Abstract
Representing 15% to 20% of all invasive breast cancers, adjuvant systemic treatment for early-stage, high-risk triple-negative breast cancer (TNBC) is preferentially done in the neoadjuvant setting based on a chemotherapy backbone of anthracyclines and taxanes. Pathological complete response to neoadjuvant treatment constitutes the main objective, regarding its correlation with oncological outcomes. The optimal neoadjuvant regimen to achieve the highest rates of pathological complete response is still under investigation, with the increasing knowledge on the molecular pathways, genomic sequencing, and immunological profile of TNBC allowing for the development of a wide array of new therapeutic options. This review aims to summarize the current evidence and ongoing clinical trials of new therapeutic options for the neoadjuvant treatment of TNBC patients.
Introduction
Triple-negative breast cancer (TNBC) occurs more frequently in younger women (<40 years) and has an aggressive behaviour with shorter relapse-free and overall survival (OS) rates. According to GLOBOCAN 2020, female breast cancer surpassed lung cancer as the leading cause of cancer globally in 2020, with 2.3 million new cases worldwide.1,2 Presenting symptoms can be classified into 3 different categories: breast lump, nonlump breast, and nonbreast symptoms. Breast lump is the most common presentation sign allowing for more frequent diagnosis at a curable stage. 3
Expression of hormonal receptors and/or human epidermal growth factor receptor 2 (HER2) are the main elements guiding clinical management of breast cancer, allowing for the subclassification as hormone receptor positive, HER2 positive, or TNBC. 1
Patients with TNBC are usually diagnosed at an earlier median age and face a more aggressive disease’s course with a peak in recurrence between 1 to 3 years after diagnosis.4,5 Integration of histological, molecular, and genetic profiles is crucial for a better clinical management of TNBC.
Lehmann et al. 6 defined a molecular classification for TNBC of 6 subtypes with basal-like 1 (BL1), immunomodulatory (IM), and molecular (M) subtypes predominantly enriched with high-grade tumours; luminal androgen receptor (LAR) and mesenchymal stem-like (MSL) subtypes mostly associated with low-grade tumours. In 2016, Lehman et al. 7 revised the proposed classification into 4 molecular subtypes: BL1, BL2, M, and LAR. Regarding clinical outcome, LAR subtype has worst prognosis, and IM has better outcomes.7,8 Recent reviews have correlated the different molecular and transcriptomic profiles of TNBC to better understand tumoral behaviour and develop new targeted therapies. 9
In the last 5 years, the development of new drugs in the neoadjuvant setting such as immune-checkpoint inhibitors (ICIs), poly-ADP-ribosyl polymerase inhibitors (PARPi), phosphatidylinosi-tol-4,5-bisphosphate 3-kinase catalytic subunit alpha (PIK3CA) inhibitors, androgen receptor (AR)-targeted therapies or anti-VEGF agents have been analysed in several trials. 10
Rationale for Neoadjuvant Treatment on TNBC and Patient Selection
Neoadjuvant chemotherapy is standard of care for locally advanced/inoperable TNBC, with clinical response to treatment predicting clinical outcomes in this subset of patients. 11 Triple-negative breast cancers have clinicopathologic features that support its aggressive behaviour when compared to non-TNBC: high mean tumour size, higher grade, and high proliferation index at diagnosis. 12
Patients with TNBC tend to present higher rates of polymerase chain reaction (pCR) after neoadjuvant chemotherapy compared to luminal subtypes. 13 Triple-negative breast cancer are more commonly associated to breast cancer gene (BRCA) mutations with over 80% of BRCA1-mutated and almost 10% of BRCA-2-mutated breast cancer classified within this subgroup. 5 Breast cancer genes encode crucial components of DNA damage repair mechanisms such as homologous recombination deficiency (HRD) and DNA stability. 5 Homologous DNA recombination defects render TNBC more susceptible to DNA-damaging agents including platinum-based cytotoxic agents or poly (ADP-ribose) polymerase (PARP) inhibitors. 14 These agents cause DNA strand cross-links that are neither recognized nor repaired by the homologous recombination defective system, leading to cell death.
