Abstract
Background:
Childhood diarrheal diseases remain highly endemic in India, but the emergence of extended-spectrum β-lactamase (ESBL)-producing
Materials and methods:
The stool samples were collected everyday throughout the year for 2 consecutive years. In our study, we collected stool samples randomly from every fifth patient. Stool samples were collected from a total of 633 randomly selected diarrheal children (age: 0-60 months) belonging to 17 communities and screened for identification of virulent diarrheagenic
Results:
Diarrheagenic
Conclusions:
This study revealed that EAEC (72.1%) is the predominant pathotype in Bihar, significantly high in ≤12 months age group children (
Introduction
Diarrheal disease is the second leading cause of death in children under 5 years of age and is responsible for killing around 5 25 000 children every year. Globally, there are nearly 1.7 billion cases of childhood diarrheal disease every year.
1
In India, 212 000 deaths were reported due to diarrhea in children less than 5 years of age.2,3 The cause of diarrhea has been studied in both developed4–6 and developing7–10 countries and it was shown that enteropathogens implicated in the cause of diarrhea may vary among regions and populations, even when the same epidemiological and microbiological methods are used.
7
The causative agents may be an array of bacterial, viral, or parasitic pathogens. Globally, 0.5 million children die annually due to rotavirus-associated diarrhea,
11
whereas national approximate from India revealed that rotavirus is the reason for 0.1 million deaths and 0.4 to 0.8 million hospitalizations in children <5 years of age.
12
About 125 million cases of endemic shigellosis caused by enteroinvasive
Diarrheagenic
Discrimination of virulent DEC strains from the normal fecal flora is difficult using conventional phenotypic methods. In recent years, with the introduction of polymerase chain reaction (PCR) technology, it has become possible to detect presence of different chromosomal and/or plasmid-encoded virulence genes, which are absent in the commensal
Multidrug-resistant (MDR) DEC strains pose a serious clinical challenge. The MDR phenotype can emerge due to the presence of transmissible metallo-β-lactamase genes in the pathotypes as these β-lactamases can hydrolyze all β-lactams except aztreonam.
31
However, the predominant mechanism of resistance in DEC to β-lactam antibiotics is due to production of plasmid-encoded extended-spectrum β-lactamases (ESBLs). The ESBL-producing organisms have become widespread globally.32,33 The ESBL producers are resistant for most β-lactams, with the exception of cephamycin and carbapenems, but remain susceptible to β-lactam inhibitors.
34
In addition, ESBLs comprise diverse group, among which the CTX-M group (so-called cefotaximases) is now the most prevalent ESBLs in most parts of the world.35,36 It is very important to determine the prevalence of ESBLs within a geographic area as emergence of ESBL-producing
Methods
Study subjects
In this study, a total of 633 stool specimens were collected throughout the year from acute diarrheal pediatric outpatients of the preschool age (0-60 months), hospitalized either at the Child Care Hospital or Nalanda Medical College and Hospital, Patna, for 2 consecutive years. Every fifth child, with diarrhea, was enrolled in the study with informed consent from the guardian.
Information about symptoms (eg, number of loose/watery stools, bloody stools, vomiting, fever ≥39°C) was collected by parental discussion and from medical records. Only the patients of Indian origin and resident of selected area of the Bihar province (Patna) were included in the study. If the parents of any child refused to participate, those patients were excluded from the study. Other exclusion criteria included travelers’ diarrhea, chronic diarrhea, antibiotic consumption, immunodeficiency, and chemotherapy treatment in infants.
Stool samples were collected from children of the preschool age with acute community-acquired diarrhea requiring hospitalization in the pediatric ward. Passage of 3 or more watery/loose stools in 24 hours, with or without clinical symptoms of an enteric ailment (nausea, vomiting, abdominal pain or cramps, dehydration, fecal urgency, or dysentery), was considered as diarrhea. Other clinical features and risk factors recorded from patients included vomiting, nausea, abdominal pain, body temperature ≥38.0°C, duration of diarrhea, feeding type, diarrhea type, drug use, etc. Severity of the disease was based on the WHO classification of dehydration.
