Abstract
Background:
Estrogen-receptor (ER), progesterone-receptor (PR), and human epidermal growth factor-2 (HER2) are performed on pre-neoadjuvant chemotherapy (NAC) biopsies of invasive breast carcinoma (IBC). However, they are not done routinely on post-NAC IBC with residual tumor.
Objectives:
This study helps in understanding the effect of alteration in expression of ER, PR, and HER2 in pre and post NAC IBC with residual tumors in terms of disease outcome, such as disease-free survival (DFS) and overall survival (OS).
Design:
This was a Cohort study (Prospective and Retrospective Cohort).
Methods:
All newly diagnosed cases of IBC who had undergone surgery after NAC with pre-NAC biopsies available with residual tumors in the breast, from January 2017 to January 2020 were enrolled in the study (n = 174) and were followed up till July 2022.
Results:
There were 174 cases included in this study. Of the 174 cases, 77 (44%) ER+ cases turned −ve, 10 ER +ve cases remained +ve and 87(50%) cases which were ER−ve remained the same. 48(27%) of PR +ve cases turned −ve, 10 PR +ve cases remained +ve and 116 (67%) cases remained −ve. 64(36%) of HER2 +ve cases turned −ve, 4 (2%) of HER2 +ve cases remained +ve; whereas 103(59%) cases remained −ve. The cases with changes in ER and HER2 status from positive to negative showed a longer DFS and OS which was statistically significant.
Conclusion:
There is a change in the HR and HER2 status in cases of IBC with residual tumor post NAC and patients with changes in the receptor status post-NAC have a better OS and DFS than those whose receptor status do not show a change.
Plain Language Summary
Introduction: Estrogen-receptor (ER), progesterone-receptor (PR), and human epidermal growth factor-2 (HER2) are performed on pre-neoadjuvant chemotherapy (NAC) biopsies of invasive breast carcinoma (IBC). However, they are not done routinely on post-NAC IBC with residual tumor. This study helps in understanding the effect of alteration in expression of ER, PR, and HER2 in pre- and post-NAC IBC with residual tumors in terms of disease outcome, such as disease-free survival (DFS) and overall survival (OS). Methods: This was a Cohort study (Prospective and Retrospective Cohort). All newly diagnosed cases of IBC who had undergone surgery after NAC with pre-NAC biopsies available with residual tumors in the breast, from January2017 to January 2020 were enrolled in the study (n = 174) and were followed up till July 2022. Results: There were 174 cases included in this study. Of the 174 cases, 77 (44%) ER + cases turned −ve, 10 ER +ve cases remained +ve and 87(50%) cases which were ER−ve remained the same. 48 (27%) of PR +ve cases turned −ve, 10 PR +ve cases remained +ve and 116 (67%) cases remained −ve. 64 (36%) of HER2 +ve cases turned −ve, 4 (2%) of HER2 +ve cases remained +ve; whereas 103 (59%) cases remained −ve. The cases with changes in ER and HER2 status from positive to negative showed a longer DFS and OS which was statistically significant. Conclusion: There is a change in the HR and HER2 status in cases of IBC with residual tumor post NAC and patients with changes in the receptor status post-NAC have a better OS and DFS than those whose receptor status do not show a change.
Keywords
Introduction
Breast cancer is the most commonly diagnosed cancer and a leading cause of cancer deaths among females worldwide. Currently, neoadjuvant chemotherapy (NAC) is the initial treatment for all patients with locally advanced breast cancer, followed by surgical management, including modified radical mastectomy or breast-conserving surgery. 1 This study aimed to assess the effect of changes in estrogen-receptor (ER), progesterone-receptor (PR), and human epidermal growth factor-2 (HER2) expression on disease outcomes among patients with residual invasive ductal carcinoma who underwent NAC in a tertiary care center in southern India.
