Abstract
Breast cancer remains the most prevalent malignant tumor and a leading cause of cancer-related mortality among women worldwide. The disease comprises distinct molecular subtypes that influence tumor behavior, metastatic potential, and therapeutic response. This review presents recent advances in breast cancer research, with a particular emphasis on molecular and epigenetic mechanisms that contribute to tumor development, progression, and treatment resistance. Key signaling pathways—including estrogen receptor (ER), human epidermal growth factor receptor 2 (HER2), Notch, fibroblast growth factor receptor (FGFR), Wnt, and Hedgehog—play essential roles in the regulation of mammary stem cells and oncogenesis. Increasing evidence highlights the significance of epigenetic alterations such as DNA methylation, histone modifications, and microRNA expression in modulating gene activity relevant to tumor initiation and therapy resistance. Epigenetic molecular targets, including DNA methyltransferases (DNMTs), histone deacetylases (HDACs), EZH2, and non-coding RNAs, are gaining attention for their potential use in diagnosis, prognosis, and targeted therapy. Integration of multigene panel testing with epigenetic biomarkers has facilitated improved risk assessment and the development of individualized treatment strategies. Moreover, novel therapeutic approaches—such as CAR-T cell therapy, nanoparticle-mediated drug delivery systems, and the involvement of circular RNAs (circRNAs) in immune modulation—offer promising directions for precision medicine. This review consolidates current insights into the molecular and epigenetic landscape of breast cancer to provide a comprehensive understanding of disease complexity and to inform the development of more effective, personalized treatment options.
Introduction
Recently, there has been a steady increase in the prevalence of breast cancer (BC) globally, especially in developed countries, with incidence rates of 1% and 99% in men and women, respectively. Globally, one in 7 women is likely to develop BC throughout their lives. 1 Important risk factors for BC include hormonal contraception, excessive alcohol intake, inadequate diet, and excessive ultraviolet (UV) radiation exposure at an early age. Other risk factors include a history of ductal carcinoma, high body mass index, first delivery at a young age, nulliparity, relatively early menarche (before age 13), delayed menopause, and use of postmenopausal hormone treatment. In addition, it is hypothesized that both endogenous and exogenous factors, including aging, food, cigarette smoking, and UV exposure, whether individually or in combination, affect bodily tissues and increase the risk of cancer development. Notably, BC-related morbidity and mortality vary considerably across geographical locations. For example, women with stable weight and large mammary tissue are at a higher risk than others among nulliparous hormone therapy adopters. In addition, women undergoing breast radiotherapy are highly vulnerable. 2
Currently, BC treatment consists of multiple therapies, including hormone treatment for hormone-positive diseases, chemotherapy, and human epidermal growth factor receptor 2 (HER2) targeted therapy. However, over 15% to 20% of BC cases are triple-negative BC (TNBC), indicating that the HER2 protein is either not generated enough or not present in these cases, or there is a shortage of progesterone and estrogen receptor (ER) sites. 3 In this review, we provide a comprehensive overview of recent findings on the molecular mechanisms and pathology of BC, emphasizing the role of multiomics techniques, which integrate genomics, transcriptomics, proteomics, and metabolomics to provide a holistic understanding of the disease. 4 This review aims to provide a comprehensive analysis of the molecular mechanisms and epigenetic modifications that contribute to BC heterogeneity, progression, and therapeutic resistance. By synthesizing recent findings from multiomics studies, we highlight key signaling pathways, emerging biomarkers, and novel therapeutic strategies that could improve early diagnosis and personalized treatment approaches. 5 Understanding these molecular and epigenetic factors is crucial for advancing targeted therapies and enhancing patient outcomes. Tamoxifen resistance, a major obstacle in hormone receptor-positive BC, has been increasingly linked to epigenetic alterations. Recent studies highlight the role of DNA methylation, histone deacetylases (HDACs), and non-coding RNAs such as miR-221/222 in modulating ER signaling and driving therapeutic resistance. 6 Aberrant expression of EZH2 and alterations in chromatin remodeling factors have also been implicated in sustaining tamoxifen-resistant phenotypes. Incorporating epigenetic therapies targeting these regulators may offer new hope in overcoming endocrine resistance and restoring drug sensitivity in affected patients. 6
Methodology
A comprehensive literature search was conducted using PubMed, Scopus, Web of Science, and Google Scholar to identify relevant studies published up to January 2025. The research identified the following keywords for the search: (Breast cancer OR Mammary carcinoma OR Tumor heterogeneity* AND Molecular pathways* OR Epigenetic regulation* OR Gene expression* OR Biomarkers* OR Signaling pathways* AND Targeted therapy* OR Prognostic markers* OR Multigene panel testing* OR Personalized medicine* OR Epigenetic modifications* OR MicroRNA* OR DNA methylation* OR Histone modifications* OR Cancer progression* OR Metastasis). The research included analyses of BC molecular mechanisms while studying epigenetic alterations and developing new treatment approaches. All methods of data extraction along with quality assessment and findings synthesis followed the PRISMA guidelines.
Types of Breast Cancer According to Molecular Analysis
The BC is classified into molecular subtypes based on genetic factors that define its phenotypic behavior. Notably, tumors with comparable physical traits exhibit a wide range of clinical outcomes owing to their distinct gene profiles. Currently, BC is classified into 5 molecular subtypes based on gene expression: luminal A (progesterone receptor [PR] positive, HER2 negative, and ER positive), luminal B, non-luminal HER-2, TNBC, and basal-like cancer with unique histological forms. The TNBC and basal carcinoma with basal feature (BLBC) are comparable and closely linked, and both have poor prognosis and considerable metastatic rates. However, most BLBC cases lack the ER; therefore, the PR is the HER2 receptor site. Moreover, BLBC is frequently called TNBC, as both cancers exhibit similar immunophenotypes, as indicated by pathological criteria. However, BLBC and TNBC are not necessarily the same based on gene expression. Specifically, only 71% of TNBCs appeared basal, while 77% of BLBCs exhibited similar characteristics with TNBC. 7 Notably, the primary treatment modalities are surgery and radiation, with subtype-specific treatment approach. Currently, patients with BC are diagnosed and classified based on molecular subtypes using accessible immunohistochemistry tests, which are a crucial part of therapy selection. Luminal type A is characterized by increased gene expression linked to ER activity and poor expression of genes linked to HER2 receptor activation and proliferation. Luminal type B is characterized by a positive ER status and reduced ER gene expression (but higher than in luminal type A). Proliferation is determined using the marker protein Ki-67 and is indicated by a positive ER status. 8 To distinguish between the types A and B luminal tumors, researchers have identified differences in the variables used in the formula for the calculation of Ki-67, which is crucial for predictive treatment evaluation. 8 Overall, the laminal type A and B possess similar characteristics to those of TNBC, as they do not express HER2 receptor as well as genes related to estrogen, progesterone, and HER2 receptors. Although biochemical markers (cytokeratin [CK]5/6, HER1, and c-KIT) can be used to identify both comparable and distinct basal subtypes, their therapeutic efficacy is debatable. 7 Currently, the only available systemic treatment is chemotherapy, as cancer cells are deficient in HER2, making medications that influence HER2 ineffective. In addition, although TNBC initially responds to treatment, it has a higher recurrence rate than other BCs. Currently available treatments for TNBC include chemotherapy and tumor excision. However, there is no targeted therapy to stop the recurrence of TNBC due to its molecular heterogeneity, poor cell differentiation, high malignancy, lack of molecular targets, rapid metabolism, metastasis, and susceptibility to chemotherapy.
