Abstract
Background:
Sustainable injectable drug delivery devices, such as prefilled syringes, multi-chamber syringes, and reusable systems, are gaining attention due to their potential to decrease resource use and waste. Despite these considerations, the integration of sustainability metrics into healthcare systems and pharmaceutical manufacturing is inconsistent, necessitating a systematic review of existing practices and outcomes.
Methods:
To address this gap, a systematic literature review (SLR) was conducted in June 2024, utilizing the databases of PubMed/MEDLINE and Embase. The studies were examined for sustainability outcomes related to environmental impact, economic efficiency, and public health benefits of injectable drug delivery systems. Cost adjustments were standardized to their respective 2024 currency along with U.S. dollars using price indices for the United States, United Kingdom, Netherlands, and Italy.
Results:
The literature search returned 2384 unique articles, with 24 studies meeting the inclusion criteria. The reviewed studies highlighted some of the potential benefits of sustainable injectable drug delivery devices. Environmental assessments reported reductions in waste and CO2 emissions, with examples from hospital implementations of prefilled syringes cutting waste by 26% to 86%, and modular insulin pumps showing a 44% increase in recycling efficiency compared to conventional systems. Economic evaluations revealed potential cost reductions of up to 26% of total drug expenditures, attributed to inefficient drug preparation processes and unused medications. Public health outcomes ranged from identified challenges in disposal practices to safer sustainable strategies, including a 57% reduction in hazardous waste that enhanced safety in a resource-limited setting. While the included studies varied in methodological rigor, most were rated as fair quality, with 2 studies rated as poor – indicating a need for more robust evidence to strengthen future evaluations.
Conclusion:
This review suggests that sustainable injectable drug delivery devices can reduce environmental impact, improve economic efficiency, and contribute to public health. By limiting waste, reducing carbon emissions, and supporting safety, these devices can offer measurable contributions for advancing sustainability in healthcare. However, the lack of clear sustainability standards and guidance from health systems and governments remains a key barrier to broader adoption.
Keywords
Introduction
Pharmaceuticals, medical technology, and drug delivery devices can affect the environment at multiple stages of a product’s lifecycle.1,2 Sustainability is gaining attention in healthcare for its potential to support environmental, economic, and public health improvements. In this context, sustainability generally refers to reducing environmental impact, minimizing material waste, supporting reusability and recyclability through devices such as prefilled or multi-chamber syringes, and reusable systems. Injectable drug delivery devices have become a focus for environmental outcomes due to their widespread use and contribution to medical waste. Sustainability considerations now extend beyond reducing ecological harm to also include alignment with evolving frameworks like health technology assessments, which evaluate the clinical, economic, and sometimes environmental value of health interventions.
Health technology assessments are beginning to incorporate environmental criteria, reflecting a broader shift toward sustainability in healthcare decision-making. However, the degree of integration varies across regions and programs, and no single structure currently guides this process.3,4 For example, the Health Technology Assessment Core Model in the European Union (EU) considers environmental safety as a domain in product evaluations, 5 while the UK NICE Strategy for 2021 to 2026 and similar programs in France, Spain, Netherlands, Sweeden, and Canada acknowledge integrating sustainability needs in emerging technologies to decrease healthcare’s carbon footprint.6 -11 These frameworks show an overall trend to integrate sustainability into healthcare systems, providing additional incentives to adopt sustainable practices, even in the absence of specific regulatory mandates.
The push to develop sustainable drug delivery devices is driven by the need to reduce carbon emissions, minimize waste, and lower costs. As a result, sustainability assessments and metrics are becoming increasingly important for improving drug delivery systems. 2 By incorporating health technology assessments alongside these metrics, healthcare systems can better evaluate the environmental, economic, and public health effects of medical technologies – and potentially improve sustainability outcomes.
Consequently, interest continues to grow in developing sustainable drug delivery systems that aim to limit the environmental impact through more efficient design and sustainable practices.12,13 Additionally, product lifecycle assessments are increasingly being used to evaluate the ecological footprint of healthcare products such as pharmaceuticals, medical technologies, and drug delivery devices.14,15 Both corporate sustainability considerations and evolving regulatory initiatives continue to influence sustainable practices across the healthcare spectrum.16,17 However, standards can vary from country to country, and even from state to state within countries,3,4 which shows the importance of having more consistent regulatory frameworks to support sustainability in drug delivery devices.
