Abstract
Approximately 10% to 15% of breast cancer cases in young women are diagnosed in patients harbouring germline (g) pathogenic or likely pathogenic variants (PVs) in the BReast CAncer 1 (BRCA1) or BReast CAncer 2 (BRCA2) genes. Preclinical and clinical studies showed a potential negative effect of germline BRCA1/2 (gBRCA1/2) PVs on ovarian reserve and reproductive potential, even before starting anticancer therapies. The aim of this article is to summarize the current literature on the fertility potential of young gBRCA1/2 PVs carriers with breast cancer and the risk of gonadotoxicity associated with anticancer treatments. Moreover, we describe the available evidence on the efficacy of fertility preservation techniques in young gBRCA1/2 PVs carriers and the safety data on having a pregnancy after breast cancer treatment.
Introduction
Approximately 10% to 15% of breast cancer cases in young women are hereditary conditions being diagnosed in patients harbouring germline (g) pathogenic or likely pathogenic variants (PVs) in the BReast CAncer 1 (BRCA1) or BReast CAncer 2 (BRCA2) genes.1,2 The prevalence of germline BRCA 1/2 (gBRCA1/2) PVs in the general population has been estimated to be 1:300 to 500 cases, but this can be higher in specific area of the world. 3 Harbouring gBRCA1 and/or gBRCA2 PVs is not only associated with an increased risk of breast cancer but also with an increased risk of ovarian cancer and other malignancies. 4 The lifetime risk of developing breast cancer in women with PV of the gBRCA1 and gBRCA2 gene is 72% and 69%, respectively. 4
The BRCA1 and BRCA2 genes belong to the family of ataxia-telangiectasia-mutated (ATM)-mediated deoxyribonucleic acid (DNA) repair genes that play a key role for the integrity of the chromosomes. 5 The BRCA genes are tumour suppressor genes, and their activity consists of repair double-stranded DNA breaks by homologous recombination. 6 Impaired DNA damage secondary to inappropriate repair of double-stranded breaks can lead to apoptosis of follicles, premature ovarian insufficiency (POI), and early menopause. 5 Preclinical evidence reported that BRCA1/2 PVs could lead to oocyte apoptosis causing accelerated ovarian aging.7,8 Notably, an indirect effect of carrying gBRCA1/2 PVs on fertility potential is represented by the indication from international guidelines to undergo Risk-Reducing Salpingo-Oophorectomy between the age of 35 and 40 years in gBRCA1 carriers or between the age of 40 and 45 years in gBRCA2 carriers as soon as family planning is completed. 9
