Abstract
Background:
Over the last few decades, tremendous progress has been achieved in the early detection and treatment of breast cancer (BC). However, the prognosis remains unsatisfactory, and the underlying processes of carcinogenesis are still unclear. The purpose of this research was to find out the relationship between myocardial infarction–associated transcript (MIAT), FOXO3a, and miRNA29a-3p and evaluated the expression levels in patients compare with control and their potential as a noninvasive bioindicator in whole blood in BC.
Methods:
Whole blood and BC tissue are taken from patients before radiotherapy and chemotherapy. Total RNA was extracted from BC tissue and whole blood to synthesize complementary DNA (cDNA). The expression of MIAT, FOXO3a, and miRNA29a-3p was analyzed by the quantitative reverse transcription-polymerase chain reaction (RT-qPCR) method and the sensitivity and specificity of them were determined by the receiver operating characteristic (ROC) curve. Bioinformatics analysis was used to understand the connections between MIAT, FOXO3a, and miRNA29a-3p in human BC to develop a ceRNA (competitive endogenous RNA) network.
Results:
We identified that in ductal carcinoma BC tissue and whole blood, MIAT and FOXO3a were more highly expressed, whereas miRNA29a-3p was lower compared with those in nontumor samples. There was a positive correlation between the expression levels of MIAT, FOXO3a, and miRNA29a-3p in BC tissues and whole blood. Our results also proposed miRNA29a-3p as a common target between MIAT and FOXO3a, and we showed them as a ceRNA network.
Conclusions:
This is the first study that indicates MIAT, FOXO3a, and miRNA29a-3p as a ceRNA network, and their expression was analyzed in both BC tissue and whole blood. As a preliminary assessment, our findings indicate that combined levels of MIAT, FOXO3a, and miR29a-3p may be considered as potential diagnostic bioindicator for BC.
Introduction
With an estimated 2.3 million new cases of breast cancer (BC) annually (11.7%), it surpassed lung cancer as the most commonly diagnosed cancer in 2020. 1 Both BC incidence and mortality rates are increasing among Iranian women. 2 Iranian women develop this disease at least a decade sooner than women in developed countries and are usually diagnosed with this disease at the age of 40 to 49 years. 3 Overcoming chemoresistance and distant-site metastasis is critical for improving outcomes in BC patients, as these factors continue to pose significant challenges to effective management and treatment of the disease. 4 Despite advances in treatment options, metastatic disease relapses persist and are a leading cause of patient mortality. 5 As a result, increased insight into the molecular mechanisms involved in BC progression could lead to the discovery of more accurate prognostic biomarkers and the development of targeted therapies with enhanced efficacy.
Breast cancer prognosis can be assessed based on classical clinicopathological features including cancer tumor size, histological subtype and grade, lymph node metastases, and lymphovascular invasion, all of which necessitate thorough histological analysis. 6 Although they can provide valuable prognostic information for some BC patients, their utility is limited by their low prognostic capacity and may not be applicable to all individuals. 7 Biopsy-based cancer detection can be uncomfortable, hazardous, costly, time-intensive, and reliant on pathologist proficiency. As such, alternative approaches are noninvasive, pain-free, easy to collect, and potentially cost-effective are gaining increasing attention. 8 Biomarkers are measurable signals that can identify malignancy or provide insight into tumor behavior, prognosis, or response to treatment. 9 The diagnosis of BC relies primarily on imaging, pathology, and serological markers. 10 In spite of their advantages, they do have limitations, including invasiveness, inconvenience, high costs, and a high rate of false-positive results. Mammography is widely accepted as the most reliable method for diagnosing BC, but it has some limitations, such as the harmful effects of ionizing radiation and low sensitivity in detecting early-stage cancer. Although a needle biopsy or surgical biopsy is commonly used to confirm BC, it is not needed for most benign tumors, and the procedure can be invasive and uncomfortable. The circulating tumor biomarker-based method is a promising alternative to the methods mentioned earlier, as it is simple, convenient, and cost-effective for detecting BC at an early stage and predicting its progression or recurrence.11 -13 Therefore, further research into the molecular etiology of BC is required to identify novel biomarkers and therapeutic targets.
