Abstract
Background:
Breast cancer is the most common cancer among women, and chemotherapy is a key treatment option. Doxorubicin is frequently used for breast cancer but poses a risk of cardiotoxicity. Recently, memantine, an N-Methyl-D-aspartate antagonist, has shown both antitumor and cardioprotective effects.
Objectives:
We conducted a study to examine the combined effects of doxorubicin and memantine on the 4T1 cell line in a breast cancer animal model and to assess memantine’s potential in reducing doxorubicin-induced cardiotoxicity in breast cancer model mouse.
Design:
The 4T1 cell line was cultured and subsequently inoculated into mice. After that animals were randomly divided into four groups: (1) control, (2) memantine, (3) doxorubicin, and (4) memantine + doxorubicin.
Methods:
After 30 days, mice were sacrificed, and their lungs, liver, and heart were collected for histological analysis. Myeloperoxidase, Malondialdehyde, and TNF-α levels in cardiac tissues were measured, and the TUNEL test was conducted for breast tumors.
Results:
Our results showed that memantine + doxorubicin reduced MDA activity and TNF-α levels in cardiac tissue in comparison to the doxorubicin group. TUNEL test revealed that the memantine + doxorubicin group demonstrated significantly more tumor cell apoptosis than the mice in other groups (P-value < 0.05), although tumor volume reduction was not significantly greater than that in the doxorubicin group.
Conclusion:
This study is the first to demonstrate that memantine can enhance the therapeutic efficacy of doxorubicin chemotherapy while also reducing doxorubicin-induced cardiac oxidative stress and inflammation in a breast cancer model.
Introduction
Breast cancer is the most prevalent cancer among women. It is also a leading cause of cancer-related deaths in females. Female breast cancer is the fifth leading cause of cancer mortality globally and exceeded lung cancer as the most diagnosed cancer in 2020, accounting for 11.7% of all cases. It is estimated to represent 15.2% of new cancer cases in 2023. 1 Previous studies have reported varying 5 year survival rates (ranging from 40% to 80%) for women with breast cancer, with patients in developed countries exhibiting better survival rates than those in other regions.2,3 Patients diagnosed with breast cancer have several treatment options, including hormonal therapy, targeted therapy, surgery, radiation therapy, and chemotherapy. Anthracyclines, a class of antibiotics, are utilized as chemotherapy drugs in breast cancer treatment.4,5 Doxorubicin (DOX), also known as Adriamycin, is a widely used chemotherapy drug for breast cancer, either alone or in combination with other medications. Its cytotoxic effects are effective against breast tumors, but it may cause considerable side effects, including alopecia, nausea, vomiting, and myelosuppression.6,7 The most critical and potentially life-threatening complication of this drug is cardiotoxicity, which has resulted in its restricted use. This dose-dependent side effect can damage cardiomyocytes, decrease ejection fraction, and ultimately lead to heart failure. 8 Doxorubicin-induced cardiotoxicity arises from several mechanisms, including increased free radical production that causes oxidative stress and lipid peroxidation. It can trigger nitric oxide production, leading to apoptosis, and increase p53 and caspase-3 expression or elevate intracellular calcium, contributing to cell death. DOX triggers inflammation and elevates TNF-α levels while disrupting mitochondria and promoting autophagy. Recent studies have shown promise in using various drugs to mitigate doxorubicin’s cardiotoxicity. 9 Memantine, an NMDA (N-methyl-D-aspartate) antagonist commonly used for Alzheimer’s patients, also affects NMDA receptors located in the heart and other organs.10,11 Activating these receptors increases intracellular calcium ions, releases oxygen free radicals, and causes tissue damage. Memantine, by antagonizing these receptors, can reduce cardiac myocyte damage, decrease free radicals, and prevent cardiac remodeling. 12 Additionally, previous research indicates that memantine can inhibit excessive glutamate production within cells, thereby slowing cancer progression. 13 Studies have identified the antitumor properties of memantine in lung and breast cancers. Recent research on the 4T1 cell line in breast cancer model rats demonstrated memantine’s antitumor effects both in vivo and in vitro.14,15 Previous studies have demonstrated memantine’s antitumor and cardioprotective properties; however, its protective effect against doxorubicin’s cardiotoxicity remains unexamined. Thereby, we conducted a study to explore the combined effects of doxorubicin and memantine on the 4T1 cell line in breast cancer model mice, as well as memantine’s role in mitigating doxorubicin-induced cardiotoxicity in an animal model.