The aim of this article is to summarize the current literature on the fertility potential of young gBRCA1/2 PVs carriers with breast cancer and the risk of gonadotoxicity associated with anticancer treatments. Moreover, we describe the available evidence on the efficacy of fertility preservation techniques in young gBRCA1/2 PVs carriers and the safety data on having a pregnancy after breast cancer treatment.
Impact of Carrying gBRCA1/2 PVs on Fertility Potential
The BRCA1 and BRCA2 genes play a crucial role in maintaining DNA integrity. They function as tumour suppressors, participating in DNA repair processes to prevent the accumulation of genetic defects. The BRCA genes are also involved in several steps of the female process of reproduction, including oocyte development. In fact, BRCA1 is highly expressed in human germ cells and in blastocysts. 10 Furthermore, a genetic analysis study showed that BRCA genes are also associated with age at menopause. 11 Several studies have shown that the presence of a PV in the BRCA genes (BRCA1 and/or BRCA2 PV) can accelerate ovarian aging. In addition, preclinical studies comparing between the specific BRCA gene have found that the effect of carrying a gBRCA2 PV on ovarian aging is less pronounced than the effect of carrying a gBRCA1 PV. 7
There are 2 main parameters used to estimate a woman’s individual ovarian reserve: the anti-Müllerian Hormone (AMH) and the antral follicular count (AFC). The AMH is a hormone that regulates the pool of primary follicles and its value correlates with the number of oocytes retrieved following ovarian stimulation. 12 It is known that the AMH level decreases constantly until the age of menopause, but its decrease could be hastened by different conditions including after the use of gonadotoxic treatments. 13
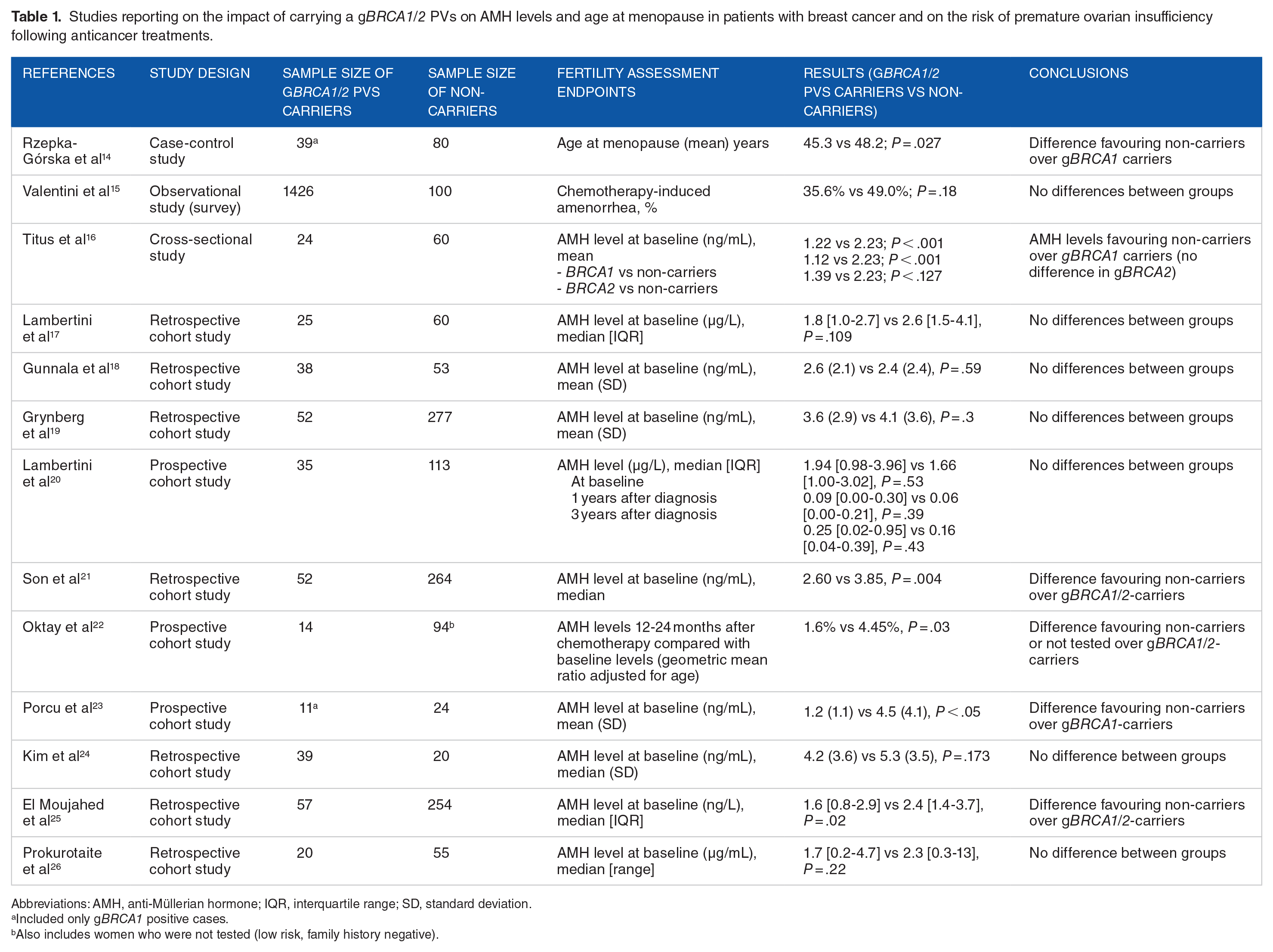
Many studies have investigated whether gBRCA1/2 PVs carriers with breast cancer have lower AMH and/or lower oocyte yield after controlled ovarian stimulation (COS), as well as a higher risk of chemotherapy-induced POI. However, results are conflicting (Tables 1 and 2).14-30
Studies reporting on the impact of carrying a gBRCA1/2 PVs on AMH levels and age at menopause in patients with breast cancer and on the risk of premature ovarian insufficiency following anticancer treatments.