Apoptosis is critical for maintaining a balance between cell death and cell division; thus, deregulation of this pathway leads to uncontrolled cell proliferation, which is involved in many different diseases, such as cancer. 14 The intrinsic pathway of apoptosis is regulated by various factors, such as the Bcl-2 family and the PI3K/AKT pathway. 15 Numerous studies have shown that the PI3K/AKT pathway is involved in the progression of a variety of tumors through regulating cellular growth and blocking apoptosis. 16 It has been reported that AKT interferes with cell death pathways through the phosphorylation of FOXO3a.17,18 FOXO3a, a transcription factor of the Forkhead box O (FOXO) family, possesses a vital role in regulating several cellular processes, such as proliferation, apoptosis, DNA damage, and cell-cycle progression. 19 Notably, in the PI3K/AKT pathway, activation of FOXO3a induces apoptosis, cell-cycle arrest, and stress resistance in most tissues, whereas FOXO3a inactivation triggers cell survival, proliferation, and stress sensitivity. 20 In BC, the deregulation of the PI3K/AKT pathway and its increased activity are associated with reduced diagnosis in patients. 21
Long noncoding RNAs (lncRNAs) are a sort of noncoding RNA (ncRNA) that are described as transcripts with a length of more than 200 nucleotides. 22 Recent research has demonstrated that lncRNAs are critical regulators of a variety of biological processes, including innate immunologic responses, genetic expression regulation, post-transcriptional processes, proliferation, invasion, metastasis, and angiogenesis of cancer cells. 23 Accordingly, there is significant evidence suggesting lncRNAs function as competing endogenous RNAs (ceRNAs) to restrict microRNA (miRNA) expression or activity. Thus, lncRNA has the potential to be a sensitive cancer diagnostic biomarker. 24 Myocardial infarction–associated transcript (MIAT) is involved in the control of cancer cell apoptosis, cell-cycle regulation, migration, and invasion. 25 In BC cells, knocking down MIAT inhibited cell growth and promoted apoptosis. It has been reported that, because of the strong invasive and metastasis ability of MIAT, it has increased in BC cell lines than normal breast cell lines. Moreover, proliferation, migration, invasion, and epithelial-to-mesenchymal transition (EMT) of BC cells were inhibited by the knockdown of MIAT, whereas the rate of apoptosis was promoted. In a xenograft model, the lowest expression of MIAT was associated with decreasing tumor growth and delaying tumor formation, thus indicating MIAT acts as an oncogene. 26
MicroRNAs are classified as noncoding mRNAs with approximately 22 nucleotides in length. Recently, a growing body of research has established that miRNAs exclusively attach to the 3’-untranslated region (3’-UTR) of messenger RNA; hence, gene expression is controlled at the post-transcriptional level. 27 Therefore, changing microRNA expression levels are involved in the onset and progression of a variety of diseases, such as cancer. 28 miRNA-29a-3p has a critical role in different biological processes such as proliferation, apoptosis, and cell-cycle regulation. It has been reported that miRNA-29a-3p has a significant impact on cancer development by acting as tumor suppressors. 29
In addition, miRNA-29a-3p expression levels have been discovered to be abnormally low in a variety of human malignancies, including papillary thyroid carcinoma (PTC), hepatocellular carcinoma, and gastric cancer. 30 Previous research found that miRNA-29a-3p is involved in the progression of BC. Moreover, circRNA ACAP2 (circACAP2) increases BC metastasis and proliferation via sponging miRNA-29a-3p. 31
The purpose of the study was to evaluate the expression levels of MIAT, FOXO3a, and miRNA-29a-3p as a ceRNA network in the BC tissues compared with the whole blood of BC patients, investigate their relationship with the clinical features of the tumor, and examine their potential as noninvasive biomarkers in BC.
Materials and Methods
Bioinformatics analysis
Protein-protein interaction analysis
The STRING server (https://string-db.org/) is used to investigate protein-protein interaction networks. We searched for FOXO3a in the STRING database and found the top 10 proteins that are related to FOXO3a. Then, we sorted out these connections based on text mining, experiments, and databases.
Analysis of STRING enrichment
The Gene Ontology (GO) is divided into 3 categories: biological process (BP), molecular function (MF), and cellular component (CC). Along with GO, the Kyoto Encyclopedia of Genes and Genomes (KEGG) is a valuable resource for studying biological pathways. Thus, we used the KEGG and the databases to investigate functions and pathways to determine their biological meaning.
In this study, after finding the protein-protein interaction of FOXO3a, GO enrichment and KEGG pathway related to FOXO3a protein-protein network from the STRING database were downloaded. They were categorized based on adjusted P value, and the top 10 were chosen. Enrichment analysis of FOXO3a was visualized in R 4.0.5 software (https://www.r-project.org/) based on the ggplot2 package (https://cran.r-project.org/web/packages/ggplot2/index.html).
The interaction between MIAT, FOXO3a, and miRNA29a-3p
The interaction between MIAT, FOXO3a, and miRNA29a-3p was determined using TargetScan (http://www.targetscan.org/) and the Starbase database (https://starbase.sysu.edu.cn/).
Construction of the competitive endogenous RNA network
We predicted the miRNAs that interacted with MIAT and FOXO3a using the miRNet (https://www.mirnet.ca/) and the miRDB (http://mirdb.org/) databases, respectively. Using miRNet, 46 miRNAs were found for MIAT, and using miRDB, 90 miRNAs based on target score were found for FOXO3a.
We combined MIAT-miRNAs and FOXO3a-miRNAs and applied them to Cytoscape software (v3.0.9) (https://cytoscape.org/), and then the ceRNA network was constructed based on the results.
Patients and sample collection
A total of 120 samples were taken from the Imam Khomeini Hospital (Tehran, Iran) among which 70 samples were collected from BC patients (35 blood samples, 35 tissue samples), and 50 cases were collected from healthy women (25 blood samples, 25 normal breast tissue). The mean age of the patients was 47 ± 3.1 years. All patients had neither chemotherapy nor radiotherapy treatment before surgery. The healthy individuals with no family history of any cancers and diseases and no history of alcohol consumption were selected. Also, their age (mean age 45 ± 5.4 years) and sex were consistent with the patient samples. The pathological features of patients were indicated in Supplementary Table 1.
RNA extraction
Total RNA was isolated from whole blood samples of BC patients and healthy controls according to the manufacturer instructions, using Trizol (Cat YT9066-YT9065-YT9064, Yekta Tajhiz Azma, Iran). The xylene-ethanol technique was used to eliminate paraffin from formalin-fixed, paraffin-embedded (FFPE) tissue, and afterward, overall RNA was extracted using Trizol Reagent (Cat YT9066-YT9065-YT9064, Yekta Tajhiz Azma) according to the manufacturer’s instructions. RNA quantification and the 260/280 nm ratio were evaluated by NanoDrop NP80 (Implen, Germany). Subsequently, total RNA was treated by RNase-free DNase (Cat MO5401, SinnaClon, Iran) to eliminated genomic DNA.
