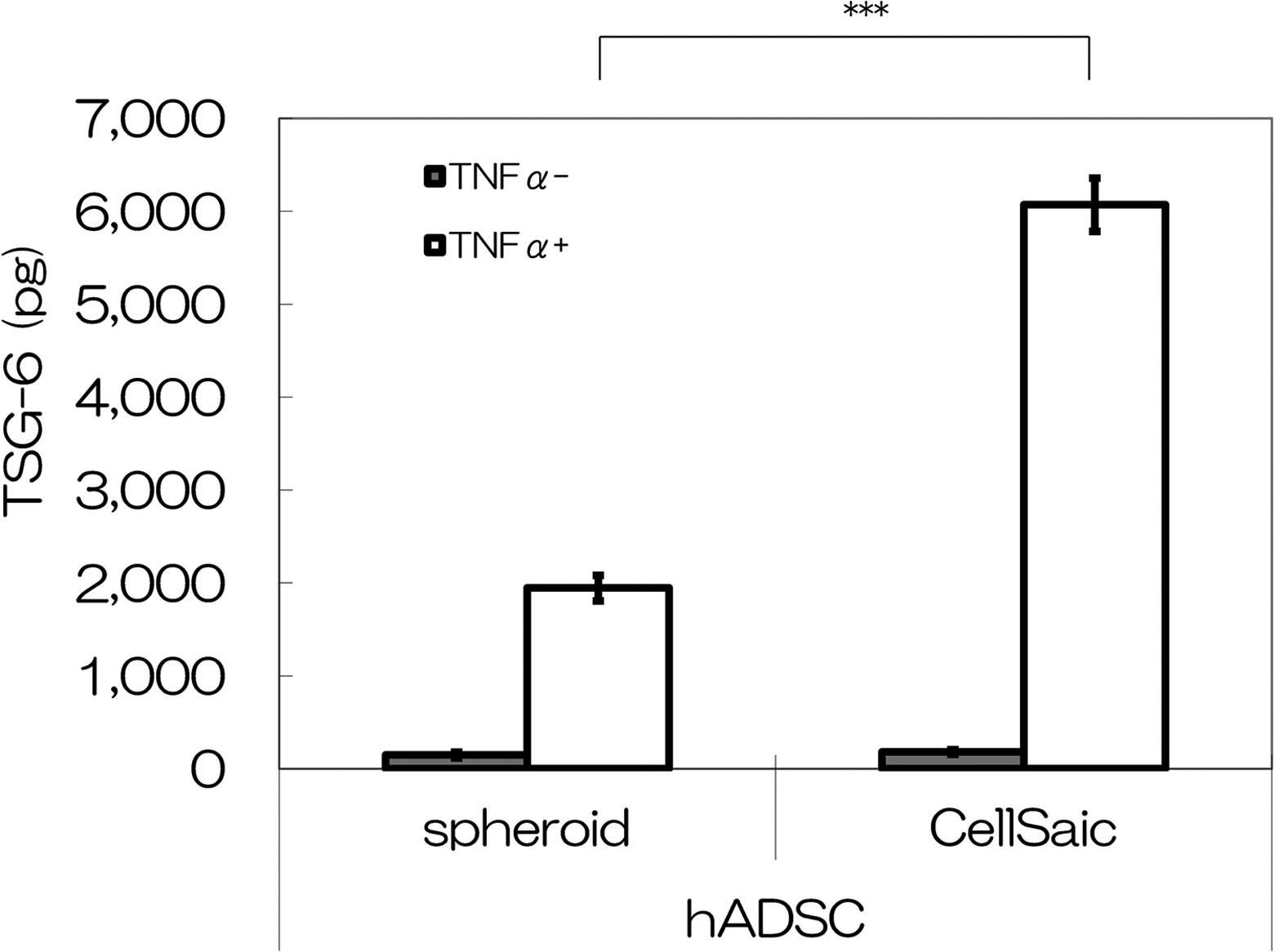
Comparison of TSG-6 levels
cADSC CellSaic has no Tumorigenicity
The safety of subcutaneous administration was assessed by conducting a tumorigenicity test in NOG mice. Nodules were observed in female and male mice in the CellSaic group at days 1, 4, and 7, reaching maximum volumes of 70.21 mm2 in male mice and 40.45 mm2 in female mice at day 4. However, similar to the physiological saline control group and the culture medium control group, subcutaneous nodules were not visible in the male and female mice of the cADSC CellSaic group at day 11 (Figs. 6A and 6B). Histological evaluation of the administration sites at 4 and 8 weeks revealed no cell division despite the presence of a remnant at the administration site; this indicates that no tumor development occurred in this group. In addition, no significant intergroup differences were observed in biochemistry testing, hematological testing, organ weight, food consumption, or body weight. No abnormalities were found during pathological evaluation of the organs in the cADSC CellSaic group at 4 and 8 weeks (Supplemental results; all Figures S1–S4 and Tables S4–S29).