Based on recommendations at International Breast Cancer Conference, treatment strategies for patients with early-stage BC have been revised. In addition, therapy for HER2 positive and TNBC after preoperative therapy has been highlighted in recent St. Gallen recommendations. 9
Furthermore, BC is classified into 2 main types based on histological appearance: extensive tumor and localized ductal carcinoma (ductal carcinoma in situ). Notably, there are several phenological variations of invasive carcinomas, such as tubular carcinoma, cribriform carcinoma, and invasive lobular carcinoma, which affect prognosis. In addition to morphological subtyping, other traditional prognostic indicators include lymphovascular emboli, lymph node metastasis, and histological grading. 10
Field Cancerization in Breast Cancer: Determinants
Molecular genetic testing that focuses on the original tumor and pathological analysis are currently used to ascertain treatment strategies and recurrence risk in patients. Despite extensive research, cancer recurrence is still observed in patients after breast-conserving surgery and postoperative radiation. The field cancerization theory postulates that the formation of cellular molecularly changed regions and the environment has the intrinsic propensity to lead to cancer progression. Field cancerization can occur regardless of morphological change, with a recurrence rate ranging from approximately 3% to 15% after 10 years. 3 According to the tumor microenvironment theory, the development and spread of tumors are considerably influenced by microenvironmental factors. These hypotheses are unlikely to coexist in opposition but rather coexist in a complex interaction that may have an impact on the manifested phenotypes. Notably, molecular changes and tumor-prone cells are found in histologically normal (HN) tissues around chest tumors; therefore, such cells are susceptible to cancer. 11 Overall, these findings contribute to the understanding of mechanisms of tissues around tumors.
Molecular pathogenesis
Recent findings indicate molecular changes in tissue patches and tumor-prone cells around HN tissues near tumor tissues. Therapeutic treatment is guided by the histopathological examination of the tumor, as shown in Table 1. However, tissues near malignancy have been found to exhibit genetic or epigenetic and transcriptomic alterations. 12 In addition, environmental risk factors, such as diet, human papillomavirus and Helicobacter pylori infections, and physiological conditions, can further alter the transcriptome of peritumoral tissues, creating a favorable environment for tumor growth. 13 Research evidence suggests that HN tissue excised from the tumor microenvironment may possess prognostic value. 14 Molecular aberrations exist in HN tissues removed 24 cm or more from the primary tumor microenvironment. 15 Collectively, these findings imply that evaluating cancers and HN tissues in the tumor microenvironment may enhance prognostic and treatment decisions. A thorough assessment of the molecular properties and alterations in peritumoral tissues in various cancers indicated the increased immune response and inflammation, metabolism, and cell proliferation. 16 Although extensive studies have been performed on cancer tissues, the details of alterations in peritumoral tissues and cells are limited. Notably, the cellular characteristics of peritumoral tissues mirror those of the tumor and therefore have unique basic characteristics.
Advances in molecular biology and pathology of breast cancer.

Collectively, these findings indicate the clinically important molecular processes in cancer prognosis, thus showing considerable heterogeneity in the canceled regions. Extensive studies have been performed to identify the number of molecular abnormalities that contribute to an environment predisposed to cancer. A prognostic tool that incorporates a combination of genomic and transcriptomic events may have the highest predictive value because they are inextricably interwoven. 17
Pathological characteristics
A well-defined sequence of proliferative alterations in the breast tissue increases the risks of BC development and related disorders, with the risk of abnormal ductal hyperplasia being the highest. 18 Apart from well-known lesions that are mild and uncommon, the histology of healthy breast tissue varies considerably among individuals. Moreover, subtle alterations in breast tissues are usually missed by regular diagnostics but could be indicative of a field cancerization effect. 19 Morphological heterogeneity in breast tissue affects the risk of cancer, as it is a phenotypic reflection of the individual’s genetic and hormonal environment. For example, the biological mechanism of lobular engraftment results in a decrease in the number of acinar cells in the distal acinar lobular unit and a reduction in the total percentage of lobules for each given volume. Although the prevalence of lobular involution varies considerably across individuals in growth stage and degree of expansion, it is most noticeable during the perimenopausal years. 20
Longitudinal examination of the complex structure of lobes in women who have undergone several biopsies showed that a delay in lobular involution is linked to an increased risk of breast malignancy. 21 In addition, heterogeneity in the histology of the breast also seems to affect the risk associated with known proliferative lesions. For example, women with abnormal epithelium, without the complex structure of the lobes in the breast cells, are at a higher risk of BC than those with full lobular involution. 22 In addition, excessive collagen deposition may affect mammographic density remarkably, resulting in tissue stiffness and improved periductal collagen alignment, which may promote tumor development and progression. 23 Notably, estrogen is linked to lobular involution and mammographic density. In addition, mammographic density is affected by reproductive and menstrual variables, with decreased density linked to pregnancy and menopause. 24 Moreover, the administration of extrinsic hormones has a similar effect as menstrual variables on breast density. 25 Although there is evidence of a connection between the percentage of average screening mammography intensity and estrogen receptor 1 (ESR1) genetic polymorphisms, research linking internal enzyme balance and breast compactness remains inconsistent, presumably because of several confounding variables. 26
Sexual and cyclical variables affect mammographic density, particularly lowering the density, which is associated with menopause and pregnancy. Breast density is comparably impacted by exogenic hormone delivery. Despite indications of a correlation between mammography intensity and frequency of single nucleotide polymorphisms (SNPs) in the ESR1 locus, studies on the relationship between endogenous hormone balance and breast density are inconsistent, most likely as a result of multiple confounding factors. 27 However, recent advances in diagnostic technologies and machine learning provide an opportunity to obtain extensive information from histological sections, which may improve prognosis and treatment outcomes. 28 As a result, it may be possible to accurately evaluate field cancerization in tissues that seem normal to enhance patient treatment.
Signaling Pathways Involved in Breast Cancer Pathology
Complex signaling networks tightly regulate normal cell and human growth. These pathways enable cells to exchange information with one another or even with their surroundings. 29 Several signaling pathways, such as cancer cell trafficking, are obstructed by various cell types and diseases. Particularly, genetic and epigenetic changes that enable cells to bypass mechanisms that regulate their multiplication, survivability, and migration contribute to the development of cancer. Overall, these numerous changes correspond to signaling networks that control cell motility, differentiation, growth, and cell death. 30 Mutations in proto-oncogenes may cause a dysfunction in these signaling pathways, resulting in cancer progression. Collectively, these signaling pathways are crucial for balancing cancer stem cell (CSC) self-renewal and differentiation into other cells. 31 In the preceding sections, we discussed the contributions and molecular mechanisms of some signaling pathways in BC.
Human epidermal growth factor receptor 2 signaling
The HER2 and epidermal growth factor receptor (EGFR), members of the human epidermal growth factor receptor group, actually belong to the family of tyrosine kinase receptors and are expressed in cancerous and normal cells. 32 Several downstream signaling, including mitogen-activated protein kinase or MAPK and phosphatidylinositol bisphosphate 3-kinase (PI3K) signal transduction pathways, is stimulated following ligand binding and phosphorylation of tyrosine in the cytoplasmic domain of HER2. 33 Breast tumorigenesis is strongly correlated with these signaling pathways, as shown in Figure 1. 34 Upregulation of HER2 signaling induces HER2 protein overexpression, which is connected to the growth of tumor cells and cancer development. 35

Schematics of signaling pathways (MTDH/AEG-1) involved in promoting tumor progression.
In addition, the precancerous effects of HER2 are related to inflammation and the growth of CSCs in breast melanoma. 36 Research findings suggest that a recently discovered enhancer at the 3’ gene body of the HER2 gene is the target locus of the HER2 regulator TFAP2C. 37 Moreover, this process is also influenced by other epigenetic processes, such as deoxyribonucleic acid (DNA) methylation and histone mutations. Notably, HER2-expressing BC cells have a higher risk of metastasis than other cells. Progesterone and progesterone-induced paracrine signals probably drive migration in primary cancer cells, resulting in the activation of MSCs or mammary stem cells. 38 The HER2 analysis has been performed on select patients to identify individual who may benefit from potentially resistant therapy, as shown in Table 2. Molecular analysis of HER2 has become a crucial component of the diagnostic workup for patients with BC to increase treatment specificity. 39
Signaling pathways involved in breast cancer.