Quantitative reverse transcription-polymerase chain reaction
High-capacity complementary DNA (cDNA) reverse transcription kit (Cat RP1400, Smobio, Taiwan) was used for cDNA synthesis. Specific primers were designed for MIAT, glyceraldehyde 3-phosphate dehydrogenase (GAPDH) using oligo primer analysis software (version 0.7.0), a specific primer for FOXO3a was used from primer bank Harvard (Gen Bank Accession: NM_001455), and stem-loop RT primer was used for the reverse transcription of miRNA29a-3p and is taken from Wang et al. 32 We normalized our data to U6 expression levels as a reference gene. The primer sequences are shown in Supplementary Table 2.
The relative expression was carried out by SYBR Green RealQ Plus 2× Master Mix Green (CatA325402, Ampliqon, Denmark) using the Applied Biosystems StepOnePlus™ (Waltham, MA, USA). Polymerase chain reaction reactions of FOXO3a and GAPDH were performed by applying the following thermal protocol: 95°C for 10 minutes, followed by 40 cycles of amplification (95°C for 15 seconds, 60°C for 45 seconds, and 60°C for 60 seconds). Polymerase chain reaction reactions of miRNA29a-3p, MIAT, and U6 were incubated at 95°C for 30 seconds, followed by 40 cycles of 95°C for 10 seconds, 60°C for 15 seconds, and finally 72°C for 30 seconds.
The specificity of our RT-qPCR amplification was validated by melting curves analyses. The GAPDH and U6 levels were used as reference genes as earlier research on GAPDH and U6 expression levels demonstrated consistent expression in multiple organs, good stability, and little fluctuation in circulation. Thus, our data were normalized by GAPDH and U6. The PCR products were confirmed on 1.5% agarose gel.
Western blot analysis
Total proteins were extracted from tissues by lysis buffer (radioimmunoprecipitation assay buffer [RIPA]) (Cat 89900, Thermo Fisher Scientific, Waltham, MA, USA). The protein concentration was detected by the Bicinchoninic Acid Assay (BCA) method (Cat 23225, Thermo Fisher Scientific). 40 μg of lysates protein were separated by 10% sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to the polyvinylidene fluoride (PVDF) membranes. These membranes were blocked with 5% skim milk powder for 1 hour and then incubated with rabbit polyclonal antibodies to FOXO3a (Cat 720128, Thermo Fisher Scientific) overnight at 4°C. After washing, these members were followed by incubation with horseradish peroxidase (HRP)–conjugated secondary antibody (Cat 15165, Thermo Fisher Scientific) for 2 hours at 4°C. The bound antibodies were detected using an ECL kit (Cat 32106, Thermo Fisher Scientific). Protein bands were visualized and quantified using Quantity-One software (Bio-Rad, Hercules, CA, USA). β-actin, as an internal control, was detected with antibeta-actin monoclonal antibody (1:1000) (Cat AM4302, Thermo Fisher Scientific).
Statistical analysis
All reactions were run in duplicates. The amplification efficiency was analyzed using LinReg PCR software (version 0.1.0). The 2−ΔΔCt method was used to calculate the relative level of gene expression. The analytical distinction between the groups was evaluated by the 1-way analysis of variance (ANOVA) and independent sample T-test, which was executed by the Graph Pad 8.0.2 software (GraphPad Software Inc, San Diego, CA, USA), and P value < .05 was considered statistically notable. Indeed, to evaluate the diagnostic ability of MIAT, FOXO3a, and miRNA29a-3p and in combination receiver operating characteristic (ROC) curve and area under the curve (AUC) were used. The appropriate cut-off values for MIAT, FOXO3a, and miRNA29a-3p were determined, and based on these cut-off points, the sensitivity and specificity were evaluated.
Results
Bioinformatics analysis
Protein-protein interaction analysis
The interaction analysis revealed that FOXO3a protein-protein network is related to nicotine adenine dinucleotide (NAD)–dependent protein deacetylase sirtuin-1 (SIRT1), cyclin-dependent kinase 2 (CDK2), catenin beta-1 (CTNNB1), NAD-dependent protein deacetylase sirtuin-3 (SIRT3), 14-3-3 protein zeta/delta (YWHAZ), RAC-alpha serine/threonine-protein kinase (AKT1), E3 ubiquitin-protein ligase Mdm2 (MDM2), cellular tumor antigen p53 (TP53), histone acetyltransferase p300 (EP300), and proto-oncogene protein (MYC) (Figure 1A). These genes that are involved in protein-protein networks have an important role in the regulation of the apoptotic signaling pathway.

(A) Protein-protein interaction network of FOXO3a with 10 partners. (B) Enrichment analysis of FOXO3a protein-protein network.
Enrichment analysis based on protein-protein interactions that were constructed in the string database indicated that these genes that are part of protein-protein network in BP in regulation of response to DNA damage stimulus, apoptotic signaling pathway, cellular senescence, and according to MF in disorder domain-specific binding, androgen receptor binding, and DNA-binding transcription factor binding are involved. Moreover, CC showed that they are in the cytosol, nucleoplasm, and protein-containing complex. The KEEG pathway has shown the role of theme in various cancer such as BC, gastric cancer, and nonsmall cell lung cancer (Figure 1B).
miR29a-3p has a common binding site with MIAT and FOXO3a
Bioinformatics investigation revealed that FOXO3a (Figure 2A) and MIAT (Figure 2B) shared a common miR-29a-3p binding site. Moreover, both MIAT and FOXO3a had a conserved miR-29a-3p binding sites on their 3’-UTRs (Figure 2C).