Abbreviations: AMH, anti-Müllerian hormone; IQR, interquartile range; SD, standard deviation.
Included only gBRCA1 positive cases.
Also includes women who were not tested (low risk, family history negative).
Studies reporting on the impact of carrying a gBRCA1/2 PVs on oocyte yield following ovarian stimulation in patients with breast cancer.

Abbreviations: AMH, anti-Müllerian hormone; IQR, interquartile range; SD, standard deviation.
Defined as retrieval of ⩽4 oocytes in women younger than 38 years.
Also includes women who were not tested (low risk, family history negative).
Included only gBRCA1 positive cases.
Included 9 women who were positive for other gene mutations (ATM, CHK2, PALP 2, NF, MUTYH.c.536A, P53) but not gBRCA1/2.
Ovarian reserve and AMH levels
Several studies with conflicting results have investigated the association between gBRCA1/2 PVs carriers with breast cancer and AMH levels (Table 1).16-25 Some authors found lower AMH levels in gBRCA1/2 PVs carriers with breast cancer compared with young women with non-hereditary breast cancer,16,21-23,25 whereas other authors could not find any negative association.17-20,24 All these studies measured AMH level at the time point of cancer diagnosis (baseline AMH), whereas only 2 studies have assessed its values over time (ie, after gonadotoxic therapy and during follow-up).20,22 The retrospective cohort study of Son et al is one of the largest studies on this topic, including 52 gBRCA1/2 PVs carriers with breast cancer and 264 non-carriers with breast cancer. The authors found a difference in median baseline AMH value of 32% between gBRCA1/2 PVs carriers with breast cancer and non-carriers with breast cancer (2.60 ng/mL vs 3.85 ng/mL, P = .004). The linear regression analysis showed a significant negative association between the presence of gBRCA1/2 PVs and AMH levels (r = −0.30, P = .04). There was no significant difference in AMH levels between gBRCA1 and gBRCA2 carriers. 21 In the retrospective study by El Moujahed et al in patients with breast cancer candidates to receive anticancer therapy, there was a statistically significant difference in median AMH value between gBRCA1/2 PVs carriers and non-carriers (1.6 vs 2.4 ng/mL favouring non-carriers, P = .02). No difference in AMH level was observed between gBRCA1 PV carriers and gBRCA2 PV carriers (1.6 vs 2.4 ng/mL, P = .27). 25 In contrast, the study of Grynberg et al, evaluating 52 gBRCA1/2 PVs carriers with breast cancer and 277 non-carriers with breast cancer, found no difference between the mean AMH levels of gBRCA1/2 PVs carriers and non-carriers (3.6 ng/mL vs 4.1 ng/mL, P = .3). 19
In 2021, Turan et al published an individual patient-level data meta-analysis including 4 studies comparing serum AMH levels in gBRCA1/2 PVs carriers with breast cancer (n = 157) vs non-carriers (n = 524). After adjusting for the effect of centre, age, body mass index, smoking, and oral contraceptive pill use, gBRCA1/2 PVs carriers with breast cancer had significantly lower AMH levels compared with non-carriers (25% lower; 95% confidence interval [CI] = 9 to 38; P = .003). In a subgroup analysis according to the specific BRCA gene, a statistically significant difference in serum AMH levels was found between non-carriers and gBRCA1 PV carriers (32% lower, 95% CI = 14 to 46, P = .001) but not with gBRCA2 PV carriers (14% lower, 95% CI = 34 to −12 higher, P = .25). 31
In summary, given the controversial results on this topic, it remains unclear to date if gBRCA1/2 PVs carriers with breast cancer have a lower ovarian reserve in serum AMH level. However, it cannot be excluded that gBRCA1/2 PVs carriers with breast cancer may have a reduced ovarian reserve when compared with non-carriers. Large multicenter prospective studies are needed to clarify the impact of gBRCA1/2 PVs on baseline ovarian reserve and the potential different effect of gBRCA1 vs gBRCA2 PVs in this regard.