An analysis of public reports published in 2020 and/or 2021 from 20 pharmaceutical manufacturers showed that 95% (19 out of the 20) of them had set internal targets to reduce their greenhouse emissions. 16 Additionally, half had publicly committed to becoming carbon neutral, along with 40% aiming for net zero emissions. 16 While the pharmaceutical and biotech industries are making efforts to adopt sustainable practices throughout the product lifecycle – including development, manufacturing, and packaging – they continue to face challenges. 17 A collaborative, multistakeholder approach remains important for moving toward more sustainable healthcare products and systems. 18
As part of broader sustainability efforts, drug delivery devices may help reduce waste and contribute sustainability efforts in medical settings, though their impact can differ compared to other waste management strategies like minimizing single-use devices in operating rooms. Within the pharmaceutical and medical device industry, there is a growing attention being given to environmental sustainability with a particular emphasis on cleaner production and reduced material wastage.17,19,20
Overall, sustainability in drug delivery devices is characterized by an increased attention to developing of sustainable processes and using consistent metrics. This review evaluates sustainability metrics across the environmental, economic, and public health domains to assess the impact of these devices. To narrow the scope of the research, it focuses specifically on injectable drug delivery devices – such as prefilled syringes, multi-chamber syringes, and reusable delivery systems – and their role in supporting sustainability in healthcare settings.
Methods
Search Strategy
A systematic literature review (SLR) was performed to assess sustainability of injectable drug delivery devices. Searches were conducted in PubMed/MEDLINE and Embase while using appropriate database syntax in June 2024; some relevant studies from other databases may not have been captured. The respective search terminology and search strategy are available in Appendix Tables 1 and 2 in the Supplemental Material. The literature search was conducted using key terms related to sustainability outcomes and injectable drug delivery devices: wastage, energy efficiency, CO2 footprint, greenhouse emissions, recycling, cost savings, reusable, disposable devices, prefilled syringes and auto-injectors, among others. This SLR was conducted according to industry best practices21 -23 and was reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (Appendix Table 3 in the Supplemental Material).24,25 A review of citations was also conducted of all full-text reviews to identify studies that were not indexed in the searched databases but met the SLR’s inclusion criteria.21 -23
Study Selection
Eligibility for inclusion was predefined according to Population, Intervention, Comparator, Outcome, Timing, and Setting (PICOTS) Study Design criteria (Table 1). Studies were eligible for inclusion if they were published between January 1, 1970 and June 1, 2024. Analyses to be considered included prospective and retrospective observational studies, economic analyses, real-world evidence (RWE) studies reporting results of injectable drug delivery devices, laboratory analyses, modeled analyses, and surveys. Economic models and budget analyses were required to be informed by rigorous proprietary information, literature-reported values, and/or expert opinion. Conference abstracts were allowed to be included if they provided enough detail surrounding the study methodology, or if a conference poster of the study was publicly available that provided this information. After removing duplicates, the search consisted of a review of titles and abstracts, and then a review of full texts for studies that appeared to meet the inclusion criteria. The search and appraisal were conducted by 2 reviewers. For studies with conflicting decisions regarding the inclusion/exclusion status, a third author reviewed the studies of interest and made the final decision. This was also done to reduce potential bias given that all authors are affiliated with 1 corporation. The review was conducted by 3 independent reviewers from different departments and regions (Medical Affairs and Health Economics and Outcomes Research; United States and Europe). All screening and data abstraction steps followed PRISMA guidelines, including independent verification.
SLR Eligibility Criteria.

Data Abstraction
This review qualitatively synthesizes findings from various studies, selected based on inclusion criteria that focus on sustainability of injectable drug delivery devices. Abstraction of data from the studies was conducted by a single reviewer with a separate reviewer verifying the accuracy and completeness of the abstracted data. Quality assessments were conducted by 2 reviewers for all included studies with the National Institutes of Health’s Quality Assessment Tool for Observational Cohort and Cross-Sectional Studies being utilized for observational cohort and cross-section studies, 26 and the Consensus on Health Economic Criteria (CHEC) for economic models. 27 For identified studies that were laboratory analyses, no qualitative assessment was conducted due to not being able to identify a consensus quality assessment tool or checklist for these types of studies. Readers are urged to use caution when interpreting the results of the laboratory analyses, and to refer to the original study since quality assessments were unable to be conducted. Qualitative synthesis was conducted to summarize findings and where possible, similar outcomes were grouped to provide frequency/rate ranges. For economic data, the costs were adjusted to their respective 2024 currency as well as to 2024 US$ using exchange rates. For costs in the United States, they were adjusted to 2024 US$ using the medical consumer price index. 28 For costs in the United Kingdom (UK), costs were adjusted using the Personal Social Services Research Unit inflation indices. 29 For costs in the Netherlands, they were adjusted using the Netherlands health services annual consumer price index, 30 while costs in Italy were adjusted using the Italy health services annual consumer price index. 31 For studies where the year of the specified cost is not mentioned, the year of the publication was used as the reference year to be conservative. The literature review aimed to assess sustainability from environmental, economic, and public health perspectives. Some studies highlight outcomes that span multiple of these categories and were included in each relevant section.