(A) The predicted binding sites of FOXO3a to miRNA-29a-3p sequence were illustrated. (B) The putative binding sites between MIAT and miRNA-29a-3p according to the starBase. (C) Bioinformatics analysis revealed that MIAT and FOXO3a shared a common miRNA-29a-3p-binding site. The red box represented the binding site for miR-29a-3p on the FOXO3a mRNA 3’-UTR, and the blue box represented the binding site for miR-29a-3p on MIAT.
The construction of MIAT and FOXO3a competitive endogenous RNA network
Long noncoding RNA can decrease the suppression of mRNA expression by competitively binding to miRNA (23). According to this theory, negative connections exist between miRNAs and lncRNAs or mRNAs, whereas positive correlations exist between mRNAs and lncRNAs.
MIAT and FOXO3a in this study were subjected to further bioinformatics analyses to identify the most commonly predicted target miRNAs. The miRNet and miRDB databases were used to predict target miRNAs of MIAT and FOXO3a, respectively. The combination of miRNAs according to 1 MIAT node, 1 gene FOXO3a node, 136 miRNA nodes, and 136 edges, which were visualized using Cytoscape. So, as it was demonstrated in Figure 3, miRNA29a-3p is only common target between MIAT and FOXO3a. This finding showed that the MIAT-miRNA29a-FOXO3a network acts as a sponge based on competitive endogenous RNA (ceRNA). According to this theory, MIAT can act as ceRNA network to regulate FOXO3a expression by sponging miR-29a-3p (Figure 3). However, further research is needed in the future.
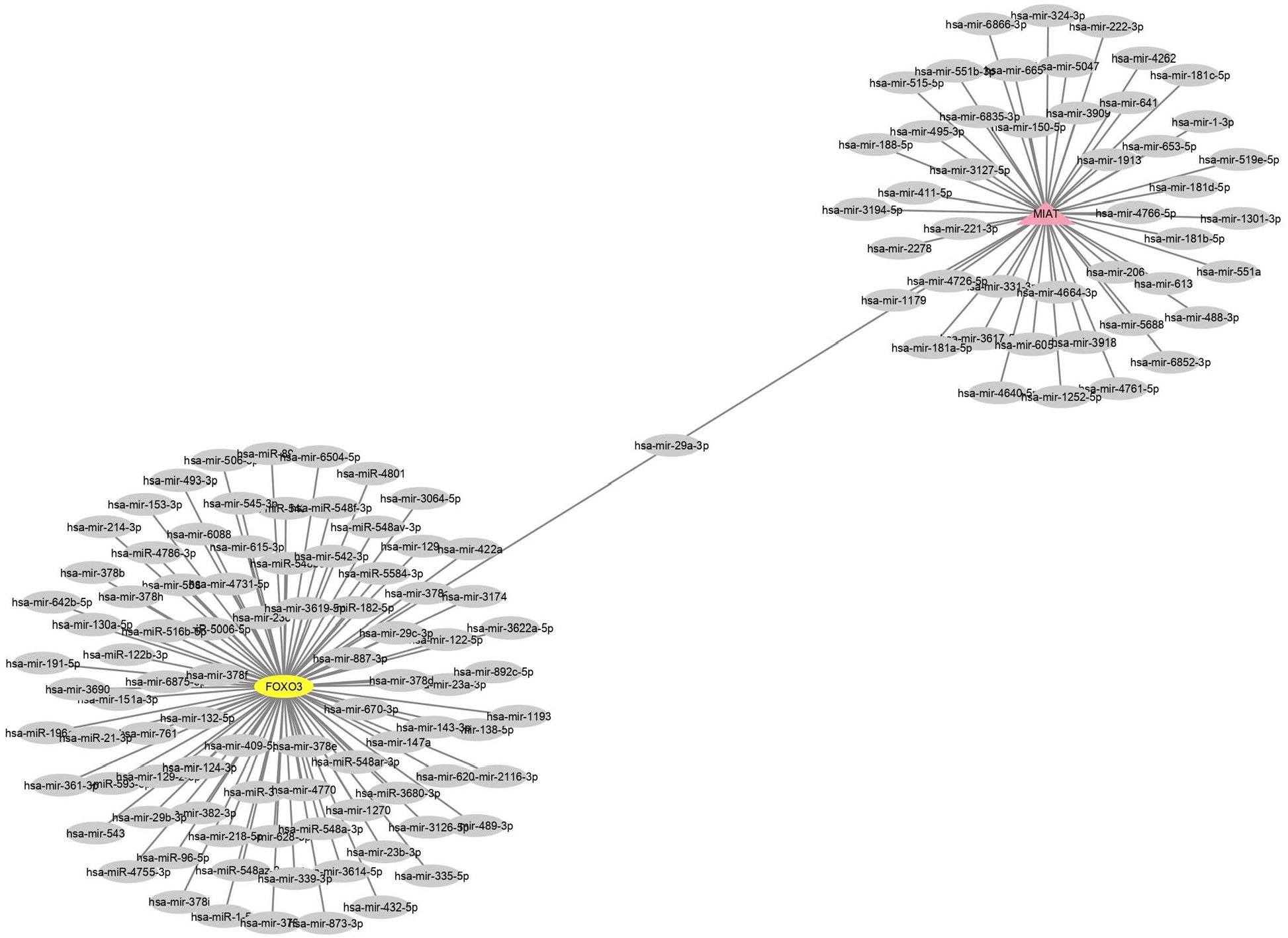
ceRNA network for MIAT, miRNA29a-3p, and FOXO3a (pink = MIAT, yellow = FOXO3a, and gray = miRNAs).
Differential expression of MIAT, FOXO3a, and miRNA29a-3p in patients’ whole blood and healthy blood
Our studies showed that the expression levels of MIAT and FOXO3a are remarkably higher in the whole blood of BC patients compared with healthy individuals (P < .04 and P < .02, respectively). In contrast, the expression level of miRNA29a-3p (P < .001) was substantially lower in the patient group compared with the control groups (P < .001) (Figure 4).