Premature Ovarian Insufficiency with Anticancer Treatments in Patients Harbouring gBRCA1/2 PVs
It is well established that anticancer therapy can lead to POI and subsequent subfertility/infertility. The commonly used chemotherapy regimens in breast cancer treatment include cyclophosphamide, anthracyclines and taxanes which are well-known agents associated with POI risk.32,33
The risk of developing treatment-induced POI depends on the type, dose and number of cycles of chemotherapy used as well on the age of the patients. However, there is no absolute threshold of exposure to anticancer therapies that determines gonadal failure and infertility, so every patient receiving systemic anticancer therapy should be considered at potential risk of developing treatment-related gonadotoxicity. 33 Moreover, considering the reduced capacity to repair DNA damage in gBRCA1/2 PVs carriers, it could be hypothesized that in such groups of patients, the gonadotoxic impact of anticancer treatments may be even more accentuated.
A recent study by Oktay et al showed a statistically significant increased chemotherapy-induced ovarian reserve loss in gBRCA1/2 PVs carriers as compared with non-carriers. In this study, gBRCA1/2 carriers with breast cancer lost significantly larger ovarian reserve (reported as geometric mean AMH level) than the non-carriers (P = .03). 22 A more recent work published by the same group found similar results. They investigated the risk of amenorrhea in patients carrying gBRCA1/2 PVs treated with chemotherapy. Of 102 patients included, 12 carried gBRCA1/2 PVs. More than 90% of the patients received anthracycline-cyclophosphamide–based chemotherapy followed by a taxane. Amenorrhea was defined as the absence of vaginal bleeding for 4 consecutive menstrual cycles at 6, 12, and 18 months after the end of chemotherapy. Multivariable-adjusted logistic regression analysis demonstrated that the presence of gBRCA1/2 PVs was a significant predictor of amenorrhea at 18 months (P = .03). The author concluded that, despite the small simple size of gBRCA1/2 carriers with breast cancer included in the study, gBRCA1/2 PVs carriers may have a higher risk of chemotherapy-induced amenorrhea. 34
In another retrospective study, Lambertini et al evaluated the serum AMH level of 35 gBRCA1/2 PVs carriers with breast cancer at diagnosis as well as 1 year and 3 years afterwards. All patients received chemotherapy as part of breast cancer treatments. Chemotherapy regimens consisted of either 6 cycles of fluorouracil, epirubicin, and cyclophosphamide (FEC) or 3 cycles of FEC followed by 3 cycles of docetaxel. No differences in AMH levels were observed between gBRCA1/2 PVs carriers and non-carriers either at diagnosis nor 1 or 3 years afterwards. 20
Valentini et al conducted an observational study to evaluate chemotherapy-induced amenorrhea in young patients with breast cancer. Of 1954 premenopausal gBRCA1/2 PVs carriers with breast cancer, 1426 received chemotherapy. Data were collected through a survey. Treatment-induced POI was defined as amenorrhea for ⩾2 years commencing within 2 years from the initiation of chemotherapy and without resumption of menstrual function. There was no significant difference in chemotherapy-induced amenorrhea between gBRCA1/2 PVs carriers and non-carriers (35.6% vs 49%, P = .18). 15
In conclusion, the results of the studies analysing risk of treatment-induced POI are conflicting. Larger studies including also currently adopted therapies such as platinum agents and/or poly ADP ribose polymerase (PARP) inhibitors are needed to shed light on this issue. 35
Strategies for Fertility Preservation in Patients Harbouring gBRCA1/2 PVs
Taking into account the risk of treatment-induced gonadotoxicity in patients with breast cancer, the role of fertility preservation is critical. There are 3 main possibilities of fertility and/or ovarian function preservation in young patients including those with breast cancer harbouring gBRCA1/2 PVs. Considering specific gBRCA1/2-related issues, special counselling is required in this setting.
Oocyte and/or embryo cryopreservation
Oocyte and/or embryo cryopreservation are widely adopted strategies for fertility preservation in patients with cancer.