Materials and Methods
Cell line
4T1 breast cancer cell line was obtained from the National Cell Bank of Iran (Pasteur Institute of Iran, Tehran, Iran). Cells were cultivated in RPMI-1640 medium (Gibco, Paisley, UK) in the presence of 100 units/ml penicillin, 100 μg/ml streptomycin (Gibco), 10% fetal bovine serum (Gibco), and then incubated in an atmosphere of CO2 which was humidified at 37°C.
Animals
Twenty healthy female BALB / c with approximately 6 to 8 weeks (16-23 grams) were obtained from Pasteur Institute laboratory animal, Tehran, Iran. Animals were kept in polyethylene cages, five per cage, with water and food available ad libitum at room temperature and 50 ± 10% relative humidity under a 12 h light/12 h dark cycle. To minimize potential confounders, treatments were administered in a randomized order across groups each day to avoid systematic bias from treatment timing and all groups were maintained under identical environmental conditions (temperature, humidity, light cycle) to reduce location-based variability.
Reagents
Doxorubicin (EBEDOXO®, EBEWE Inc., Austria), Memantine (Tasnim pharmaceutical Co.), Ketamin (Alfasan®, Netherland), Xylazine (Rompun®, USA), Formaldehyde (Merck, Germany), and TUNEL assay kit (Elabscience Biotechnology Inc, Houston, USA) used for TUNEL test.
4T1 tumor model
For the injection, the mammary fat pad of the fourth mammary gland on the right side was chosen, and 106 prepared 4T1 cells were injected directly into the mammary fat pad for each animal on the first day of trial. In about a week masses were observed clearly, and tumor size could be measured by caliper.
Experimental protocol
When tumors reached about 100 mm3, animals were assigned randomly into four groups consisting of 5 mice each. We decided on the sample size based on previous studies, ethical considerations and institutional guidelines. Mice in the first group received 0.1 ml IP (intraperitoneal) saline 15 times every other day as the control group. In the second group, animals received 20 mg/kg IP 15 times with one-day interval memantine as the memantine group (300 mg/kg cumulative dose). Animals in the third group were injected with 5 mg/kg IP doxorubicin on days 1, 7, 14, 21, and 28 as the doxorubicin group (25 mg/kg cumulative dose). Animals in the last group were injected with memantine and doxorubicin as memantine + doxorubicin group with the same schedule (5 mg/kg IP doxorubicin on days 1, 7, 14, 21, 28, and 20 mg/kg IP memantine 15 times every other day). Animals’ tumor size and weight were measured twice a week. Tumor size was measured by caliper and calculated by the formula of Tumor volume:
Animals were monitored daily and humane endpoint criteria included: 1. Loss of > 30% of body weight, 2. severe lethargy and unresponsiveness and 3. impaired mobility. After 30 days, none of the animals met the predefined humane endpoint criteria and all animals were euthanized under anesthesia with ketamine (80 mg/kg) and xylazine (10 mg/kg). Animals’ hearts were isolated, weighed, washed with PBS, and the left ventricle was isolated from each heart and longitudinally divided into two parts, one for molecular evaluation and the other one for histopathological examination. Tumors were isolated for the TUNEL test and weighted. Lungs and livers also were isolated for metastasis evaluation. This study was performed following the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH publication No 85-23, revised 1996) and was approved by the committee of Care and Use of Laboratory Animals of Tabriz University of Medical Sciences, Tabriz, Iran (Ethical code: IR.TBZMED.AEC.1402.013). The reporting of this study conforms to the ARRIVE 2.0 guidelines for reporting animal research 16 (Supplementary file 1).
