Abstract
Background:
Breast cancer with low expression of human epidermal growth factor receptor 2 (HER2) has emerged as a new category benefiting from anti-HER2 therapies. This study evaluated the prevalence of HER2-low expression and concordance of HER2 scoring among pathologists at King Hussein Cancer Center (KHCC) in Jordan.
Objectives:
To determine the prevalence of HER2-low breast cancer tumors and evaluate the interpathologist concordance in HER2 scoring using the 2018 American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) guidelines.
Design:
This is a retrospective observational study.
Methods:
In total, 116 breast cancer samples were randomly selected from routine practice at KHCC. Each tumor was scored for HER2 expression by 3 pathologists independently using the 2018 ASCO/CAP guidelines. Concordance among pathologists was evaluated using Fleiss’ kappa and Cohen’s weighted kappa statistics.
Results:
The original HER2 scoring revealed 7 tumors (6%) as HER2-negative (0 immunohistochemistry [IHC] score), 84 (72.4%) as HER2-low (including tumors with 1+ IHC score or 2+ IHC score with negative fluorescence in situ hybridization [FISH]), 21 (18.1%) as HER2-positive (including tumors with 3+ IHC score or 2+ IHC score with positive FISH), and 4 (3.4%) as equivocal (2+ IHC score with no FISH). Consensus scoring showed 1+ as the most frequent HER2 score. Complete agreement among all 3 pathologists occurred in 78 tumors (67.2%), while high agreement (agreement among 2 pathologists) occurred in 38 tumors (32.8%). Cohen’s weighted kappa ranged from 0.691 to 0.849, indicating substantial agreement. The Fleiss kappa of overall agreement on HER2 scoring was 0.645.
Conclusion:
This study highlights a significant prevalence of HER2-low tumors among the randomly selected sample of Jordanian breast cancer patients and demonstrates substantial interpathologist agreement in HER2 scoring. These findings underscore the importance of standardized HER2 scoring and the potential impact of emerging therapies on a large patient population in Jordan.
Introduction
Breast cancer is the most common cancer among women and the second most common cancer globally, accounting for 11.6% of all new cancer cases in 2022. 1 The expression of human epidermal growth factor receptor 2 (HER2) is crucial for categorizing breast cancer subtypes, predicting prognosis, and guiding treatment decisions. 2 The HER2 status is primarily determined through immunohistochemistry (IHC) and fluorescence in situ hybridization (FISH) tests, which classify tumors into either HER2-positive or HER2-negative categories. 3 Classically, breast cancer is considered HER2-positive for tumors with a 3+ score tested by IHC assay or a 2+ IHC score with positive gene amplification. 3 In Jordan, HER2 expression in breast cancer has been previously documented in 2 studies, with prevalence rates ranging from 18% to 24%.4,5
Until recently, anti-HER2 agents were only recommended for use in HER2-positive breast cancer patients, as they were the only cohort who demonstrated positive outcomes when treated with these agents.3,6,7 However, a specific subgroup of HER2-negative patients (1+ IHC score or 2+ IHC score without gene amplification) was shown to benefit from new generations of anti-HER2 conjugate therapy (trastuzumab deruxtecan therapy). 8 This subtype is now recognized as HER2 low-expression group.8,9 This has led to changes in standard treatment recommendations to include HER2 low-expression cancer patients, allowing them to be treated with the new generation of anti-HER2 agents.10,11
Currently, HER2-low expression is not clinically recognized by pathologists as a distinct subtype from HER2-negative breast cancer, raising concerns about the reliability and accuracy of HER2-low scoring among pathologists. 12 In this study, we aimed to estimate the prevalence of HER-low-expression tumors and the concordance of reporting HER2-low expression among 3 pathologists to guide our clinical practice.