The National Comprehensive Cancer Network (NCCN) 2022 Breast Cancer Guidelines standard of care for TNBC comprises a combination of anthracyclines plus taxanes in the neoadjuvant setting. Pathological complete response after neoadjuvant treatment is associated with improvement in disease-free survival (DFS). 11 Furthermore, patients who do not have a pathological complete response after neoadjuvant chemotherapy have a 20% to 30% higher risk of relapse. Therefore, the degree of pathological response after neoadjuvant chemotherapy allows the definition of a subset of TNBC patient which benefit from adjuvant chemotherapy, as stated in CREATE-X trial. 15
Early TNBC cT1a (<0.5 cm) or cT1b (0.6-1.0 cm) without lymph node involvement has good prognosis without neoadjuvant treatment. Vaz-Luis et al performed an observational study of 363 TNBC patients with T1a-b N0 disease treated with adjuvant chemotherapy versus those without adjuvant therapy to assess the distant recurrence-free survival (DRFS). The subgroup of untreated T1a and T1b had a 5-year DRFS of 93% and 90% compared to treated T1a and T1b DRFS of 100% and 96%. 16
The NCCN guidelines on breast cancer define candidates for neoadjuvant treatment patients with inoperable disease or with operable disease if one of the following is present: cT ⩾ 2 or cN ⩾ 1 HER2-positive or TNBC; large primary relative to breast size if breast conservation is desired; cN+ likely to become cN0 following neoadjuvant treatment; delay of definitive surgery. Therefore, for patients with TNBC tumours >1 cm N0/N+, systemic chemotherapy is recommended. Standard chemotherapy regimens for TNBC consist of a combination of anthracyclines, taxanes, and alkylating agents.
Sequential use of anthracyclines plus taxanes is mainly used based on retrospective evidence from subgroup analysis before 2010. First reports for anthracycline-based regimens had pCR rates ranging from 14% to 47% and sequential regimens of anthracyclines plus taxanes rates ranging from 17% to 39%. 17 The phase III BCIRG-005 clinical trial assessed the efficacy and safety of sequential versus concurrent use of taxane-anthracycline-based adjuvant chemotherapy. Ten years follow-up analysis confirmed that doxorubicin, cyclophosphamide, and docetaxel (TAC) and sequential doxorubicin, cyclophosphamide plus docetaxel monotherapy (AC→T) had similar efficacy outcomes. Doxorubicin, cyclophosphamide, and docetaxel regimen with granulocyte colony-stimulating factor (G-CSF) support provided a shorter treatment duration with less acute toxicity. 18
The GeparTrio trial assessed pCR rate after neoadjuvant anthracyclines, cyclophosphamide, and taxanes. The subgroup analysis of this cohort revealed rates of pCR up to 57%. 19 Since these first trials, the main goal has been to define which agent or combination of agents achieves the highest pCR in this setting.
TNBC Molecular Heterogeneity as Therapeutic Target
Molecular heterogeneity of TNBC was first formally classified in 2011 by Lehman et al. with the proposal of 6 molecular subtypes, each presenting unique molecular characteristics that can be used as therapeutic targets (Table 1). Owing to the histological landscape of tumour specimens, IM and MSL subtypes were fused into the same molecular subtype reflecting infiltrating lymphocytes and tumour-associated mesenchymal cells, originating a 4-subtype classification (BL1, BL2, M, and LAR). 7 Each subtype has a specific molecular signature that contributes to the differential response to each treatment strategy. Basal-like 1 has increased expression of cell cycle and DNA damage response genes, 8 contributing to an increased sensitivity to platinum agents and better response to neoadjuvant chemotherapy. Homologous recombination deficiency sensitizes tumour cells to platinum-induced cell death.
Molecular pathways in TNBC subtypes as therapeutic targets.