Histopathological examination
Tissues from the liver, heart, and lungs were fixed in a 10% formalin solution, dehydrated in ascending alcohol grades, and then embedded in paraffin. Hematoxylin and eosin (H & E) were used to stain the paraffin blocks after they were cut using a microtome. Cardiac tissues also were stained with Masson’s trichrome. A pathologist who was not aware of the treatment plan looked at each section under a light microscope.
Myeloperoxidase (MPO) assay
MPO cardiac activity was measured in accordance with Mullane et al’s 17 previous description to assess neutrophil infiltration into the myocardium. Briefly, three cycles of freeze-thaw and sonication were carried out after heart tissues (left ventricle) were homogenized in phosphate buffer(pH = 6) containing 0.5% hexadecyltrimethylammonium bromide (HTAB). All samples were then centrifuged at 2000 g for 45 minutes at 4°C. Then, 2.9 ml of solution containing 0.167 mg/ml of O-dianisidine hydrochloride and 0.0005% H2O2 was mixed with 100 μl of supernatant. After five minutes, 100 μl of a 1.2 M hydrochloric acid solution was added, and the mixture was shaken once more to stop the reaction. Finally, a spectrophotometer (Cecil 9000, UK) was used to measure changes in absorption at 400 nm.
Malondialdehyde (MDA) assay
In order to assess lipid peroxidation, MDA level in cardiac tissues was measured. Following the homogenization of cardiac tissues, samples were centrifuged for 15 minutes at 2000 g. Following this, 3 ml of phosphoric acid (1%) was mixed with 250 µl of the supernatant. Afterward, the samples were incubated for 45 minutes at 90°C after 1 milliliter of thiobarbituric acid (TBA, 0.67 %) was added. After the samples had cooled, 3 milliliters of n-butanol was added, and the centrifuge was run once more for 15 minutes at 2000 g. The absorption changes were finally measured using a spectrophotometer (Cecil 9000, UK) at 532 nm. The units used to express all results are nMol/mg of protein. 18
TNF-α
The cardiac levels of TNF-α were assayed by ELISA Kit (R&D, DuoSet, McKinely Place NE Minneapolis, MN) according to the manufacturer’s recommendations. It is assessed by capturing the microplate using antibody and then detecting it with an enzyme-linked secondary antibody and measuring the resulting color change to determine its concentration.
TUNEL assay
To determine whether memantine with doxorubicin can promote more apoptosis compared to other groups in a 4T1-induced breast cancer mouse model, assessment with TUNEL test was performed. Briefly, after tumors were obtained and fixed in formalin, all samples were dehydrated in ascending alcohol grades and embedded in paraffin. Tissue samples were cut into 7-micron sections using a sharp microtome and were placed in the water bath. Then sections were placed on the 20% alcohol-soaked slides and incubated for 3 days at 60°C. After that, all samples were deparaffinized. Sections were incubated with Proteinase k Tris / Hcl, pH = 7.4, for 30 minutes at 37°C shortly after deparaffinization. Following two PBS washes of each slice, 50 µl of TUNEL reaction mixtures were added, and the slices were left in the dark at 37°C for an hour. Following a final rinse with PBS buffer, the sections were examined using the Citation Cell Imaging system (Biotek) at the range of 515–565 nm (GFP).
Statistics
Data were presented as mean ± standard deviation wherever possible. Normality was checked using the Shapiro-Wilk test. One-way ANOVA was used to make a comparison between the groups followed by LSD post-hoc test for data meeting the normality assumption. When the assumption of normality was not met, the Kruskal-Wallis test was applied. P-value less than 0.05 (P < 0.05) was considered significant. All statistical analyses were performed using IBM SPSS Statistics 27 software.