The strategy consists of performing COS, based on injections of follicle-stimulating hormone (FSH) with/without luteinizing hormone (LH). At the end of the hormonal stimulation phase, the mature oocytes are collected and either directly frozen or fertilized to become embryos to be frozen afterwards. Controlled ovarian stimulation leads to increased oestradiol (E2) levels in serum due to hormonal manipulation; this was historically a concern in patients affected by breast cancer. In 2005, Oktay et al reported the results of a COS protocol that included the inclusion of an aromatase inhibitor (ie, letrozole) in conjunction with gonadotropins. Letrozole inhibits the aromatase enzyme that is responsible for converting androgens into E2. With this protocol, peak E2 levels were similar to those detected in unstimulated cycles. After a follow-up time of 24 months, the breast cancer recurrence rate was similar between patients who underwent ovarian stimulation and those who did not. 36 These results were later confirmed by other authors.37-40 As shown in a large meta-analysis, concomitant use of letrozole as part of COS reduced circulating E2 levels without impairing the efficacy of ovarian stimulation. 38 Another more recent meta-analysis has also shown that performing COS before anticancer treatments in young women with breast cancer did not seem to be associated with detrimental prognostic effect in breast cancer recurrence or mortality. 40
The study by Moravek and colleagues showed no difference in recurrence and survival rates between patients with breast cancer who underwent COS for oocyte/embryo cryopreservation without letrozole (n = 150) and those who did not (n = 160). 41
There is less available literature on the topic concerning patients with gBRCA1/2 PVs breast cancer. 37 In a prospective single-centre study, Kim et al compared the relapse free-survival rate between 120 breast cancer patients undergoing COS and 217 who did not undergo any fertility preservation procedure. After a mean follow-up of 5 years, no difference in relapse free-survival was found between 120 breast cancer patients who underwent COS and 217 who did not undergo any fertility-preserving procedure (hazard ratio [HR] = 0.77, 95% CI = 0.28 to 2.13; P = .61). Among the 54 gBRCA1/2 PVs carriers with breast cancer included in the analysis, 29 underwent COS whereas the other 25 did not receive any fertility-preserving procedure. Even in the gBRCA1/2 PVs population, no significant difference in relapse-free survival was observed between the 2 groups (P = .57). 37 In another study, 31 of the 123 patients who underwent fertility preservation were gBRCA1/2 PVs carriers. The overall survival rate was similar among women with breast cancer who underwent fertility preservation and women with breast cancer who declined to perform any procedure (3 years overall survival: 95.5% vs 93.5%, P = .85; 5 years overall survival: 84.2% vs 81.4%, P = .7). However, no subgroup analysis in the specific cohort of gBRCA1/2 PVs carriers with breast cancer was performed. 42
Multiple studies compared oocyte yield after COS between patients with breast cancer carrying or not gBRCA1/2 PVs (Table 2).17-19,23-30 The studies are heterogeneous and reported inconsistent results; importantly, most of them were single-centre retrospective studies including a very small number of gBRCA1/2 PVs carriers with breast cancer. Lambertini et al reported a trend for lower number of oocytes (6.5 vs 9.0, P = .145) and cryopreserved oocytes (3.5 vs 6.0, P = .121) in gBRCA1/2 PVs carriers compared with non-carriers. In addition, a low response rate (defined as retrieval of ⩽4 oocytes in women younger than 38 years) occurred more frequently in gBRCA1/2 PVs carriers (40.0% vs 11.1%, P = .147) after COS, and these patients required higher doses of FSH (2775 vs 2025 IU, P = .085) and longer stimulation (11.5 vs 9.0 days, P = .110). The authors claimed that although these differences could have a meaningful clinical impact, they were not statistically significantly different. 17 Grynberg et al found no difference in the number of collected oocytes after COS in gBRCA1/2 PVs carriers with breast cancer over non-carriers (8.9 [±6.9] vs 9.9 [±8.1], P = .5). The study of Arab et al found similar results: mean number of collected oocytes was 13.32 (±8.63) vs 11.72 (±8.63), P = .34 in gBRCA1/2 PVs carriers vs non-carriers. In contrast, Porcu et al found a statistically significant difference in the number of collected oocytes after COS favouring non-carriers over gBRCA1/2 PVs carriers with breast cancer (mean number of collected oocytes: 7.2 [±4.4] vs 3.1 [±2.3], P < .05). Another study in line with these results showed a statistically significant difference in the number of collected oocytes after COS favouring non-carriers over gBRCA1/2 PVs carriers with breast cancer (median number of collected oocytes): 8.7 (±7.9) vs 4.7 (±4.2), P = .02. 24 Similarly, another study showed a statistically significant difference in the number of collected mature oocytes after COS between gBRCA1/2 PVs carriers with breast cancer and non-carriers with breast cancer (median number of collected mature oocytes 7.0 vs 9.0, P = .04). 25