Results
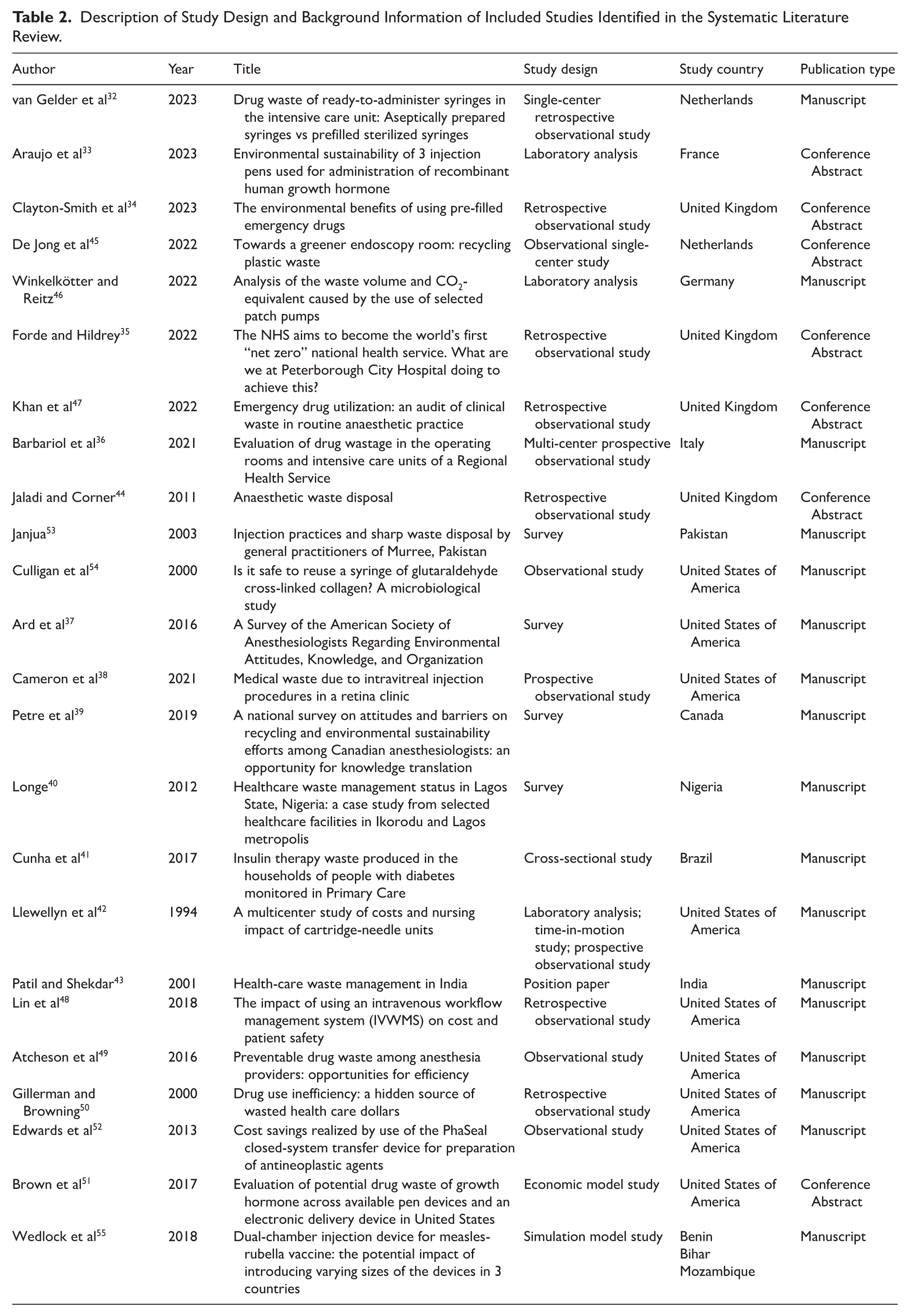
The literature search returned 427 studies in PubMed/MEDLINE and 1982 articles in Embase, and after removing duplicates there were a total of 2384 unique studies reviewed in this literature search (Figure 1). Following title/abstract screening, there were 67 studies that underwent full-text review. In total, 24 studies met our inclusion criteria and were included in this literature review (Table 2).32 -55 Of the 24 included studies, 7 were conference abstracts33 -35,44,45,47,51 and 17 were peer-reviewed manuscripts.32,36 -43,46,48 -50,52 -55

Study identification flowchart.
Description of Study Design and Background Information of Included Studies Identified in the Systematic Literature Review.
These studies showed the multifaceted impacts of implementing sustainable practices in healthcare, spanning 3 main domains: environmental, economic, and public health domains (Figure 2). The quality assessment of included studies found that 20 studies were rated as fair quality,32,34 -39,41,42,44,45,47 -55 and 2 were rated as poor (Appendix Tables 4 and 5 in the Supplemental Material).40,43 Two additional studies were not assessed due to their study design not being applicable to the quality assessment frameworks.33,46 The interpretation of study findings should be balanced against the risk of bias associated with quality grading; lower-quality studies may carry higher risk of bias, while higher-quality studies are more likely to reflect lower risk.