Results
Effects of memantine on weight loss
Table 1 shows that the group receiving doxorubicin experienced the most weight loss. The weight difference between the mice on days 22 and 30 was significant (P-values: 0.012 and 0.002). Mice in the control group weighed more than those receiving doxorubicin and those in the Dox + Mem group (P-value < 0.05). However, there was no statistically significant difference between the memantine group and the Dox + Mem group on days 22 and 30 (P-values: 0.087 and 0.195).
Weight loss during the study.

P-value < 0.05 compared to control group, #P-value < 0.05 compared to memantine group. Data are shown in mean ± standard deviation.
Effects of memantine on tumor volume
Table 2 presents tumor volumes on days 10, 16, and 22, and at the end of the study. The control group demonstrated the highest tumor volume increase, averaging 2428.74 ± 382.71 mm³, up from 424.73 ± 736.12 mm³ by the end of the study. ANOVA test indicated no statistically significant differences in tumor volume among groups on days 10 (P-value: 0.671) and 16 (P-value: 0.110). However, on day 22, the control group’s tumor volume was significantly greater than that of the other three groups (P-value < 0.001), with no significant differences among the other groups. On the last day, the control group again showed significantly higher tumor volumes compared to the other groups (P-value < 0.001). Additionally, tumor volumes were significantly lower in the doxorubicin group and the Dox + Mem group compared to the memantine group (P-value: 0.014 and P-value: 0.001).
Mean tumor volume during the study.
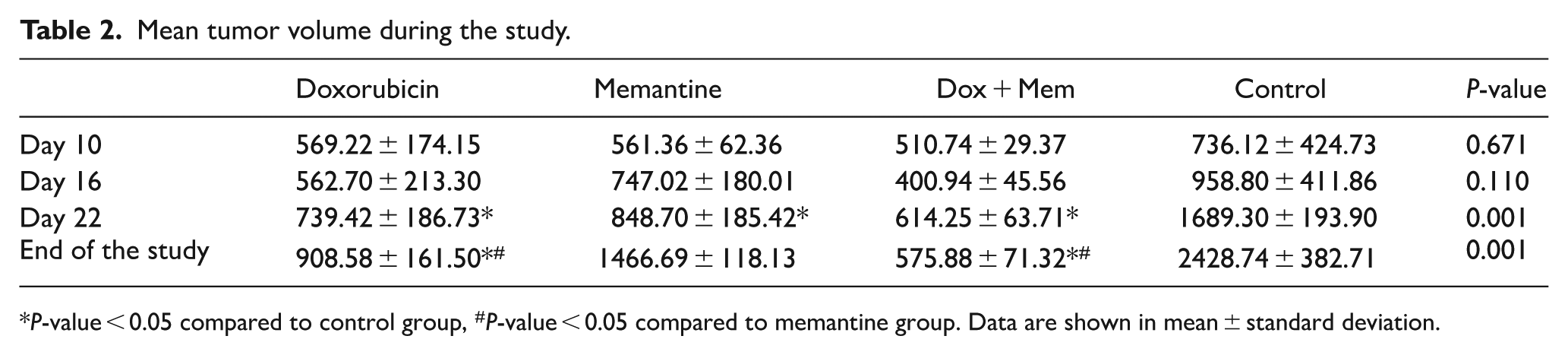
P-value < 0.05 compared to control group, #P-value < 0.05 compared to memantine group. Data are shown in mean ± standard deviation.
Effects of memantine on tumor apoptosis
DNA fragment detection in the TUNNEL test images confirmed cell apoptosis (Figure 1A). Image-J software quantified fluorescent light, showing that the Dox + Mem group had the highest average radiation (M: 11.22 ± 2.14) as seen in Figure 1B. This indicates greater tumor apoptosis in this group compared to the control and memantine groups (P-value < 0.001). Additionally, the Dox + Mem group demonstrated significantly more cell apoptosis than the mice treated with doxorubicin alone (P-value = 0.001).