Expression of MIAT, jFOXO3a, and miRNA29a-3p in the whole blood of breast cancer patients in comparison to the normal control group (P < .04, P < .02, and P < .001, respectively).
Differential expression of MIAT, FOXO3a, and miRNA29a-3p in ductal carcinoma tissue and normal breast tissue
Our outcomes revealed that the total mean expression levels of MIAT and FOXO3a were dramatically much higher in BC tissue than normal breast tissue (P < .03 and P < .006, respectively). In comparison with the control groups, miRNA29a-3p expression levels were significantly lower in the patient group (P < .05) (Figure 5A).
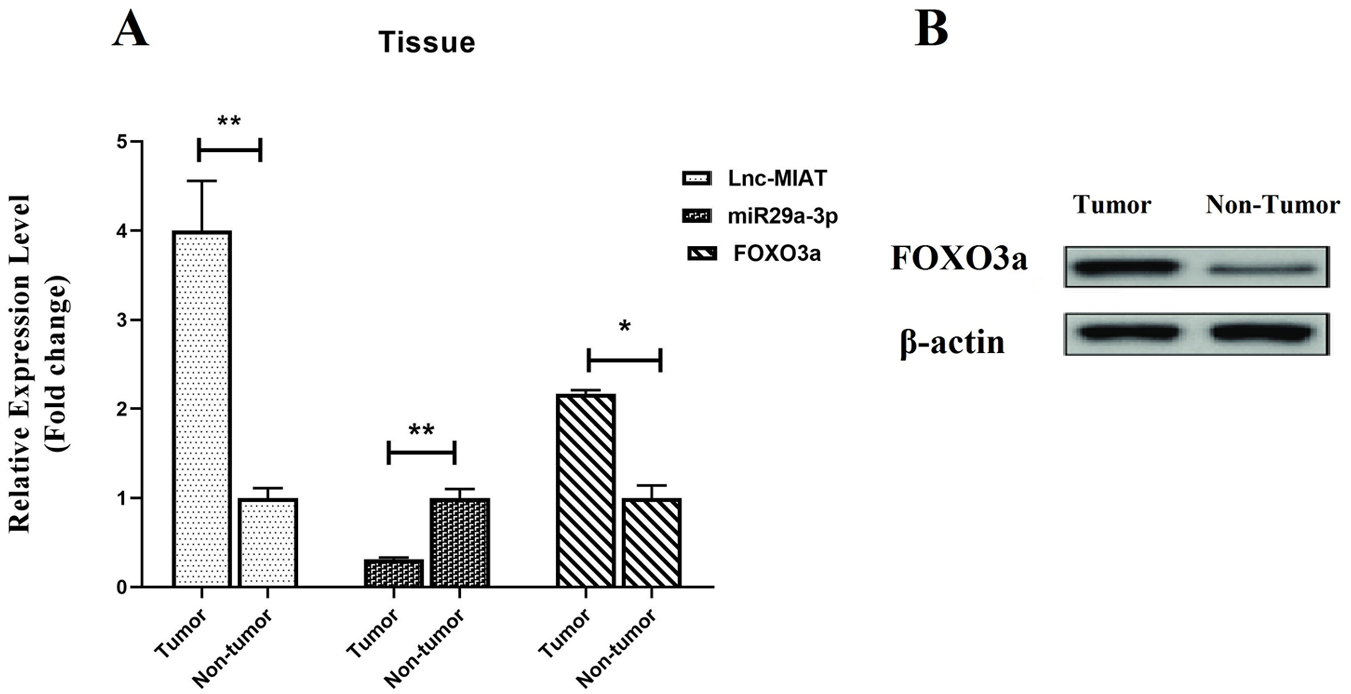
(A) Expression of MIAT, FOXO3a, and miRNA29a-3p in BC tissue in comparison with normal breast tissue (P < .03, P < .006, and P < .05, respectively). (B) Western blot analysis of Foxo3a abundance in BC tissues and normal breast tissue. The total protein extracted from breast tissues was analyzed using a polyclonal antibody against human FOXO3a. β-actin was used as a loading control.
Western blot analysis of FOXO3a protein in ductal carcinoma breast cancer tissue and normal breast tissue
Our result indicated that the expression level of FOXO3a in freshly collected ductal carcinoma BC tissue was significantly higher than normal breast tissue, as shown in Figure 5B. Collectively, these data suggest that the expression of FOXO3a might be a frequent event in BC tissues.
Differential expression of MIAT, FOXO3a, and miRNA29a-3p based on clinical features of the tumor in ductal carcinoma breast cancer tissue and whole blood
The result of this study indicated that overexpression of MIAT was associated with stage I and stage II breast tumors and lymph node involvement in both BC tissue (P < .01 and P < .03, respectively) and whole blood (P < .04 and P < .002, respectively). There was no significant difference in estrogen receptor (ER), human epidermal growth factor receptor 2 (Her2), progesterone receptor (PR) status in BC tissue, and whole blood. However, upregulation of FOXO3a was positively associated with ER-positive (P < .02), lymph node involvement (P < .01), and tumor, node, and metastasis (TNM) stage (P < .004) in BC tissue. Moreover, there was a significant difference in the upregulation of FOXO3a and ER-positive (P < .04), lymph node involvement (P < .01), and TNM stage (P < .007) in whole blood, but there was no significant difference in Her2, PR status, and ER-negative in BC tissue and whole blood.
In contrast, low expression of miRNA29a-3p was clearly associated with ER-positive in both BC tissue (P < .006,) and whole blood (P < .004), but significant difference has not been seen between downregulation of miRNA29a-3p and other features of the tumor.