Sustainability of drug delivery devices framework.
Environmental Impact
Outcomes examining environmental impact were evaluated in 15 of the included studies (Table 3).32 -46 These studies report some environmental considerations of reducing injectable drug delivery device waste, and promoting recycling and reusability through sustainable practices.
Outcomes and Results of Studies Included in the Systematic Literature Review.

Waste Reduction
Several studies highlighted the role of ready-to-administer (RTA) prefilled syringes, cartridges or devices in reducing medical waste, decreasing preparation steps, and improving storage efficiency. For example, a prospective observational study conducted in 12 hospitals in northern Italy found an overall mean drug wastage rate of 38% (95% CI: 37.2-38.9) of prepared syringes, and estimated a yearly waste of 139 531 syringes and 4968 kg of medical waste, which is significant in the context of the overall medical waste in these hospitals. 36 Another retrospective analysis of 8 years of data (August 2015-May 2023) of a 32-mixed bed medical-surgical intensive care unit (ICU) evaluated the impact of replacing manually prepared syringes with RTA prefilled syringes. The study found that after the introduction of prefilled syringes, drug waste was reduced by 26-percentage points, decreasing from 31% to 5% (P < .001). 32
An audit of departmental practices for drawing up emergency drugs showed that switching to prefilled syringes led to an 86% reduction in syringes wasted (6.1-0.84 kg) and a 92.5% decrease in CO2e (40-3 kg) in 1 hospital system, suggesting that prefilled syringe implementation leads to environmental benefits and CO2e reduction. 34 A hospital in the UK also reported a 78.5% reduction in desflurane (a high carbon footprint product) waste through better anesthetic practices, minimizing the average number of 50 ml propofol vials used per month from 82 to 44. 35 Similarly, a multicenter study of 6 hospitals in the U.S. showed that use of cartridge-needle units reduced waste by 22% compared to conventional vials and syringes. 42
Several studies also provided broader evidence supporting the implementation of sustainable practices by showing the current environmental burden. For example, a study that included an analysis of hospital waste data in Calcutta, India, found that 0.43% (standard deviation [SD]: 0.31) of waste by weight was from disposable syringes. 43 Another observational case study of 20 hospitals in Nigeria also showed that 8.9% of healthcare waste was from sharps and pathological waste (including syringes). 40 Although syringes contribute to medical waste, their overall contribution appears to be limited. Studies previously mentioned suggest that prefilled syringes reduce drug overfill and preparation waste compared to manually prepared syringes,32,34,35,42 contributing to overall waste reduction, although they remain single-use devices.
Further examining waste management, a prospective observational study of a retina clinic by Cameron et al found an average 190 g of medical waste produced with each intravitreal injection, including sharps and biohazard waste (1.5%), waste from administering the procedure (16%), and waste from temperature-controlled shipping on orders of medication (83%). 38 Lastly, a cross-sectional study of 105 primary care patients with diabetes in Brazil showed that patients who were instructed on proper disposal were 21 times more likely to dispose of waste properly than those who did not receive instruction (prevalence ratio: 21.5, P < .0001). 41
Recycling and Reusability
A survey of 2189 anesthesiologists in the U.S. found that 80.1% (95% confidence interval [CI]: 78.2-81.9) were interested in recycling, but only 27.7% (95% CI: 25.7-29.9) reported recycling occurring in the operating rooms where they worked, showing a gap between awareness and action. 37 Another survey of anesthesiologists in Canada by Petre et al also indicated that more than half supported recycling practices (52%) while barriers such as lack of leadership (63.5%) and inadequate education (62.8%) hindered their implementation, 39 indicating the need for leadership and education initiatives to improve recycling programs. Furthermore, an audit of a trust in the UK identified that 11.3% of their anesthetic waste could be recycled, and extrapolated over the entire organization, a potential 4628 kg of waste could be recycled annually. 44 They also estimated that by segregating their waste, they could recycle the equivalent of 39 trees, 4 m3 landfill, and 47 barrels of oil, and if applied to the entire UK could equate to 14 083 trees, 1905 m3 landfill, and 16 825 barrels of oil, equating to saving 70 000 t carbon footprint from just anesthetic waste alone. 44 Additional evidence from clinical settings includes a single-center study in the Netherlands, where a gastrointestinal (GI) endoscopy unit found that 14.6 kg of waste collected during 15 procedures, with 9.6% of waste being recyclable plastics. 45
Modular systems used in medical devices have also indicated efficiencies, with an analysis of insulin pump systems by Winkelkötter and Reitz reporting that modular systems generated a 44.3% recycling percentage (of 5.5 kg/a total waste) compared to 2 nonmodular systems, which generated 14.6% of recycling percentage (of 4.9 kg/a total waste) and 16.0% of recycling percentage (of 5.1 kg/a total waste). 46 Modular systems had a 12% less global warming potential (GWP) with 13.6 kg CO2e compared to nonmodular systems with 15.5 kg CO2e. 46
In another study, a laboratory test of specific growth hormone (GH) therapy products by Araujo et al found that over a 3-year treatment period, the energy consumed producing reusable pens specifically was lower (1.18 kW/hour) than disposable pens (149.36 and 115.45 kW/hour), resulting in CO2e savings of 105 or 81 kg, and indicating that reusable devices can provide both energy efficiency and reduced carbon emissions over time. 33
Economic Impact
Of the studies included in this literature review, 12 had outcomes assessing economic impact.36,38,42,44,45,47 -52,55 These studies show the economic considerations of reducing medical device waste through cost reductions and efficiency improvements in medical practices.
