Abstract
Background:
Multiplex polymerase chain reaction (PCR) testing for viral acute respiratory infections (ARI) at the Point of Care (POC) has demonstrated clinical and economic value in secondary care, yet its impact in primary care remains uncertain. United Kingdom (UK) guidelines make conflicting recommendations on the use of testing in primary care settings.
Objectives:
This study provides expert consensus on the potential clinical and economic implications of rapid PCR testing at the POC in primary care settings.
Design:
A modified Delphi consensus panel approach was employed, with consensus statements developed from existing literature and evaluated through two rounds of questionnaires. Open-ended questions were posed to explore potential barriers to implementation, evidence generation, and suitable settings for testing.
Methods:
A multistakeholder panel of 9 experts was purposely recruited, representing stakeholders from seven areas. A narrative literature review was conducted to generate consensus on the potential value of implementing rapid PCR testing at the POC for ARIs in primary care settings. Two Delphi rounds were completed, with participants rating their level of agreement with presented statements on a Likert scale from 1 to 5.
Results:
Seventeen statements were generated based on the results of a narrative literature review, with eight achieving consensus and further evidence generation recommendations developed for six statements. Two statements were removed due to non-agreement and two were merged into a single statement, which later achieved consensus. The lack of cost and clinical effectiveness data was ranked as the greatest barrier to implementation. Primary care settings with high and low risk patients, such as general practices and care homes, were considered ideal for implementation.
Conclusion:
There is potential value in rapid multiplex viral PCR testing for ARIs in primary care settings and care homes. While existing evidence and expert consensus indicate a likely benefit, further real-world evidence trials are recommended to evaluate the cost-effectiveness of this approach.
Keywords
Introduction
Winter pressures on the United Kingdom (UK) healthcare system are largely driven by seasonal viral infections. Influenza typically plays the most significant role, often following an initial surge of respiratory syncytial virus (RSV), while COVID-19 continues to contribute an unpredictable and compounding challenge. 1 The COVID-19 pandemic further highlighted the critical importance of rapid diagnosis and differentiation of acute respiratory infections (ARI) for timely and effective patient management. 2 While COVID-19 is no longer classified as a public health emergency of international concern (PHEIC) by the World Health Organization (WHO), 2 it remains an endemic disease, and all ARIs continue to pose a significant risk. The National Institute of Health and Care Excellence (NICE) defines ARIs as an acute illness (present for 21 days or less) affecting the respiratory tract with symptoms such as cough, sore throat, fever, sputum production, breathlessness, wheeze, chest discomfort or pain, and no alternative explanation. 3 ARIs can lead to severe respiratory symptoms and hospitalisations, particularly among vulnerable groups. National Health Service England (NHS England) highlights that ARIs are a major cause of emergency department (ED) visits and remain a common cause of poor health outcomes, with many individuals seeking medical consultations for respiratory issues. 4 Furthermore, The European Centre for Disease Prevention and Control (ECDC) has reported a steady rise in primary care visits for respiratory illnesses since September 2023, with test results confirming the continued prevalence of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), RSV, and seasonal influenza in the community. 5 Though optimal treatments of each ARI differ, symptomology may overlap, making diagnosis difficult in practice.
Point-of-care testing (POCT) utilizing polymerase chain reaction (PCR) technology may provide a rapid and reliable diagnosis of viruses linked to winter illnesses. 6 Multiplex viral PCR panels allow for the rapid and simultaneous identification of multiple viral pathogens, enhancing the efficiency of diagnosis and treatment. Studies of rapid PCR testing for multiple ARIs (from here referred to as multiplex viral PCR) in the hospital setting have demonstrated widespread impacts on patients and the healthcare system, including a faster and more accurate diagnosis, reduced hospital stays, improved use of infection control facilities and more appropriate antiviral use. 7 A range of respiratory pathogens can be detected simultaneously using PCR, which can reduce the time to test results, distinguish between diseases with similar presentations, 7 and are more cost effective than conducting single PCR tests for individual respiratory viruses. 8 Additional advantages of these tests include minimising inappropriate antimicrobial prescriptions and reducing pathogen transmission.9,10 An Australian study showed that following a positive PCR result, incorrect antimicrobial prescriptions were discontinued in 34% of patients, 9 while a UK study found that patients who underwent laboratory testing spent significantly more time in an open ward compared to those who received point-of-care polymerase chain reaction (POC PCR) testing, with a mean difference of 16.9 hours. 10 These studies highlight the practical benefits of rapid PCR testing on patient outcomes and healthcare resource management.
POCT in community care settings (predominantly primary care) offer the potential to improve the management of winter respiratory infections. A key advantage of multiplex viral testing is its ability to identify the most appropriate treatment, thereby reducing unnecessary antibiotic prescriptions. While the impact in primary care has not yet been clearly established, it remains to be seen whether the use of POCT can meaningfully improve prescribing practices in this setting.
NHS England has issued guidance (2023) supporting the integration of POCT into urgent community response services within primary care. 11 The guidance outlines several key benefits of using POCT in community settings, including improved clinical decision making, reduced need for hospital admissions, and lower re-contact rates following on scene patient discharge. 11 Though NHS England supports this integration, much of the supporting evidence they have utilised comes from the broader community or urgent care contexts rather than routine general practice. 11 Therefore, while the guidance provided is relevant, more primary care-specific research is needed to establish its direct applicability in primary care.
In contrast, NICE has recently issued guidelines (2023) recommending that clinical assessment alone should guide decisions on prescribing antimicrobials for suspected ARIs at first presentation, without relying on PCR testing. 12 NICE’s position is based on a lack of robust economic evidence on the use of point-of care (POC) single pathogen tests, with existing data limited and demonstrating no cost-effectiveness, with no evidence identified for the use of multiplex viral PCRs. 12 Due to these gaps, the NICE committee called for further evidence on the accuracy of POCT to provide a more definitive recommendation in the future, concluding that until such evidence is available, clinical assessment remains the most reliable indicator for guiding decisions on prescribing antimicrobials for suspected ARIs. 12
Differing guidelines from NHS England and NICE highlight a gap in the current evidence base and underscores the need for further research. Although NHS England supports the integration of POCT into urgent community response services within primary care, primary care-specific research is needed to further establish the effectiveness of POCT in this setting. Moreover, NICE’s recommendation that clinical assessment alone should guide decisions on suspected ARIs at first presentation is based on lack of evidence. The NICE committee clearly stated further research is needed to define the ‘role of point-of-care microbiological testing for guiding management in people with symptoms and signs of an ARI, taking into account clinical and cost-effectiveness of the tests, good antimicrobial stewardship cost, and time taken to do the test and get a result’. 12
As a result of conflicting guidance and unmet evidence needs, the purpose of this study is to use a modified Delphi methodology to obtain consensus on how multiplex viral PCR-based POCT might best be incorporated into clinical workflow in primary care in the UK for viral ARIs. This is achieved by addressing three objectives through the collaboration of a multidisciplinary panel:
(1) To explore the potential benefits of multiplex viral PCR testing at or near the POC for ARIs in community care settings, which includes general practice surgeries, care homes and other decentralised diagnostic centres across the UK, considering the opinions of all relevant stakeholders. From this point forward, the terms ‘community care’ and ‘primary care’ will be used interchangeably where applicable, as the community care settings predominantly referred to in this study are primary care settings.
(2) To identify barriers to implementing this approach (should there be agreement on its potential value).
(3) To provide recommendations for future research that could clarify uncertainties in the evidence and allow for a more informed decision on adopting multiplex viral PCR POCT in a community setting.