Overall, limited and conflicting data are available to date in the specific population of young breast cancer patients harbouring gBRCA1/2 PVs. Even though gBRCA1/2 PVs carriers with breast cancer may have a reduced performance in of oocyte yield after COS, oocyte/embryo cryopreservation is the first fertility preservation strategy to be discussed also for gBRCA1/2 PVs carriers. Notably, this strategy also allows the access to pre-implantation genetic testing (PGT) for those who are interested. 43
Cryopreservation of ovarian tissue
Cryopreservation of ovarian tissue is an effective fertility preservation procedure for those patients receiving cytotoxic therapies that are not eligible to undergo oocyte/embryo cryopreservation. 32 The indications for this procedure are mainly: (1) patients who are prepubertal (not applying to breast cancer), (2) adults who cannot delay their gonadotoxic therapies for at least 2 weeks, and (3) any other contraindication to COS. 32
One of the advantages of this procedure is the preservation of endocrine ovarian function. However, the success of this technique is highly dependent on the ovarian reserve of the patient at the time of the procedure as well as on the expertise of the centres and operators involved in the cryopreservation technique.
In breast cancer patients harbouring gBRCA1/2 PVs, extremely limited data are available about ovarian tissue cryopreservation. Considering the high risk of ovarian cancer in these patients, it should not be considered in the first choice for fertility preservation but should be offered only in very selected cases. 43 Importantly, after the desired pregnancy has been achieved or when family planning is concluded, the transplanted ovarian tissue should be completely removed. 4
One study reported a live birth after transplantation of ovarian tissue in a gBRCA2 PVs carrier. 44 In this case, the ovary was cryopreserved prior to chemotherapy and the ovary was transplanted orthotopically after the end of the breast cancer treatments. Lambertini et al reported on 2 other women, one with gBRCA1 PV and the other one with gBRCA2 PV, who cryopreserved ovarian tissue prior to gonadotoxic therapy. One conceived spontaneously after transplantation of the tissue and gave birth to a healthy baby boy. The other patient carrying a gBRCA1 PVs performed an In-Vitro-Fertilization and PGT after transplantation of the ovarian tissue. The embryo was affected by gBRCA1 PVs, and the patients decided not to proceed further with conception projects. 17 To our knowledge, 2 children have been born so far after ovarian tissue transplantation in gBRCA1/2 PVs carriers with breast cancer.
For gBRCA1/2 PVs carriers with breast cancer, cryopreservation of ovarian tissue should only be considered for very young patients who cannot pursue oocyte and/or embryo cryopreservation. It should be noted that most women who undergo ovarian tissue cryopreservation do not know their gBRCA1/2 status at the time of diagnosis during oncofertility counselling. However, BRCA status should be known before attempting any transplantation procedure.
Ovarian function suppression with a gonadotropin-releasing hormone agonist (GnRHa) during chemotherapy
The use of a GnRHa during chemotherapy is a standard strategy to preserve ovarian function during chemotherapy, with most of the evidence available in the setting of breast cancer. The GnRHa reversibly suppresses ovarian function by primarily blocking the pituitary gland. Pituitary desensitization, by causing a GnRH receptor down-regulation, drastically reduces the secretion of FSH and LH, inducing a state of hypogonadotropic hypogonadal anovulation, sometimes referred to as ‘pseudomenopause’. 45 Once the long acting application is stopped, the ovarian function, if not irreversibly damaged by gonadotoxic therapies, is expected to return.