The immunohistochemistry markers ER, PR, and HER2 are useful for subtyping, therapeutic planning, and tailoring chemotherapy regimens of patients with breast cancer. Estrogen-receptor is a nuclear protein expressed in 65% to75% of breast cancers. Estrogen-receptor status is a significant factor in breast cancer management, 2 as ER-expressing breast cancers tend to show better differentiation, favorable prognosis, and responsiveness to hormone therapy. Progesterone-receptor is a transcription factor expressed in about 55% to 65% of breast cancers. Patients with loss of PR show more aggressive cancer behavior and often do not respond to hormone therapy. Human epidermal growth factor-2 is a transmembrane tyrosine kinase receptor that initiates a signaling cascade leading to cell proliferation. Although only 12% to 20% of breast cancers show either over-expression of HER2 protein or HER2 gene amplification, HER2 subtypes exhibit aggressive tumor behavior, poor disease outcome, and decreased response to hormonal therapy. 3 Only hormone-positive tumors receive adjuvant endocrine therapy post-surgery. However, there are no clear guidelines about adjuvant endocrine therapy for patients who have ER and PR conversion after NAC. The effects of changes in hormone receptors and HER2 status on the prognoses of these patients remain unclear.4-6
Material and Methods
This intradepartmental study was conducted in the department of pathology in collaboration with the departments of medical oncology and general surgery at a tertiary care referral facility. It is a cohort study that includes both retrospective and prospective components. Participants included all patients with newly diagnosed invasive ductal carcinoma from January 2017 to January 2020 who underwent modified radical mastectomy or breast-conserving surgery after NAC showed residual tumor in the breast or lymph node and for whom a pre-NAC biopsy was available. Patients who had a pathological complete response were excluded. Additionally, patients were excluded if tissue blocks could not be retrieved from the pathology department or if the tissue was exhausted.
The total number of cases received in our tertiary care center, with pre-NAC and post-NAC biopsies available, was an average of 32 cases over 6 months. The targeted sample size of 174 was estimated using the statistical formula for comparing 2 dependent proportions (estimated at a 5% level of significance and 80% power). The expected change in the proportion of immunohistochemistry positivity to negativity was 0.09 and negativity to positivity was 0.02.
Data collection
Clinical details (eg, age, disease stage, site of metastasis, chemotherapy regimen, and dates of diagnosis, most recent follow-up, relapse, and death, as applicable) were obtained from patient records. Fluorescence in situ hybridization (FISH) reports were collected where available for HER2 equivocal cases with a score of 2 or higher. Histopathological data were collected from the department of pathology and hospital information system. Pathological TNM staging, according to the American Joint Committee on Cancer guidelines, was noted. Histologic tumor subtyping was done according to the World Health Organization (WHO)’s classification of breast tumors. Histological grading of the Tru-cut biopsy using the Modified Bloom Richardson Scoring System also was noted. Since the study was performed in a de-identified manner, wavier of consent was obtained by Institutional ethics committee
Immunohistochemistry was done manually in the laboratory on the tissue sections with residual tumors. The molecular subtypes based on immunohistochemistry were as follows: ER (+), HER2Neu (±), Ki67 (luminal), ER (–), HER2Neu (+), HER2-enriched, and ER (–) HER2Neu (triple negative). Surgical specimens were assessed for pathological response to NAC according to guidelines from the 2018 ASCO-College of American Pathologists report (“yp” T and N stages). The post-NAC modified radical mastectomy or breast-conserving surgery blocks were retrieved from the department of pathology.
Estrogen-receptor and PR were classified using Allred scores (% (0 to 5) + intensity (0 to)), with any score ⩾ 3 considered positive. HER2 status was graded as follows: a grade of 0 indicated no staining or incomplete, faint, or barely perceptible membrane staining in ⩽ 10% of tumor cells; a grade of 1+ indicated incomplete, faint, or barely perceptible membrane staining of > 10% of invasive tumor cells; 2+ indicated weak to moderate complete membrane staining in > 10% of tumor cells; and 3+ indicated complete, intense, circumferential membrane staining in ⩾ 10% of invasive tumor cells. For HER2, scores of 0, 1+, and 2+ with FISH negativity were considered HER2 (–). A grade of 3+ or 2+ grade with positive FISH result was considered HER2 (+). In the absence of a FISH test, a HER2 score of 2+ was considered positive in post-NAC cases.