Cost Reductions
The previously mentioned study of 12 hospitals in northern Italy showed the potential cost reductions of €83 087 (US$92 935) was achieved by using prefilled syringes, avoiding the need for advance drug preparation. 36 In a related study, an interactive model found that a reusable electronic auto-injector resulted in lower expected GH waste, with the expected annual cost in waste per-patient of $118.99 compared to costs ranging from $316.49 to $2745.98 for the single use auto-injector. 51 The previously mentioned study of 6 U.S. hospitals demonstrated that cartridge-needle units offered 22% cost savings compared to conventional systems, with lower disposal costs. 42
Cost reductions were also observed in a 12-month retrospective analysis of a single institution, which found that improving drug efficiency and preventing avoidable drug waste translated into savings, associated with waste reduction observed in anesthesia practices. 49 They also estimated that the annual cost of preventable drug waste for the institution for the 10 drugs assessed was $245 574. 48 Similarly, a 3-month pre-post retrospective analysis of a hospital information system showed that IV workflow management systems reduced wasted doses significantly, from 20 448 (18.43%) to 6272 (6.16%), a 64% reduction (P < .001), leading to cost savings of $182 251 over the 3-month post-period. 48
Similar findings were reported in an observational study of 3 hospital sites (adult inpatient, children inpatient, and adult ambulatory) in Rhode Island, which assessed 6 drugs for tracking and analysis of drug waste (atracurium, midazolam, propofol, rocuronium, succinylcholine, and thiopental) and revealed that the annual departmental drug expenditure of unadministered cost from prepared syringes was $387 856, with waste accounting for at least 26% of the total annual pharmaceutical expenditure. 50 Lastly, a 5-day observational assessment of 2 sites in the UK in 2021 showed that the proportion of drawn-to-wasted drugs was 90% for propofol, 54% for ephedrine, metaraminol, 97% for atropine, 81% for glycopyriym, and 100% for amethonium, with £10 281 (US$13 571) being the annual predicted vial cost for these drugs. 47
Efficiency Improvements
Efficiency improvements extended beyond cost reductions. For example, an analysis of closed-system transfer devices for preparation of hazardous antineoplastic medications at Walter Reed National Military Medical Center (WRNMMC) in the U.S. found that portions of the unused single-use vials could be salvaged and their beyond-use date extended. 52 Of the 21 medications assessed, with 57.03% were being wasted and could have been saved instead, representing cost reductions of $131 682 and an estimated annual saving of $960 878. Similarly, a trust in the UK spent £646 964 (US$853 998) on the disposal of clinical waste. 44 Furthermore, the single-center study in the Netherlands mentioned before also found that waste processing costs were 28% higher for recycling of plastics €2.40 (US$2.68) than all residual waste €1.87 (US$2.09). 45
Another example comes from vaccine delivery. A study on dual-chamber injection devices for measles-rubella vaccines showed efficiency improvements by reducing preparation time, eliminating reconstitution steps, and minimizing vaccine wastage. 55 The integrated design allowed precise dosing and decreased handling errors while contributing to a streamlined administration workflow.
Public Health Impact
Six studies included in this literature review had outcomes assessing public health impact.37,40,41,52 -54 These studies emphasize the public health and social benefits of improving disposal practices and implementing sustainable practices in medical settings.
Disposal
A qualitative study of 20 general practitioners in Pakistan revealed that 60% of practitioners discarded syringes in open places, posing public health risks while only 25% used municipal waste bins, thus increasing the potential for needle-stick injuries and the spread of infectious diseases. 53 Another observational study found that the reuse of glutaraldehyde-crosslinked collagen syringes for specific individual patients could be safe and reduce waste, highlighting that this practice could mitigate the risks associated with improper syringe disposal. 54 Further evidence came from a case-study of 20 hospitals in Nigeria referenced before, which showed that most facilities in Lagos lacked appropriate treatment systems for waste management due to inadequate funding and low prioritization. 40
Sustainable Practices
The study at WRNMMC also found that the 57.03% of antineoplastic waste can be salvageable while ensuring the drugs remain sterile and safe to use. 52 As noted in the environmental impact, a cross-sectional study of 105 primary care patients with diabetes in Brazil showed that patients who were instructed on proper disposal were more likely to dispose of waste, with proper disposal practices reducing the risk of public health issues. 41 Finally, the previously mentioned survey of 2189 anesthesiologists in the U.S. supported practices benefiting public health such as reprocessing equipment (48.4%), using prefilled syringes (56.6%), and donating equipment and supplies to medical missions (65.1%). 37
Discussion
This review of 24 studies highlights the diverse impacts of sustainability in injectable drug delivery devices, particularly in waste management practices, carbon footprint, recyclability, lifecycle management, and others.32 -55 These studies provide insights into the environmental, economic, and public health considerations of reducing injectable drug delivery waste and the need for sustainable injectable drug delivery practices to minimize environmental harm while aligning with evolving expectations and goals for efficiency and responsible resource use.