(A) Diagram of TUNNEL test for evaluation of apoptosis. (B) Fluorescent index in different groups at the end of the study.
Effects of memantine on tumor weight
Figure 2A illustrates the tumor weight after the mice were sacrificed, and the tumors were separated from surrounding tissues. The control group had an average tumor weight of 2283.66 ± 807.00, significantly higher than the other groups (P-value < 0.05). In the other three groups, the treatment regimen resulted in reduced tumor weights, with the lowest weight found in the Dox + Mem group (1020.33 ± 83.54). However, the differences among the three groups were not statistically significant (P-values: 0.582, 0.532).

(A) Tumor weight after the experiment. (B) Cardiac MPO level at the end of the experiment. (C) Cardiac MDA level at the end of the experiment. (D) Cardiac TNF-α level at the end of the experiment.
Effects of memantine on MPO activity
Neutrophil infiltration in cardiac tissue was assessed by measuring cardiac myeloperoxidase (MPO) levels. The results indicated that average enzyme activity rose from 1.03 ± 0.15 in the control group to 4.33 ± 1.24 in the doxorubicin group (P-value < 0.001). Treatment with memantine reduced MPO activity in cardiac tissue to 3.13 ± 0.58 (Figure 2B), but this decrease was not statistically significant (P-value: 0.067).
Effects of memantine on MDA activity
Malondialdehyde (MDA) levels, a marker of lipid peroxidation in myocardial tissue (Figure 2C), were significantly higher in the doxorubicin-treated group compared to controls (P-value < 0.001). However, adding memantine to the treatment regimen significantly lowered MDA levels to a mean of 8.16 ± 1.27 (P-value = 0.002).
Effects of memantine on myocardial TNF-α level
Doxorubicin significantly increased TNF-α levels in myocardial tissue (P-value < 0.001, Figure 2D). In the group treated with both memantine and doxorubicin, TNF-α levels in heart tissue were decreased to 58.76 ± 11.20, which was statistically significant (P-value = 0.04).
Effects of memantine on cardiac histopathology
Histological evaluation via light microscopy revealed that all groups, except the doxorubicin group, demonstrated normal alignment of myofibrils and preserved morphology of nuclei and cells. In the doxorubicin group, however, atrophy and thinning of myocardial fibers were noted. Masson’s trichrome staining assessed fibrosis development across all groups but showed no changes in any of them (Figure 3). Besides, at the end of the study heart weights did not have any significant difference across treatment groups (Supplementary file 2).
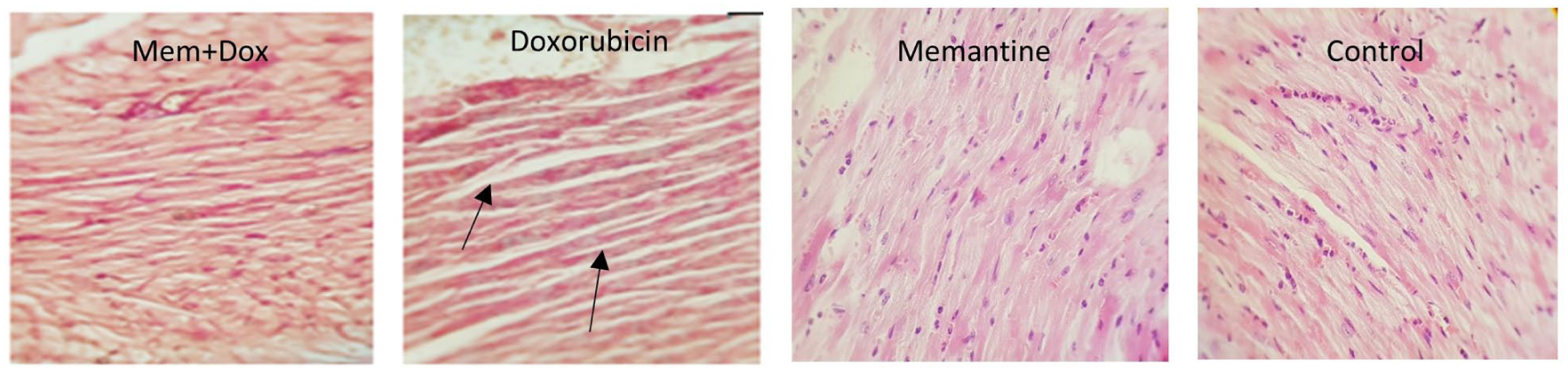
H&E staining was performed on heart sections from each of the groups and evaluated by light microscopy (magnification of ×400). Atrophy in myocytes is shown with the black arrows in the doxorubicin group. Myocardial atrophy was not seen in other groups.