38
Children with no dehydration were classified as mild diarrhea and those with mild to moderate dehydration were classified as moderate diarrhea. Children who required intravenous therapy for correction of dehydration and/or electrolyte disturbance were classified as severe. However, the children whose diarrhea could be due to classic pathogens such as
Specimen collection and transport
The specimens were collected in clean, sterile wide-mouthed containers with tight-fitting leak-proof lids and stored at Microbiology Laboratory at the Hospital. Later, the samples were transported to the Microbiology Department of All India Institute of Medical Sciences (AIIMS)-Patna and/or Molecular Biology Department of RMRIMS (Rajendra Memorial Research Institute of Medical Sciences) for further processing within 2 hours of receipt.
Reagents
All media and chemicals were obtained from BD-Difco (Sparks, MD, USA) or Oxoid Inc (Thermo Fisher, Ottawa, ON, Canada), unless otherwise indicated. Oligonucleotide primers used in the study were synthesized from Integrated DNA Technologies (IDT, Coralville, IA). All PCR reagents including
Conventional screening and identification of DEC
Stool samples were first inoculated into selective enrichment medium (lauryl sulfate tryptose broth) at a ratio of 1:10. After overnight incubation, it was subcultured onto MacConkey agar and sorbitol MacConkey agar plate containing cefixime (0.05 mg/L) and tellurite (2.5 mg/L) and incubated for 24 hours at 37°C to isolate
Molecular identification of DEC strains
Diarrheagenic
Polymerase chain reaction experiments were performed with a 20-µL reaction mixture containing 3 µL of template DNA and 2.0 µL of 10× PCR buffer containing 25 mM MgCl2, 2 µL of a 2.5 mM mixture of deoxynucleoside triphosphates, 0.25 µL of 5 U/µL of
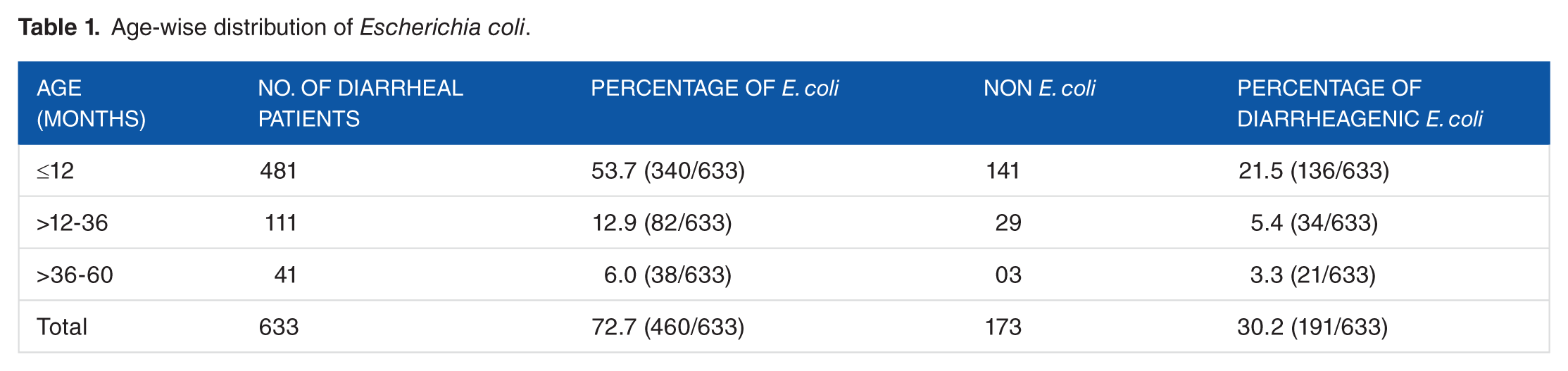
Age-wise distribution of
Antibiotic sensitivity test
The
Characterization of ESBLs and PCR detection of bla determinants
Presence of ESBLs was determined by standard disk diffusion method on Mueller-Hinton agar which is useful for quantitative determination of susceptibility of bacteria to antibacterial agents, recommended by the CLSI guidelines.47,48 Here, ESBL activity was determined by the double-disk diffusion method using ceftazidime (30 μg), ceftazidime plus clavulanate (30/10 μg) disks and cefotaxime (30 μg), and cefotaxime plus clavulanic acid (30/10 μg) disks (Oxoid Limited, Basingstoke, UK) on Mueller-Hinton agar (Oxoid Limited, Basingstoke, UK).