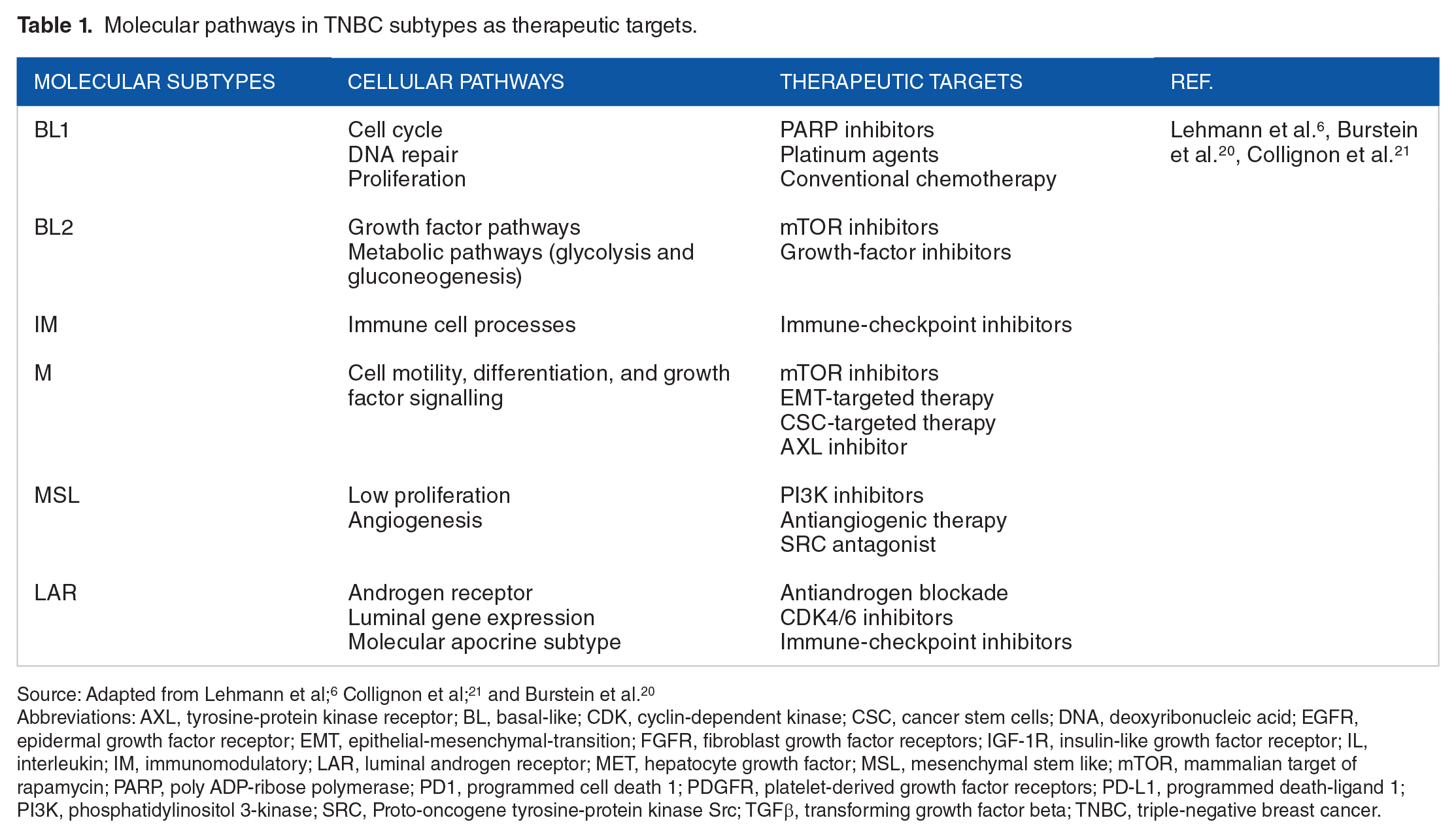
Abbreviations: AXL, tyrosine-protein kinase receptor; BL, basal-like; CDK, cyclin-dependent kinase; CSC, cancer stem cells; DNA, deoxyribonucleic acid; EGFR, epidermal growth factor receptor; EMT, epithelial-mesenchymal-transition; FGFR, fibroblast growth factor receptors; IGF-1R, insulin-like growth factor receptor; IL, interleukin; IM, immunomodulatory; LAR, luminal androgen receptor; MET, hepatocyte growth factor; MSL, mesenchymal stem like; mTOR, mammalian target of rapamycin; PARP, poly ADP-ribose polymerase; PD1, programmed cell death 1; PDGFR, platelet-derived growth factor receptors; PD-L1, programmed death-ligand 1; PI3K, phosphatidylinositol 3-kinase; SRC, Proto-oncogene tyrosine-protein kinase Src; TGFβ, transforming growth factor beta; TNBC, triple-negative breast cancer.