Assessment of the diagnostic performance of MIAT, FOXO3a, and miRNA29a-3p in whole blood and ductal carcinoma breast cancer tissue
MIAT, FOXO3a and miRNA29a-3p were differently expressed in the whole blood of BC and healthy individuals; hence, ROC curve was applied to predict the potential of these genes in differentiating BC from healthy individuals. MIAT at the cut-off 0.8 with a sensitivity of 78%, a specificity of 100%, and AUC values 0.830 (Supplemental Figure 6A), FOXO3a at the cut-off 0.9 with a sensitivity of 90%, a specificity of 100%, and AUC values 0.900 (Supplemental Figure 6B), and miRNA29a-3p at the cut-off 0.8 with a sensitivity of 100%, a specificity of 80%, and AUC values 0.910 (Supplemental Figure 6C) detect BC from healthy women, respectively.
In addition, ROC curve was used to assess potential of MIAT, FOXO3a, and miRNA29a-3p to distinguish between BC tissue and normal samples. MIAT at the cut-off 0.6 with a sensitivity of 88%, a specificity of 80%, and AUC values 0.880 (Supplemental Figure 6E), FOXO3a at the cut-off 0.8 with a sensitivity of 75%, a specificity of 100%, and AUC values 0.853 (Supplemental Figure 6F) and miRNA29a-3p at the cut-off 0.8 with a sensitivity of 80%, a specificity of 100%, and AUC values 0.940 (Supplemental Figure 6G) distinguish BC from healthy individuals, respectively.
Furthermore, the diagnostic accuracy of the combination of MIAT, FOXO3a, and miRNA29a-3p in whole blood and BC tissue was performed. In the whole blood, the combination of MIAT, FOXO3a, and miRNA29a-3p diagnoses BC from healthy women at the cut-off 0.9 with a sensitivity of 95%, a specificity of 90%, and AUC values 0.990 (Supplemental Figure 6D), and in BC tissue, they detect BC from normal samples at the cut-off 0.9 with a sensitivity of 90%, a specificity of 100%, and AUC values 0.100 (Supplemental Figure 6H).
Discussion
The existing diagnosis approach cannot detect cancer early and it influences the life quality of patients. As a result, it is critical to find a biomarker that would be accessible, cost-effective, and sensitive enough to detect and monitor BC patients. The purpose of this research was to determine the interaction between MIAT, FOXO3a, and miRNA29a-3p and compare the expression levels of them in BC tissue with whole blood to see if they could be useful bioindicator for ductal carcinoma BC diagnosis and management in clinical practice. This study revealed the overexpression of MIAT and FOXO3a and the downregulation of miRNA 29a-3p in the tissues and whole blood of Iranian women with BC ductal carcinoma (Figures 4 and 5A and B). The AUC values for MIAT, FOXO3a, and miRNA29a-3p indicated that they are effective candidates with a high degree of specificity and sensitivity for the diagnosis of ductal carcinoma BC.
Moreover, MIAT-miRNA29a-3p-FOXO3a have been seen as the ceRNA networks in BC. One of the main strengths of this study is the construction of the ceRNA network using bioinformatics analysis based on MIAT-targeting and FOXO3a-targeting. According to Figure 3, miRNA29a-3p was considered the only common target between FOXO3a and MIAT.
The MIAT/miRNA-29a-3p/FOXO3a ceRNA network has been described for the first time, which may help us identify a novel ceRNA network involved in the regulation of BC.
The apoptotic pathway is critical for tumor growth and metastasis at all stages. Apoptosis is a BP that contributes significantly to the growth and survival of multicellular organisms by eliminating damaged, old, or autoimmune cells through a regulated cell death mechanism. 33 Apoptosis includes a large number of signaling pathways, which is considered a precise regulatory mechanism. 34 Therefore, any sort of alternation in these pathways leads to tumorigenesis, metastasis, and resistance to anticancer drugs. Thus, cell-cycle control mechanisms have emerged as a potential therapeutic strategy. 35
In this study, the STRING database and enrichment analysis based on BP and KEEG pathway indicated that FOXO3a protein-protein network has a critical role in the regulation of the apoptosis signaling pathway and has both positive and negative effects on the regulation of the intrinsic apoptosis pathway (Figure 1A and B). Among them, MYC, TP53, CDK2, AKT, and MDM2 are important regulators in cell-cycle progress and the apoptosis pathway.
It has been reported that FOXO3a acts as a metastasis suppressor because it increases the expression level of E-cadherin and downregulates EMT transcription factors. This resulted in the reversal of the invasive behavior of BC cells. 36 Song et al 37 reported that upregulation of FOXO3a substantially inhibited BC cell migration and invasion in vitro. In this study, we have shown significant upregulation of FOXO3a in BC tissues and whole blood compared with normal groups. In accordance with our results, the overexpression of FOXO3a in breast tumor tissues and cell lines has been shown in some studies. 38 It also has been shown that upregulation of FOXO3a promotes growth of cancer cells and tumor progression. 39 However, recent studies have suggested that the low expression of FOXO3a induces EMT and subsequently promotes cancer cell invasion and proliferation, which are associated with BC development and poor response to therapy. 40
Estrogen receptor has a critical role in growth, proliferation, and differentiation in BC. There is abundant evidence to show crosstalk between FOXO3a and ER signaling pathways. 41 Sisci et al 42 demonstrated that, in BC cell lines, invasive phenotype of ERα+ had been reversed by activation of FOXO3a. Whereas in ERα− cell lines, tumor cell invasion had promoted significantly. Moreover, they proposed that according to functional interaction between FOXO3a and ERα, cell migration and invasion had reduced in ERα+ tumors. However, in the absence of receptor, FOXO3a triggered many pathways that lead to opposite consequences. Jiang et al 43 established a clear correlation between FOXO3a expression and ER-positive in human breast tumors and recognized FOXO3a as a promising prognostic marker. They proposed that a high level of FOXO3a expression was shown to be strongly associated with long-term survival in ER-positive cell lines. Thus, according to ER status, patients who were identified with FOXO3a+/ER+ had shown better prognoses compare with ER−/FOXO3a+. On the contrary, loss of ER in patients with downregulation of FOXO3a has shown that overall survive was being worse compared with ER+ patients. Moreover, they confirmed that FOXO3a expression was associated with lymph node involvement and TNM stage. Chen et al 44 observed that upregulation of FOXO3a was correlated with increased AKT expression and lymph node metastases. Along with these findings, our analysis showed that, overexpression of FOXO3a was positively associated with ER-positive, lymph node involvement, and TNM stage in BC tissue and whole blood.