The findings suggest that sustainability practices are connected across environmental, economic, and public health impacts. The sustainability benefits seen on environmental impact, such as waste reduction, can lead to a reduction in operational costs of waste disposal, thus providing economic efficiencies and cost reductions. Economic gains seen through sustainable practices allow the potential to improve patient health outcomes. This can be done as safer waste disposal can reduce the risks of infection and contamination. Healthcare systems and institutions that adopt more sustainable approaches may see benefits across all 3 domains.
While these findings highlight the potential benefits of existing sustainable injectable drug delivery, evolving treatment trends bring new challenges. For instance, the rise of GLP-1 treatments for self-injection at home is increasing the use of disposable systems that cannot be easily dismantled or recycled, creating a growing waste burden. This shows the importance of considering design choices early, especially incorporating reusable components and eco-design principles to help reduce their long-term environmental impact.
Environmental Considerations in Sustainable Drug Delivery
Injectable drug delivery systems can impact the environment through the duration of their lifecycle. For example, Araujo et al showed the use of reusable pens resulted in energy savings compared to disposable alternatives. 33 Similarly, Winkelkötter and Reitz highlighted the environmental advantages of modular insulin pumps, which indicated greater recycling potential and lower global warming impacts than non-modular systems. 46 Overall, these findings suggest that sustainable injectable drug delivery systems may offer both environmental and cost-related benefits, which could support their broader use in healthcare settings aiming to reduce their environmental footprint.
Newer healthcare models such as home-based care can also help further contribute to sustainability and greener practices. Providing medical care and drug administration in patients’ homes or nearby facilitates can potentially reduce the environmental impact. This is due to minimizing transportation for patients, thus decreasing emissions and resource use. 56 As a result, home-based care models can lower the carbon footprint associated with traditional healthcare delivery methods.
Economic Implications for Sustainable Practices
Implementing sustainable practices may help reduce costs for healthcare institutions, primarily by lowering waste and resource use. However, financial incentives may be necessary to support the adoption by encouraging engagement from key stakeholders. The cost savings from waste reduction and efficiency improvements may appeal to decision makers. Hospitals and health systems that have implemented sustainable practices saw reductions in costs from more efficient drug preparations,36,49,50 cartridge needle units, 42 and IV workflow management systems. 48
However, surveys of anesthesiologists in the U.S. and in Canada included in this review found that, while a majority supported recycling and sustainable practices, only a portion of their hospitals or facilities had implemented these practices.37,39 This trend has been reported throughout the literature, with reasons for lack of uptake in sustainable practices including not having buy-in from leadership, not understanding the issues at hand, and lack of training.37,39,57 Regulatory inconsistency, particularly around reusability standards and sterilization protocols, was also noted as a barrier.
These findings align with broader research indicating that reusability and recyclability are needed in reducing healthcare sector’s carbon footprint. 15 While most of the studies included in this review centered around syringes or similar products, these are not the only delivery system products that can contribute to environmental waste, nor the easiest to make sustainable. The use of recycled materials is limited and/or prohibited for certain delivery systems based on regulatory and safety requirements and guidelines. Therefore, efforts should be tailored to the specific challenges of differing drug delivery rather than applying a one-size-fits-all approach to improving sustainable practices in this setting.
Bridging Sustainability and Public Health
The adoption of reusable devices and the subsequent reduction in waste not only contributes to improved economic and environmental metrics but also to public health metrics as there are lower risks associated with healthcare waste exposure. This is particularly critical in developing countries, where inadequate waste disposal can lead to the spread of infectious and communicable diseases.40,53 As several of the studies included in this review indicated, proper waste management is needed for mitigating these risks.
While the studies included in this review help document the effectiveness of sustainability practices on key metrics, how well these efforts are put into practice may depend on existing infrastructure and the education provided within institutions. A study of hospitals in Nigeria found that inadequate funding and poor prioritization of waste treatment systems hindered their sustainability initiatives. 40 Improving infrastructure and providing education in advance is important to ensure these efforts result in the intended benefits. Education also plays a role in sustainability adoption. A study in Brazil found that patients who received proper disposal training were more likely to follow correct waste management protocols, reducing contamination risks. 52 These findings highlight the importance of integrating sustainability education into routine healthcare practices to support both environmental and public health goals.