Estrogen-receptor, PR, and HER2 expression changes in the pre-NAC biopsy and post-NAC cases from January 2017 to January 2020 were analyzed retrospectively and further correlated with disease outcomes, including disease-free survival (DFS) and overall survival (OS) rates in a 24- to 48-month follow-up period. Studied outcome variables included DFS from date of diagnosis to documented relapse or last follow-up and OS calculated from date of diagnosis to date of death from any cause or last follow-up.
Statistical analysis
All categorical variables, such as age, pathological staging, clinical staging, grading, molecular subtype, ER expression, PR expression, and HER2 status are expressed in frequency and proportion. All quantitative variables are expressed as mean (SD) and median (IQR). These include age, size of tumor, and survival outcomes such as rates of DFS and OS. Associations of ER expression, PR expression, and HER2 status with the categorical variables mentioned above were assessed using the chi-square or Fisher’s exact test, and statistical significance of changes in ER, PR, and HER2 expression was assessed using the McNemars test. Survival function was studied using the Kaplan-Meier product moment estimator, and comparison of survival outcomes in relation to changes in ER expression, PR expression, and HER2 status was assessed using the log rank test. All statistical analyses were conducted at a 5% significance level, and P values < 0.05 were considered significant. Statistical Package for Social Science (SPSS), version 20, was used for statistical analysis.
Results
The study enrolled 174 women from January 2017 to January 2020 who were diagnosed with invasive ductal carcinoma, received NAC with residual tumor in the breast, and underwent either modified radical mastectomy or breast-conserving surgery. Follow-up was conducted from January 2020 to July 2022. All participants had tissue blocks available in the department of pathology and case records available from the department of medical oncology and hospital information system. The findings of this study will encourage clinicians to retest ER, PR, and HER2 status on residual tumors post-NAC, aiding in the assessment of tumor evolution or treatment resistance. This approach can help create a personalized therapy plan, ultimately improving the patient’s chances of survival.
Baseline clinicopathological parameters
The patients’ breast cancers were classified into molecular subtypes based on immunohistochemical assessment of ER, PR, HER2, and Ki67 measurements. Of 174 participants, 101 (58.2%) had hormone positive (luminal type) cancer; however, further subtyping into luminal A or luminal B was not done as Ki-67 was not completed for 80 (45.9%) participants. Sixty-three (36.1%) women had triple negative breast cancer, and 10 (5.7%) had HER2 enriched breast cancer. Table 1 presents a summary of the results, along with details of the hormonal therapy administered to the participants. The hormonal therapy was given based on the HR status prior to NAC.
Details of baseline clinicopathological parameters.