The real benefit of GnRHa during chemotherapy in breast cancer patients has been debated for some time in the field of oncofertility.46-48 Nevertheless, nowadays, the efficacy of this approach in breast cancer patients has been confirmed by many randomized studies.49,50 A meta-analysis including 12 randomized trials showed that use of GnRHa during chemotherapy is associated with a reduction in the risk of POI (odds ratio = 0.36; 95% CI = 0.23 to 0.57; P < .001) and increase in the probability of having a pregnancy (odds ratio = 1.83; 95% CI = 1.02 to 3.28; P = .041). No negative consequences on patients’ prognosis were observed in the 3 trials reporting on disease-free survival (HR = 1.0; 95% CI = 0.49 to 2.04, P = .939). 51
Given these results, current guidelines support the use of temporary ovarian function suppression during chemotherapy as a strategy for ovarian function preservation.33,52,53 The evidence on the efficacy of this technique in gBRCA1/2 PVs carriers with breast cancer is limited. In the final analysis of the PROMISE-GIM6 trial at a median follow-up 12.4 years, the authors reported data on 10 patients with gBRCA1/2 PVs breast cancer. 54 Among them, 4 gBRCA1/2 PVs carriers were included in the GnRHa arm (chemotherapy combined with the GnRHa triptorelin) and 6 gBRCA1/2 PVs carriers in the control arm (chemotherapy alone). Among gBRCA1/2 PVs carriers with breast cancer, incidence of chemotherapy-induced POI was 0% and 33% in the GnRHa and control arms, respectively. These results are in the same line of the overall trial findings; however, the very small sample size for this specific analysis does not allow deriving strong conclusion on the efficacy of this strategy in the specific cohort of patients with gBRCA1/2 PVs.
Overall, considering the safety and efficacy of the procedure in the general breast cancer population, the wide availability and the low cost, it is reasonable to offer this option also to young gBRCA1/2 PVs carriers receiving chemotherapy some time before undergoing risk-reducing gynaecological surgery. However, patients should be informed about the limited available data in this setting and about the side effects of ovarian function suppression.55,56
Pregnancy After Breast Cancer Diagnosis and Treatment in Patients Harbouring gBRCA1/2 PVs
In recent years, an increasing number of data have provided evidence on the safety of pregnancy after proper treatment and follow-up for breast cancer, including for patients with hormone receptor-positive disease.57-59
To date, only 2 studies reported the prognostic impact of pregnancy in gBRCA1/2 PVs breast cancer survivors.60,61 The study of Valentini et al included 128 gBRCA1/2 PVs carriers with a pregnancy at the time or after breast cancer diagnosis and 269 matched non-pregnant gBRCA1/2-PVs survivors, who served as a control group. Among the gBRCA1/2 PVs with a pregnancy, 53 had a pregnancy after breast cancer and 75 were diagnosed with breast cancer during pregnancy. No difference in breast cancer–specific mortality was observed between patients with a pregnancy and the control group (adjusted HR = 0.76; 95% [CI] = 0.31 to 1.91; P = .56). Even when considering only patients with a pregnancy following breast cancer, similar results were obtained (adjusted HR = 0.73; 95% [CI] = 0.21 to 2.68, P = .64). 60
A second international, multicentre, retrospective cohort study investigated specifically the safety of pregnancy in patients with gBRCA1/2 PVs and a history of invasive early breast cancer diagnosed at age ⩽40 years. Of the 1252 gBRCA1/2 PVs carriers included, 195 had a pregnancy after breast cancer, resulting in a pregnancy rate at 10 years of 19% (95% CI = 17% to 22%). No difference in disease-free survival was observed between the patients with or without a subsequent pregnancy (adjusted HR = 0.87; 95% CI = 0.61 to 1.23; P = .41). 61 In 2024, an update of the previous analysis has included a biggest cohort of gBRCA1/2 PVs carriers with breast cancer. Of 4732 gBRCA1/2 PVs survivors, 659 women had at least 1 pregnancy after breast cancer. The cumulative incidence of pregnancy at 10 years was 22% (95% CI = 21% to 24%), with a median time from diagnosis of breast cancer to pregnancy of 3.5 years. After a median follow-up of 7.8 years, there was no difference in disease-free survival between patients who conceived and those who did not (adjusted HR = 0.99; 95% CI = 0.81 to 1.20, P = .90). Furthermore, the occurrence of pregnancy was associated with a lower rate of breast cancer–specific survival events (adjusted HR = 0.60; 95% CI = 0.40 to 0.88, P = .009) and of death due to any cause (adjusted HR = 0.58; 95% CI = 0.40 to 0.85, P = .005). 62
By pooling together the results of these 2 studies, no worse overall survival outcomes in gBRCA1/2-PVs carriers with a subsequent pregnancy were observed (HR = 0.56; 95% CI = 0.45 to 0.68) as compared with those without. 58
In conclusion, according to the growing literature on the topic, pregnancy after breast cancer appears to be safe also in gBRCA1/2 PVs carriers.