Methods
One hundred and sixteen breast cancer core biopsies from routine clinical practice were randomly selected from the archives of the King Hussein Cancer Center (KHCC; Amman, Jordan). Tumor-type data were collected. All breast cancer selected samples would have the predictive markers such as estrogen receptors (ERs), progesterone receptors (PRs), and HER2 routinely performed. The HER2 staining was done on the Ventana-Roche Benchmark Ultra automated platform using the rabbit monoclonal antibody 4B5 (Ventana-Roche, Tucson, Arizona) according to the manufacturer’s instructions. Appropriate positive and negative controls were used and were acceptable.
Originally, slides were evaluated by a pathologist in KHCC, which was akin to routine care, without considering the aim of this study. Then, the slides were reviewed by 2 other pathologists (one from KHCC and the third from an external academic institution [Hashemite University, Zarqa, Jordan]). All pathologists scored HER2 IHC as 0, 1+, 2+, or 3+ according to the American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) 2018 guidelines. 13 For the HER2 2+ IHC score (equivocal) tumors, the corresponding FISH amplification status was also recorded.
The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement for cross-sectional studies. 14 A completed STROBE checklist is provided as a supplementary file (Supplementary File 1).
The final HER2 scores were categorized as follows: a score of 0 was classified as negative (HER2-negative group), a score of 1+ was classified as HER2-low group, and a score of 2+ was considered equivocal. For equivocal tumors, if FISH amplification was present, they were classified as HER2-positive; if FISH was non-amplified, they were included in the HER2-low group. A score of 3+ was classified as positive.
The level of agreement of all 3 pathologists was considered as “absolute agreement” if all 3 pathologists scored the same HER2 score. If only 2 pathologists scored the same HER2 scores, the agreement was regarded as “high agreement,” and if none of the pathologists scored the same scoring agreement level, it was considered as “no agreement.” To further assess concordance, HER2 scores were analyzed using both individual categories (0, 1+, 2+, 3+) and combined categories (0 vs 1+/2+ vs 3+ and 0 vs 1+/2+/3+). In the individual category analysis, any discrepancy between scores (eg, 1+ vs 2+) was considered a disagreement. In contrast, the combined category analysis grouped adjacent scores (eg, 1+ and 2+), and such differences were not considered disagreements.
The interobserver agreement for HER2 scoring (as 0, 1+, 2+, and 3+) and pathologists’ overall agreement on HER2 scoring were calculated using the Fleiss multiple-rater kappa (κ) test. To evaluate interobserver agreement, Cohen’s weighted kappa was calculated for each pair of scoring pathologists. Statistical analysis was performed using the Statistical Package for Social Sciences (SPSS) version 25 (IBM, Armonk, New York).
Results
The original human epidermal growth factor receptor 2 scoring
At the time of reporting, the pathologist originally scored HER2 of 116 tumors as follows: 7 patients (6%) were classified as HER2-negative (score 0), 64 patients (55.2%) as HER2-low (score 1+), 32 patients (27.6%) as equivocal (score 2+), and 13 patients (11.2%) were classified as HER2-positve (score 3+) (Figure 1). Among the equivocal tumors, 8 (6.9%) were FISH positive and categorized as HER2-positive, 20 (17.2%) were FISH negative and categorized as HER2-low, and 4 (3.4%) had no FISH results (Table 1).

Representative images of HER2 immunohistochemistry. (A) 0 HER2 IHC score; (B) 1+ HER2 IHC score; (C) 2+ HER2 IHC score; (D) 3+ HER2 IHC score.
Original HER2 scoring.