Fibroblast growth factor receptor signaling
Fibroblast growth factor receptors (FGFRs) are tyrosine kinase receptor subtypes that span a particular region of the biological membrane, making them single-pass membrane proteins. The FGFRs consist of an internal portion containing tyrosine kinase motifs that can be phosphorylated and a carboxyl-terminal end, as well as 3 distinct subdomains, including immunoglobulin (D1, D2, and D3), at their N-terminal extracellular domains. 47 In humans, the FGFR family comprises 6 receptors (FGFR1 to FGFR6) that bind to 18 fibroblast growth factor ligands or FGFs. 48
Notably, substances that attach to these cellular membrane receptors cause cells to react to extracellular signals. Once coupled, the receptor is activated, leading to a chain of events that activates more molecules. The greatest class of these kinds of receptors that have such functionality is the receptor tyrosine kinase (RTK) family. 49 Specifically, the binding of ligands induces monomers of RTK to create dimers; the monomers tether the tyrosine residues and cause them to inter-phosphorylate, directly activating one another. 50 This process of dimerization and inter-phosphorylation allows other surrounding molecules, which are known as adaptors, to adhere and phosphorylate proteins in the cytoplasm. One such adaptor is FGFR substrate 2, also known as FRS2. Following the binding of FGF, FGFRs assemble into dimers and link to the FRS2 adaptor, which triggers several downstream signaling events that activate major tumorigenic pathways, 51 including the PI3K protein kinase A (PI3K-AKT) and MAPK. 52
Research evidence indicates that dysregulated FGFR signaling pathways can act as cancer-driving oncogenes. 48 Dysregulation of the FGFR pathway prevents cell apoptosis, enhances mitogenesis, and promotes epithelial-to-mesenchymal transition (EMT). 53 The following mechanisms are involved in the dysregulation of FGFR pathway: the appearance of fusion proteins with FGFR, which is caused by the translocation of genes that activate the kinase activity of FGFR 54 ; FGFR mutations; overexpression of genes and post-transcriptional events that eventually boost FGFR protein thresholds; FGF overexpression inside stromal and tumor cells and intracellular spaces that stimulate the paracrine pathway; and alternative splicing of FGFR. 48 Several internal cellular processes regulate FGFR signal transduction, which in turn triggers cell apoptosis, mitogenesis, multiplication, EMT, and the development of oxygen delivery channels during cancer. 55
Notch signaling
The Notch signal transduction pathway involves 5 ligands that act as single transmembrane proteins. 56 The Notch signaling pathway is based on cell-to-cell interaction 57 and is activated when Notch ligands bind to receptors on nearby cells. 58 The Notch extracellular and intracellular domains are produced by a series of important proteolytic fragmentations of the Notch ligand-receptor complex. Notably, the Notch intracellular domain behaves as a transcription factor and becomes responsible for the regulation of downstream genes of interest. 59 In BC, the activation of the Notch pathway induces aberrant proliferation of breast cancer stem cells (BCSCs). Notch-1, a Notch signaling cascade receptor, can regulate BCSC self-renewal, multiplication, and apoptosis as well as epithelial-mesenchymal transition to BC. 60 Aberrant Notch signaling pathway activation contributes to the progression of BC largely by regulating angiogenesis. The activity and destiny of BC cells during the metastatic cascade may be controlled by the Notch signaling pathway in conjunction with other signal transduction pathways. 61 Moreover, the HER2 signaling pathway, which is active in approximately 20% of breast tumors and linked to a more aggressive disease, also interacts with the Notch signal transduction pathway.
Hedgehog signaling
The Hedgehog (Hh) signaling pathway plays a crucial role in the development of ducts in the mammary glands, regulates BCSCs, and is essential for cancer development. 62 Severe abnormalities in morphogenesis (ductal) are caused by disruption of downstream transcriptional targets, which include protein patched homolog 1 (PTCH-1) and glioma-associated oncogenes zinc finger 2 (GLI-2). Mechanistically, the transcription factor GLI-1 interacts with the CXC motif chemokine 12/14 (CXCL12-CXCR4) axis to promote lung invasion of BC cells in a mouse model. 63 The dysregulation of Hh signaling in breast melanoma may induce the activation of other signal transduction pathways. Transforming growth factor (TGF) and Hh promoted liver tumor lung metastasis in animal models of HCV or hepatocellular carcinoma. 64 Patients with TNBC characterized by Hh and Wnt signaling coactivation have lower symptom and overall life expectancy rate. 65 Activation of the Hh pathway contributes to the exacerbation of BC and may help in the prediction of postoperative deterioration, as high expression of PTCH-1 and GLI-1 correlates with the invasiveness of BC. 66
Estrogen receptor signaling
The ERs are classified as nuclear or membrane-bound receptors. 67 The ERα and β are nuclear receptors that possess the ability to inhibit or activate target genes. 68 Both receptors have 6 functional domains and possess several similarities and the ability to generate heterodimers. 69 The ERα plays a substantial role in the progression and pathogenesis of BC, as its expression is upregulated in BC. 70 Recent findings indicate that approximately 75% of patients with BC show positive ERα expression. 71 Notably, ERβ inhibition activates BC type 1 (BRCA1) gene, resulting in the suppression of tumor cell proliferation. 72 However, ERβ expression decreases with increasing proliferation of BC cells. Notably, ERβ exerts tumor-suppressive effects in a mouse model by interacting with p53.
In contrast, ERα promotes the progression of BC cells by interacting with cyclin D1. 73 Cyclin D1 regulates cell cycle transition from the G1 phase to the S phase in some tumor cells. 74 Therefore, the interaction of ERα with cyclin D1 is considered to be the most significant mechanism through which ERα promotes the proliferation of BC cells. 38
The ERα is classified into various isoforms based on the structure, 75 among which the ERα36 isoform promotes metastasis and exacerbates disease severity in patients with BC. In contrast, ERα tamoxifen isoform is targeted in the treatment of BC and has an antagonistic effect on ERα36 due to the upregulation of aldehyde dehydrogenase 1 family member A1 (ALDH1A1) gene. 76
Wnt signaling
Wnt signaling pathway or β-catenin pathway plays a crucial role in mammary gland tumorigenesis. 77 β-catenin overexpression induces the activation of the Wnt signaling pathway. Compared with that in normal cells, β-catenin is highly activated in BC cells. 78 In addition, the Wnt coreceptor LRP6 is overexpressed in TNBC. 79 Proteins of the Wnt family are divided into 2 types functionally, with 1 group capable of activating the Wnt pathway and the other group capable of activating the polarity of planar cells and Wnt/calcium processes. 80 Wnt5a has been shown to promote tumor growth and migration in TNBC. 81 Notably, some studies revealed that Wnt5a might slow the spread of BC and that its absence is linked to a bad prognosis. Both canonical and non-canonical Wnt pathways can be activated by Wnt5a. 82 However, further research is required to understand the specific contributions of each Wnt member to BC growth and metastasis.