Furthermore, all ESBL-producing isolates were investigated for the presence of the β-lactamase (
Primer sequences and expected size of polymerase chain reaction–amplified gene targets of the pathogenic strains of
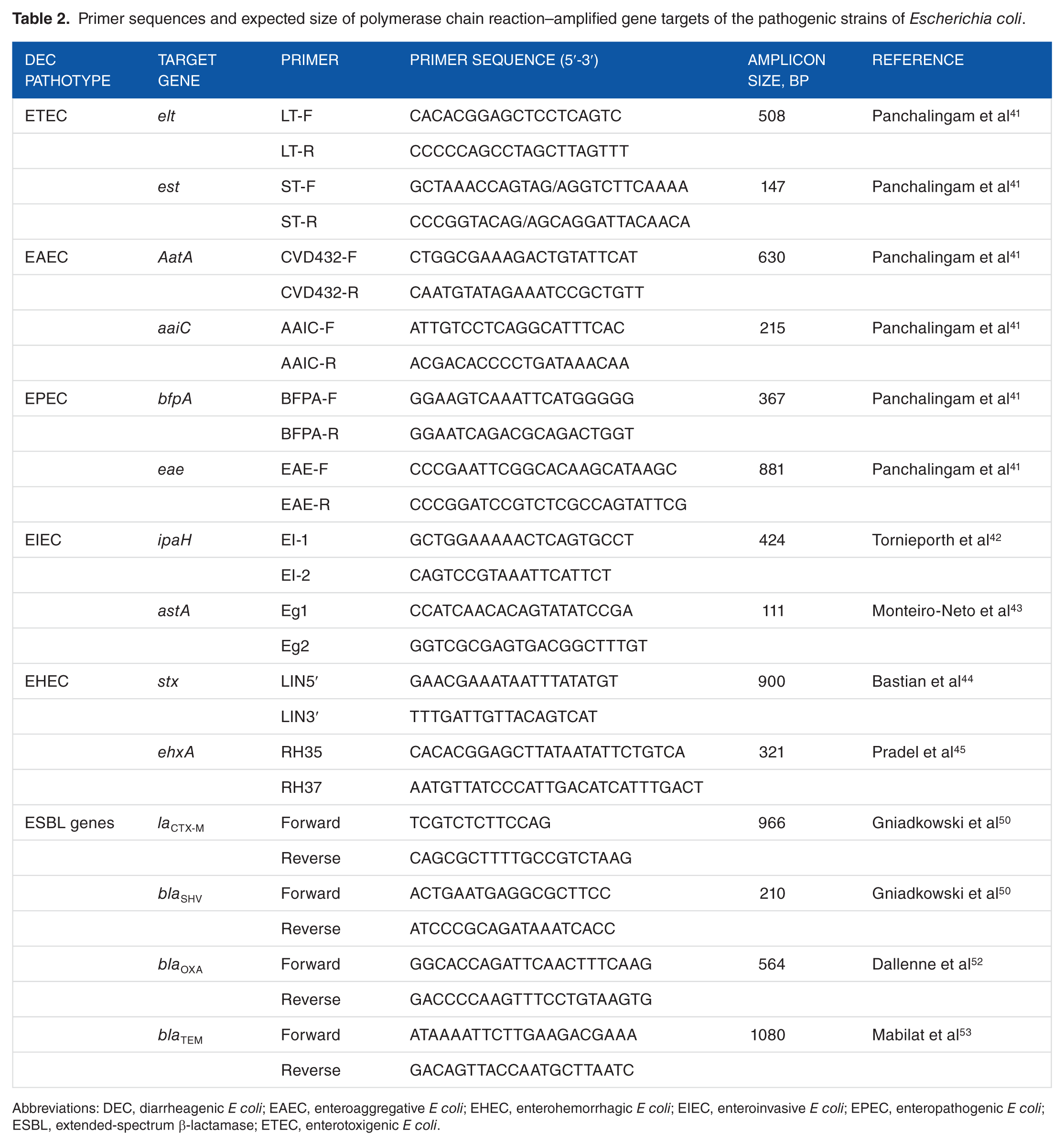
Abbreviations: DEC, diarrheagenic
Statistical analysis
Associations between categorical variables were obtained using χ2 and Fisher exact tests, with the analysis of variance and Kruskal-Wallis tests used for comparison of medians using GraphPad Prism Software (Version 6) (GraphPad Software, Inc., La Jolla, CA, USA). A
Results and Discussion
In India, diarrhea still remains the most common cause of death among children under 5 years of age. Every year, an estimated 2.5 billion deaths occur due to diarrhea; among them, 30% to 40% is contributed only to DEC.26,54 Diarrheagenic
Geographical location of the area of Study. In this study only the patients from the Patna district of Bihar state in India were enrolled.
Biochemically confirmed
Age-wise distribution of ETEC, EAEC, EPEC, EIEC, and EHEC among total DEC isolates.
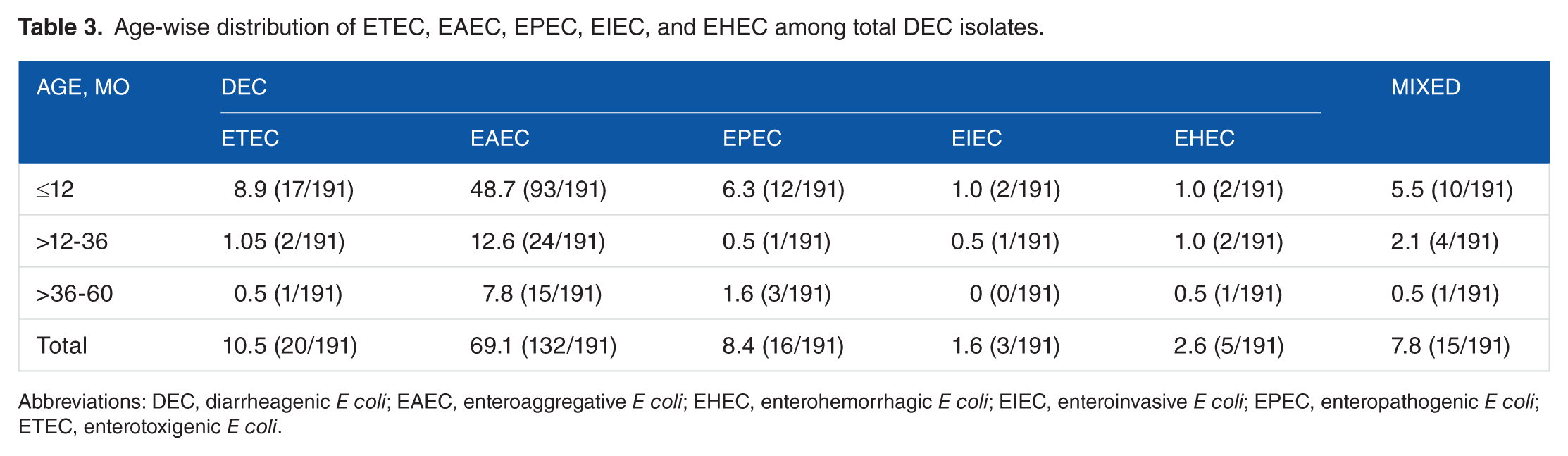
Abbreviations: DEC, diarrheagenic