Prefilled Syringes Impact
Building on these findings, another theme identified in several of the included studies was the impact of prefilled syringes in reducing drug waste and supporting patient safety.32,34,37 Prefilled syringes are a potential efficient alternative to manually filled single-use syringes, as they remove steps from the drug preparation process that can contribute to waste. They may also help provide improved dosing accuracy, which can lead to a reduction in both drug waste as well as medication errors.32,34 An important note about prefilled syringes is that since they are still single-use devices, their impact on waste reduction is primarily through improved efficiency. While prefilled syringes demonstrate measurable sustainability benefits, these outcomes may not be generalizable across all injectable drug delivery devices due to differences in format, use case, and regulatory pathways.
Implications for Manufacturers
The findings of this literature review also have implications for pharmaceutical and biotech manufacturers, who may need to consider developing products that reduce the environmental impact throughout the product lifecycle and attempt to measure this impact more consistently. For example, Winkelkötter and Reitz and Araujo et al observed waste reduction through changes in packaging and design.33,46 Another opportunity lies in reducing packaging waste by supporting material choices that allow for reuse and reprocessing.58 -60 When feasible, promoting recyclability – even for non-medical applications – could contribute to broader sustainability efforts. For instance, an analysis found that PVC blister-packaging is more environmentally friendly than aluminum blister-packaging as aluminum packaging consumes 63% more energy and has 70% higher global warming potential. 60
Innovation
Recent innovations in sustainable drug delivery, such as green packaging technology that reduces waste, and advanced injectable drug delivery systems, such as devices that have sequential stoppers or chambers with multiple drugs in the same device, further support the shift toward environmentally conscious healthcare solutions.61,62 These developments aim to reduce material use and limit waste, while maintaining efficiency in drug administration. When thoughtfully integrated, such innovations may offer both environmental and economic benefits.
Green innovation in injectable drug delivery devices extends beyond end-stage waste management and includes upstream design choices that reduce environmental impact across the product lifecycle. Examples include the use of biodegradable or compostable packaging materials, refillable or modular device systems that enable reuse, and design approaches that minimize material inputs or energy use during manufacturing. 63 Manufacturers can play a critical role by integrating sustainability criteria – such as carbon footprint, recyclability, or ease of disassembly – into early-stage R&D and device engineering. Doing so not only enhances environmental performance but may also position products favorably within future regulatory or procurement frameworks that prioritize sustainability.
Policy Implications and Future Directions
The findings from this literature review have the potential to help enhance and facilitate policy discussions centered around sustainability of injectable drug delivery. Institutions in the healthcare sector could benefit from the prioritization of sustainability practices at their facilities. Ways to prioritize sustainable practices include using recyclable and reusable materials, having efficient waste management processes, and training employees and workers regarding the benefits of sustainable injectable drug delivery practices. Government and institutional policies can further enhance the facilitation of these changes. 64
Despite the benefits of injectable drug delivery devices identified in this systematic literature review, the uptake remains low for several reasons. Firstly, many healthcare institutions face resource constraints and competing priorities, which can make sustainability efforts appear secondary, especially when upfront costs or workflow disruptions are perceived. 65 Secondly, the lack of clear and harmonized regulatory guidance on sustainability metrics limits the ability of institutions to evaluate or justify switching to sustainable alternatives. 66 This regulatory ambiguity may also deter manufacturers from investing in greener technologies. Additionally, leadership engagement and staff education remain inconsistent across settings; while there may be individual champions of sustainability, institutional inertia and limited training on device reuse, recycling, or waste management hinder broader implementation. 66 Lastly, the absence of financial incentives or reimbursement structures that reward sustainability can create economic disincentives, particularly in settings where procurement decisions are driven by short-term costs rather than long-term value. 67
Effective implementation of sustainable injectable drug delivery devices requires more than just proven outcomes, it depends on institutional leadership and workforce readiness. 67 Leadership engagement, especially the C-suite and financial leaders, is critical for setting sustainability as an operational priority and driving culture change. However, sustainability is often not embedded into leadership metrics or clinical performance goals. Parallel to this, many frontline providers lack training in sustainable device use or the downstream consequences of medical waste. 68 Embedding sustainability education into clinical training programs, continuing education, and procurement protocols can help build the internal capacity needed to implement these practices consistently.
Scaling sustainable injectable drug delivery devices will likely require better alignment of financial and regulatory levers. Policymakers and payers could consider incorporating environmental performance into health technology assessment frameworks or value-based payment models, rewarding practices that reduce waste or emissions. Reimbursement incentives – such as shared savings from waste reduction or preferential coverage of reusable devices could further accelerate uptake. Clearer sustainability standards – particularly for post-market guidance and health technology assessments – could help reduce friction for manufacturers and health systems by clarifying what matters most. Aligning these mechanisms could help shift sustainability from a “nice-to-have” to a system-supported standard. A summary is presented in Table 4.