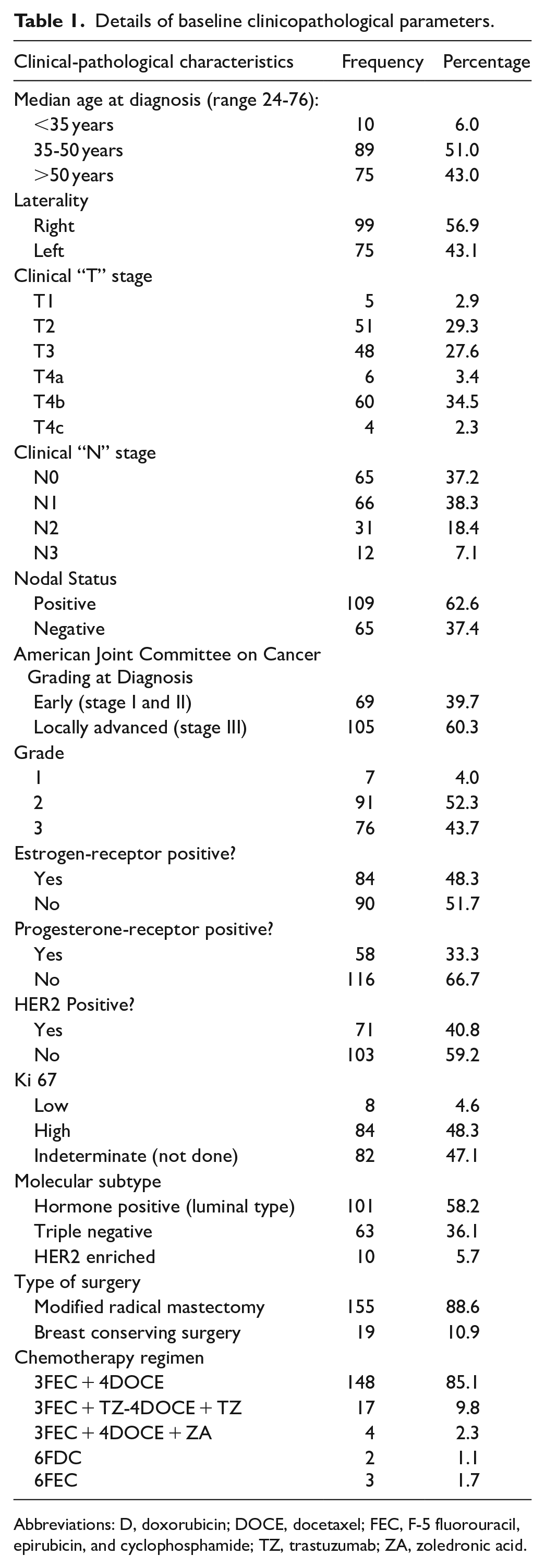
Abbreviations: D, doxorubicin; DOCE, docetaxel; FEC, F-5 fluorouracil, epirubicin, and cyclophosphamide; TZ, trastuzumab; ZA, zoledronic acid.
Clinicopathological parameters after neoadjuvant chemotherapy
Post-NAC, 84 (48.3%) of residual tumors were in the stage ypT2, followed by 38 (21.8%) in stage ypT1, 37 (21.3%) in stage ypT3, and 15 (8.6%) in stage ypT4. The mean period of follow-up was 22 months (ranging from 4 to 59 months); 81 (46.6%) did not relapse during follow-up; 50 (28.7%) were lost to follow-up; 7 (4%) relapsed; and 36 (20.7%) women died during the course of the study. Table 2 summarizes the results.
Details of clinicopathological characteristics after neoadjuvant chemotherapy.

Of the 174 cases, a switch in at least one of the receptor statuses occurred in 36.1%. Table 3 reports the distribution of these changes.
Changes in estrogen-receptor (ER), progesterone-receptor (PR), and HER2 expression after neoadjuvant chemotherapy.

Figures 1A, B and 2 show the control cases. Figure 3 shows the Invasive Ductal Carcinoma before chemotherapy and its immunohistochemical expression. Figure 4 shows a residual tumor post-NAC and its retained expression of ER, PR and HER2. Figure 5 shows another case of residual tumor post-NAC and loss of expression of ER, PR and HER2.

(A) and (B) Diffuse and strong nuclear expression of ER and PR (×400).

Circumferential complete intense membrane staining of HER2 (×400).

Invasive ductal carcinoma before neoadjuvant chemotherapy. (A) Tumor cells arranged in nests and cords: grade 2, score 7 (H&E ×400). (B), (C) Tumor cells showing strong and diffuse nuclear expression of ER and PR (immunohistochemistry ×400). (D) Circumferential complete intense membrane staining of HER2 (immunohistochemistry ×400).