Epigenetic Modifications in Breast Cancer
Histone modifications
The effects of histone modifications, such as acetylation (covalent binding of acetyl group of acetyl-CoA to the lysine amino group), deacetylation (removal of the acetyl group from the lysine tail of histone), and methylation (addition of methyl group to the histone tails of lysine and arginine), in breast malignancies have been extensively studied, as shown in Figure 2. 83 Histone deacetylation results in a tighter chromatin structure and suppression of histone gene expression. 84 In addition, indicators of histone acetylation and methylation differ between normal and cancerous cells in breast tissue. 85
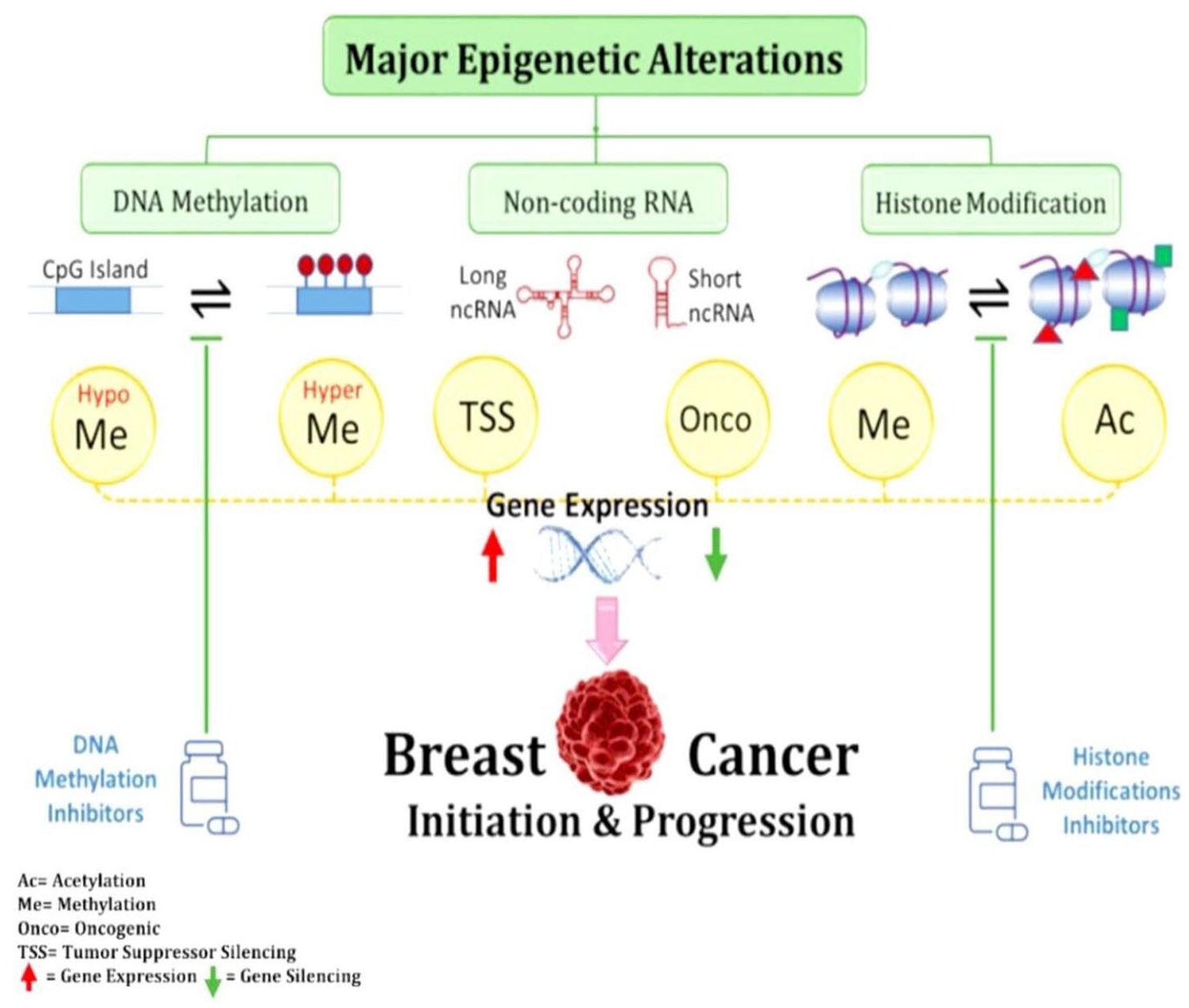
Oncogenes and tumor suppressor genes are schematically shown to be dysregulated as a result of CpG island methylation and histone alterations. 86
Moreover, markers for distinguishing BC subtypes have been identified. Breast cancer subtypes, such as luminal A and basal, exhibit H3K4me3, H3K27me3, and H3K27me3 downregulation. However, H3K27me3 overexpression was detected only in the luminal A subtype, 87 which inspired the concept of clustering within BC subtypes. Notably, the 3 H3 lysine acetylation, 14H3 lysine methylation, and 14 lysine combinatorial variations of histone alterations have been assessed in patient-derived cancerous tissue and primary carcinoma cells of the breast and BC cell lines. The type of cell lines may vary according to the changes in histone remodeling. The BC cell lines overexpress K36me1 or K9me3/K14Ac, whereas primary breast cells appear to lack the K14Ac biomarker. 88 The effects of dysfunctional histone modifications (such as H3K9ac/H3K27me3 imbalance) on carcinogenesis, tumor cell proliferation and multiplication, BC signaling pathways, and metastasis have been extensively studied. 89 In addition, a link has been shown between the H3K9ac instability and genes involved in cell division, apoptosis, cell-to-cell communication, migration, and energy metabolism. 90
Histone deacetylases are classified into 4 categories based on their similarity to yeast proteins. The HDACs regulate the expression and production of several proteins involved in BC development and progression. 91 The HDAC overexpression has been observed in BC. Matrix metallopeptidase 9 (MM9 protein-coding gene) and enhanced cell invasion have been linked to the overexpression of HDAC1, HDAC8, or HDAC6 in BC cells. 92
DNA methylation
During DNA methylation, methyl groups from S-adenosylmethionine (SAM) are reversibly transferred to cytosine at the fifth carbon position in a reaction catalyzed by DNA methyltransferases (DNMTs), such as DNMT3A, DNMT1, and DNMT3B. 93 However, these DNMTs have different functions. For example, DNMT3A and DNMT3B create novel DNA methylation patterns during embryogenesis, whereas DNMT1 only helps in the addition of methyl groups to DNA. 94 Whole-genome sequencing indicated a relationship between BC development and DNA methylation pattern, and 345 methylated genes in 40 distinct breast tumor cell lines were identified. 95 In addition, further examination of the target CpG identified over 18 700 differentially methylated genes. 96 Moreover, hypomethylation in gene bodies has been found in BC. 97 Notably, CpG methylation in the gene body might contribute to the activation of transcription, whereas CpG methylation in the promoter is inversely associated with the expression of genes. 98
Furthermore, DNA methylation affects several genes involved in transcription signaling, DNA binding, homeobox proteins, and cell differentiation. The DNA methylation also affects chromatin structure, transcriptional control, DNA repair, cell cycle regulation, apoptosis, and metabolic activities, all of which are involved in the development of malignant cells. 99 In addition, genes involved in cell adhesion, tissue invasion, and malignancy pathways are affected by variations in DNA methylation. 100 Moreover, DNA methylation capable of affecting the tumor microenvironment may alter oncogenesis. 101
MicroRNAs
Regardless of the cancer stage, microRNAs (miRNAs) are associated with the pathogenesis of breast tumors. The miRNAs possess both oncogenic and cancer-suppressive effects, playing dual roles in cancer. 99 Among 309 miRNAs examined, 133 miRNAs were found to play important roles in both healthy and malignant tissues in various breast carcinoma subtypes. In addition, miRNAs can be used to determine the stages of breast carcinoma. 102 Notably, miRNAs can regulate expression of the ER. Overall, miRNAs play a crucial role in normal breast growth and BC development. However, some miRNAs, such as miRNA-27a, function as oncogenic agents to promote ER overexpression and promote cancer development. 103
The role of microRNA in breast cancer
The miRNAs are associated with the etiology of BC, playing either tumor-suppressive or -promoting roles. Some miRNAs can regulate the expression of ER and can be used to determine the phases of BC. The miRNAs, such as microRNA-335-5p, microRNA-21, microRNA-142-3p, and microRNA-192-5p, suppress ER expression, whereas other miRNAs, such as miRNA-27a, promote ER overexpression to facilitate cancer development. In MCF-7 cells, miRNA-148a upregulates ER expression by decreasing DNMT1 expression. Altogether, miRNAs play a crucial role in both normal breast formation and the development of breast tumor tissues. 61
The miRNAs are involved in cell growth, EMT, energy metabolism, Wnt/-catenin signal transduction pathways, immunological responses, and apoptosis during breast carcinoma development. In contrast, microRNA-1207-5p is associated with increased cell proliferation via the long non-coding RNA PVT1. Several microRNAs, including microRNA-543, microRNA-455, microRNA-143, microRNA-424, and microRNA-26a-5p, suppress cell proliferation. 97 Due to their capacity to control genes involved in EMT and metastasis, SerpinB2 and the miR-200c/141 cluster are regarded as predictive indicators of TNBC.61,104
The microRNA-200c, microRNA-200c, and microRNA-141 upregulation was observed in patients in the malignant phase. MicroRNA-122 promotes metastasis by enhancing the accessibility of nutrients to pre-metastatic sites, and miR-148 prevents the advancement and spread of breast carcinoma cells. In addition, miRNAs are involved in energy metabolism. Several miRNAs, including microRNA-421, microRNA-193a, microRNA-133a, microRNA-211-5p, microRNA-335, microRNA-145, and microRNA-124, can inhibit tumor spread.105,106 The 3’-UTR of the CD274 cluster can be targeted by miRNAs, such as microRNA-195 and microRNA-497, which may promote immune evasion and suppress immune response.105,107,108 Moreover, miRNAs contribute to the ability of tumor cells to avoid the apoptotic response, which is crucial.