Methods
Study Design
The study utilised the Delphi technique – a systematic process of facilitating consensus among panel members by combining anonymous questionnaires with in-depth discussions. 13 The method has played a significant role over past decade to develop best practice guidance using expert consensus where research is lacking, limited, or evidence is conflicting. 13 In this study, the modified Delphi method was comprised of three stages: two Delphi rounds of anonymous questionnaires followed by a consensus panel. Typically, this design involves multiple survey rounds, with the exact number determined a priori. 13 While there are no universally standardised protocols for modified Delphi studies, it is common practice for investigators to first conduct a literature review and design targeted questionnaires to guide the panel toward consensus. In our modified Delphi study, a narrative literature review was conducted to summarise existing evidence on multiplex viral PCR testing and identify research gaps, informing the development of the first Delphi questionnaire. A narrative literature review was chosen over other review types, such as systematic or scoping reviews, due to the limited amount of existing research on the benefits of rapid multiplex viral PCR testing for ARIs. Conducting a narrative literature review provided an opportunity to highlight existing information on the topic, while also identifying gaps in the literature. 14 The second Delphi questionnaire was designed utilizing results from the first questionnaire.
The remaining information presented in the methods section describes the steps taken to obtain consensus on how multiplex viral PCR-based POCT might best be incorporated into clinical workflow in primary care in the UK for viral ARIs.
Step 1: Panel Recruitment and Composition
In this step, the expert panel was purposively selected to reflect a range of UK-based stakeholders with demonstrable expertise in infectious diseases, respiratory diagnostics, and health system decision-making. Given the narrow scope of the study, which focused on infectious diseases within England’s primary care context, a purposive sampling approach was considered the most appropriate method. Purposive sampling is consistent with best practices in Delphi methodology, where the goal is to recruit individuals with relevant knowledge and experience rather than apply random sampling techniques, ensuring participants can provide informed and meaningful input based on their direct knowledge and practical engagement with the subject matter. 15 This may include general practitioners with experience in diagnostic testing, researchers with a focus on infectious disease management and acute respiratory infections, and other stakeholders with domain-specific expertise.
Step 2: Panel Identification and Shortlisting
Next, an initial longlist of 124 individuals was compiled based on secondary research, and included professionals working across integrated care systems, pathology networks, public health bodies, provider organisations, and trade associations. From this list, 38 individuals were purposively shortlisted based on previous experience in managing ARIs, evaluating diagnostic technologies, or holding roles relevant to service planning and policy. Seven further panellists who were not on the initial longlist were recommended through professional networks and invited to participate based on their unique expertise and recognised leadership in relevant domains.
Additional criteria for shortlisting panellists included geographic representation, with an emphasis on ensuring perspectives from across England. We also considered progressional expertise, prioritizing individuals with prior publications in the field of rapid multiplex PCR testing and with approximately 10 to 15 years of experience in infectious disease to increase the likelihood that their depth of knowledge would provide meaningful insights to the study. While we recognised the importance of demographic diversity, we did not apply specific criteria related to age or gender, as the initial pool of eligible panellists was already limited. Applying further restrictions would have constrained our ability to assemble a panel with necessary expertise.
Preliminary discussions were held with shortlisted individuals to introduce study aims, explore their fit for the panel, and solicit recommendations for additional stakeholders. These conversations also provided insight on how the final composition of the panel should be balanced in terms of roles, specialisms, and practical experience.
Step 3: Panel Recruitment, Composition, and Search Strategy
To broaden representation and mitigate potential gaps (such as perspective and role gaps), panel members and study investigators nominated suitable colleagues through snowball sampling, yielding one additional candidate outside the original search strategy.
After compiling a list of suitable panellists, 46 invitations were issued, and nine individuals consented to participate. We recognise nine panellists is a small number of panellists for a Delphi panel, however, this number aligns with Delphi guidelines for studies involving homogeneous, expert groups in specialised areas. 15 Recruitment was limited by the niche nature of the topic and its geographic focus, resulting in a constrained number of qualified applicants within England. Participating panellists were likely familiar with PCR POCT technology, which may introduce selection bias; however, many also had experience with alternative diagnostic platforms, offering balanced and comparative insights.
The final panel ensured broad and relevant expertise across the clinical, academic, commissioning, and diagnostic spectrum of infectious diseases within the UK, while also reflecting a diversity of perspectives through representation from different areas of England. Many held multifaceted roles that spanned multiple domains of healthcare, enhancing the depth of perspectives. A panel chair was appointed based on their experience leading similar expert groups and their expertise in respiratory infectious disease strategy. Refer to Table 1 for an overview of the demographic characteristics of the panel.
Sample Characteristics of Nine Finalised Panellists (n = 9).
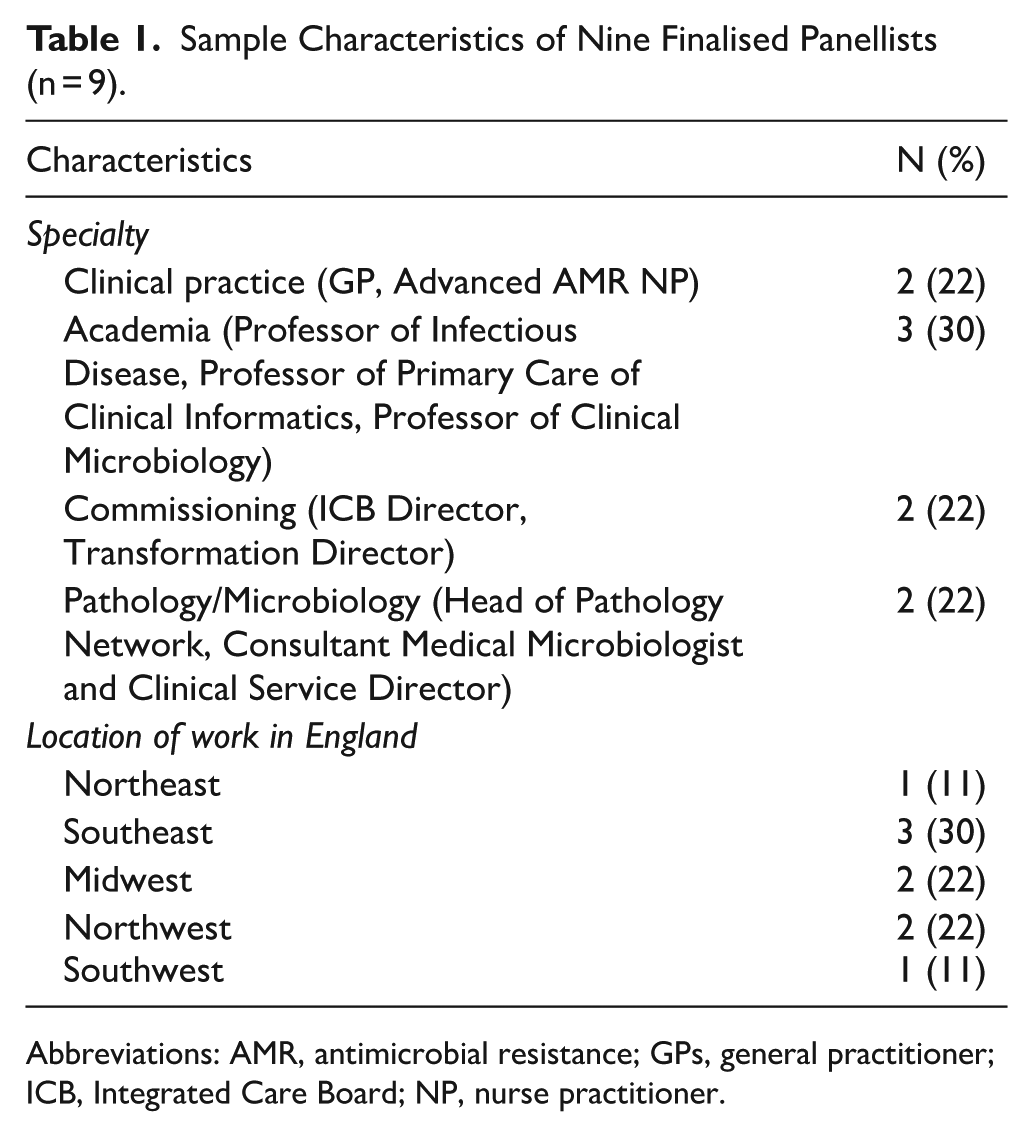
Abbreviations: AMR, antimicrobial resistance; GPs, general practitioner; ICB, Integrated Care Board; NP, nurse practitioner.