Differential subtype response to neoadjuvant therapy was first analysed in a retrospective study of 125 TNBC patients that analysed the addition of carboplatin to neoadjuvant chemotherapy with a combination of anthracyclines plus taxanes, anthracyclines, or taxanes. Basal-like 1 tumours had the highest pCR under carboplatin regimens than the remaining subtypes (BL2, M, IM, MLS, and LAR) summed (80% vs. 23%, p = .027), and LAR tumours had the lowest pCR despite treatment regimen (14.3% vs. 42.7%, p = .045). 24
Despite being chemo-resistant, LAR subtype has molecular evidence of oestrogen plus AR activation, 20 which seems to contribute to its lower proliferative index and response to anti-oestrogen or anti-androgen therapies.4,23
Mesenchymal subtype (MES) was characterized by pathways of mismatch repair and DNA damage repair. Basal-like immunosuppressed (BLIS) showed downregulation of immune and cytokine pathways, being associated with worse outcomes. Basal-like immune-activated (BLIA) showed upregulation of immunoregulatory pathways and proved to be the subtype with best clinical outcomes. 24
Relevance of BRCA Status
DNA repair mechanisms play an important role on the stability and integrity of the genome. Homologous recombination, end joining, mismatch repair, nucleotide/base excision repair, and telomere metabolism are the most relevant DNA repair mechanisms. 25
From the wide range of lesions occurring to the DNA molecule, DNA double-strand break (DSB) poses the greatest threat to genome stability. 25 BRCA1 and BRCA2 are crucial for the homologous recombination repair, and inherited defects to one of these genes can lead to cancer. Breast cancer gene loss translates in genomic instability leading to oncogenic transformation of nontumorigenic cellular populations into cancer stem cells and tumour development. As classic tumour suppressor genes, BRCA genes tend to rely on the ‘2-hit’ hypothesis to initiate cancer progression. Individuals predisposed to BRCA-associated hereditary cancers carry a germline mutation in the BRCA genes, needing a second hit to the wild-type BRCA allele to develop cancer. 22
Five percent of all breast cancers arise from BRCA1/BRCA2 germline mutations due to cellular impairment to repair DSB through homologous recombination.26,27 Germline mutations for BRCA genes occur in 10% to 20% of TNBC patients and somatic mutations in 3% to 5%. 28 Breast cancer gene 1/2 status seems to affect tumoral phenotype, with germline-mutated BRCA1 carriers mainly developing TNBC at younger ages (median age at diagnosis of 47 years for BRCA1 versus 58 years for BRCA2) and BRCA2 carriers usually presenting with hormone receptor–positive disease.29,30
Recently, the concept of BRCAness status has emerged comprising sporadic cancers that do not have germline BRCA mutation but share phenotypic characteristics and defective HR. 31 Somatic mutations, large deletions, DNA hyper-methylation of BRCA1/2, germline PALB2 mutations, and RAD51C hyper-methylation are the most common associated mechanisms. 31 Breast cancer gene 1 mutation increases sensitivity to interstrand crosslinking agents (platinum or alkylating agents), topo-isomerase II inhibitors (anthracyclines), or PARP inhibitors.32,33
Neoadjuvant Platinum Agents for TNBC
Despite being put aside in the early studies on neoadjuvant treatment for TNBC, increasing knowledge on the association between BRCA mutation and triple-negative phenotype renewed the interest on this subset of drugs. Platinum salts have increased efficacy in cells with a defected DNA repair system, inducing single-strand (SSBs) and DSBs. 34 Several trials and retrospective studies demonstrated the effectiveness of platinum-based regimens for TNBC BRCA-mutated patients in a preoperative setting. A retrospective analysis of 6903 patients included 102 patients with BRCA1 germline mutation, which received neoadjuvant chemotherapy with cyclophosphamide, methotrexate, fluorouracil (14 patients), docetaxel plus doxorubicin (25 patients), doxorubicin without docetaxel (51 patients), or cisplatin (12 patients). Twenty-four of 102 patients achieved pCR, with highest pCR rate for gBRCA1 carriers who received neoadjuvant cisplatin therapy (83%; 10 out of 12 patients), no survival data was reported. 35
Platinum-based neoadjuvant regimens showed superior pCR rates when compared to standard chemotherapy regimens in several randomized clinical trials (Table 2). The CALGB 40603/Alliance was a randomized phase II trial designed to assess the addition of carboplatin and/or bevacizumab to neoadjuvant regimens in stage II-III TNBC patients. Patients were randomized to paclitaxel 80 mg/m2 weekly for 12 weeks followed by dose-dense doxorubicin plus cyclophosphamide (ddAC) for 4 weeks or assigned to carboplatin (AUC 6) every 21 days for 4 weeks with/without bevacizumab 10 mg/kg each 2 weeks during 9 cycles. Carboplatin AUC 6 arm had a greater percentage of pCR (41%-54%: odds ratio [OR]:1.71; p = .0029). Despite better pCR, the addition of carboplatin did not improve long-term OS.36,37
Neoadjuvant trials of platinum-based chemotherapy in early-stage TNBC.