The role of circular RNA in breast cancer
Research evidence suggests that circular RNAs (circRNAs) are involved in the onset and spread of mammary melanoma. In addition, circRNA population is lower in breast tissues than in normal tissues, and circRNAs in samples collected from nearby normal tissues may be indicators of breast carcinoma. The circRNA microarray analysis identified differentially expressed circRNAs in patients with BC, including the upregulated circRNAs hsa, circular 100219, and hsa circular 406697 circular 006054, and the downregulated circRNAs circular 103110, circular 104689, and circular 104821. 109
In addition, certain protein-coding alleles that produce large amounts of circRNAs have been associated with tumors. Currently, research is ongoing to elucidate the function of circRNAs in melanoma.
The role of circular RNA in gene regulation
Most biological activities, including cancer development, depend on the regulation of gene expression. The circRNAs may act as microRNA sponges and control the proliferation, metastasis, and migration of cancer cells. For example, circHIPK3 sponges 9 miRNAs, including miR-124, a tumor repressor involved in the development of mammary tumor tissues. In addition, hsa-circ 0001982 knockdown in mammary cancer cells prevented invasion and proliferation and induced apoptosis. Similarly, circ-ABCB10 knockdown promoted apoptosis and reduced the proliferation of BC cells by sponging miR-1271. 110
The circGFRA1 may stimulate proliferation and prevent apoptosis and is favorably linked with tumor size, TNM staging, lymph node metastasis, and TNBC histological grade. The circGFRA1 may act as a microRNA-34a sponge to control GFRA1 production via a competing endogenous (ceRNA) mechanism. Collectively, these findings indicate that circRNAs can control cancer development by trapping specific miRNA species linked to tumorigenesis, proliferation, differentiation, and motility. 111
Epigenetic regulation of radioresistance and mitochondrial oxidative stress in breast cancer
Radiotherapy remains a key component of BC treatment, particularly in localized and post-surgical settings. However, the development of radioresistance significantly limits its long-term efficacy. Epigenetic modifications have emerged as critical mediators of radioresistance, often through altered DNA repair capacity, chromatin remodeling, and dysregulated expression of non-coding RNAs. The DNMTs and HDACs are commonly overexpressed in resistant tumors, resulting in the silencing of tumor suppressor genes and impaired radiation-induced apoptosis. 112
Furthermore, mitochondria are central to both oxidative stress regulation and epigenetic signaling. Ionizing radiation induces mitochondrial dysfunction, elevating reactive oxygen species (ROS) levels that contribute to genomic instability and promote tumor survival. Mitochondrial metabolites, such as α-ketoglutarate and acetyl-CoA, influence nuclear and mitochondrial epigenetic marks, thereby modulating gene expression involved in stress response and energy metabolism. 113 Studies have shown that alterations in mitochondrial DNA methylation patterns and histone acetylation affect the transcription of oxidative phosphorylation genes, thereby promoting treatment resistance. Targeting redox-sensitive transcription factors and mitochondrial epigenetic regulators holds potential in sensitizing resistant tumors to radiotherapy. Thus, integrating knowledge of mitochondrial and oxidative stress-driven epigenetic mechanisms is essential for understanding radioresistance and developing more effective therapeutic strategies in BC management. 114
Microenvironmental Elements
Mammary cancer cells metastasize to tissues, such as the bone, liver, lung, and brain, and several interactions occur between indigenous stromal cells and dispersed tumor cells in colonized tissues. 115 Growth factors, immune cells, chemokines, extracellular matrix (ECM), cytokines, tumor-associated macrophages, tumor-associated fibroblasts, and other unconfirmed components contribute to the development of the tumor microenvironment. 116 Both organ-specific variables and the invasion of various stromal cells can affect the metastatic microenvironment.
Interactions between indigenous stromal cells and disseminated tumor cells in various host tissues are intricate and complicated. Several factors may affect the metastatic microenvironment, particularly in organs and various stromal cells. 117
Cells of the immune system
Tumor recurrence and tumorigenesis are inhibited by the immune system. In addition to immune response to infections, leukocytes play a role in both the development and spread of cancer. Distant tumor tissue locations can cause metastasis by altering the subclinical leukocyte composition. Neutrophils, a class of leukocytes, may play a role in the initiation of metastatic disease.118-120
Specific neutrophils, such as CXCR2+ neutrophils, have been reported to affect the activities of mesenchymal cells of the stroma after metastasis in a murine model of breast carcinoma. Analysis of the pre-metastatic lung microenvironment in a murine model of BC murine showed that neutrophils function as modulators of metastasis initiation, demonstrating that immune cells control the establishment of malignant environments. 121 Specifically, the pro-metastatic action of mesenchymal stromal cells is induced by CXCR2+ neutrophils in a lung melanoma. 121 In addition, researchers have characterized the involvement of neutrophils in the onset of metastasis by altering the pre-malignant lung environment in mice models of BC, indicating that cells of the immune system might control the development of malignant environments.
Notably, the immune system both inhibits and promotes the tumorigenesis depending on the type and stage of cancer. For instance, alterations in leukocyte content at locations remote from the initial tumor can cause metastasis, making leukocytes key immune cells that contribute to tumor growth and metastasis.
Transforming growth factor beta
The TGF-β assists in the creation of a pulmonary microenvironment before metastasis by modifying several inflammatory cytokines (S100A8/A9) and growth factors, such as vascular endothelial growth factor A (VEGF) and angiopoietin 2 ANGPT2, and aberrant production of TGF-β promotes mammary cancer progression by changing the microenvironment.122-125 For example, the TGF-1 receptor kinase (ALK5) inhibitor IN-1130 has the potential to prevent lung cancer in an orthotopic murine model of 4T1 melanoma. In addition, EW-7197, another ALK-5 inhibitor, prevents lung metastasis in patients with BC. Based on these findings, it could be speculated that inhibiting TGF-β signaling, either alone or in conjunction with immunotherapy, may be a potential treatment for lung cancer caused by melanoma. 126 A change in the microenvironment caused by aberrant expression of TGF-β may affect the development of melanoma. Notably, the mechanisms by which TGF-β affects specific cytokines (S100A8/A9) and growth factors to contribute to the development of a pre-metastatic ambiance (VEGF, Angpt2) have been elucidated using a 4T1 xenograft model. Collectively, these findings suggest that immunotherapy combined with the suppression of TGF-β signaling may be an efficient treatment strategy for lung metastasis of breast melanoma. 127
Extracellular matrix proteins
During the early phases of BC invasion, ECM proteins, such as versican, tenascin-C (TNC), and periostin (POSTN), are crucial for the establishment of metastases. The BCSCs express TNC, which is typically generated by fibroblasts. In addition, abnormal TNC expression in BCSCs has a metastasis-initiating effect in the lung. Another stromal-derived component that can bind to Wnt ligands is POSTN. 128 The POSTN has been demonstrated to encourage the growth of cancerous stem cells during lung carcinoma development. Similarly, infiltrating CDllb+/Ly6C myeloid cells produce versican at malignant sites in the lungs to promote lung metastasis. 129 Moreover, ECM components may promote tumor metastasis development by providing a suitable microenvironment for cancer cells to spread throughout the body. Natural killer cells, macrophages, and other immune cells express 41 integrin, which is linked to fibronectin and binds to vascular cell adhesion molecule-1 (VCAM-1). Notably, VCAM-1 is aberrantly produced in mammary cancer cells. Melanomas expressing VCAM-1 favor the parenchyma of lung tissues, which contain collagen and elastin fibers, for homing. 117
Breast Cancer Gene Mutations
Homology-directed DNA repair promotes the collaboration of BRCA proteins to decrease and prevent tumors. Therefore, deletion mutations or damage of BRCA genes may diminish DNA repair capacity, which can increase the risk of carcinogenesis. For example, the risk of breast neoplasm increased by 5 to 6 times following deletion mutations and loss of function in the BRCA genes, increasing the possibility of malignant cell growth.1,130 The BRCA1 and BRCA2 have a total of 1650 and 1731 distinct mutations, respectively.131,132 The most frequent mutation in BRCA1 and BRCA2 genes is a frameshift mutation, which frequently results in the premature appearance of stop codons and lower levels of mature RNAs and functional proteins. Research findings suggest that young patients with more severe subcategories of mammary cancer have a larger proportion of BRCA mutations.133,134 Moreover, clinical evidence suggests a connection between basal epithelial marker expression and breast tumors with BRCA1 mutations that do not show ERBB2 or ER overexpression.132,135
These indicators are frequently linked to neoplasms that do not show expression of ERBB2 or ER. Although these cancerous cells exhibit a basal-like phenotype, BRCA-1 basal-like mammary melanoma cells and spontaneous basal-like mammary cancers originate from luminal epithelial precursors rather than from basal progenitors. Notably, tumor phenotypes may not always reflect the histology of cells with phenotypic variation. Breast cancer frequently activates the PI3K/AKT/mechanistic target of rapamycin (mTOR) pathway, which is regarded as a carcinogenic pathway. 136
Approximately 30% of the time, PIK3CA alterations and Phosphatase and TENsin homolog (PTEN) protein loss are the most common PI3K activation pathways.137,138 Since their discovery in several solid tumors in 2004, somatic mutations in PIK3CA, which encodes p110a, have been described. Even before PIK3CA mutations were identified, the PIK3CA gene overexpression has been observed in several neoplasms, including roughly 10% of mammary cancer cases.139,140 However, only approximately 3% of cancers have PIK3R1 mutations.136,141,142 Overall, the PIK3R1 gene product p85a functions as a negative tumor regulator by maintaining p110a.