In our study, the enrolled patients are of uniform sex distribution. The overall sex distribution among the study subjects was 348 (54.9%) men and 285 (45.02%) women. Next, the preschool children patient data were stratified into 3 age groups, viz, group A: ≤12 months, group B: >12 to 36 months, and group C: >36 to 60 months. Analysis of age-wise distribution revealed that 481 (74.6%) children were of ≤12 months of age, 111 (17.3%) were >12 to 36 months of age, and the rest 41 (8.2%) were >36 to 60 months of age (Table 1). The distribution of different types of DEC pathotypes among different age groups was found to be significant (
Enteroaggregative
Enterotoxigenic
Low prevalence of EHEC infection has been reported in developing countries. 69 Consistently, we found presence of EHEC in only 2 cases (1%) of the ≤12 months age group, 2 cases (1%) in the >12 to 36 months age group, and 1 case (0.5%) in the >36 to 60 months age group (Table 3). Stool samples containing EHEC were both of bloody and nonbloody types. Therefore, the absence of blood in the stool does not rule out presence of EHEC pathotypes. Acute diarrhea has been often related to EIEC pathotypes. 70 In our findings, EIEC was identified in the 2 cases (1%) in the ≤12 months age group, 1 case (0.5%) in the >12 to 36 months age group, and no cases (0%) in >36 to 60 months age group (Table 3).