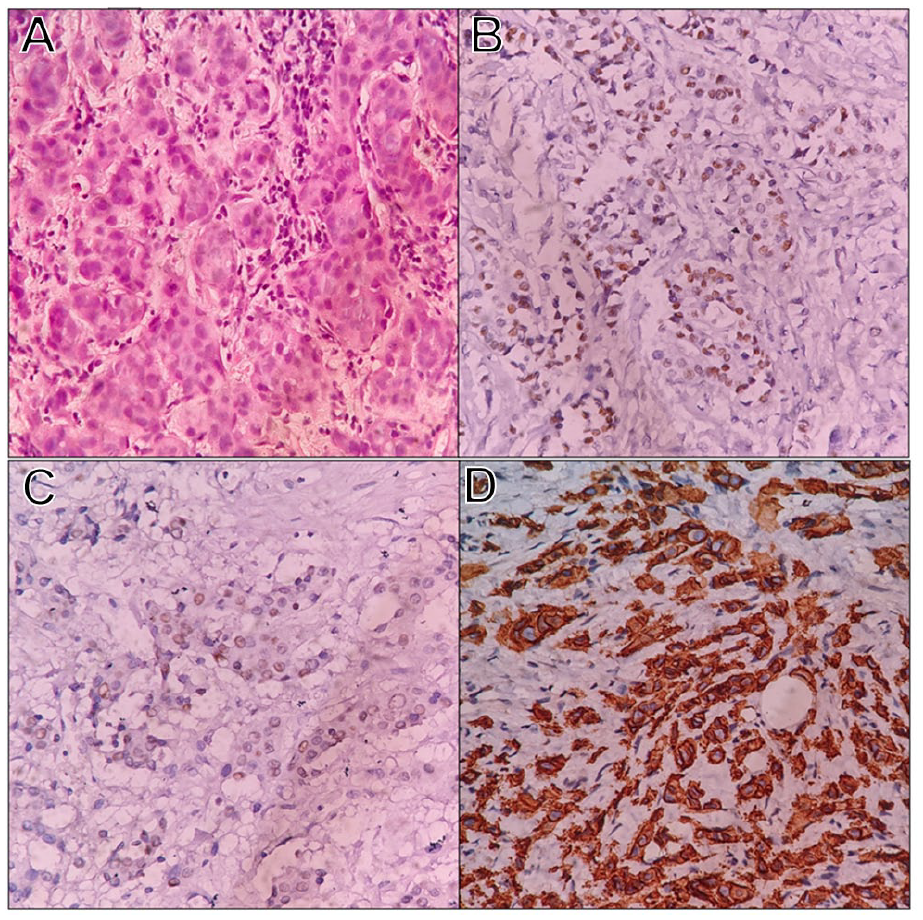
Invasive ductal carcinoma after neoadjuvant chemotherapy. (A) Residual tumor cells in nests (H&E ×400); (B)–(D) Tumor cells showing retained ER, PR & HER2 expression (immunohistochemistry ×400).

Invasive ductal carcinoma after neoadjuvant chemotherapy. (A) Residual tumor cells in cords and nests (H&E ×400). (B)–(D) Tumor cells showing loss of estrogen receptor, progesterone receptor, and HER2 expression (immunohistochemistry ×400).
Association of changes in ER, PR, and HER2 expression after NAC with baseline clinicopathological characteristics
Discordance in the ER, PR, and HER2 status was studied and compared with the other clinicopathological parameters using a chi-square or Fisher’s exact test. There was no statistically significant correlation between the studied baseline clinicopathological characteristics and changes in the ER, PR and HER2. The clinical stage and molecular subtype also had no significant correlation with the changes in ER, PR, and HER2. Tables 4 to 6 depict the results of these tests with p-values.
Association of estrogen receptor (ER) expression with other baseline characteristics.

Values expressed as frequencies followed by percentage and p-values generated by Fisher’s exact test.