Notably, 38 230 AKT1 mutations (E17K) are present in 1.4% to 8% of mammary tumors that express both ER and PR. The ER-positive tumors can develop resistance to hormone therapy if they have alterations that activate the PI3K cascade, such as the downregulation of PTEN, upregulation of HER2 or insulin-like growth factor 1 receptor (IGF-1R), or stimulation of mutant AKT1. Valid prognostic biomarkers, such as PIK3CA mutations, can aid in the clinical care of mammary cancer patients.142,143
The Role of Stem Cells in Breast Cancer
Stem cells play important roles in various aspects of BC development, progression, and treatment resistance. Cancer stem cells, a subset of cells within a tumor, can self-renew and differentiate into various cell types within the tumor. The CSCs are thought to be responsible for tumor initiation and maintenance. In BC, CSCs contribute to tumor heterogeneity, as they can give rise to different cell types with varying levels of aggressiveness. The CSCs are often resistant to conventional cancer treatments, such as chemotherapy and radiation, leading to tumor recurrence after initial treatment. The inherent properties of CSCs, including their efflux pump activity, contribute to their ability to survive and resist the effects of therapy. Stem cells are involved in the process of tumor metastasis to distant sites in the body. 144 In addition, CSCs play a crucial role in seeding and initiating metastatic tumors, contributing to the spread of BC to other organs. The tumor microenvironment, which includes blood vessels, immune cells, and supporting cells, interacts with CSCs. Signals from the microenvironment can influence CSC behavior, including self-renewal, differentiation, and invasion. Stem cells, including CSCs, can undergo EMT, contributing to the ability of cancer cells to invade surrounding tissues and migrate to other parts of the body. Currently, research is ongoing to specifically target CSCs as a strategy for effectively treating BC. Targeted therapies that focus on disrupting pathways involved in CSC self-renewal and survival hold promise for improving treatment outcomes. Understanding the role of stem cells in BC can facilitate the development of personalized treatment approaches with improved outcomes. In addition, identifying and targeting CSCs within an individual’s tumor can potentially lead to tailored therapies that address the specific characteristics of the cancer. Stem cells, particularly CSCs, play essential roles in BC development, progression, therapeutic resistance, and metastasis.
Molecular Diagnostic Approaches for Breast Cancer Detection
Breast cancer detection along with its classification and treatment planning has received major improvements through recent molecular diagnostic developments. Through molecular techniques, researchers obtain vital tumor information that helps doctors make exact medical diagnoses and create specific therapy plans for patients. The fundamental role in BC subtype categorization belongs to gene expression profiling technologies including microarray analysis and RNA sequencing, which distinguish between luminal A, luminal B, HER2-enriched, and basal-like types. 145 The PAM50 gene signature serves as a fundamental tool that supports treatment decision-making. Bioinformatics tools and excellent RNA quality remain essential components of this method, so its access might become restricted in resource-limited settings. The prognostic tools Oncotype DX and MammaPrint use gene expression profiles to determine recurrence risks and help decide whether chemotherapy should be provided to patients. 146
Immunohistochemistry (IHC) provides widespread use in protein detection for hormone receptor (ER and PR) and HER2 analysis in tissue specimens. The results obtained from this method establish eligibility for patients who need endocrine and HER2-targeted therapies. 147 Cost-efficient IHC does exist, yet it has limitations that lead to inconsistent measurements that standard operating procedures help resolve. Doctors use fluorescence in situ hybridization (FISH) to examine HER2 gene amplification as a method for deciding which patients will gain benefit from anti-HER2 medicine. Ordinary health care operations face barriers in using FISH due to its cost and technical requirements, although this technique provides results that exceed IHC accuracy.
Liquid biopsy techniques revolutionize cancer diagnostics through their ability to detect circulating tumor DNA (ctDNA) together with circulating tumor cells (CTCs) as well as biomarkers present in bloodstream samples. The analysis method of liquid biopsies provides an effective substitute for tissue assessments since it enables scientists to track cancer changes directly and observe how patients respond to therapies and when diseases come back. Currently, there are technical hurdles blocking the standardization process of ctDNA detection systems and establishing clinical evidence for global adoption. The main role of next-generation sequencing (NGS) in BC genomics is its ability to analyze every mutation present in important cancer-linked genes such as BRCA1, BRCA2, and TP53. 148 The benefits of NGS for therapy selection extend to hereditary BC patients, yet the data analysis demands specialized expertise and high expense.
Two new molecular diagnostic methods emerged for cancer-risk assessment: one combines testing multiple related cancer genes simultaneously through multigene panel testing and the other uses epigenetic marker analysis for studying DNA methylation patterns that indicate BC progression. The study of SNPs that exist in connection to mammographic density helps scientists develop new risk assessment methods. 144 Additional validation procedures must be completed before implementing these methods in standard clinical procedures.
The application of molecular diagnostics in BC detection and management has revolutionized both diagnostic procedures and therapeutic solutions because of their ability to generate highly specific clinical evaluations. 149 There are differences between each approach but also specific advantages that require individualized application depending on clinical needs. Future investigators should work on improving diagnostic technique efficiency as well as making testing more convenient and implementing multiomics methodology for better accuracy in identification.
Therapies for Breast Cancer Treatment
Chemotherapy
According to the form and phase of cancer, contemporary therapies incorporate several approaches, including surgery, radiation, immunotherapy, hormone therapy, and chemotherapy, as shown in Figure 3. Utilizing tailored chemotherapy enables the drug to be delivered to the cancer mass in a targeted manner, reducing the adverse effects of therapy. To deliver targeted chemotherapy, the use of nanocarriers for anticancer medications may be a wise choice. Currently, several paclitaxel and doxorubicin nanoparticles have received regulatory approval or are undergoing clinical trials for the treatment of BC mutations. 150 Due to the absence of formulation solvents, these nanomedicines are less hazardous than traditional formulations and more specific to the cancer site. 150 Chemotherapy is frequently used in combination with surgery for treating BC clinically. Owing to advances in biology and immunology, the treatment strategies for BC are continuously changing. Recently, neoadjuvant chemotherapy has become a crucial component of BC treatment. 151 Moreover, systemic chemotherapy is usually applied as a neoadjuvant chemotherapy prior to the use of local therapeutic modalities, such as surgery and radiation. Patients with locally progressive and mid-stage BC are major targets of systemic chemotherapy. According to Wang et al, 152 neoadjuvant chemotherapy intends to change inoperable BC into operable BC and serves as a pharmacological basis in follow-up treatment to enhance the prognosis of patients. Trastuzumab is typically included in the neoadjuvant chemotherapy regimen for BC subtypes with HER2 overexpression, and improved outcomes have been observed in clinical usage. 153 Several preclinical studies indicate that brief fasting as well as fasting-mimicking diets can increase the susceptibility of cancer cells to chemotherapy and other treatments while shielding healthy cells from chemotherapy. 154

Treatment strategies for breast cancer.