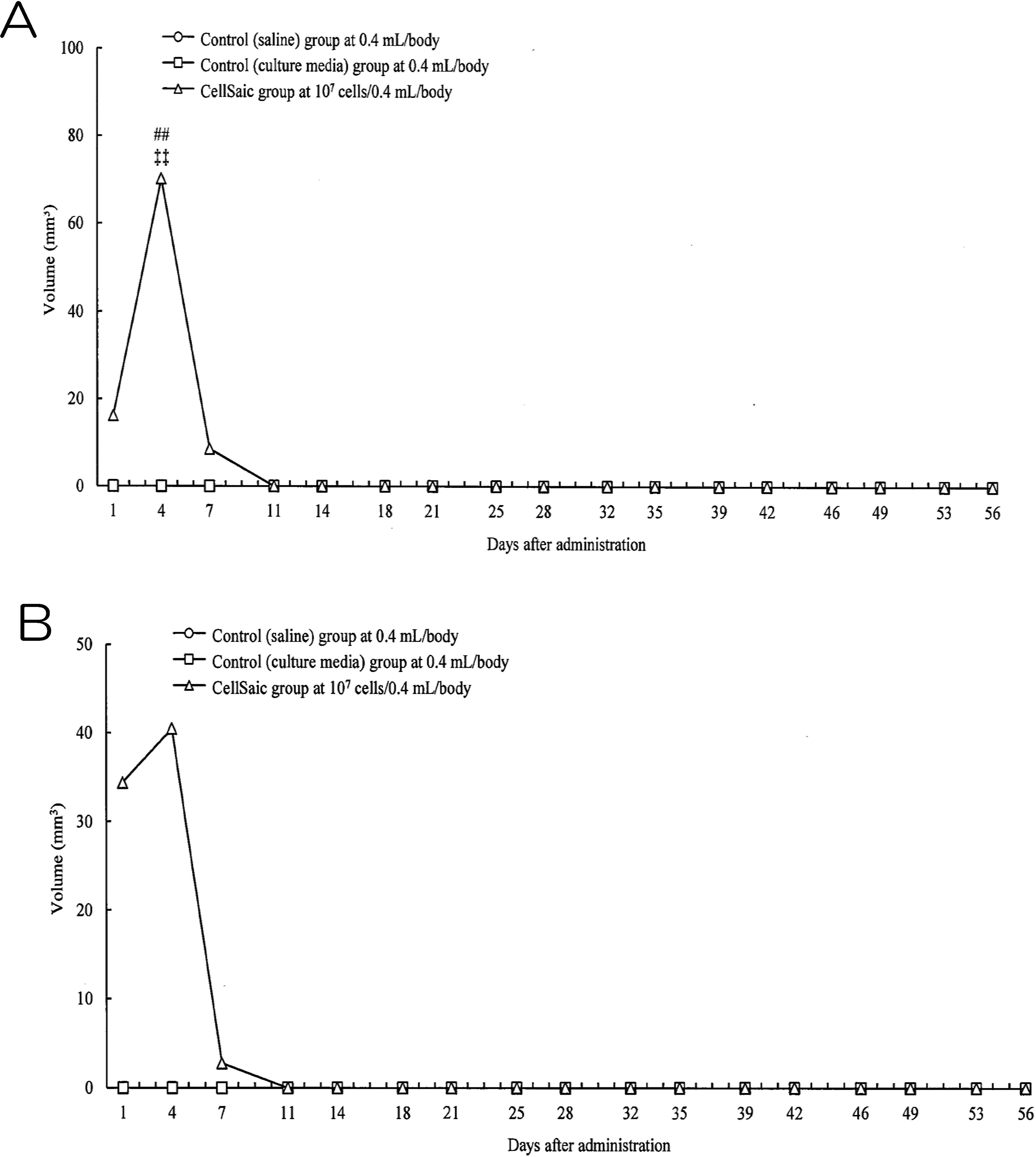
Volume of nodules in tumorigenicity test. (A) Volume of nodules in male mice. Control (saline) group; circle, Control (culture media) group: square, CellSaic group; triangle. CellSaic group was significantly different from the control (saline) group (##:
Discussion
Our mouse models of DSS-induced colitis demonstrated that human and canine ADSC CellSaic exerted higher therapeutic effects than cells injected alone. A previous study reported the effect of CellSaic 21 . The presence of a scaffold for cells can prevent anoikis, which is apoptosis of adherent cells induced by the absence of a scaffold.