Effects of memantine on tumor metastasis
Lung slide examination in the control group revealed multiple metastatic nodules. The doxorubicin and memantine groups each had 2 and 4 nodules, respectively, while only a single nodule was found in the Dox + Mem group (Figure 4). In the liver slides, no metastatic nodules were detected in the Dox + Mem, whereas other groups displayed nodules of varying sizes (Figure 5).

Histological appearance of lungs sections stained with Hematoxylin and Eosin evaluated by light microscopy (magnification of ×100) with metastasis (arrows); (A) control: showing large metastatic nodule, (B) doxorubicin: showing large metastatic nodule, (C) memantine: showing several metastasis, (D) Mem + Dox: showing.

Histological appearance of liver sections stained with Hematoxylin and Eosin evaluated by light microscopy (magnification of ×100) with metastasis (arrows); (A) control, (B) doxorubicin, (C) memantine, (D) Mem + Dox. Metastatic nodules were not seen in the Mem + Dox group.
Discussion
Numerous hypotheses have been proposed regarding cardiomyopathy caused by doxorubicin, with most studies agreeing on the increased levels of tissue free radicals, lipid peroxidation, and reduced antioxidants in cardiac tissue.19,20 Research has demonstrated that doxorubicin induces programmed cell death in endothelial cells and cardiac myocytes. 21 While doxorubicin’s cytotoxic effects in tumor cells are beneficial, its apoptotic effects in cardiac tissue may result from different mechanisms including the generation of reactive oxygen species (ROS), lipid peroxidation, mitochondrial dysfunction, and ferroptosis.22,23 Numerous compounds have been utilized to prevent and treat doxorubicin-induced cardiomyopathy, with notable studies demonstrating significant results using statins, metformin, sildenafil, and erythropoietin.24 -26
NMDA receptors are crucial in the cardiovascular system; their activation in cardiac myocytes elevates mitochondrial calcium levels and promotes apoptosis, autophagy, and oxidative stress. 27 Existing antagonists that block these receptors can reduce myocardial pathologies. 12 A study found that stimulating NMDA receptors in rat cardiac tissue led to oxidative stress, reduced blood flow, and impaired cardiac hemodynamics. 28 Another study on chronic receptor stimulation indicated increased tissue fibrosis and a higher risk of arrhythmia. 29 Research on the protective effects of NMDA antagonists showed that dizocilpine, an NMDA receptor antagonist, reduced inflammatory cytokines and overall cardiac inflammation. 30 A study found that dizocilpine, an NMDA receptor inhibitor, decreased lipid peroxidation caused by homocysteine. 31 Abbaszadeh et al 11 reported that memantine treatment in isoproterenol-induced heart failure rats significantly reduced MPO enzyme activity in cardiac myocytes at a dose of 20 mg/kg (P-value < 0.05). In contrast, our study did not observe this effect, likely due to variations in study design and the prophylactic use of memantine in their study (P-value: 0.067). Also, their study demonstrated that memantine significantly reduced MDA levels in serum and cardiac tissue of heart failure model, aligning with the current findings. 11 This suggests that memantine’s anti-lipid peroxidation effects and reduction of inflammatory responses may contribute to its cardioprotective properties. Similarly, Jannesar et al 10 found that memantine decreased MDA, MPO, and TNF-α, further supporting our results.