Radiotherapy for breast cancer
Radiation therapy (RT) is a crucial component of multidisciplinary approaches for treating BC. Recent findings suggest that localized lymph node radiotherapy may offer greater targeted exposure and a decrease in long-term toxicity, leading to a modest improvement in overall survival. In addition, 2 studies supported the use of partial breast irradiation in patients with a minimum risk of BC. 155
Moreover, advances in RT methods, such as 3-dimensional (3D) scheduling of treatments, have reduced radiation exposure to the heart. Piroth et al 156 reported that mean RT dosage exposure to the heart decreased from 13.3 Gy in the 1970s to 4.7 Gy in the 1990s and 2.3 Gy in 2006.
Traditional adjuvant radiation for BC is time-consuming, expensive, and requires daily administration for several weeks. Mastectomy may be the only option available for certain patients, and some may decide to completely forego radiation. 157
Immunotherapy for breast cancer
The BC immunotherapy faces a considerable challenge in improving response rates. Given the relatively limited T-cell infiltration in most BC subtypes, the development of novel approaches capable of improving lymphocyte infiltration and inducing de novo T-cell responses may be crucial to the success of immunotherapy in patients with BCs. 158 Oncolytic viruses have emerged as a promising method to boost treatment efficacy when used in combination therapy and as a distinctive platform for individualized treatment among various strategies currently under consideration. Regardless of their limited effectiveness when administered as a monotherapy, oncolytic viruses have a higher safety and capacity for attacking and killing cancerous cells as well as promoting immunotherapeutic effects in patients, as shown in Table 3.159,160 The development of novel macrophage-targeted BC treatment strategies, such as inhibition of macrophage selection, improvement of macrophage-mediated tumor cell phagocytosis, and macrophage repolarization from tumor to antitumor phenotype, has been made possible by studies on the functions of tumor-associated macrophages in BC development, advancement, therapy resistance, and metastasis. 161 Although promising results have been obtained in preclinical trials, these forms of treatment have demonstrated low clinical success, indicating the need for further studies to improve clinical efficacy.
Treatment strategies for breast cancer.

However, findings from adoptive cell immunotherapies, such as chimeric antigen receptor T-cell therapy or CAR therapy and Tumor-Infiltrating Lymphocyte therapy or TIL therapy, which involve the separation of antitumor T cells from the main tumor, ex vivo growth and stimulation, and the subsequent reintroduction of these cells into the patient, have also been demonstrated in both clinical and preclinical research for the treatment of patients diagnosed with breast carcinoma. 162 The development of neoantigen vaccines that trigger neoantigen-specific immune responses via the stimulation of APCs has also been facilitated by NGS and bioinformatic advances in technology. 163 Similarly, neoantigen recognition and the enhancement of personalized neoantigen-based translational immunotherapy research have been made possible by these advancements.164-166
Although RTK is a useful target for T lymphocyte-based immune therapies in HER2-overexpressing tumors, the tumors may avoid cytotoxic T cell-mediated destruction by suppressing human leukocyte antigen (HLA)-I, as the expression levels of both receptors correlate negatively with BC cells. 167 Similarly, ER expression and HLA-I production are adversely associated with normal and malignant breast tissue. This relationship could be explained by the small proportion of lymphocytes that invade tumors and the ineffectiveness of T-cell cytotoxic reactions. 168 Notably, the administration of kinase inhibitors could be an effective strategy to boost the antitumor effects of T-lymphocyte-based immunotherapies in the event that drugs targeting various protein kinases, including MAPK or HER2, might boost HLA-I expression in BC cells. Accordingly, methods designed to promote HLA-II expression in cancerous cells could prove useful for improving patient response and survival. Improved knowledge of immune escape by cancer cells and the development of specific inhibitors of immune checkpoints have opened new treatment possibilities. Given that lymphocyte infiltration is limited in most breast tumors, monoclonal antibody therapies targeting programmed death 1 (PD-1) and programmed death ligand 1 (PD-L1) demonstrate some benefits in individuals with recurrent BC. Recently, research has focused on developing combined immunotherapy and molecularly targeted therapeutics for metastatic BC. 169 Promising results have been achieved using nanoparticles, as they are effective in considerably enhancing systemic anticancer immune responses compared with currently available immunotherapy. 170 In addition, nanoparticles have shown effectiveness in overcoming immune-suppressive impact caused by tumor’s microenvironment and drug resistance, as well as in delivering neoantigens and adjuvants to cancerous cells, minimizing the adverse effects of anticancer drugs. 170 However, further studies on the effectiveness and mechanisms of nanoparticles are necessary. For instance, some nanoparticles, such as gold complexes, titanium dioxide, and silica, can cause the penetration of residual cancer cells in the vicinity’s vasculature and the development of micrometer-sized holes in endothelial walls of blood vessels, increasing the risk of metastasis.170,171
The incidence of immune-related adverse effects in various organs, such as the epidermis and the digestive tract, is another significant disadvantage of immunotherapies, especially when used in combination regimens. Although these immune-related side effects typically have minimal severity, life-threatening effects might occur and would frequently lead to a decrease in medication dose or termination of therapy. 172 Therefore, further research is necessary to develop biomarker panels for the selection of patients and the prediction of the effectiveness of immunotherapies and potential side effects.
Targeted therapies for breast cancer
Target therapies for BC involve the use of chemicals or medications to stop the spread of the disease by disrupting the functioning of molecules required for the continued existence and multiplication of cancer cells. The BC cells may excessively express certain receptors that can initiate downstream communication when triggered, inducing the production of genes necessary for cancerous cell movement, angiogenesis, expansion, development, and longevity. 173 The BC progression is largely influenced by ERs. This is the rationale for the long-standing procedure of targeting estrogen to inhibit the estrogen signaling pathway in women with estrogen-positive breast tumors. Tamoxifen was the first medicine licensed for advanced estrogen-positive BC and was effective in reducing recurrence by approximately 40% to 50%. 149 Selective ER modulators or SERM have been utilized to restrict tumor expansion in metastatic estrogen-positive BC. Another treatment option for estrogen-positive breast tumors is the use of aromatase inhibitors, such as anastrozole, exemestane, and letrozole, which reduce the levels of estrogen in cancerous cells by blocking androgen production via inhibition of the enzyme aromatase.
Currently, several treatments are available for other subtypes of BC that are not influenced by hormones. The most prevalent overexpressed receptor in BC is the HER2 protein, which is regarded as a useful biomarker for therapy. The first medication licensed by the Food and Drug Administration (FDA) in 1998 for the treatment of HER2 receptor-positive breast tumors was the recombinant antibody trastuzumab, which specifically targets HER2. Additional medications, such as pertuzumab and lapatinib, were not effective against drug-resistant cancers and had adverse clinical effects. However, the conjugated monoclonal antibody TDM1 may be employed in patients with HER2 receptor-positive breast tumors, as it can effectively deliver the microtubule inhibitor DM1 into BC cells to limit growth. 174 The HER1 may be a possible target for HER2- and hormone receptor-negative TNBC that responds to treatments, such as PARP1 inhibitors. For example, combined treatment with the monoclonal antibody cetuximab and the chemotherapeutic drug cisplatin produced promising results in a phase II research, indicating that some subtypes of TNBC may be susceptible to EGFR suppression. Currently, it is common practice to treat patients with TNBC using chemotherapy with anthracycline and taxol analogs. 175 PACS08 phase III trials are currently ongoing to evaluate potential therapeutic strategies for TNBC that is resistant to anthracycline and taxane medications. Recent investigations suggest that combined treatment with the microtubule-stabilizing drug ixabepilone and capecitabine may be effective in treating TNBC. 176
Furthermore, treatment with a combination of bevacizumab and paclitaxel or docetaxel has beneficial effects in patients with advanced metastatic BC. Moreover, targeted medicines against growth factors, such as VEGF, have been approved for BC treatment. In addition to drugs that target other tyrosine kinases, such as SRC (proto-oncogene tyrosine-protein kinase Src), insulin-like growth factor receptor (IGFR), PARP enzyme inhibitors, and MMPs (matrix metalloproteinases), agents inhibiting pathways such as PI3K/AKT/mTOR (phosphatidylinositol 3-kinase/protein kinase B/mammalian target of rapamycin) and RAS/MEK/ERK (rat sarcoma/mitogen-activated protein kinase kinase/extracellular signal-regulated kinase) are readily accessible for BC treatment. 177 The mTOR inhibitor everolimus interacts with the cellular receptor FKBP12 to create a complex called everolimus-FKBP12, which has a strong inhibitory effect on TREM signaling in cancerous cells. In addition, combined treatment with everolimus and exemestane can help improve hormonal sensitivity in patients with BC. The main obstacles to current studies in the field of BC-targeted treatments are resistance mechanisms. Combining various molecules to target different signaling pathways to block cancer cell escape routes and reduce drug resistance is an exciting area for further study. However, it is important to note that molecular targeting, especially for advanced relapses, should be conducted to better understand the causes of resistance. As shown in Table 4, alternative molecular mechanisms that may lead to the emergence of therapeutic resistance in cancerous breast cells may include compensatory survival routes, elevated PI3K signaling, signaling of RTKs outside of the ErbB/HER family, and other HER receptors. 178
Summary of clinical trial outcomes for targeted therapies in breast cancer.