With respect to preventing anoikis, we considered that hADSC CellSaic showed good survival intraperitoneally compared with hADSCs in suspension delivered as single cells due to adhesion to the µ-pieces in hADSC CellSaic. Indeed, we intraperitoneally injected the hADSC CellSaics with labeled cells and confirmed their behavior
In our
Angiogenesis contributes to repair the tissue. During chronic inflammatory diseases, inhibition of angiogenesis attenuates further inflammation and disease pathology 25 . Angiogenesis plays an important role as a protective factor during the regeneration of injured tissues. Intraperitoneal administration of recombinant human hepatocyte growth factor, HGF, which has been shown to be a potent angiogenic factor, can accelerate colonic mucosal repair in rats with DSS-induced colitis 26 . In pathological evaluation, repair of ulcer and epithelial tissue structure was promoted more in the hADSC CellSaic group than in the DSS control group. In addition, BMSC CellSaic promotes angiogenesis as compared with using BMSCs alone 21 . Thus, the promotion of angiogenesis may have contributed to the repair of intestinal tissue, although firm evidence was not obtained in this study. The cADSC CellSaics had the same effect as hADSC CellSaics. In contrast, i.v. administration of the cADSC suspension showed no effect either at the clinical dose or same cell count as that used with the CellSaic. This observation would be caused by the reason mentioned in hADSC above.
Tumorigenicity analysis using NOG mice administered cADSC CellSaics showed no formation of subcutaneous nodules and cell division of administered cells after 4 and 8 weeks, suggesting that there is no serious risk of tumor development. Moreover, there were no abnormal findings including tumor development in systemic organs, indicating no serious risk of toxicity. In summary, canine ADSC CellSaics may be safe for veterinary therapies.
We found that human and canine ADSCs, administered as CellSaic, are effective for DSS colitis therapy. In addition, we confirmed no tumorigenicity or toxicity for cADSC CellSaic. Based on these findings, these results may be clinically applied for colitis therapy of dogs; moreover, clinical studies of companion dogs will accelerate the clinical development of therapies for humans.
Supplemental material
Supplemental Material, Supplemental_material_1 - The Therapeutic Effects of Adipose-Derived Stem Cells and Recombinant Peptide Pieces on Mouse Model of DSS Colitis
Supplemental Material, Supplemental_material_1 for The Therapeutic Effects of Adipose-Derived Stem Cells and Recombinant Peptide Pieces on Mouse Model of DSS Colitis by Reiko Iwazawa, Sayako Kozakai, Tsukasa Kitahashi, Kentaro Nakamura, and Ken-ichiro Hata in Cell Transplantation
Supplemental material
Supplemental Material, Supplemental_material_2 - The Therapeutic Effects of Adipose-Derived Stem Cells and Recombinant Peptide Pieces on Mouse Model of DSS Colitis
Supplemental Material, Supplemental_material_2 for The Therapeutic Effects of Adipose-Derived Stem Cells and Recombinant Peptide Pieces on Mouse Model of DSS Colitis by Reiko Iwazawa, Sayako Kozakai, Tsukasa Kitahashi, Kentaro Nakamura, and Ken-ichiro Hata in Cell Transplantation
Footnotes
Acknowledgments
The authors thank Mr. Tadashi Ito of Nihon Bioresearch Center, Inc. The authors would like to thank the members of the technical staff of Regenerative Medicine Research Laboratories, FUJIFILM Corporation, whose opinions and knowledge were very helpful throughout the completion of this work.
Ethical Approval
The Animal Care Committee of the FUJIFILM Corporation approved the experimental protocol, and all experimental procedures used in animal studies were performed.
Statement of Human and Animal Rights
All experiments used animals were performed in accordance with international guidelines. Human subjects were not used in this study.
Statement of Informed Consent
Statement of Informed Consent is not applicable to this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