Following panellist finalisation, inclusion and exclusion criteria were developed for the narrative literature review. Included studies focused on POC testing using multiplex viral PCR to detect multiple respiratory viruses or strains and were published in the English language. Additionally, eligible study designs included randomised control trials (RCT), as well as experimental, observational, qualitative, review, (eg, systematic reviews and meta-analysis), diagnostic accuracy, evaluation, pilot, mixed methods, and real-world evidence (RWE) studies.
Studies were excluded if they were not available in full text, did not involve research from a POC setting, and were not published in the English language. We acknowledge potential for bias when studies are limited to the English language, though the inclusion of non-English language studies may require increased resources, cost, time and expertise. 16
Moreover, study protocols, clinical guidelines, editorials, opinion pieces, and grey literature were excluded from the review. Additionally, studies were excluded if they focused exclusively on the economic or clinical benefit of antibody testing, vaccination strategies, or medical imaging, without incorporating a multiplex viral PCR test component. Studies were also excluded if they focused only on the incidence of respiratory viruses rather than detection methods, or on the outcomes or success of treatment methods rather than the type of treatment provided.
No restrictions were placed on study population, geographic location, participant demographics, or study setting to capture all relevant literature on multiplex viral PCR testing. While the primary focus of this review is community care settings, studies conducted at the POC in a secondary care setting were also included, acknowledging economic and clinical benefits of POC testing in other settings may be applicable to community care. 17
Step 4: Generation of Search Strategy and Initial Search
Following the finalisation of inclusion and exclusion criteria, a comprehensive search strategy was developed and applied to Pubmed and Embase (April and May 2024). Search terms spanned four categories: clinical and economic outcomes, healthcare settings, rapid PCR tests, and respiratory infections. The final search strategy included 30 terms, and can be found in Appendix A.
Step 5: Study Assessment for Eligibility and Data Extraction
Next, a two-step screening process was undertaken to identify relevant studies that could be included in the final narrative review. The initial search strategy yielded a total of 2461 studies, comprising 1619 results from PubMed and 2070 from Embase. An external search was conducted through Google Scholar, which identified two additional relevant studies. In total, 2461 records were screened during this phase, and 2396 records were excluded, leaving 65 records eligible for full-text review. The full-text screening phase involved the screening of 64 studies, as one study could not be retrieved. Following this full text review, 27 studies met the predefined inclusion and exclusion criteria and were included in the final narrative review.
Step 6: Data Collection from Included Studies
After screening was complete, three reviewers extracted data from included studies, identifying up to 42 outcomes per study. Extracted information was converted into statements and grouped by topic in a Microsoft Excel (Microsoft, Washington [WA], USA) spreadsheet. Statements related to the same topics, such as turnaround time and time to isolation, were grouped together to identify their context and highlight areas of agreement or discrepancies.
Step 7: Designing Delphi Questionnaires and Defining Consensus
Next, after the finalisation of statements, two Delphi questionnaires were developed in Microsoft Forms (Microsoft, WA, USA) and sent to panellists by email. Each questionnaire consisted of three sections. The questionnaire was not formally validated, as no established frameworks or pre-validated instruments were deemed suitable to guide questionnaire development due to the novelty of the research topic. The first-round questionnaire was informed by findings from the literature and served as the foundation for the second round, which was refined based on expert feedback. As such, the questionnaire was developed specifically for the purpose of this study and evolved iteratively through the Delphi process.
In our study, two Delphi rounds was considered sufficient given the existing evidence available in the literature and a planned in-person panel discussion, expected to generate in-depth insights and address remaining uncertainties from the questionnaire rounds. To reduce bias in panellist responses, the identity of the responding panellist in each questionnaire was blinded from survey investigators to encourage open discussion during the final consensus panel.
Although there is no universally accepted cut-off for defining consensus in Delphi studies, a scoping review of 287 studies reporting how Delphi studies in health studies define consensus provided useful insight into common practices. Of the studies analysed, 43% were modified Delphi panels. 15 Moreover, among the 81% of studies which reported how consensus was defined, the most frequently used thresholds were 70% (n = 87), followed by 80% (n = 67), and 75% (n = 49). 15 Based on these findings, our study defined consensus as a threshold of ⩾80%, the highest of the three reported percentage cut offs. This decision aligns both with the existing literature and the need for robust expert agreement, given the limited research to date on the impact of PCR testing for ARIs in primary care.
Scale-based questions in each questionnaire were presented on a Likert scale from 1 to 5. Consensus was defined as ⩾80% of panellists selecting either ‘agree’ or ‘strongly agree’ (4 or 5) or ‘disagree’ or ‘strongly disagree’ (1 or 2). While the study did not differentiate between ‘agree/disagree’ and ‘strongly agree/disagree’ responses, a 5-point Likert scale was employed to allow for granularity in responses. Grouping higher and lower ends of the scale together for analysis assumed that the overall direction of opinion, rather than intensity of agreement, was the primary focus in defining consensus. The 5-point Likert scale was adopted based on the results of the scoping review, which revealed that a five point Likert scale is most commonly used within Delphi studies in health sciences, utilised in 40% of included studies. 15 Additionally, based on the number of panellists who responded to each questionnaire, the 80% consensus threshold did not correspond to a whole number in either Delphi questionnaire 1 or 2. Therefore, the number of panellists required to reach 80% consensus varied by questionnaire and was rounded to the nearest whole number.
Step 8: Delphi Questionnaire 1
After the development of the first-round questionnaire based on insights from the narrative review, the questionnaire was piloted by one of nine (11%) panellists. This panel member was selected based on their broad clinical expertise in managing infectious diseases and extensive research in diagnostic testing. The panel member provided valuable feedback on several aspects of the questionnaire, including the clarity of section descriptions, the content of each statement based on available literature, and the use of uniform terminology throughout the questionnaire. Additionally, they reviewed the phrasing of statements to ensure conciseness, provided feedback on the direction of rankings (eg, positioning statements with the highest added value at the top), and provided constructive feedback regarding the list of study types considered most suitable to address evidence gaps identified by the panel. Following the incorporation of feedback, the first Delphi questionnaire was sent to panellists in May 2024.
The first two sections of the questionnaire consisted of statements derived from the results of the narrative literature review. The first section of the questionnaire asked panellists to rate the applicability of eight statements derived from evidence on the impact of rapid PCR testing in secondary care, with no corresponding evidence in community care, to community care settings. In the second section of the questionnaire, panellists were presented with four statements derived from conflicting evidence in secondary care and asked to rate their level of agreement with these statements in secondary care settings. Additionally, panellists were presented with five statements outlining potential benefits of rapid PCR testing in community care, with weak or no supporting evidence in the literature, and instructed to rate their agreement with these statements in community care settings to identify key evidence gaps. The third section of the questionnaire asked panellists to identify the main barriers to community-based PCR testing for ARIs and rank the most ideal setting for its implementation. For the complete questionnaire, see Supplementary File 1.
Step 9: Delphi Questionnaire 2
After receiving responses to the first Delphi questionnaire, a second-round questionnaire was distributed to panellists in June 2024. The questionnaire was developed based on the results and feedback from the first questionnaire, alongside input from survey investigators. The first section of the questionnaire consisted of five statements on rapid PCR testing in secondary care, while the second section of the questionnaire consisted of six statements combining the benefits of rapid multiplex viral PCR testing at the POC in secondary care with conflicting evidence and the benefits of rapid multiplex viral PCR testing in community care with weak/no evidence. The third section of the questionnaire was refined to limit panellists to selecting their top two ideal settings for community-based PCR testing, aiming for wider agreement. A new setting was also added based on panellist feedback. For the complete questionnaire, see Supplementary File 2.