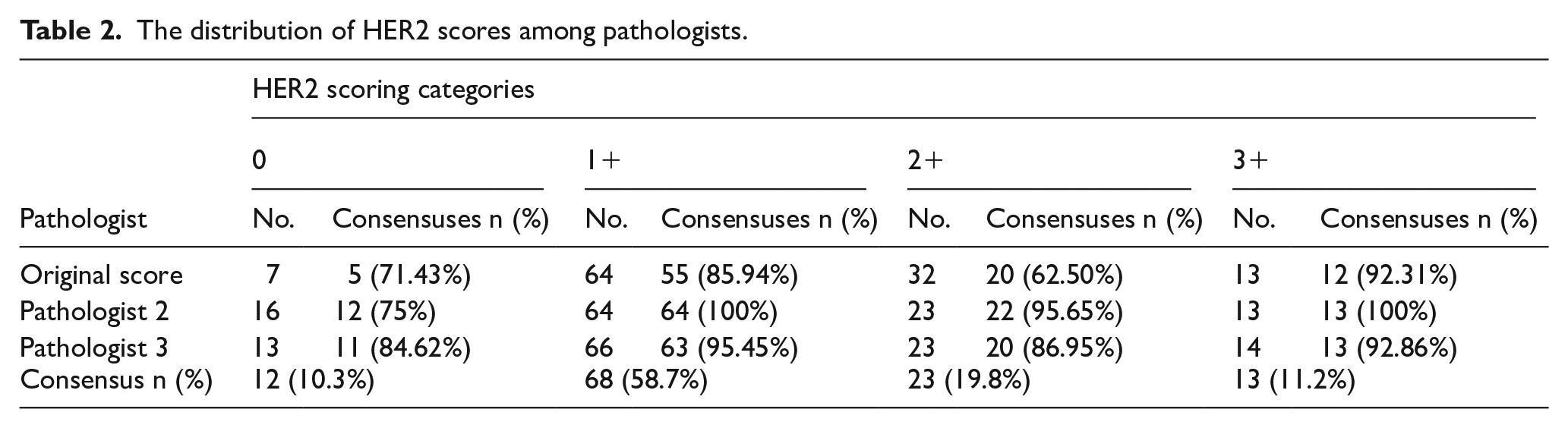
Consensus scoring
The original and the 2 study pathologists scored 116 tumors, with a total of 348 scores recorded. The most frequently assigned HER2 score was 1+, with a total of 194 scores (55.7%) across all pathologists (Table 2). Based on the majority scoring (agreement between at least 2 pathologists), consensus was reached for 12 tumors (10.3%) with an HER2 score of 0, 68 tumors (58.7%) with a score of 1+, 23 tumors (19.8%) with a score of 2+, and 13 tumors (11.2%) with a score of 3+.
The distribution of HER2 scores among pathologists.
Agreement
Absolute agreement for HER2 scoring was achieved in 78 (67.2%) tumors, while high agreement was achieved in 38 (32.8%) tumors. None of the tumors were deemed challenging for HER2 scoring (Table 3). Original pathologist and pathologist 2 had a kappa of 0.691 (substantial agreement), and original pathologist and pathologist 3 had a kappa of 0.652 (substantial agreement). Finally, pathologist 2 and pathologist 3 had the highest agreement with a kappa of 0.849 (almost perfect) (Table 4).
Level of agreement among pathologists.

Cohen’s weighted kappa for pairwise agreement between pathologists.

For individual HER2 categories, 67.2% of tumors achieved an absolute agreement, 32.8% showed high agreement, and none were categorized as challenging. When 1+, 2+, and 3+ HER2 scores were combined into a single category, absolute agreement increased to 86.2%, and high agreement decreased to 13.8%. Similarly, when only 1+ and 2+ HER2 scores were combined, absolute agreement rose to 85.3%, with high agreement at 14.7% (Table 5). This increase in agreement is because, in the grouped analysis, score differences like 1+ vs 2+ are no longer considered disagreements, which leads to higher absolute agreement and decreased high agreement.
Agreement among combined and individual categories.

The kappa values were 0.504 for the 0 score, 0.604 for the 1+ score, 0.620 for the 2+ score, and 0.915 for the 3+ score, all with P-values < .0001 (Table 6). The Kappa values indicate a moderate to substantial agreement for lower HER2 scores (0, 1+, and 2+) and almost perfect agreement for the highest HER2 score (3+).
Fleiss’ multirater kappa of agreement.