MIAT regulates a variety of signaling pathways in cancer. In the study by Yang et al, 45 it was reported that MIAT has been identified as a critical factor in cell invasion, migration, and proliferation through the PI3K/AKT signaling pathway. MIAT dramatically increased PI3K and AKT phosphorylation and stimulated the production of C-MYC and cyclin D1. Recent literature indicated that the expression level of MIAT was higher in BC cells than in normal cell lines, as well as suggesting that MIAT may serve as an oncogene in BC, sharing miRNA-155-5p response element with DUSP7 and promoting BC progression. 26 Alipoor et al 46 established for the first time that MIAT is implicated in the incidence and progression of BC, presenting it as a potential tumor marker for BC detection and therapy. Their findings have suggested MIAT was upregulated in BC tissues and cell lines, and this study showed that the expression level of this gene in the whole blood was consistent with its expression in the BC tissues and confirmed the previous data, so it has the potential to be considered as a noninvasive biomarker in the whole blood for BC. They discovered that MIAT expression was considerably greater in high-grade breast ductal carcinoma than in surrounding nontumor tissues and was associated with clinic pathological characteristics of tumors, such as the Her2, the p53 gene, the ER, and the PR. Their findings indicated that the expression level of MIAT was significantly increased in ER-positive and PR-positive tumor tissues. Moreover, MIAT has recently been found to be overexpressed in p53-negative tumor tissues. In addition, it was shown that inhibiting MIAT expression resulted in G1-phase arrest and apoptosis in BC cells. These findings suggest that MIAT may act as a cell-cycle regulator. Besides, MIAT inhibition prevents BC cell migration and decreases the expression of EMT genes. A recent study showed that the expression level of MIAT is increased in ER-positive BC tissue and cell lines. 47 It has been reported that MIAT is significantly expressed in stage I and stage II breast tumors. 48 In a study by Ye et al, 49 it was shown that higher expression of MIAT was positively related to lymph node status and TNM stage in BC, and they proposed that MIAT serves as a noninvasive biomarker for the diagnosis of BC. The result of this study indicated that the expression level of MIAT was increased in BC tissue and whole blood of patients. Furthermore, overexpression of MIAT was clearly associated with stage I and stage II breast tumors and lymph node involvement in both BC tissue (P < .01 and P < .03, respectively) and whole blood (P < .04 and P < .002, respectively).
A substantial amount of experimental data have demonstrated that miRNAs play a critical role in cancer cell death regulation. Numerous strategies have been developed to either inhibit the expression of oncomiRs or to increase the expression of tumor suppressor miRNAs in an attempt to re-establish miRNA activity in apoptotic pathways. 50 In BC cells, miRNA-29a-3p has been demonstrated to have a tumor suppressor function by interrupting the cell cycle during the G0/G1 phase via the negative regulation of the expression of CDC42. 51
In the study by Li et al, 52 it was shown that miRNA-29a-3p mimic promoted the proliferation of BC cell lines (MCF-7 and T47D). The inhibition of miRNA-29a-3p was shown to suppress the proliferation of these cell lines. It has been reported that miRNA29a-3p has a negative effect on N-MYC, which leads to upregulation of the mesenchymal phenotype and promotes tumor invasion in BC cells. 53 Pei et al 54 proposed that higher miRNA-29a-3p expression increased cell proliferation, whereas decreased miRNA-29a-3p expression suppressed cell growth. Wu et al 55 found that in BC cells, the expression level of miRNA29a-3p was decreased. Moreover, miRNA-29a-3p inhibited cells in the G0/G1 phase and restricted tumor development through decreasing the expression of B-MYB. Yan et al 56 revealed that miRNA29 family members (miR-29a, miR-29b, and miR-29c) increase p53 levels and trigger apoptosis in a p53 pathway. In this research, miRNA-29a-3p was downregulated in BC tissue and whole blood of patients compared with control groups. In assessing the relationship between miRNA29a-3p expression and clinical features of tumor, we found that overexpression of miRNA29a-3p was clearly associated with ER-positive in both BC tissue (P < .006) and whole blood (P < .004).