Abbreviations: A, anthracyclines; AC, doxorubicin + cyclophosphamide; Bev, bevacizumab; Cp, carboplatin; D, docetaxel; DFS, disease-free survival; EC, epirubicin + cyclophosphamide; EP, epirubicin + paclitaxel; FEC, 5-fluorouracil + epirubicin + cyclophosphamide; Gem, gemcitabine; NabT, nab-paclitaxel; OS, overall survival; pCR, pathological complete response; T, paclitaxel; TNBC, triple-negative breast cancer.
The GeparSixto/GBG 66, a phase II clinical trial, randomized previously untreated, nonmetastatic, stage II/III TNBC patients to 18 weeks of paclitaxel once a week, nonpegylated liposomal doxorubicin plus bevacizumab simultaneously to the backbone regimens, either carboplatin (AUC 1.5) each week or no carboplatin. First analysis showed that adding carboplatin resulted in better pCR rates (53.2% vs 36.9; p = .005). 38 The final report confirmed a significantly better DFS (hazard ratio [HR]: 0.56; 95% confidence interval [CI]: [0.34, 0.93]; p = .022) without statistically significant improvement of OS. 39
Efficacy and safety of combining taxanes and platinum agents in the neoadjuvant setting was assessed in a recent phase II trial that analysed 59 stage II/III TNBC that underwent 4 cycles of carboplatin AUC 6 and weekly nab-paclitaxel 100 mg/m2 for 16 cycles. Patients with DNA repair defect achieved higher pCR rates (overall response rate [ORR]: 4.76; p = .03). 47
TBCRC 030 was an open-label, randomized, phase II trial aimed to evaluate the impact of HRD on pathologic response to single-agent cisplatin or paclitaxel. Patients were randomized to either cisplatin (75 mg/m2 every 3 weeks) during 4 cycles or weekly paclitaxel (80 mg/m2) for 12 weeks, with a pCR rate of 15.3% for cisplatin and 11.9% for paclitaxel. 48
Regarding the influence of BRCA status on platinum sensitive and the new targeted drugs for BRCA-mutated TNBC, BrighTNess trial compared the impact on pCR of adding veliparib to carboplatin or carboplatin alone to standard neoadjuvant chemotherapy in TNBC. A phase III, randomized, placebo-controlled trial randomly assigned 634 patients (2:1:1) to receive 1 of 3 segment 1 regimens: paclitaxel (80 mg/m² weekly for 12 cycles) plus carboplatin AUC6 (every 3 weeks, 4 cycles) plus veliparib 50 mg per os twice a day; paclitaxel, carboplatin, and veliparib placebo (twice a day); paclitaxel, carboplatin placebo, and veliparib placebo. Following segment 1, all received doxorubicin plus cyclophosphamide every 3 weeks for 4 cycles. Results stated higher pCR rate when combining paclitaxel, carboplatin, and veliparib than in patients receiving paclitaxel alone (53% vs 31%; p < .0001). This was not true for the comparison with patients receiving paclitaxel plus carboplatin (58%; p = .36). 49
The phase III GeparOcto trial compared efficacy and safety of sequential treatment with dose-dense epirubicin, paclitaxel, and cyclophosphamide (iddEPC) or weekly paclitaxel plus nonpegylated liposomal doxorubicin with additional carboplatin (PMCb) in TNBC. Final analysis demonstrated a pCR of 48.3% for iddEPC and 48% for the carboplatin arm, showing that PMCb did not improve pCR rate compared to iddEPC. 50
GeparOLA trial documented a similar pCR rate to GerparSixto trial in HRD TNBC patients treated with neoadjuvant paclitaxel plus carboplatin (59.3% vs 63.5%). 51