Step 10: Consensus Panel
Following the two Delphi questionnaires, a consensus panel discussion took place in July 2024 at the Royal Society of Medicine in London, England, where outstanding areas of uncertainty from the questionnaires were addressed. Seven of nine panellists (78%) were in attendance. Follow-up calls were conducted with two panellists who were unable to attend.
The subsequent consensus panel focused on four core objectives: (1) discussing statements from section one of the second questionnaire that lacked consensus, (2) recommending evidence generation methods for statements in section two of the second questionnaire that did not achieve consensus in the first questionnaire, (3) identifying the ideal setting for POC testing for ARIs in the UK, and (4) provide recommendations for future research.
This in-person discussion fostered a deeper and more dynamic exchange of perspectives, allowing for the resolution of outstanding disagreements. As a result, consensus was ultimately reached on all statements that had initially lacked agreement, further reinforcing an exploratory rather than prescriptive tone. The combination of two rounds of questionnaires followed by the interactive, expert discussion provided the necessary depth for consensus without the need for additional Delphi rounds. Despite the small panel size, the agreement observed during the Delphi questionnaire rounds, coupled with expert panel, lead to robust conclusions. Though this approach diverges from the traditional Delphi format, the combination of two questionnaire rounds followed by an in-person consensus meeting effectively achieved the study’s objectives.
For a high-level overview of the consensus process, including the number of statements that reached agreement at each stage, please refer to Figure 1.
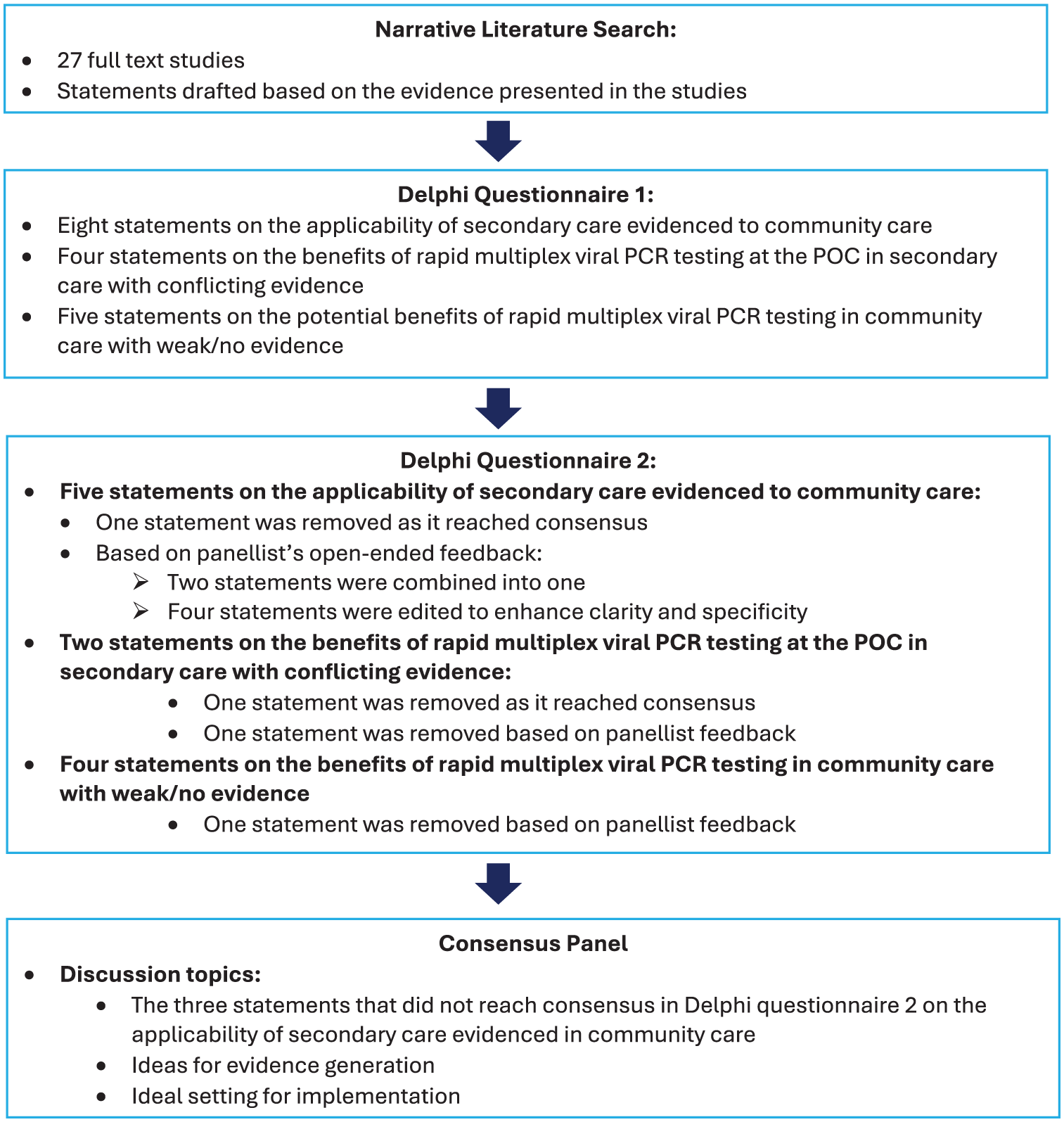
Overview of the modified Delphi methodology, including the narrative literature search, Delphi questionnaire 1, Delphi questionnaire 2, and consensus panel.
Results
Narrative Review Process and Statement Development
The search strategy created for this narrative literature review yielded 2461 results from the selected databases: PubMed and Embase. 65 studies were eligible for full-text screening, and 27 studies advanced past the full text screening phase and were included in the final review. The main reason for the exclusion of a study was the multiplex viral PCR test utilised in the study detected only a single virus (including multiple strains of the same virus) (n = 23), the study was not focused on ARIs (n = 12) and/or the study was focused on the outcomes or success of treatment methods, rather than the type of treatment provided (n = 2). For information on how an AI-assisted review software (Nested Knowledge, MN, USA) was involved in the screening process, see Supplementary File 3.
Statements were generated based on relevant information in included studies, and categorised into three groups:
(1) Statements with non-conflicting evidence in secondary care.
(2) Statements with conflicting evidence in secondary care.
(3) Statements deemed important but lacking supporting evidence.
These categorised statements formed the basis for the first questionnaire.
Delphi Questionnaire 1
Of the nine experts recruited for the panel, eight (89%) completed the first Delphi questionnaire (see Supplementary File 1 for question format). As eight panellists completed the questionnaire, consensus was defined as at least six of eight participants selecting ‘agree’ or ‘strongly agree’ (4 or 5), or ‘disagree’ or ‘strongly disagree’ (1 or 2). Though this equates to 75% of panellists, it is the closest number of panellists to the predefined consensus threshold of 80%.
In first section (see Table 2) of the first questionnaire, 2 out of 8 statements reached consensus and open-ended feedback was provided for the remaining statements (see Figure 2).
Overview of Topics Covered in the Three Sections of Delphi Questionnaires 1.