Values expressed as frequencies followed by percentage and p-values generated by chi-square test.
Association of progesterone receptor (PR) expression with other baseline characteristics.

Values expressed as frequencies followed by percentage and p-values are generated by Fisher’s exact test.
Values expressed as frequencies followed by percentage and p-values are generated by chi-square test.
Association of HER2 expression with other baseline characteristics.

Values expressed as frequencies followed by percentage and P values are generated by Fisher’s exact test.
Values expressed as frequencies followed by percentage and P values are generated by chi-square test.
Association of changes in ER, PR, and HER2 expression with OS and DFS rates
Overall survival is defined as the time (in months) between the dates of diagnosis and last follow-up or death due to any cause. Disease-free survival is defined as the time (in months) from treatment until recurrence of disease (or death). The median OS time was 42 months. Changes in ER, PR, and HER2 were compared with the OS and DFS of patients using Kaplan-Meier survival graphs. These tests revealed a statistically significant correlation between changes in ER and HER2 with OS and DFS, whereas changes in PR status did not have a statistically significant effect on OS or DFS. Figures 6 and 7 indicate the significant correlation between changes in ER and HER2 with the DFS of patients respectively along with their p-values. Those patients with a switch in ER and HER2 from positive to negative had longer OS and DFS.

Survival curve depicting changes in estrogen receptor (ER) status with disease-free survival of patients (P < 0.001).

Survival curve depicting changes in HER2 status with disease-free survival of patients (P = 0.003).
Discussion
The prognostic factors in the current WHO classifications of breast tumors include HR status, HER2 status, and NAC regimen. Many studies have correlated these factors with the patients’ DFS and OS. It is well established that HR (+) patients who receive NAC have better survival, compared with those who are HR (–). Although multiple recent studies have attempted to decipher how changes in hormone status and HER2 status following NAC influence OS rates, few have examined the Indian setting. In this study, we assessed 174 patients being treated for invasive ductal carcinoma in southern India. In comparison, similar studies have included groups sizes ranging from 32 5 to 459 6 patients. We have further tried to identify changes in ER, PR, and HER2 status following NAC, as well as any associations of these changes with the DFS and OS rates. We also evaluated associations of HR and HER2 changes with relevant clinicopathological variables. Other factors may have also influenced OS and DFS. Further studies with larger sample sizes could provide a clearer understanding.
Baseline clinicopathological parameters (pre-NAC)
In our study population of women with invasive ductal carcinoma, the median age of participants was 50 (35-50), which is in accordance with the latest Indian statistics of breast cancer. All patients in our study were females. Most presented with locally advanced breast cancer (60.3%), likely due to a lack of social awareness and screening programs. 7 The most common histologic grade in our study was grade 2 (52.3%), similar to previous observations of 59% 8 and 40.8%. 9 In this study, 58.2% of cases were HR (+), 36.2% were triple-negative breast cancers, and 5.7% were HER2-enriched, similar to a study done by Parinyanitikul et al, 10 showing 49.1% of participants as having the HR (+) breast cancer type, 32.6% having the triple-negative type, and 18.3% having the HER2-enriched type.
Clinicopathological parameters post-NAC
In the current study, 88.6% of patients underwent modified radical mastectomy, and the remaining 10.9% underwent breast-conserving surgery following NAC. These findings align with that of Ozmen et al, 11 in which 87.8% underwent modified radical mastectomy and 12.2% underwent breast-conserving surgery. Most (85.1%) patients in our study received taxane- and anthracycline-based chemotherapy, similar to the 70.8% who received similar regimens in Parinyanitikul et al. 10 After NAC treatment, 48.3% of patients in the current study were staged as ypT2, and 50% had nodal positive status, which is in agreement with Al-Saleh et al, 12 who found 54% of cases were nodal positive. No other significant correlation between changes in HR or HER2 status with other clinicopathological parameters (eg, age, laterality, clinical stage) were observed. Studies by Hirata et al, 6 and Enomoto et al, 13 also found no statistically significant correlation between these variables.