Gene therapy
Gene therapy has attracted considerable interest in BC research. Increased clinical applications of RNA interference (RNAi)-based medicines are necessary, particularly when used in conjunction with conventional therapies. Innovative approaches, such as stem cell-based systems and genome editing, may facilitate the clinical application of gene therapy. Immunotherapies have become attractive treatment options for some patients with BC. Before considering gene therapy as a viable alternative to chemotherapy for BC treatment, several issues must be resolved. 179
Genetic targets (oncogenes and tumor suppressor genes) play important roles in tumor development. Two key molecules, Brk/PTK6 and mTOR, control several signaling processes that affect cell growth, proliferation, angiogenesis, survival, invasion, metastasis, apoptosis, and autophagy. The Brk/PTK6 and mTOR can be employed as therapeutic targets for the treatment of BC, as they are highly elevated during mammary tumorigenesis. However, studies on both proteins are limited. 180
Furthermore, non-replicative and conditionally cytotoxic gene therapies are effective against metastatic BC. Herpes virus, reovirus, adenovirus, poxvirus, and Newcastle disease virus are examples of oncolytic vectors that can multiply inside breast tumor cells while sparing healthy cells. Armed oncolytic vectors attack angiogenesis and tumor immunity as additional mechanisms. Baculoviruses may be effective BC gene therapy delivery vehicles. In addition, ex vivo genetic alterations of immune cells, such as in dendritic cell (DC) vaccines and adoptive T-cell treatment, are appropriate when using viral gene therapy techniques. Viral delivery systems may improve the effectiveness of gene therapy considerably. Moreover, neural and mesenchymal stem cells may enhance the distribution of gene therapies in metastatic BC. The application of viral delivery systems may improve the efficacy of chemotherapy in BC. However, further research is required to develop improved gene carriers with enhanced targeting and transfection efficiency, reduced toxicity, and improved biostability. Extensive research using the right tumor models may facilitate the adoption of experimental gene therapy techniques in clinical practice. 181 Free nucleic acid gene therapy has the potential to modulate important candidate genes in BC, but its impact is limited by poor cell uptake and instability in circulation. Nanomedicine is a rapidly developing science that aims to ameliorate BC through tailored drug/gene delivery and address the drawbacks of traditional treatments. For BC gene therapy, nanoparticles have the potential to act as game-changers because they can effectively transport and deliver specific drugs or genes to tumor sites by extending the period of flow, increasing bioavailability, and decreasing the likelihood of immune system-based detection. 182
The role of circular RNA in therapy
Recent studies have uncovered certain circRNAs that are crucial for the development of mammary cancer and could be potential therapeutic target. For instance, circRNA-000911 promotes the activity of Notch 1 or nuclear factor kappa B (NF-kB) signaling pathway by acting as a sponge for microR-449a, whereas circ-FOXO3 triggers cell apoptosis and slows the spread of mammary cancer. 183 The CircVRK1 suppresses the ability of BCSCs to spread and self-renew, making it a prospective therapeutic target for reducing mammary cancer metastasis and relapse. The BCSC elimination is essential for reducing the spread and recurrence of breast melanoma. 110 Overall, extensive studies have been performed to elucidate the role of circRNAs as potential therapeutic targets since they were discovered to contribute to the growth and advancement of mammary cancer.
Hormone therapy
Hormonal therapy is necessary for patients with hormone receptor-positive breast neoplasms and is effective in both adjuvant and metastatic disease settings. Although endocrine therapy is typically effective in hormone receptor-positive tumors, its long-term benefits can be limited by primary and acquired resistance. However, periodic fasting or a fasting-mimicking diet enhances the effectiveness of the endocrine therapeutics tamoxifen and fulvestrant in murine models of hormone receptor-positive BC by lowering circulating insulin-like growth factor 1 (IGF-1), insulin, and leptin levels and inhibiting AKT-mTOR signaling via the upregulation of EGR1 and PTEN. 184 In addition, periodic cycles of fasting-mimicking diet in combination with fulvestrant and palbociclib (a cyclin-dependent kinase 4/6 inhibitor) treatment promote long-lasting tumor regression and reverse acquired drug resistance. Moreover, fasting and fasting-mimicking diet prevented tamoxifen-induced endometrial hyperplasia. Studies on patients with hormone receptor-positive BC receiving estrogen therapy indicated that cycles of fasting-mimicking diets cause metabolic changes, including reduced levels of insulin, leptin, and IGF-1, with leptin and IGF-1 remaining low for extended periods. These long-lasting effects are associated with long-term anticancer activity in mice, suggesting that a fasting-mimicking diets could serve as an adjuvant to estrogen therapy in hormone receptor-positive BC. Currently, tamoxifen is the only active adjuvant hormonal therapy for pre- and postmenopausal women, and the duration of treatment can affect disease-free survival, risk of contralateral BC, and overall survival. Aromatase inhibitors, such as anastrozole, letrozole, and exemestane, are only used in postmenopausal women. Fulvestrant is used to treat recurrent disease after or during tamoxifen treatment. Luteinizing hormone-releasing hormone analogs are used in adjuvant therapy in premenopausal patients and sometimes in cases of recurrence. Approximately 50% of hormone receptor-positive breast neoplasms become resistant to hormone therapy; however, certain molecules involved in tumor cellular growth pathways can reverse this resistance. 184
Conclusions
This review highlights critical molecular and epigenetic mechanisms driving BC initiation, progression, and therapy resistance. Breast cancer is now molecularly classified into 5 subtypes, each characterized by distinct gene expression and signaling profiles. Among the most promising molecular targets are ER, HER2, Notch, Wnt, Hh, and FGFR, which regulate key processes such as cell proliferation, apoptosis, and metastasis. In addition, epigenetic regulators including DNMTs, HDACs, enhancer of zeste homolog 2 (EZH2), and non-coding RNAs like miR-221/222 and circRNAs have been strongly associated with hormone resistance, particularly tamoxifen-resistant BC. Mitochondria-mediated epigenetic changes and oxidative stress further contribute to radioresistance and disease aggressiveness. Advances in multiomics approaches and molecular profiling have led to the identification of predictive biomarkers and therapeutic targets. Novel strategies such as CAR-T cell therapy, nanoparticle-based delivery systems, and CRISPR-Cas9 genome editing offer new directions in personalized medicine. Future research should focus on integrating these molecular insights to develop targeted interventions that address resistance mechanisms and improve clinical outcomes in BC patients.
Footnotes
Acknowledgements
The authors acknowledge Dr Manzoor Hussain for helping them in the formatting of the article.
Ethical Consideration
N/A.
Consent for publication
N/A.
consent to participate
N/A.
Author contributions
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data will be available on request.