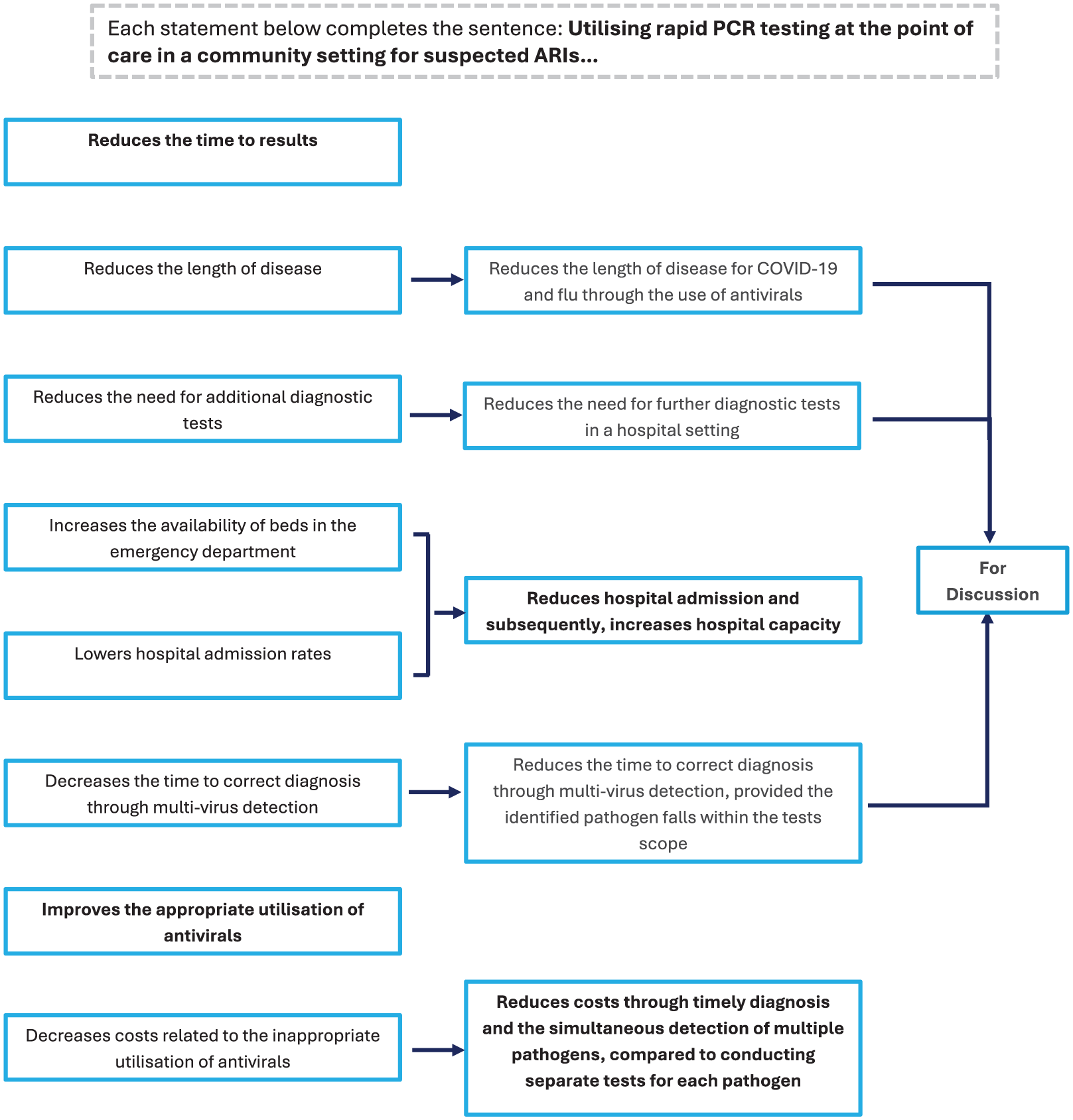
Refinement of eight statements on the applicability of secondary care evidence to community care from Delphi questionnaire 1 to consensus panel.
In second section (see Table 2) of the first questionnaire, 1/9 statements reached consensus, and two additional statements were removed (see Figure 2). Panellists felt these statements were not plausible in the given context, had no proven benefits, and failed to consider additional factors.
The results of the third section highlighted panellists’ views on primary barriers (see Figure 3) and risks associated with multiplex PCR testing implementation, as well as ideal settings for community-based PCR testing (see Figure 5a). 18 Open ended feedback from panellists lead to the removal of emergency departments/hospitals from the list of ideal settings and the addition of care/nursing homes.
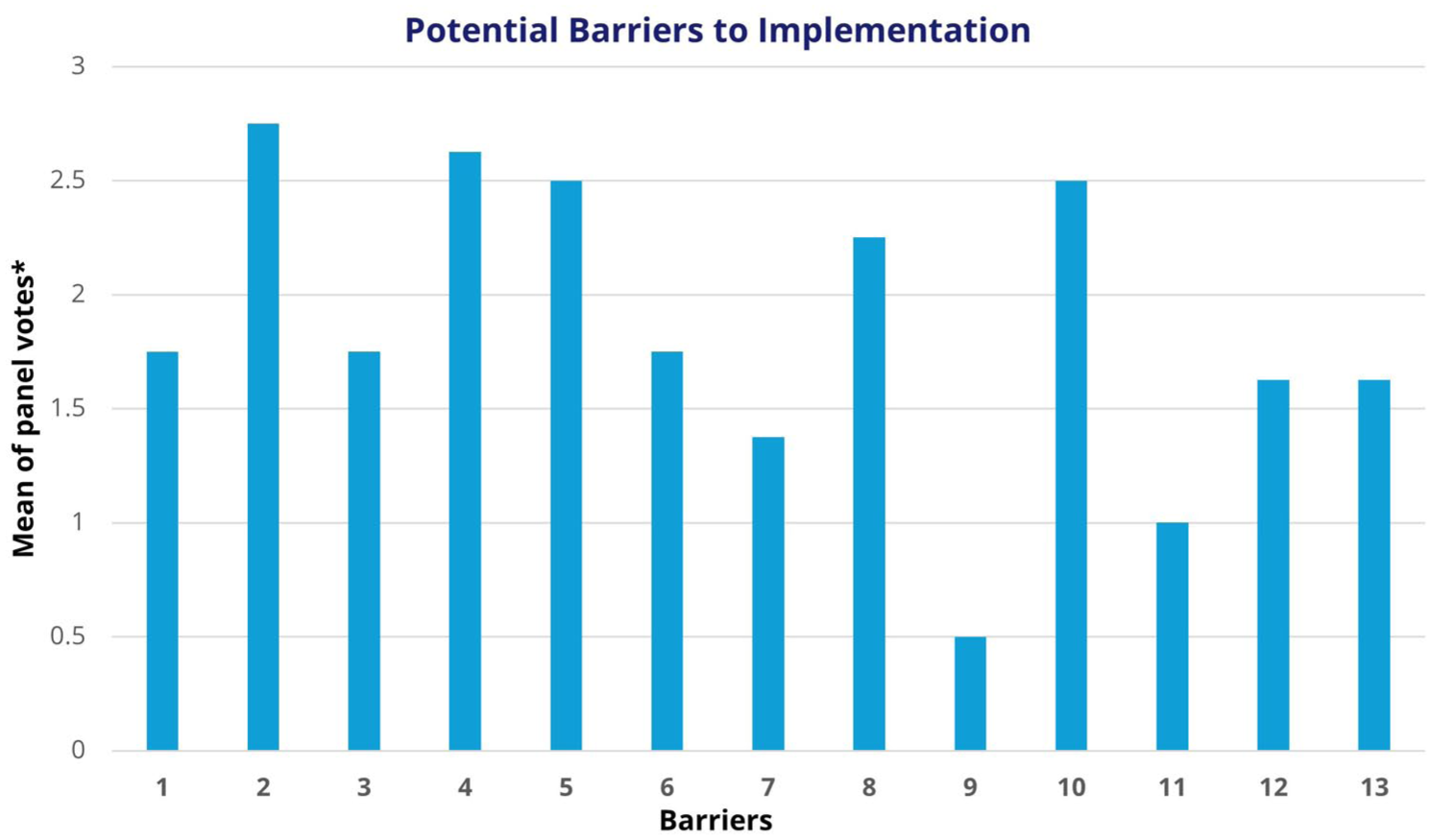
Panellists’ results from Delphi questionnaire 1 on the primary barriers associated with utilising PCR testing for ARIs at the POC.