Discussion
This is the first study in Jordan and the Arab world to examine the concordance of HER2 scoring in breast cancer. While our study was not designed to estimate true population-level prevalence, the original HER2 scoring showed that HER2-low accounted for most tumors examined in our study (N = 84, 72.4%), highlighting the potential benefit for a large portion of breast cancer patients in Jordan from anti-HER2 therapies. Globally, of all breast cancer patients, around 50% to 67.2% are categorized as HER2-low patients.15,16 The higher prevalence of HER2-low tumors in our study may be due to the small sample size. In our larger studies, HER2-positive tumors made up 24%, while in this study, they accounted for only 18%. This suggests an underrepresentation of HER2-positive tumors, which may have led to an overrepresentation of HER2-low tumors. 4 A previous retrospective study of breast cancer patients in Jordan revealed that patients with HER2-zero scores had better ribociclib and endocrine treatment outcomes compared with HER2-low patients. 17 These findings underscore the importance of recognizing HER2-low expression as a distinct category in Jordan, given the emerging evidence that patients with HER2-low breast cancer can benefit from novel anti-HER2 therapies, specifically trastuzumab deruxtecan. 8
The Fleiss multirater kappa values suggested moderate to substantial agreement for lower HER2 scores (0, 1+, and 2+) and near-perfect agreement for the highest score (3+). The HER2 IHC score of zero showed the lowest kappa value (0.5), reflecting moderate agreement and highlighting the difficulty in distinguishing negative from low HER2 expression. This variability is expected because, by definition, HER2-low tumors exhibit faint, barely perceptible, and incomplete membrane staining, which can be challenging to accurately assess. In addition, the cutoff between a score of 0 and 1+ is arbitrarily set at 10%, meaning tumors with staining near this threshold may be scored differently, as either 0 or 1+, by different observers or even by the same observer at different times, reflecting both interobserver and intraobserver variability. This is consistent with previous studies showing greater variability in the 0 and 1+ categories. 12 A study using CAP survey data from over 1400 laboratories found poor concordance in distinguishing HER2 scores of 0 vs 1+, with less than 70% agreement in 19% of tumors. This discordance was further highlighted in a separate analysis within the same study, with 170 breast cancer biopsies reviewed by 18 pathologists, where only 26% concordance was achieved between scores of 0 and 1+. 18 Another global retrospective study of 789 patients with HER2-negative breast cancer found that, following HER2-low-expression training, 67.2% of tumors were rescored as HER2-low, including over 30% of tumors originally classified as IHC 0. 16
Recent evidence highlights the potential for tumors with very low HER2 expression to benefit from emerging therapies. 19 With these findings, guidelines may redefine the HER2-zero score to represent the complete absence of HER2 expression, without or with membrane staining in 10% or less of tumor cells. This change could significantly reduce discrepancies in scoring, particularly in tumors with low HER2 expression levels. 20
Most tumors (N = 78, 67.2%) were concordantly classified by all 3 pathologists, and no tumors had complete disagreements among the 3 pathologists. This may be due to the fact that discordance among pathologists scoring based on well-established guidelines is usually only a 1-step difference (eg, 2+ vs 1+ or 1+ vs 0). Therefore, it would be extremely unlikely for 3 pathologists to assign 3 different scores when only 2 categories are possible, especially since the 3+ score has almost perfect concordance among the 3 pathologists.