Due to advancements in high-throughput sequencing and novel computing technologies, lncRNA has been recognized as a key molecule in the regulation of gene expression at the post-transcriptional level in recent years. 57 Increasing evidence indicates that lncRNA functions as a ceRNA, inhibiting the expression or activity of miRNA. Micro RNA has been recognized as a critical regulatory element in the ceRNA network and has a negative impact on regulating RNA gene expression by interacting with the target region of mRNA 3’-UTR, causing adenosine acidification, decreasing mRNA stability, and limiting translation. 58 The bioinformatics analysis of this study suggested that miRNA29a-3p has a common binding site with MIAT and FOXO3a (Figure 2). Microarray research identified a binding site between miRNA-29a-3p and FOXO3a, showing a similar targeting connection between miRNA-29a-3p and FOXO3a. Growing evidence is emerging to connect miRNA-29a-3p downregulation to FOXO3a overexpression. 59 In another similar study, dual-luciferase demonstrated miRNA-29a-3p has a targeting relationship with FOXO3a in OC (ovarian cancer) and, according to western blot, overexpression of miRNA-29a-3p inhibited the expression of FOXO3a and downregulation of miRNA-29a-3p elevated the expression of FOXO3a. Based on these results, they showed that FOXO3a could be targeted by miRNA-29a-3p. 60
In addition, it has been reported that MIAT has complementary base pairing sites with miRNA-29a-3p, and it may function as an endogenous miRNA sponge to inhibit the expression of miRNA-29a-3p in gastric cancer. 61 However, further research is needed to determine the exact mechanism of miRNA29a-3p activity in BC. In this study, we showed that the expression levels of MIAT and miRNA29a-3p in whole blood and ductal carcinoma BC tissues have a negative correlation. In fact, overexpression of MIAT is associated with decreased miRNA29a-3p and increased FOXO3a expression. Following these data, it can be concluded that MIAT, miRNA-29a-3p, and FOXO3a levels have a significant relationship with the pathogenesis of ductal carcinoma BC.
To confirm the endogenous connection between miRNA-29a-3p, MIAT, and FOXO3a in BC, we propose that future investigations use a dual-luciferase test.
Considering the fact that an ideal biomarker should have high sensitivity and specificity, this study confirmed that MIAT, FOXO3a, and miRNA29a-3p, as well as the combination of them, have shown high sensitivity and specificity in BC tissue and whole blood compared with healthy individuals. Together, these findings indicate that they can be potential bioindicator for BC patients in the whole blood with better sensitivity and specificity.
In summary, it is appealing to demonstrate that MIAT and FOXO3a with high expression and miR-29a-3p, with down expression as a ceRNA network, can be potentially effective bioindicators for the detection of BC in clinical practice. The crosstalk between ncRNAs may provide hope for an accurate diagnosis of BC in the future.
Supplemental Material
sj-docx-1-bcb-10.1177_11782234231184378 – Supplemental material for Evaluation of the Potential Diagnostic Role of the Lnc-MIAT, miR-29a-3p, and FOXO3a ceRNA Networks as Noninvasive Circulatory Bioindicator in Ductal Carcinoma Breast Cancer
Supplemental material, sj-docx-1-bcb-10.1177_11782234231184378 for Evaluation of the Potential Diagnostic Role of the Lnc-MIAT, miR-29a-3p, and FOXO3a ceRNA Networks as Noninvasive Circulatory Bioindicator in Ductal Carcinoma Breast Cancer by Shokufeh Razi, Hossein Mozdarani and Roudabeh Behzadi Andouhjerdi in Breast Cancer: Basic and Clinical Research
Supplemental Material
sj-docx-2-bcb-10.1177_11782234231184378 – Supplemental material for Evaluation of the Potential Diagnostic Role of the Lnc-MIAT, miR-29a-3p, and FOXO3a ceRNA Networks as Noninvasive Circulatory Bioindicator in Ductal Carcinoma Breast Cancer
Supplemental material, sj-docx-2-bcb-10.1177_11782234231184378 for Evaluation of the Potential Diagnostic Role of the Lnc-MIAT, miR-29a-3p, and FOXO3a ceRNA Networks as Noninvasive Circulatory Bioindicator in Ductal Carcinoma Breast Cancer by Shokufeh Razi, Hossein Mozdarani and Roudabeh Behzadi Andouhjerdi in Breast Cancer: Basic and Clinical Research
Supplemental Material
sj-docx-3-bcb-10.1177_11782234231184378 – Supplemental material for Evaluation of the Potential Diagnostic Role of the Lnc-MIAT, miR-29a-3p, and FOXO3a ceRNA Networks as Noninvasive Circulatory Bioindicator in Ductal Carcinoma Breast Cancer
Supplemental material, sj-docx-3-bcb-10.1177_11782234231184378 for Evaluation of the Potential Diagnostic Role of the Lnc-MIAT, miR-29a-3p, and FOXO3a ceRNA Networks as Noninvasive Circulatory Bioindicator in Ductal Carcinoma Breast Cancer by Shokufeh Razi, Hossein Mozdarani and Roudabeh Behzadi Andouhjerdi in Breast Cancer: Basic and Clinical Research
Supplemental Material
sj-docx-4-bcb-10.1177_11782234231184378 – Supplemental material for Evaluation of the Potential Diagnostic Role of the Lnc-MIAT, miR-29a-3p, and FOXO3a ceRNA Networks as Noninvasive Circulatory Bioindicator in Ductal Carcinoma Breast Cancer
Supplemental material, sj-docx-4-bcb-10.1177_11782234231184378 for Evaluation of the Potential Diagnostic Role of the Lnc-MIAT, miR-29a-3p, and FOXO3a ceRNA Networks as Noninvasive Circulatory Bioindicator in Ductal Carcinoma Breast Cancer by Shokufeh Razi, Hossein Mozdarani and Roudabeh Behzadi Andouhjerdi in Breast Cancer: Basic and Clinical Research
Footnotes
Acknowledgements
The authors thank all patients for their involvement in our project, and Ms Fatemeh Rajabi, for making contribution to proofreading of the article. The earlier version of the manuscript was presented as preprint in the following link “21203/rs.3.rs-1437285/v1.”
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