The phase II INFORM Trial aimed to compare pCR after neoadjuvant single-agent cisplatin (75 mg/m2 for 3-4 cycles) or doxorubicin-cyclophosphamide (60 mg/m2-600 mg/m2 for 3-4 cycles) every 2 (dose dense) or 3 weeks according to oestrogen receptor status in stage II/III BRCA carriers HER2-negative patients. For TNBC, dose-dense schedule was mandatory, with 23% achieving pCR with cisplatin and 29% with AC. 52 Pathologic complete response rate for cisplatin versus AC was consistent with results from GeparSixto, GeparOcto, and BrightNess trials. Among BRCA carrier’s addition of platin did not significantly improve pCR. The lack of additional benefit among BRCA carriers could be explained by the greater sensitivity of BRCA breast cancer carriers to DNA-damaging agents (CDDP or anthracyclines plus alkylating agents). Higher sensitivity to DNA-damaging agents leads to a better response to anthracyclines plus alkylating agents, reducing the potential benefit of adding a platinum agent. Therefore, gBRCA1/2 status and HRD may better predict chemosensitivity and higher pCR but not platinum benefit in the neoadjuvant setting. 53
Conclusion
Addition of a platinum agent to standard neoadjuvant anthracycline and taxane-based chemotherapy has been matter of extensive investigation, with most clinical trials showing a benefit in the rate of pathological complete response. Moreover, KEYNOTE-522 and BrighTNess trial showed that improvement on pCR rate led to an increase on event-free survival (EFS) for TNBC patients treated with a platinum agent. Despite increases in EFS, no clinical trial demonstrated OS benefit. A recent meta-analysis of clinical trials comparing addition of carboplatin to the standard anthracycline-taxane regimen showed benefit in DFS (HR: 0.60; 95% CI: 0.47-0.78; p < .001) and OS (HR: 0.69, 95% CI: 0.50-0.95; p = .02). 54 Therefore, addition of a platinum agent to standard neoadjuvant chemotherapy should be a shared informed decision, integrating the grade of toxicity that patient is able/willing to tolerate and the clinicopathological features of the disease.
Neoadjuvant PARP Inhibitors for TNBC
In 2005, Farmer et al pivotal study demonstrated that BRCA1/BRCA2 dysfunction increased cell sensitivity to inhibition of PARP enzymatic activity, involved in base excision repair. This repair mechanism represents a key pathway in DSB repair, and its inhibition leads to chromosomal instability and cell death by apoptosis. 55
A subsequent study used the concept of synthetic lethality to prove that, despite loss of one gene is not lethal, concurrent inactivation leads to cell death. Poly-ADP-ribosyl polymerase is involved in single-strand DNA breaks repair, and its inhibition alone is not lethal because SSBs can be repaired by other mechanisms such as homologous repair. In the absence of BRCA1/BRCA2, homologous repair mechanisms cannot compensate for PARP inhibition, leading to increased cytotoxicity and subsequent cell death. 56 The promising results of a proof-of-concept trial for Olaparib in germline BRCA1/2-mutated advanced breast cancer in 2009 was the first step for the development of PARPi as a valid therapeutic option for breast cancer (Table 3). 57 OlympiAD and EMBRACA trials showed positive results for olaparib and talazoparib in the metastatic setting for germline BRCA-mutated HER2-negative breast cancer patients.58,59 Following PARPi approval in this setting, several clinical trials aimed to compare pCR and long-term outcomes between PARPi and standard neoadjuvant chemotherapy.
Neoadjuvant clinical trials of PARP inhibitors in early-stage TNBC.

Abbreviations: AC, doxorubicin + cyclophosphamide; Cp, carboplatin; EC, epirubicin + cyclophosphamide; Pac, paclitaxel; PARP, poly ADP-ribose polymerase; pCR, pathological complete response; T, docetaxel; TNBC, triple-negative breast cancer.