Delphi Questionnaire 2
Of the 9 experts recruited for the panel, seven (78%) completed the second questionnaire (see Supplementary File 2 for question format). As seven panellists completed the questionnaire, consensus was defined as at least six of seven participants selecting ‘agree’ or ‘strongly agree’ (4 or 5) or ‘disagree’ or ‘strongly disagree’ (1 or 2). Although this equates to 78% of panellists, it is the closet number of panellists to the predefined consensus threshold of 80%.
First section of the second questionnaire consisted of five statements on the applicability of secondary care evidence to community care (see Figure 2). The statements that did not reach consensus in the second questionnaire were carried forward for further discussion at the consensus panel (see Figure 3). Notably, the three statements that moved forward for discussion received five out of seven votes, meaning just one more vote would have brought them to consensus. All statements reached consensus among panellists at the discussion.
Second section of the second questionnaire consisted of six statements (see Figure 4), where panellists outlined the type of evidence required to determine the relevance of the provided statement, as well as how evidence would be collected (see Figure 4).

Nine statements with conflicting or weak/no evidence presented in Delphi questionnaire 1 and panellists’ suggestions for evidence generation for statements that progressed to Delphi questionnaire 2.
Lastly, section three of the second questionnaire instructed panellists to select two of five settings as the ideal setting for POC testing for ARIS in the UK (see Figure 5b).

(a) Panellist responses from Delphi questionnaire 1 on the most appropriate setting for POCT and (b) Panellist responses from Delphi questionnaire 2 on the most appropriate setting for POCT.
Consensus Panel
Panel Logistics and Consensus Statements
The first part of the consensus panel addressed the three statements on the applicability of secondary care evidence to community care, which had not reached consensus in the second questionnaire. Panellists agreed to remove caveats from statements and include the word ‘may’ in each statement, reflecting the lack of sufficient evidence to support them definitively at this stage.
Panellists also agreed on the need to establish definitions for what constitutes ‘high risk’ and ‘community settings’. One panellist highlighted the need to clarify PCR testing is not part of primary care, and its introduction in primary care would supplement existing testing mechanisms rather than replace them. Refer to Table 3 for detailed panellist feedback on each statement, including final consensus statements.
Refinement of the Three Statements That Did Not Reach Consensus in Delphi Questionnaire 2, from the Original Statements Presented in Delphi Questionnaire 1 to the Final Statement Agreed Upon at the Consensus.

Evidence Generation
The second part of the consensus panel discussion focused on evidence generation. Panellists were presented with six statements of uncertain evidence previously introduced in the second section of the second questionnaire. The six statements were grouped into the following three categories based on similarity:
(1) improve patient flow and lower testing threshold,
(2) reduce emergency department attendance and length of stay, and
(3) patient and clinician satisfaction.
Panellists discussed how evidence could be obtained for each category of uncertainty to allow for a more informed decision about technology adoption. This was done through the Population, Intervention, Comparison, and Outcome (PICO) framework – a model used to structure research questions and guide both the generation and evaluation of evidence. Panellists agreed that evidence should investigate the redirection of resources from reactive to proactive care settings, as well as reduce the duplication of testing in secondary care.
Discussion
Discussions with nine experts in the field of ARIs led to a consensus on the potential value of implementing rapid multi-virus testing at the point of care in community settings, with the general direction in favour of implementing the test as a standard. However, strong clinical and economic evidence is essential to ensure certainty in its value. Standardised testing across the healthcare system and ease of use are crucial factors in ensuring the introduction of testing in primary care contributes to reducing winter hospital pressures and curbing inappropriate antibiotic use, influencing the fight against antimicrobial resistance (AMR).
Multiplex viral PCR testing has the potential to help address the overprescription of antibiotics, a major driver of AMR, which the WHO has identified as one of the most significant global public health threats. 19 In response, the UK government has developed strategies to reduce infection rates, optimise antimicrobial use, and advance diagnostic tools to support these efforts. 20
Due to differing guidelines from NHS England (2023) and NICE (2023) and a clear unmet evidence need on POC testing for ARIs using PCR testing, we used a modified Delphi methodology to generate expert consensus on the topic. Three statements on the applicability of secondary care evidence to primary care, which lacked consensus after two Delphi questionnaires, were discussed by the panel. The panel recommended removing caveats from the statements and adding the word ‘may’ to make the statements less definitive. Panellists were then presented with six statements of uncertain evidence and utilised a PICO framework to generate an evidence framework for a single study. Emphasis was placed on the importance of generating evidence that could influence clinical guidelines. Although an RCT provides stronger evidence for outcomes, panellists concluded that a RWE study is essential in demonstrating the feasibility of multi-virus testing in a tangible setting. Lastly, the panel agreed that two common barriers to the implementation of such testing are the cost/financing of the test, and the implementation of the test.
During the consensus discussion, panellists agreed that most consensus statements generated from evidence in community care were plausible. When considering the applicability of statements based on secondary care evidence, the panel did not identify specific reasons why such evidence would not be transferable to community setting. However, they acknowledged that evidence derived from community care is still needed to fully understand these impacts. Panellists also provided constructive feedback on each statement, including potential obstacles of test implementation such as testing accuracy outside hospital settings, potentially leading to repeat testing in secondary care. To minimise this risk, panellists believed maintaining consistent sampling standards and platforms across care settings could be beneficial. In any case, further evidence would be needed to make any informed decisions about the potential value of multiplex viral PCR POCT in community care. For statements derived from conflicting evidence (secondary care) and weak/no evidence (community care), the panel agreed that these were logical hypotheses based on clinical and managerial experience but required validation by high-quality research.
Panellist consensus and insights can be used to inform updates to NHS England and NICE guidance by providing expert driven direction in the absence of definitive evidence. While both guidelines highlight a gap in the current evidence base and emphasise the need for further evidence generation to support concrete recommendations, consensus highlights likely clinical and operational values of multiplex viral PCR POCT in community care and align with national health priorities within the NHS. As a result, they can help shape the development of interim or conditional recommendations and assist in emphasizing the urgency of future research to bridge the evidence gap to support broader adoption.
Panel Recommendations for Future Research
Panellists discussed potential research study designs that could clarify uncertainties in the evidence and allow for more informed decision making in adopting multiplex viral PCR POCT in a community setting. They agreed studies could be completed in a stepwise manner, where a RWE study could be completed first (with data collected during routine practice) and would suit the needs of a commissioner (for example an integrated care board in England), while a multi-centre RCT should be completed second and would suit the needs of a health technology assessment (HTA) body or clinical guideline development group.
NICE has established a robust framework for RWE, which enhances quality, guides decision making, and offers comprehensive guidelines outlining different principles of RWE. 21 The panel proposed a RWE study could be completed as a pre-post observational study through a technology pilot, where sufficient data is collected to account for potential variations in outcomes due to seasonality. Such a study could analyse retrospective data collected before the technology is implemented into community care and compare this to prospectively collected data. This study could offer initial data on cost and clinical outcomes in real practice to support a business case and test patient acceptance. Commercial sponsorship may be explored at this stage to support the scalability of the intervention.
Next, a RCT would have strong impact and robustness if facilitated by a research group independently of industry involvement. However, these studies require external grant funding often contingent on the completion of a successful feasibility phase. Therefore, the RWE study can demonstrate feasibility for a RCT and can be used to perform a power calculation to determine RCT sample size. Though an RCT is more rigorous, it provides stronger outcome evidence and generates high-quality research, essential for demonstrating cost-effectiveness. Panellists agreed that measurable effect sizes should be a key focus of the RCT, with robust data collection.
The panel also addressed key barriers to test implementation, including funding for tests and affordability during winter surges, reinforcing the need for further research (RWE/RCT) to generate the clinical evidence needed for informed decision-making. Next, panellists noted that the value of multiplex PCR POCT is likely to vary by risk group, with higher-risk populations (eg, those eligible for influenza vaccination) expected to benefit most. Consequently, a key future consideration should be determining whether the technology should be broadly implemented (ie, also to lower-risk subgroup populations).