HR receptor and HER2 status conversion rates post-NAC
Out of the 174 cases that received NAC, 77 (44%) ER-positive cases became negative, while 10 remained positive; 87 (50%) ER-negative cases stayed negative; 48 (27%) PR-positive cases turned negative, and 10 remained positive; 116 (67%) PR-negative cases continued to be negative; 64 (36%) HER2-positive cases became negative, and 4 (2%) stayed positive; and 103 (59%) HER2-negative cases remained negative. These conversion rates are higher than those observed in other studies. For instance, Chen et al, 14 reported conversion rates of 16% for ER, 22.2% for PR, and 15.2% for HER2; Hirata et al, 6 found conversion rates of 14.9% for ER, 29.1% for PR, and 22.6% for HER2; and Yang et al, 15 reported conversion rates of 13% for ER, 19.5% for PR, and 22.6% for HER2.
We observed conversion in at least one of the 3 markers in 36.6% of participants, concordant with an overall conversion rate of 40.7% observed in Parinyanitikul et al, 10 and of 23.1% in Al-Saleh et al, 12 Other studies suggest no significant changes in HR and HER2 status after NAC. Arens et al, 16 for example, found no significant change in any of the 3 hormonal markers after NAC, perhaps due to the lower number of patients, compared with studies reporting a change. Kasami et al, 17 also found no significant change in ER and HER2 status, whereas PR status changed in 36% of their cases; they focused on patients with advanced local disease with regional and distal metastases, and thus their results might be more representative of the response rate for NAC in advanced cases.
In our study, 6 (60%) cases with retained ER and PR expression were of histologic grade 3, and the remaining 4 (40%) were grade 2. In cases with retained HER2 expression, 3 (75%) were grade 2, and 1 (25%) was grade 3. We attributed these results to tumor heterogeneity.17,18 The mechanism of HR and HER2 status conversion after NAC is complex. Intertumoral heterogeneity can lead to multiple clones with distinct phenotypes within individual tumors. Some clones within the same tumor are HR (+), and others are HR (–). Similarly, HER2 (+) cells are not evenly distributed within individual tumors. Sensitivity to chemotherapy also varies between clones: HR (–) tumor cells are more sensitive to chemotherapy than are HR (+) tumor cells, and HR (+) cells, known as insensitive tumor cells, are left behind after NAC as part of the residual disease. Wang et al 18 and Thor et al 19 also have shown that HER2 (+) tumor cells are more likely to be eradicated by chemotherapy, and patients benefit from this.
One recognized mechanism for the conversion of HR status is the downregulation of the estrogen receptor after NAC. Bines et al 20 and Rose and Davis 21 suggest that chemotherapy can suppress ovarian and adrenal function, leading to reduced hormone levels. This decrease in hormones may cause the HR status of remaining tumors to shift from HR-positive (+) to HR-negative (–) after NAC. This change is believed to be the main cause of the HR status shift following chemotherapy. However, false-negative results for HR and HER2 status have been reported in pre-NAC Tru-cut biopsies due to intertumoral heterogeneity. The conversion of HR-negative to HR-positive and HER2-negative to HER2-positive may be related to the limited tumor material obtained in Tru-cut biopsies, which may only capture a small portion of the tumor with different phenotypic characteristics. Other factors contributing to receptor status conversion include genetic mutations, statistical errors, and variations in staining methods. 4
Our results corroborate those of other studies indicating a significant correlation between any change in the receptor status and DFS and OS rates in patients with breast cancer as seen in studies like Vemuru et al. 22 Unlike our findings, however, Tacca et al, 23 Yang et al, 15 Jin et al, 24 and Van de Ven et al, 25 all find that OS and DFS rates are better for patients whose HR status switches from negative to positive, compared with those who do not exhibit a switch after NAC. In the current study, patients who exhibited a change in ER and HER2 status from positive to negative showed better OS and DFS rates. As none of our participants switched from negative to positive HR or HER2, the survival of such patients could not be assessed. However, Tacca et al, 23 note that patients with HR (–) tumors who switched to a positive status after NAC had better OS and DFS rates than did patients whose tumors remained HR negative after NAC.