Intermediate
Analysis of the clinical symptoms of diarrheagenic patients caused by different pathogroups of DEC was done. Watery stool was the most dominant criteria for stool consistency among the study subjects. The findings indicate that infection with EAEC caused moderate (84.8%) to mild (12.9%) dehydration among patients preferably with yellowish (43.9%) stool and watery (62.1%) consistency (Table 4). Infection with ETEC was mostly characterized by moderate (80%) dehydration status with yellow colored (55%) watery stool (65%), whereas EPEC infection resulted in moderate dehydration (62.5%) with reddish (37.5%) or yellow colored (31.2%) semisolid (37.5%) or watery (12.5%) stool (Table 4). Other clinical criteria tested were as follows: body temperature of ≥38.0°C in 3 (60%) EHEC cases, nausea and/or vomiting in 75 (56.8%) EAEC cases and in 5 (25%) ETEC cases, and abdominal pain in 15 (75%) ETEC cases (Table 4).
Clinical features and risk factors in diarrheic children infected with diarrheagenic
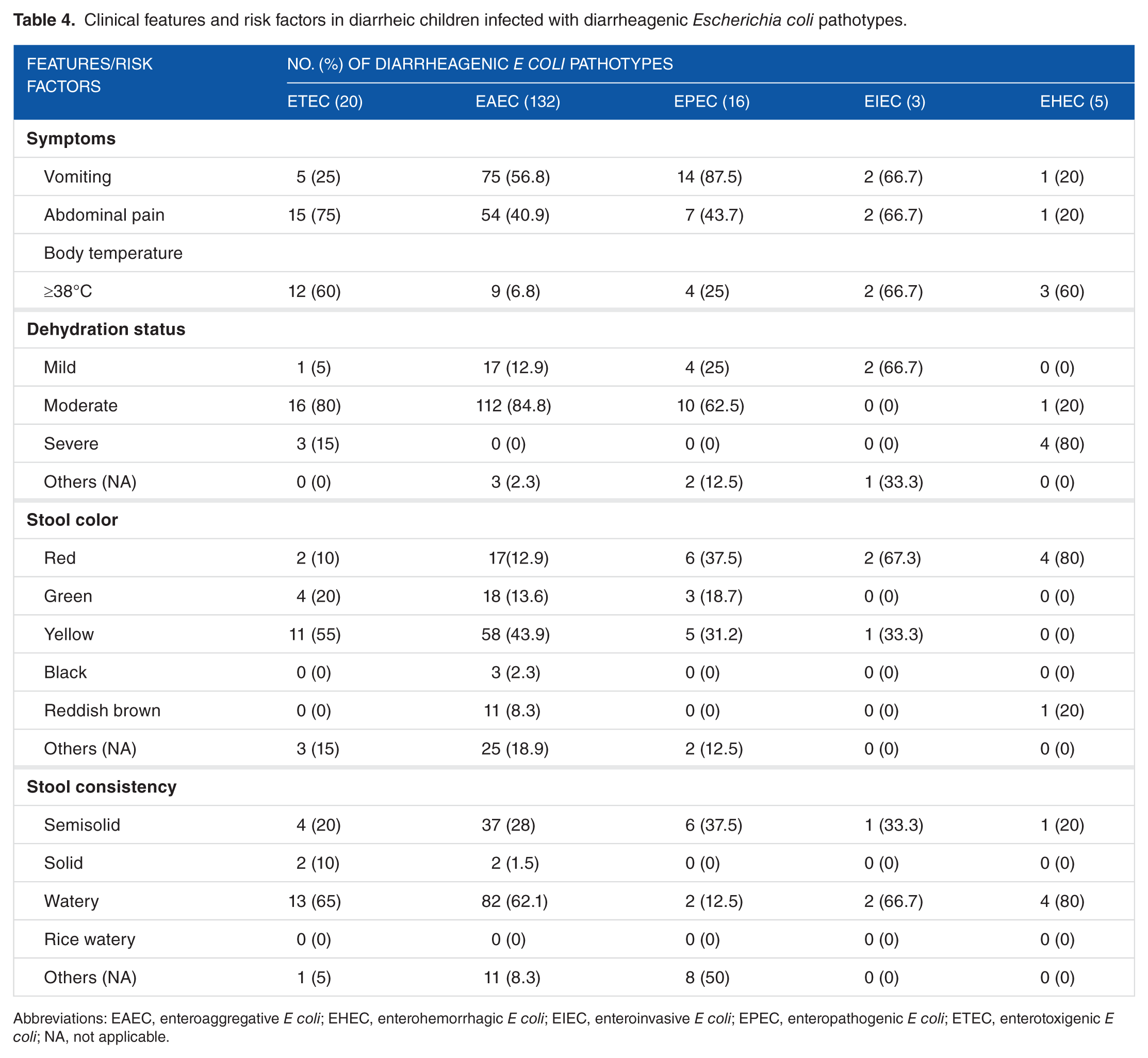
Abbreviations: EAEC, enteroaggregative
All antibiotic susceptibility results were interpreted using the break points of the CLSI guidelines. Among the study isolates, typical ETEC pathotypes were found to be sensitive to levofloxacin (48%), ciprofloxacin (53%), ceftazidime (54%), ceftriaxone (56%), ampicillin (64%), and amikacin (100%) (Figure 2). In EAEC strains, sensitivity was found mostly for amikacin (100%), whereas comparatively less sensitivity was observed for piperacillin (14.6%), levofloxacin (16.7%), ceftazidime (19.1%), gentamicin (26.5%), and ciprofloxacin (33.4%) (Figure 2). Most of the EPEC, EIEC, and EHEC strains were resistant to most of the antibiotics, except amikacin and ciprofloxacin (not shown).