Memantine, a well-known anti-Alzheimer’s drug, has been researched for additional applications, particularly in cancer treatment. Cancer cells often exhibit altered metabolic pathways to meet their increased demands for growth, 32 primarily relying on glucose and glutamine. 33 Numerous studies have investigated memantine’s antitumor effects, with one study demonstrating its anti-cancer properties in prostate cancer via the Bax protein pathway. 34 A study demonstrated that memantine, whether used alone or with the chemotherapy drug topotecan, reduced the survival of small cell lung cancer cells. 35 Given that the NMDA receptor signaling pathway promotes cancer cell growth and differentiation, North et al 36 concluded that using antagonists like memantine can diminish the survival of breast cancer cell lines. It is reported that memantine treatment led to a dose-dependent decrease in the survival of MC-7 breast cancer cells. Additionally, in vitro, cancer cell migration was reduced, and the combination of memantine with Paclitaxel had synergistic effects on MC-7 cell apoptosis. 37 In a recent in vitro study, memantine was shown to alter the expression of the Bcl-2, Bax, Casp3, and Casp-9 proteins in the 4T1 cell line. Additionally, it inhibits p-Erk production, suggesting a potential pathway for its anti-cancer effects. 38 It is also demonstrated that memantine exhibits anti-cancer properties in mice with 4T1-induced breast cancer, showing a significant reduction in tumor volume when administered at a dose of 10 mg/kg, which aligns with the current findings. 15 While in our study the dox + mem group exhibited significantly higher tumor apoptosis than the DOX-alone group, no significant additional reduction in tumor volume was observed. This could be attributed to several factors. First, the small sample size (n = 5 per group) may have limited the statistical power to detect subtle differences in tumor volume, as noted in the study limitations. Second, increased apoptosis in the dox + mem group may primarily affect the viable tumor region, reducing the number of actively proliferating cells without immediately altering total tumor volume, which includes necrotic or non-viable tissue. This is supported by prior studies suggesting that apoptosis-inducing treatments may require longer observation periods to translate into measurable volume reductions.36,38 Additionally, memantine’s antitumor effects may involve pathways (Bax/Casp3 or p-Erk inhibition, as reported by Albayrak et al 38 ) that enhance apoptosis without directly impacting tumor size in the short term. Future studies with larger sample sizes, longer durations, or assessments of viable tumor mass could clarify whether memantine’s synergistic effects extend to tumor volume reduction.
While past studies have linked memantine’s cytoprotective effects to NMDA receptors, D. Jantas and Lason 39 examined its use with doxorubicin to mitigate the drug’s adverse neurological effects. Their findings suggest that these protective effects operate independently of the NMDA receptor pathway, resulting from a reduction in caspase-3 activity, intracellular LDH, and signaling pathways like PI3 K/Akt. 39 Dexrazoxane (DXZ), the only FDA-approved drug for mitigating doxorubicin (DOX)-induced cardiotoxicity through iron chelation, has notable limitations, including exacerbation of chemotherapy-induced bone marrow suppression, potential reduction of DOX’s antitumor efficacy, and an increased risk of secondary malignancies with prolonged use. 40 In contrast, the current study demonstrates that memantine provides both cardioprotection and enhanced tumor apoptosis in a 4T1 breast cancer model, suggesting a promising alternative with a potentially favorable safety profile.
Limitations
This study had several limitations, the most significant being the small sample size of mice. While ethical considerations encourage using fewer animals, larger samples could enhance the validity of the findings. Additionally, the study did not explore various pathways or the expression of different genes and proteins, which are crucial for understanding changes in the cell cycle. The data on protein expression could inform future research. Conducting in vitro studies could further elucidate cell signaling pathways. This study utilized the TUNEL assay to detect apoptosis; future research could employ IHC analysis of apoptotic and anti-apoptotic proteins to better confirm apoptosis, metastasis, and cardioprotective mechanisms, thereby enhancing understanding of the underlying molecular pathways. Further investigations might also consider varying doses of memantine or different treatment regimens, such as prophylactic use. Finally, due to memantine’s shorter half-life in mice compared to humans, further research is needed to optimize dosing and design human clinical trials.