I-SPY trial, a phase II platform trial, assessed the impact of adding novel agents or novel combinations to standard neoadjuvant chemotherapy in high-risk primary breast cancer. Addition of velaparib and carboplatin versus weekly paclitaxel improved pCR rate in TNBC patients (52% vs 26%). 62
Following these positive results, BrighTNess trial assessed the association of veliparib plus carboplatin or carboplatin alone to neoadjuvant chemotherapy. Combination of paclitaxel, carboplatin, and veliparib did not achieve a higher pCR when compared to paclitaxel and carboplatin (53% vs 58%, P = .36). 49 Subgroup analysis stratifying pCR by germline BRCA mutation found no difference in favour of gBRCA mutation carriers (51% vs 48%). 49 Long-term outcomes presented in ESMO 2021 showed that at a median follow-up of 4.5 years after surgery, pCR and EFS were not improved by the addition of veliparib. 60
Both OlympiAD and I-SPY 2 led to the design of a trial to assess olaparib plus standard neoadjuvant chemotherapy in early breast cancer. Multicentric phase II GeparOLA trial explored the combination of neoadjuvant chemotherapy plus olaparib or carboplatin, followed by epirubicin/cyclophosphamide in HRD nonmetastatic HER2-negative breast cancer. About 55.1% on paclitaxel plus olaparib versus 48.6% on paclitaxel plus carboplatin achieved pCR. Subgroup stratification showed that TNBC patients had a pCR rate of 56.0% with olaparib and of 59.3% with carboplatin.62,63
A similar rate of pCR was observed for the use of PARPi for TNBC neoadjuvant approach both in BrighTNess and GeparOLA trials (53% vs 56%). However, monotherapy with PARPi in metastatic setting was assessed for patients with gBRCA1/2 mutation regardless of HR status and reported an improvement of overall response- and progression-free survival compared to nonplatin regimens. 51 Results from the GeparOLA phase II study did not show evidence of benefit in pCR for the combination of olaparib plus conventional chemotherapy. 51
Recent data have been published regarding the combination of olaparib plus platinum-based neoadjuvant regimens, with PARTNER phase II/III trial randomizing patients to neoadjuvant carboplatin AUC5 plus paclitaxel with or without olaparib 150 mg during 4 cycles followed by physician’s choice of anthracyclines regimen. Preliminary data on safety supports the combination, with the most common adverse events (AEs) grade ⩾ 3 being haematological. Neutropenia was reported in 19% with febrile neutropenia in 2% of patients. Other haematological AE reported were anaemia (15%) and thrombocytopenia (5%). Fatigue (7%), hypertension (3%), headache (3%), and diarrhoea (2%) were the most common grade ⩾ 3 nonhaematological AEs. 64
The PETREMAC trial is a large phase II clinical trial evaluating the use of several treatment options. Olaparib monotherapy in 32 treatment-naïve TNBC patients had a high overall response rate (56%). 65
Talazoparib has been evaluated on the neoadjuvant setting since 2017, when the first feasibility study for early breast cancer with germline BRCA mutation was performed. 62 The highest response rates and favourable toxicity profile led to the modification to a phase II design to assess pCR after 4 to 6 months of single-agent talazoparib. Litton et al designed a trial to assess pCR to monotherapy with talazoparib for 6 months in germline BRCA-positive patients. Twenty patients underwent 6 months of daily talazoparib followed by surgery, with TNBC patients achieving a pCR rate of 57% (95% CI: 29%-82%). 66
Results from a phase II, nonrandomized, open-label trial on the efficacy plus safety of talazoparib in the neoadjuvant setting for early gBRCA1/2-mutated HER2-negative breast cancer was terminated in September 2020. Forty-eight TNBC patients were treated with 24 weeks of neoadjuvant talazoparib monotherapy 1 mg/day followed by surgery, achieving a pCR of 46%. 67 Efficacy data of niraparib monotherapy in the neoadjuvant setting was shown in a phase I clinical trial where 15 patients with somatic or germline BRCA-mutated TNBC received niraparib 200 mg once daily during 2 months before surgery plus additional cycles of niraparib up to 6 months after surgery or neoadjuvant chemotherapy at treating physician’s discretion. Fifteen patients performed magnetic resonance imaging (MRI) and breast ultrasound after 2 months of treatment, with a response rate of 89% by MRI. 68
Conclusion
Although evidence for the use of PARP inhibitors in the neoadjuvant setting is not totally established, current trials suggest a role for neoadjuvant PARP inhibition in germline BRCA-mutated patients. GeparOLA trial indicates that intensity de-escalation with olaparib instead of carboplatin maybe an option for germline BRCA-mutated patients. Furthermore, results from PREMETRAC trial suggest a possible benefit for the use of PARP inhibitors in treatment-naïve TNBC patients, which are corroborated by Litton et al. phase II clinical trial that showed comparable pCR rates between 24 weeks of monotherapy with talazoparib and standard chemotherapy regimens for germline BRCA-mutated TNBC patients. Therefore, PARP inhibitors may represent an option in the neoadjuvant setting for treatment-naïve or minimally treated germline BRCA-mutated TNBC patients.