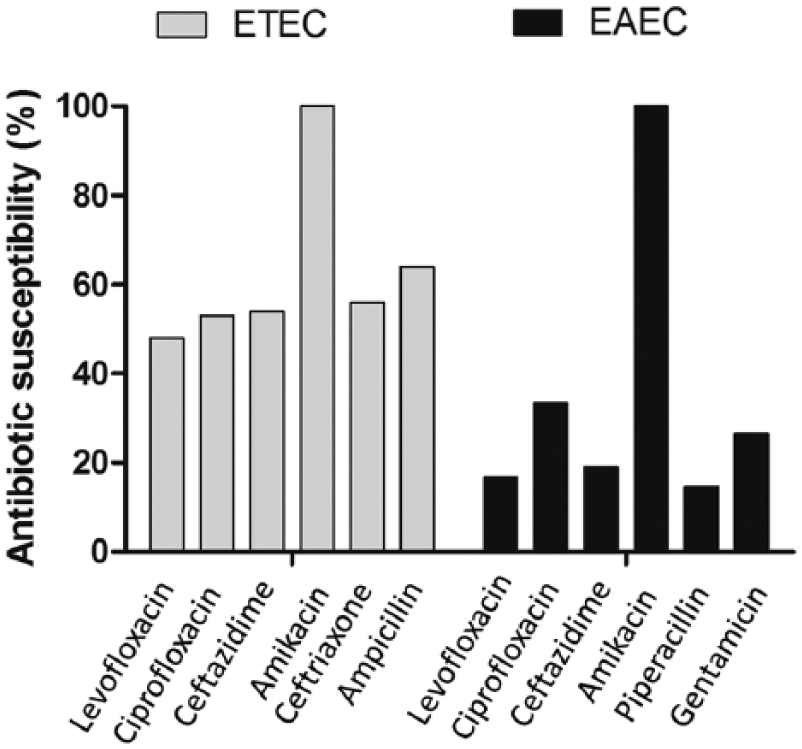
Antibiotic susceptibility pattern of ETEC and EAEC pathotypes. Antibiotic susceptibility test for ETEC and EAEC pathotype was performed according to CLSI guidelines. ETEC pathotypes were found to be sensitive to levofloxacin, ciprofloxacin, ceftazidime, amikacin, ceftriaxone and ampicillin. In case of EAEC pathotypes, comparatively less sensitivity was observed for levofloxacin, ciprofloxacin, ceftazidime, piperacillin and gentamicin, whereas maximum sensitivity was found for amikacin.
Importantly, new classes of enzymes conferring resistance to β-lactam antibiotics have emerged due to antibiotic selection pressure over the past decades, most alarming being the ESBL-producing enteric pathogens.
72
Typically, ESBLs can hydrolyze third-generation cephalosporins and aztreonam, but not carbapenems, and therefore are inhibited by clavulanic acid and tazobactam. Therefore, in this study, high-level resistance to third-generation cephalosporins were selected for detection of ESBL-producing DEC pathotypes. Notably, the results demonstrated that 37.6% of total DEC isolates of this study were ESBL producers (n = 72), being prevalent among ETEC (n = 35; 18.32%), followed by EPEC (n = 21; 10.9%), EAEC (n = 13; 6.8%), and EIEC (n = 3; 1.57%). The ESBL-producing
Results of antimicrobial susceptibility tests and resultant MICs for diarrheagenic
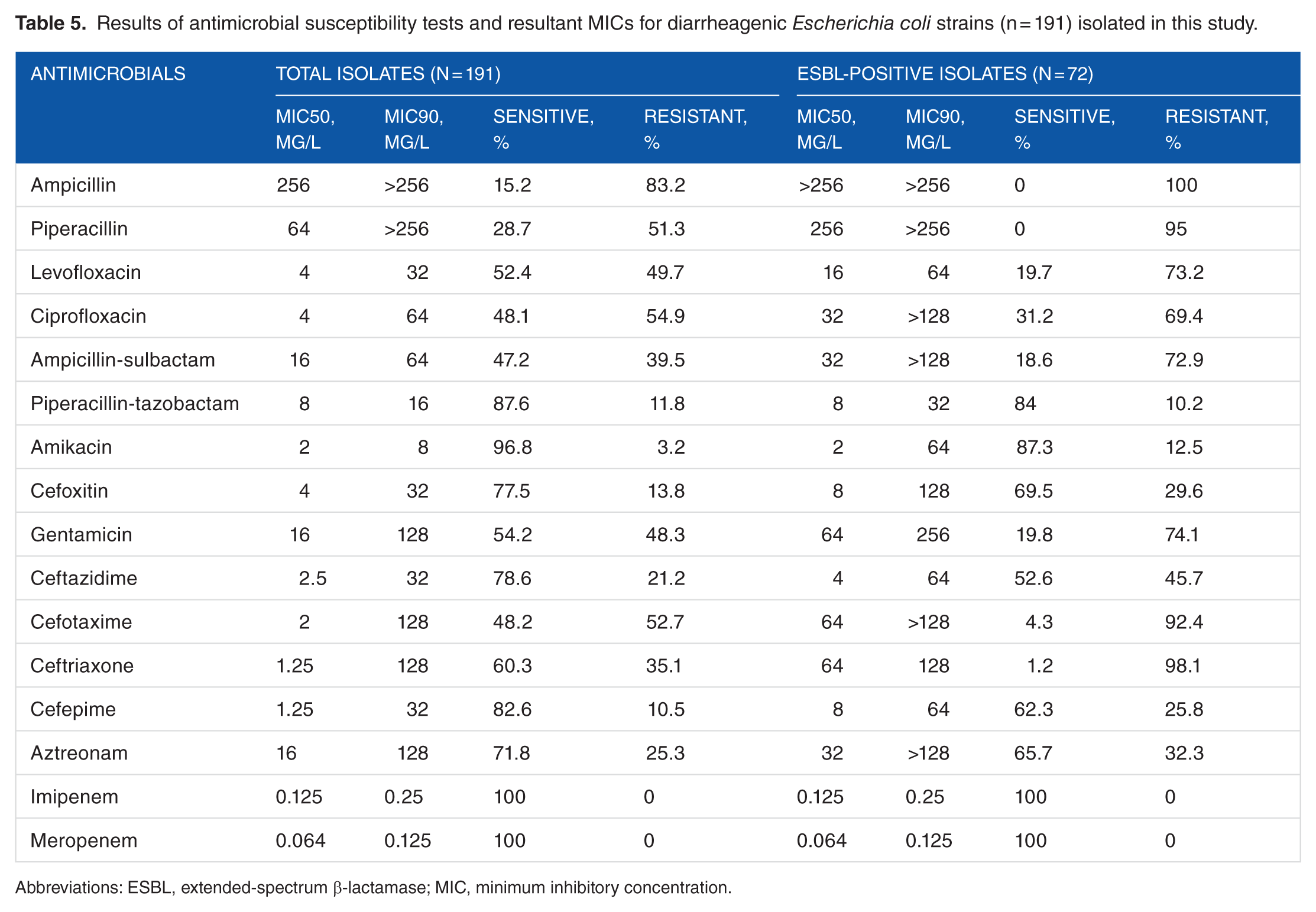
Abbreviations: ESBL, extended-spectrum β-lactamase; MIC, minimum inhibitory concentration.
The ESBL-producing pathogens frequently exhibit plasmid-encoded multidrug resistance.
48
Owing to mutational changes, more than 200 types of ESBLs are currently described in various species of the Enterobacteriaceae family and other nonenteric organisms.48,73 All ESBL-producing isolates were investigated for the presence of β-lactamase (