Conclusion
This study demonstrated for the first time the potential antitumor and cardioprotective effects of memantine in combination with doxorubicin in mice with breast cancer modeled using the 4T1 cell line. The combination treatment increased apoptosis, reduced breast tumor metastasis, reduced pro-inflammatory cytokines and lipid peroxidation, and protected cardiac tissue from doxorubicin-induced oxidative stress and inflammation. Overall, the findings highlight memantine’s potential for synergistic anti-cancer effects and cardiomyocyte protection.
Supplemental Material
sj-docx-1-bcb-10.1177_11782234251393421 – Supplemental material for Memantine, an NMDA Receptor Antagonist, Attenuates Doxorubicin-Induced Cardiac Oxidative Stress and Inflammation in Mouse 4T1 Breast Cancer Model
Supplemental material, sj-docx-1-bcb-10.1177_11782234251393421 for Memantine, an NMDA Receptor Antagonist, Attenuates Doxorubicin-Induced Cardiac Oxidative Stress and Inflammation in Mouse 4T1 Breast Cancer Model by Amirhosein Ghafouri-Asbagh, Koorosh Tabatabaei, Haleh Vaez, Ali Jafarizadeh, Hossein Saeedi, Behrooz Shokouhi and Behzad Baradaran in Breast Cancer: Basic and Clinical Research
Supplemental Material
sj-docx-2-bcb-10.1177_11782234251393421 – Supplemental material for Memantine, an NMDA Receptor Antagonist, Attenuates Doxorubicin-Induced Cardiac Oxidative Stress and Inflammation in Mouse 4T1 Breast Cancer Model
Supplemental material, sj-docx-2-bcb-10.1177_11782234251393421 for Memantine, an NMDA Receptor Antagonist, Attenuates Doxorubicin-Induced Cardiac Oxidative Stress and Inflammation in Mouse 4T1 Breast Cancer Model by Amirhosein Ghafouri-Asbagh, Koorosh Tabatabaei, Haleh Vaez, Ali Jafarizadeh, Hossein Saeedi, Behrooz Shokouhi and Behzad Baradaran in Breast Cancer: Basic and Clinical Research
Footnotes
Acknowledgements
We thank the staff of immunology research center of Tabriz University of Medical Sciences.
Ethical Considerations
Present study was performed following the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH publication No 85-23, revised 1996) and was approved by the committee of Care and Use of Laboratory Animals of Tabriz University of Medical Sciences, Tabriz, Iran. This study was approved by the Ethics Committee of Tabriz University of Medical Sciences (IR.TBZMED.AEC.1402.013).
Author Contributions
Amirhosein Ghafouri-Asbagh: Investigation, Methodology, Project administration, Writing—original draft
Koorosh Tabatabaei: Investigation, Methodology, Project administration, Writing—original draft
Haleh Vaez: Conceptualization, Supervision, Writing—review & editing
Ali Jafarizadeh: Project administration, Writing—review & editing
Hossein Saeedi: Methodology, Project administration, Writing—review & editing
Behrooz Shokouhi: Conceptualization, Project administration, Writing—review & editing
Behzad Baradaran: Conceptualization, Methodology, Supervision, Writing—review & editing
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research protocol was approved & funded by the Immunology Research Center, Tabriz University of Medical Sciences.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of Data and Materials
The datasets used and/or analyzed during this study are available from the corresponding author upon reasonable request, subject to institutional and collaborative agreement constraints.
Declaration of Artificial Intelligence (AI) Use
Grammarly was used for text refinement; no scientific data were generated or modified by AI.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