Discussion
This review summarizes the current literature on the topic of fertility and pregnancy-related issues in young women with breast cancer and harbouring gBRCA1/2 PVs. The BRCA genes proved to participate in a multitude of key cellular functions, including repair of DNA. Pathogenic variants in BRCA1/2 genes can lead to an inefficient repairing of damaged DNA. The accelerated decrease of reproductive potential can be caused by an inefficient DNA repair and increased accumulation of DNA breaks within oocytes. This can lead to reduced ovarian reserve and increased sensitivity to the toxicity of chemotherapy. However, to date, despite the biological rationale and preclinical evidence, results of different studies are discordant and it remains unclear whether gBRCA1/2 PVs carriers with breast cancer have a lower reproductive potential or fertility preservation strategies are less effective. There may be a difference between gBRCA1 and gBRCA2 PVs regarding the impact on reproductive outcomes but further studies are needed to have more precise answers in this regard.
Oocyte or embryo cryopreservation should be considered as the first fertility preservation option also in gBRCA1/2 PVs carriers with breast cancer. Ovarian tissue cryopreservation should be considered only in motivated patients who have no access to oocyte and embryo cryopreservation or for those who have contraindication to oocyte or embryo cryopreservation and are diagnosed years before the recommended age of risk-reducing surgery. Oocyte/embryo cryopreservation also allows the possibility of avoiding transmitting the PV to offspring by accessing PGT. 63
Increasingly reassuring data exist on the safety of pregnancy after cancer diagnosis and treatment also in patients carrying gBRCA1/2 PVs.
Conclusions
In patients with breast cancer and carrying gBRCA1/2 PVs, a proper oncofertility counselling is indicated and should include discussion regarding the potential impact of the genetic defect on their reproductive system (Figure 1). So far, it remains inconclusive if gBRCA1/2 PVs carriers with breast cancer have a reduced reproductive potential compared with non-carriers, and if carrying a gBRCA1/2 PVs has an additional impact of anticancer therapies, particularly for the recently approved targeted therapies. The results of current studies are controversial and derive mostly from studies with a small simple size. Importantly, considering that gBRCA1/2 PVs carriers face unique reproductive challenges, results of breast cancer studies should not be generalized to this special patient population. Further prospective multicenter research efforts are needed to provide more solid answer to the oncofertility counselling of patients with breast cancer and carrying gBRCA1/2 PVs.

Fertility issues in young women with breast cancer and carrying gBRCA1/2 PVs.
Declarations
Footnotes
Acknowledgements
Not applicable.
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors consent for publication.
Author contributions
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partly supported by the Italian Association for Cancer Research Italian Association for Cancer Research (‘Associazione Italiana per la Ricerca sul Cancro’, AIRC; MFAG 2020 ID 24698).
Competing interests:
Professor ML reports advisory role for Roche, Lilly, Novartis, AstraZeneca, Pfizer, Seagen, Gilead, MSD, Exact Sciences, Pierre Fabre, and Menarini; speaker honoraria from Roche, Lilly, Novartis, Pfizer, Sandoz, Libbs, Daiichi Sankyo, Takeda, Gilead, and Menarini; and travel Grants from Gilead, Daiichi Sankyo, and Roche; and research funding (to the Institution) from Gilead all outside the submitted work. Dr GB reports advisory role for Novartis, AstraZeneca, Daiichi Sankyo, and Menarini Stemline and speaker honoraria from Lilly, Novartis, Pfizer, Daiichi Sankyo, AstraZeneca, Gilead, and Istituto Gentili all outside the submitted work. Dr LT reports advisory role, role as honoraria speaker, and travel grants from AstraZeneca, all outside the submitted work. The other authors declare that there is no conflict of interest.
Availability of data and materials
Not applicable.