Conclusions
In summary, this study revealed that EAEC (72.1%) is the predominant pathotype in Bihar, significantly high in ≤12 months age group children (
Footnotes
Acknowledgements
The authors are grateful to all the participants for their help during collection of the isolates for this project. The authors wish to acknowledge National Institute of Cholera and Enteric Diseases for providing reference DEC strains and the Indian Council of Medical Research (ICMR) for research support. They are also thankful to Mrs Bhawana, Mr Subhash Kumar, and Mr Rakesh Mandal for technical support.
Peer review:
Four peer reviewers contributed to the peer review report. Reviewers’ reports totaled 1438 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
SD and PD designed the study. SD, AM, AS, and AK performed the experiments. SD, AS, and PD analyzed the data. SD and AM analyzed the sequences and wrote the manuscript which was corrected and approved by all the other co-authors. UKS, AKJ, PD, and SD contributed to reagents/materials/analysis tools. All authors read and approved the final manuscript.
Availability of Data and Materials
All the relevant data of this study have been fully presented in the main manuscript and tables.
Consent to Publication
We state that all the authors have contributed significantly to the work and have approved the content of this manuscript and its submission.
Ethical Approval and Consent to Participate
The study protocol and consent procedure were approved by the Institutional Ethics Committee (IEC) of All India Institute of Medical Sciences (AIIMS), Patna, and Rajendra Memorial Research Institute of Medical Sciences (ICMR), Patna. Informed written consent was obtained from legal guardians of the enrolled children. All clinical investigations were conducted according to the principles expressed in the Declaration of Helsinki.