The cases with changes in ER and HER2 status from positive to negative showed longer DFS and OS rates, and the difference was statistically significant. Change in ER (P < 0.001) or HER2 (P < 0.003) status was significantly associated with OS rates. Changes in ER (P < 0.001) and HER2 (P < 0.004) status also were significantly associated with DFS. In contrast, no correlation was observed between changes in PR with OS (P = 0.604) or DFS (P = 0.605). These results contrast with those of Jin et al, 24 who observed that changes in PR status had a statistically significant correlation with OS and DFS rates, compared with changes in ER status.
In the current study, 7 (4%) of 174 patients experienced a relapse of their breast cancer: 4 (57%) with luminal subtype, 2 (29%) with HER2-enriched cancer, and 1 (14%) with triple-negative breast cancer. Additional, 4 (57%) of those 7 were classified as grade 3 and 3 (43%) as grade 2. We observed conversion among those with HR and HER2 status of the luminal and HER2-enriched subtypes but not among those with triple-negative breast cancer.
During the course of the study, 36 (20.7%) of 174 patients died, of which 21 (58%) had triple-negative breast cancer, 11 (31%) had luminal subtype, and 4 (11%) had HER2-enriched breast cancer. Additionally, 21 (58%) were grade 3, 14 (39%) were grade 2, and only 1 (3%) belonged to the grade 1 subtype. We also observed a conversion in at least one of the 3 markers in 23 (69%) of these 36 cases. However, as mentioned earlier, no statistically significant correlation was observed between the conversion in HR and HER2 status with histologic grade or molecular subtype, which is in agreement with other similar studies by Al-Saleh et al, 12 Yang et al, 15 and Uzun et al. 26
Limitations of our study
Limitations in our study mainly were due to lack of data. Of the 4 participants with a pre-NAC HER2 score of 2 + with no FISH reports available, all 4 cases were positive and all were classified as HER2 (–) based on the post-NAC resection specimen. Thus, other types of cancer were not included in our analysis. The menopausal status of patients also was not assessed due to lack of available data. There were no cases in our study with a positive switch in any of the 3 markers after NAC. Hence, the survival of such cases could not be assessed.
Conclusion
Our study assessed the effect of changes in ER, PR, and HER2 expression on breast cancer outcomes among patients in southern India with residual invasive ductal carcinoma who underwent modified radical mastectomy or breast-conserving surgery after NAC. We found changes in HR and HER2 status in these participants, mostly switching from positive to negative in all 3 markers studied. Patients with changes in the receptor status post-NAC had better OS and DFS prognoses than those whose receptor status did not change. Thus, we conclude that post-NAC there is a change in the ER and HER2 status. Patients’ ER and HER2 status may be suitable prognostic markers for managing cases of invasive ductal carcinoma with residual tumor post-NAC.
Footnotes
Ethical Considerations and consent to participate
Institutional Ethics Committee for Observational Studies’ approval was obtained-Project no. JIP/IEC/2021/134—A Wavier of consent was also obtained for the same. Consequently, it was not necessary to obtain consent from the patients.
Consent for publication
Consent for publication is obtained from all the co-authors and their respective departments
Author contributions
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by JIPMER Medical College, Puducherry, India—Grant no. JIP/Res/Intramural/sub com/2020-21.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data and materials supporting the findings of this study are available upon reasonable request from the corresponding author. All relevant data, including patient information and experimental protocols, can be accessed through institutional repositories or directly from the authors, subject to ethical and privacy considerations. Any additional materials, such as datasets or supplementary files, will be provided as required for peer review or further research purposes.