Framework for Scaling Sustainable Injectable Drug Delivery Devices.

Recommendations to Industry, Policymakers, and Healthcare Providers
To accelerate the adoption of sustainable injectable drug delivery systems, such as prefilled syringes or multi-chamber syringes, coordinated action is needed across key stakeholder groups. For industry, incorporating sustainability metrics – such as carbon footprint, material recyclability, and end-of-life impact – early in the design process can support more informed decision-making by purchasers and regulators. Policymakers can play a key role by embedding environmental criteria into health technology assessments and procurement frameworks, creating clear incentives for sustainability-focused innovation. Within healthcare institutions, providers can support implementation by designating sustainability champions and ensuring clinical staff are trained in environmentally responsible practices, including appropriate device selection, reuse protocols, and recycling practices.
To support consistent evaluation and adoption, manufacturers and health technology assessment bodies could align around a core set of sustainability metrics. These may include carbon footprint across the product lifecycle, percentage of recyclable or biodegradable materials used, energy intensity of production, and volume of medical waste generated per use. Including such metrics in regulatory submissions and health technology assessment evaluations would enable more transparent comparisons across products and create clearer incentives for sustainable innovation.
Limitations
There were several limitations with this study. First, in terms of methodological limitations we used a targeted and systematic approach to identify articles. This may have inadvertently excluded relevant articles not indexed in the literature databases we searched, as well as industry white papers and graduate student dissertations or theses. However, by utilizing 2 databases (PubMed/MEDLINE and Embase), and reviewing citations, we likely minimized the chances of missing relevant articles.
Second, another methodological limitation was the inclusion of conference abstracts. Although this aimed to provide a comprehensive review, these abstracts may contain only preliminary data or lack methodology. Additionally, while most conference abstracts undergo peer-review, it is often less rigorous than that for full manuscript journal submissions. Most studies identified were rated with a fair quality of the evidence, which may lead to the potential for increased bias due to methodological weaknesses.
In terms of review content limitations, the scope of the study was voluntarily limited to injectable drug delivery devices, which prevented the conclusions from being applicable to all drug delivery devices. However, further real-world research and long-term studies are needed to gain a deeper understanding of the environmental impact of injectable drug delivery devices. Additional research is also recommended to better understand the benefits of drug delivery devices related to the other route of administration.
Finally, most of the studies included in this review were conducted in high-income settings, where infrastructure and procurement models may differ substantially from those in low- and middle-income countries. As such, the transferability of sustainability practices – particularly those involving reusable systems or safe disposal practices – may be limited without parallel investments in training, infrastructure, and supply chain resilience. Additionally, variation in regulatory requirements across regions (eg, EU vs U.S.) may further limit the generalizability of sustainable strategies and their adoption across health systems.
Conclusion
This review suggests that certain injectable drug delivery devices, including prefilled syringes, multi-chamber syringes and reusable systems, can advance sustainability in healthcare by helping address environmental, economic, and public health challenges. Evidence suggests that injectable drug delivery devices, such as prefilled syringes, reduce waste by 26% to 86% compared to vials and up to 26% of total drug expenditures annually, although generalizability remains limited. Achieving long-term progress may depend on clearer regulatory alignment, broader use of assessment tools, and greater adoption of innovations that improve efficiency and reduce the environmental impact across the healthcare system. This will be made possible by incorporating sustainability early in the design process, embedding environmental criteria into health technology assessments and procurement metrics, and designating sustainability champions and ensuring clinical staff are trained in environmentally responsible practices.
Supplemental Material
sj-docx-1-ehi-10.1177_11786302261416998 – Supplemental material for Sustainability in Injectable Drug Delivery Devices: A Systematic Literature Review of Environmental, Economic, and Public Health Impacts
Supplemental material, sj-docx-1-ehi-10.1177_11786302261416998 for Sustainability in Injectable Drug Delivery Devices: A Systematic Literature Review of Environmental, Economic, and Public Health Impacts by Eric P. Borrelli, Matthias Borms, Monica Telinoiu, Haymen Girgis, Cecile Frolet and Julia D. Lucaci in Environmental Health Insights
Footnotes
Author Contributions
EPB conceptualization, methodology, writing – review and editing. MB conceptualization, methodology, writing – review and editing. MT conceptualization, methodology, writing – review and editing. HG conceptualization, methodology, writing – review and editing. CF conceptualization, methodology, writing – review and editing. JDL supervision, conceptualization, data interpretation, writing – drafting, review, and editing.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by Becton, Dickinson and Company.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.P.B., M.B., M.T., H.G., C.F., and J.D.L. are employees of Becton, Dickinson and Company.
Data Availability Statement
Data analyzed during the current study is available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