Our study demonstrated substantial agreement to almost perfect among the 3 pathologists in scoring HER2 expression, with a Cohen’s weighted kappa ranging from 0.69 to 0.85. The highest agreement was observed between pathologist 2 and pathologist 3 (0.85), indicating almost perfect agreement, highlighting the reliability of HER2 scoring and consistent HER2-low classification in our institution’s clinical practice. In comparison to previous studies, the percentage of tumors with absolute agreement in our study is significantly higher. For instance, the absolute agreement was only achieved in 6% of tumors in a recent study on the concordance of HER2 scoring in the United Kingdom and the Republic of Ireland. 12 However, this study included 16 pathologists in comparison to only 3 pathologists in our study. Another study involving 6 pathologists using the HercepTest (Agilent Dako, Snta Clara, California) and PATHWAY anti-HER2 (4B5) (Ventana Medical Systems, Tuscon, Arizona) assays reported complete concordance rates of only 44.7% and 45.6% of cases, respectively. 21 In a recent study, trained pathologists had substantial interobserver agreement in scoring HER2-low and HER2-ultralow expression, with kappa values of 0.69 and 0.79, respectively. 22
The relatively high level of agreement among the 3 pathologists in our study reflects adherence to the CAP/ASCO guidelines for HER2 reporting and the experience of the pathologists, each having a minimum of 5 years of experience. However, it should be noted that the original scoring was more discrepant, as it was performed before the distinction between scores 0 and 1+ became critically important. Regarding the 2+ scores, the lower number upon rescoring may be because the second and third pathologists knew this was a study without direct consequences for patients, whereas the original pathologist might have erred on the side of 2+ in borderline cases to give patients the benefit of the doubt, allowing for further testing and potential benefit from targeted treatment if FISH was positive.
Clearly, there is a need to improve accuracy in HER2 scoring, as it has significant implications for patient eligibility to receive new antibody-drug conjugates targeting HER2-low and HER2 ultra-low breast cancers. Institutions, including ours, should adopt the updated HER2 reporting guidelines with synoptic-style detailed reporting and implement the recommended quality measures to ensure accurate scoring. In addition, the use of artificial intelligence to enhance HER2 scoring accuracy has been piloted at our institution, and we plan to adopt it more broadly in the near future.
Several limitations should be acknowledged. First, our study was conducted at a single institution, reducing the generalizability of our findings, and most importantly, because the tumors were randomly selected, the observed proportions of HER2-low tumors in our study should not be interpreted as representative of the broader Jordanian population. In addition, while the sample size was adequate for statistical analysis, larger studies with more pathologists are needed to confirm these results and explore potential variations. Furthermore, some bias may have been introduced, as the 2 pathologists who performed the rescoring were aware of the specific objectives of this study and may have paid closer attention to differentiating scores of 0 from 1+, whereas the first pathologist, who did the original scoring, conducted it at a time when misclassifying a score of 0 as 1+ was not considered impactful.
Conclusion
In conclusion, this study provides valuable insights into the observed proportion of HER2-low breast cancer within a randomly selected Jordanian cohort and demonstrates substantial agreement among pathologists in HER2 scoring. These findings underscore the importance of recognizing HER2-low expression as a distinct category and the potential benefits of the emerging anti-HER2 therapies for a large patient population. Standardized scoring protocols and continuous training are essential to ensure accurate and consistent HER2 assessment, ultimately guiding effective and personalized treatment strategies in breast cancer management. Further research involving larger and more diverse cohorts is warranted to validate these findings.
Supplemental Material
sj-docx-1-bcb-10.1177_11782234251363664 – Supplemental material for Human Epidermal Growth Factor Receptor 2-Low Breast Cancer: Prevalence Rate and Scoring Concordance Among Pathologists
Supplemental material, sj-docx-1-bcb-10.1177_11782234251363664 for Human Epidermal Growth Factor Receptor 2-Low Breast Cancer: Prevalence Rate and Scoring Concordance Among Pathologists by Maher Sughayer, Ghada Al-Jussani, Ahmed Salem, Yassin Mullahwaish, Fanar Alsmarat and Dua Abuquteish in Breast Cancer: Basic and Clinical Research
Footnotes
Acknowledgements
None.
Ethical Considerations
This study received ethical approval from the Institutional Review Boards of King Hussein Cancer Center (approval no. 24 KHCC 103).
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability
The data supporting the findings of this study are fully presented in the manuscript. Reasonable requests for further data can be made to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