Additionally, the ideal setting for PCR POCT was discussed. Some panel members recommended implementing testing in nursing homes, care homes, and multigenerational households with high-risk individuals, given the vulnerability of these populations. However, this focus may overlook younger patients, many of whom attend EDs for ARIs. During a single week in November 2022, hospitals in England experienced a 40% surge in influenza admission rates. Notably, 230 children under the age of were admitted, a significant rise from 12 children within the same group during the corresponding week in the previous year. 22 While many young patients face a low risk of complications from ARIs, their presence in EDs adds to the strain on the healthcare system. Expanding POC testing to alternative settings could help alleviate some of this pressure on EDs, highlighting the importance of including both high-risk and non-high-risk patients as target populations in the outlined studies.
The panel also explored additional study considerations, including socioeconomic status, social deprivation, and language barriers, emphasising the need to incorporate quality of life and National Health Services (NHS) resource allocation into study outcomes.
Limitations
This modified Delphi consensus study aimed to gather expert insights into the potential clinical utility and implementation of multiplex viral PCR POCT for ARIs. While this work provides valuable early-stage perspectives into the feasibility of implementation, several limitations must be acknowledged.
Firstly, we recognise methodological limitations are present in the study. The final panel did not include early-career professionals (less than 10-15 years of experience in infectious disease). Though we prioritised individuals with substantial expertise to ensure a high level of subject matter expertise, this may have limited the inclusion of emerging perspectives from individuals early on in their career. The panel was also comprised of one female, which was a result of both the small and highly specialised pool of eligible experts, and the gender of experts who agreed to participate in the panel. However, we recognise that greater gender diversity could have expanded perspectives and increased the depth of insights. Next, a narrative review was selected over systematic or scoping review approaches, due to the early, exploratory nature of the research topic, which required flexibility in identifying and synthesizing relevant literature. However, narrative reviews inherently lack a standardised methodology, which can introduce selection bias and may reduce the comprehensives of the reviewed literature. 14 Additionally, we acknowledge that Delphi questionnaire 1 was pilot tested with one of nine panel members (11%), raising concerns on potential bias. We recognise that piloting with a single individual carries the risk of bias feedback, particularly if that person is overly familiar with the content, consults external sources, or invests more time in the piloting process than other panel participants would have. However, our intent in this pilot was not to validate the questionnaire is a formal sense, but rather to conduct a pragmatic check of Delphi questionnaire 1 for clarity, flow, feasibility, and included content. Therefore, we believed one reviewer to be sufficient in identify concerns and avoid conflicting opinions at an early stage without introducing bias. While we recognise a formal piloting process involving additional panellists could have enhanced robustness by capturing a wider range of perspectives, we considered this targeted approach appropriate for the scope and aim of our study. Internal validity was strengthened through structured feedback gathered through the Delphi rounds, allowing for revisions in questionnaire structure and statements for Delphi questionnaire 2. 23 Nonetheless, a formal piloting process is an important consideration for future Delphi studies.
Next, the size of the panel and its composition presents limitations. This study did not include a formal sample size calculation or statistical power analysis to determine the ideal number of participants. While such analysis can strengthen justification, they are not standard practice in Delphi studies, which typically rely on expert consensus rather than statistical inference. Instead, sample size was determined based on a practical rationale, including the number of available experts in the field. Although the final panel comprised only nine experts, this aligns with accepted practices for Delphi studies in specialised, homogeneous fields. However, the small panel size may have limited the diversity of perspectives. 15 In addition, the panel lacked patient representation, essential for shaping the design of clinical studies and the inclusion of patient reported outcomes, as well as UK HTA representation, which are critical for understanding evidence and cost effectiveness requirements. Furthermore, none of the nine experts held direct policymaking authority. Despite this, several panellists held influential roles across relevant organisations. For example, SDeL served as a representative on the NICE Osteoporosis Working Group, and another panellist was affiliated with the UK Health Security Agency (UKHSA). The panel’s collective interdisciplinary expertise, including general practitioners who also held academic positions, helped offset the limitations of the small panel size. These overlapping roles enabled the group to contribute a broad and integrated perspective on infectious disease issues. However, future studies should prioritise the inclusion of these groups (patients, HTA professionals and policy makers) to ensure more comprehensive and policy relevant insights. Additionally, the generalisability of the study is limited by the composition of the panel, which included only experts based in England. However, this was an intentional decision, designed to reflect the structure of the NHS and national diagnostic policymaking. Although recommendations were generated as context specific, several recommendations, such as the need for evidence generation and subgroup analysis, are likely applicable to other healthcare systems.
Moreover, the study has potential for selection bias, as experts who agreed to participate may have been more familiar with PCR POCT for infectious diseases and the specific technology investigated. Nonetheless, several panellists brought experience with a range of diagnostic technologies, contributing to a balanced perspective.
Next, the nature of evidence serves as a limitation in this study. As with all expert elicitation methods, findings of this study are opinion-based and considered lower-tier evidence compared to data generated through clinical trials or RWE studies, carrying a risk of subjectivity and bias. To mitigate bias, we conducted two anonymised Delphi questionnaire rounds where panel members and study investigators were blinded, preventing the identification of panel respondents. The purpose of blinding was to encourage independent thinking and prevent dominant voices from influencing responses. However, blinding served as a limitation, as it prevented the ability to determine which specialities were underrepresented within questionnaires, or dropout patterns by specialty. Nevertheless, blinding was a deliberate choice to reduce the risk of study investigators unintentionally steering the direction of the in-person discussion based on panellists’ prior responses. As a result, the consensus panel allowed for open, transparent, and inclusive discussion, providing depth needed to strengthen study outcomes.
Lastly, the lack of RWE or trial-based evidence serves as a key limitation for informed decision making. Though panellists were positive on the potential of implementing multiplex PCR POCT, they recognised that expert opinion in the absence of supporting trials (such as RCTs) or RWE studies is not sufficient to guide implementation. Consequently, the panel adopted a more exploratory tone, deliberately adjusting the language of consensus statements to reflect possibility rather than certainty. For example, statements originally phrased as ‘can reduce’ were revised to ‘may reduce’, signalling a cautious, evidence-aware approach. The panel emphasised the need for further research to validate these perspectives and support informed implementation. This study serves as an initial step to inform, guide and prioritise future research, including clinical evaluations, RWE generation, and economic analyses. Subsequent studies outlined are essential to produce robust data needed for evidence-based decision-making.
Conclusions
Based on the current evidence of using multiplex viral PCR testing in secondary care, there is potential value for healthcare systems to utilise such testing in community care settings, predominantly primary care and care homes. While existing evidence and expert consensus indicate a likely benefit, further studies were recommended. Further evidence should be gathered in community care settings to investigate the impact and cost-effectiveness of this new approach, which can inform guideline development to ensure consistency in practice and patient’s access to these technologies. It is important to recognise that ARIs must be understood within the context of the entire healthcare system, including both primary and secondary care. Future implementations should avoid shifting pressures within the system. Quality assurance and adequate staffing are essential to effectively support any new testing procedures. 24
Supplemental Material
sj-docx-1-his-10.1177_11786329251374554 – Supplemental material for The Potential Impact of Primary Care-Based Multiplex Polymerase Chain Reaction Point of Care Testing for Viral Acute Respiratory Infections in the UK: Modified Delphi Study
Supplemental material, sj-docx-1-his-10.1177_11786329251374554 for The Potential Impact of Primary Care-Based Multiplex Polymerase Chain Reaction Point of Care Testing for Viral Acute Respiratory Infections in the UK: Modified Delphi Study by Simon de Lusignan, Sarah Nathens, Jamie Erskine, Anjali Ramkeesoon, Norbert Farkas, Michael R. Barer, Tristan W. Clark, Liz Cross, Bruce Daniel, Christopher George, Abid Hussain, David Thorne and Terry Whalley in Health Services Insights
Supplemental Material
sj-docx-2-his-10.1177_11786329251374554 – Supplemental material for The Potential Impact of Primary Care-Based Multiplex Polymerase Chain Reaction Point of Care Testing for Viral Acute Respiratory Infections in the UK: Modified Delphi Study
Supplemental material, sj-docx-2-his-10.1177_11786329251374554 for The Potential Impact of Primary Care-Based Multiplex Polymerase Chain Reaction Point of Care Testing for Viral Acute Respiratory Infections in the UK: Modified Delphi Study by Simon de Lusignan, Sarah Nathens, Jamie Erskine, Anjali Ramkeesoon, Norbert Farkas, Michael R. Barer, Tristan W. Clark, Liz Cross, Bruce Daniel, Christopher George, Abid Hussain, David Thorne and Terry Whalley in Health Services Insights
Supplemental Material
sj-docx-3-his-10.1177_11786329251374554 – Supplemental material for The Potential Impact of Primary Care-Based Multiplex Polymerase Chain Reaction Point of Care Testing for Viral Acute Respiratory Infections in the UK: Modified Delphi Study
Supplemental material, sj-docx-3-his-10.1177_11786329251374554 for The Potential Impact of Primary Care-Based Multiplex Polymerase Chain Reaction Point of Care Testing for Viral Acute Respiratory Infections in the UK: Modified Delphi Study by Simon de Lusignan, Sarah Nathens, Jamie Erskine, Anjali Ramkeesoon, Norbert Farkas, Michael R. Barer, Tristan W. Clark, Liz Cross, Bruce Daniel, Christopher George, Abid Hussain, David Thorne and Terry Whalley in Health Services Insights
Footnotes
Appendix A: Search Strategy
(Antimicrobial Resistance
Acknowledgements
We would like to thank our expert panel for their continuous and valuable support and time throughout this process, from filling out the questionnaires to serving as valuable contributors and reviewers for this manuscript.
Ethical Considerations
Ethical approval was not sought for this study. According to the Declaration of Helsinki, ethical approval is required for ‘research involving human subjects, including research on identifiable human material and data’ (Article 1). This study involved a modified Delphi panel, focusing on gathering expert opinions and did not involve the collection or use of personal health data, clinical interventions, biological materials, research of NHS service users, confidential health records, carers, or vulnerable populations. To confirm this, we used the NHS Research Ethics Committee (REC) decision tool, which is the official mechanism for determining whether ethical approval is needed in the UK. The tool is comprised of 17 questions across four domains, none of which applied to our study. Based on our responses, the tool confirmed that ethical approval was not required. Full details of the questions in the NHS Research Ethics Committee (REC) decision tool are provided in the link embedded in the citation below.
Next, participants were engaged as expert stakeholders purposively recruited for their subject matter expertise and were co-authors of the resulting paper, reinforcing their role as collaborators rather than research subjects. The study aimed to gain insight into participants professional expertise rather than personal or identifiable information. Therefore, in accordance with both the Declaration of Helsinki and NHS REC guidance, ethical approval was not required.
Citation: World Medical Association. Declaration of Helsinki: Ethical principles for medical research involving human participants. JAMA. 2025;333(1):71-74. doi:10.1001/jama.2024.21972.
Consent to Participate
All participants provided written informed consent to participate in the study. Before the study commenced and written informed consent was obtained, participants were made aware of the studies objective, methodology, and final output.
Participants signed a non-disclosure agreement (NDA) issued by Alira Health, which included a no-obligation clause, allowing either party to terminate participation at any time. Before the panel discussion, participants were anonymised to one another and were made aware that the panel would involve a group discussion where they would be encouraged to express views openly. During the consensus panel, measures were taken to encourage open discussion and minimise undue influence or groupthink, including structured moderation to ensure equal participation among all panel members.
Each participant signed an independent contractor agreement outlining study objectives, fair market value (FMV)-based remuneration (if applicable), expected time commitment, and applicable dates of the Delphi questionnaires and Delphi panel. Compensation was provided to participants as part of a standard professional consulting arrangement to acknowledge their time, effort, and professional expertise, and was not intended to pressure or influence their decision to participate or express certain views. The agreement also included confidently provisions to safeguard all data shared during the study. Specifically, the agreement stated that throughout the term of the agreement (throughout the Delphi questionnaires and Delphi panel), and for a period of 3 years after, the contract facilitator will hold all confidential information in confidence, not copy or use (or allow any of its employees, contractors or agents to copy or use) any confidential information (except as may be necessary to perform the services), use the confidential information only for the benefit of the contract facilitator (and not for the benefit of the participants or any third party), and not disclose or otherwise make available confidential information to any third party except as authorised in writing and in advance by the contract facilitator.
All digital data used for the purpose of the study (ie, survey responses, meeting notes, and transcripts) were stored securely on encrypted, access restricted symptoms managed by Alira Health. Recordings were used exclusively for analysis and were anonymised in all final outputs. No statements or opinions were attributed to individual participants.
Consent for Publication
Not applicable. No identifiable personal data, images, or other individual level information are included in this publication.
Author Contributions
SN, JE, and AR conducted the narrative literature review, recruited the Delphi panel, designed and administered the two rounds of questionnaires, collected and analysed data, facilitated the consensus panel discussion, and summarised findings to draft manuscript. All panellists - TWC, TW, AH, MRB, CG, DT, BD, SDL, LC - made valuable contributions to the panel provided feedback for submission. NF oversaw the process, offering expertise and guidance throughout.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Alira Health received consulting fees from Cepheid for the consensus panel meeting, encompassing reimbursement for travel, accommodation costs, and panel participation. The funder had no role in the design of the Delphi study including the narrative literature review; creation, distribution, and analysis of the questionnaire; the panel; and the writing of the manuscript. It is also important to note that the funder did not influence panel outcomes.
Declaration of Conflict of Interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SN, JE, AR, and NF are employees of Alira Health, which received consulting fees from Cepheid for coordinating the panel. These fees included payments to the following panellists for their contributions to the project: TWC, TW, AH, MRB, CG, DT, SDeL, and LC – for their contributions to the project.
TWC has received research grants from BioFire Diagnostics, Biomerieux, QIAGEN, Sense Biodetection, and Inflammatix. He has received speaker fees, honoraria, and travel reimbursement from BioFire, Biomerieux, QIAGEN, Cepheid, and Janssen, as well as consultancy fees from Biomerieux, QIAGEN, Cepheid, Roche, Janssen, and Synairgen. He has served on advisory boards for Cepheid, Roche, Janssen, GSK, Shiongi, Sanofi, and Seqirus and is a member of an independent data monitoring committee for a Roche-sponsored trial. He owns shares in Synairgen plc.
DT is the owner Director of Health Policy Explained Limited. He has received honoraria for lectures from iRythym, ResMed, WA Communications, Regeneron, Gedeon Richter, PA Consulting, Thornhill Events, Baxter Medical, Chase People, Consilient, Daiichi Sankyo, Dovetail, Boehringer Ingelheim, Pierre Fabre, AstraZeneca, Bayer, AbbVie, Accord, Alira Health, Amgen, Biogen, Connect Health, Convatec, Urgo, Ferring, Ipsen, Lily, Nevro Medical, Novartis, Sandoz, Tesla Radiology, Warn, and Mtech Access. He has served on advisory boards for Verpora, Wilmington, Alira, Aspire, Decisive, and Mtech Access and received research funding from AstraZeneca through his Primary Care Network Director role.
SdeL holds or has held grants in his institution for respiratory vaccine research from AstraZeneca, GSK, Moderna, MSD, Sanofi, and Seqirus. Moderna, Sanofi and Roche have supported point of care testing research. SdeL been a member of advisory boards for AstraZeneca, GSK and Sanofi.
TW, AH, BD, MRB, CG, and LC do not have any conflicts of interest to declare.
Data Availability Statement
Two rounds of Delphi questionnaires distributed to the panel are included as supplementary material. Additional data is available on request from authors.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