Neoadjuvant Immunotherapy for TNBC
Despite breast cancer being classified as a non-immunogenic cancer, TNBC IM subtype has characteristics like tumour-infiltrating lymphocytes (TILs), expression of immune evasion molecules (PD-L1) and genomic instability that turns this subtype into a ‘hot’ and immunogenic tumour. 20 Immune-checkpoint inhibitors have shown to improve outcomes compared to standard chemotherapy alone in the metastatic setting. Despite combination of atezolizumab plus paclitaxel did not improve PFS or OS in IMpassion131 clinical trial, results from KEYNOTE-355 and IMpassion130 led to the approval of the combination of conventional chemotherapy and ICIs as first-line treatment for PD-L1 ⩾ 10% and PD-L1 ⩾ 1% metastatic TNBC, respectively.69 -71 Following these results in metastatic setting and the evidence that immune escape exponential increases with disease progression, several randomized trials have been studying the use of neoadjuvant chemo-immunotherapy.72,73
Several clinical trials have been published recently, supporting the combination of ICI to standard neoadjuvant chemotherapy as a promising option in the neoadjuvant setting for TNBC (Table 4).
Neoadjuvant TNBC clinical trials with immunotherapy.

Abbreviations: AC, doxorubicin + cyclophosphamide; Cp, carboplatin; DFS, disease-free survival; EC, epirubicin + cyclophosphamide; FEC, 5-fluorouracil, epirubicin + cyclophosphamide; NabT, nab-paclitaxel; pCR, pathological complete response; T, paclitaxel; TNBC, triple-negative breast cancer.
In KEYNOTE 522, TNBC patients were randomized to 4 cycles of pembrolizumab, weekly paclitaxel followed by AC. Final estimated pCR for standard neoadjuvant chemotherapy versus pembrolizumab was 22% vs 60%, respectively. 75 Event-free survival was recently reported by Schmid et al, 78 with an estimated EFS of 84.5% (95% CI: [81.7, 86.9]) for pembrolizumab plus chemotherapy and of 76.8% (95% CI: [72.2, 80.7]) for the placebo-chemotherapy group at 36 months (HR for event or death: 0.63; 95% CI: [0.48, 0.82]; P < .001).
Pembrolizumab was further evaluated in a phase III trial that randomly assigned (2:1 ratio) stage II/III TNBC patients to receive neoadjuvant therapy with 4 cycles of pembrolizumab (200 mg), paclitaxel plus carboplatin or placebo each 3 weeks plus paclitaxel and carboplatin. Combination of pembrolizumab plus chemotherapy achieved a pCR of 64.8% (95% CI: [59.9, 69.5]) versus 51.2% (95% CI: [44.1, 58.3]) for the placebo-chemotherapy arm. 74 Pathological complete response rate was significantly higher with the combination of immunotherapy plus neoadjuvant chemotherapy in the TNBC subgroup.
GeparNuevo is a phase II double-blind placebo-controlled trial, stratifying TNBC patients by stromal TILS, to durvalumab/placebo every 4 weeks plus nab-paclitaxel followed by epirubicin/cyclophosphamide. In both arms, pCR rate was highest for PD-L1-positive tumours, with 53.4% of pCR (95% CI: [42.5%, 61.4%]) for durvalumab and 44.2% (95% CI: [33.5%, 55.3%]) for placebo. 73 NeoTRI-PaPDL1, a phase III trial, evaluated the combination of neoadjuvant carboplatin AUC2, nab-paclitaxel with or without atezolizumab followed by adjuvant anthracycline regimen with EFS as primary endpoint. In the intention to treat (ITT) population, addition of atezolizumab resulted in a pCR of 48.6% versus 44.4% (OR 1.18; 95% CI: [0.74, 1.89]; p = .48) in the chemotherapy only arm. 76 However, NeoTRIPaPDL1 was the only trial adopting a nonanthracycline-containing neoadjuvant regimen and not having pCR as primary endpoint.
IMpassion 031, phase III trial, compared efficacy and safety of atezolizumab 840 mg/placebo each 2 weeks plus nab-paclitaxel 125 mg/m² weekly up to 12 weeks followed by doxorubicin 60 mg/m² plus cyclophosphamide at 600 mg/m² each 14 days during 8 weeks in the neoadjuvant setting. A difference of 17% in pCR rate (95% CI: [6, 27]; p = .0044) was seen in favour of atezolizumab plus chemotherapy. 77
Efficacy of atezolizumab in the neoadjuvant setting is being further evaluated on the GeparDouze clinical trial. A phase III, placebo-controlled trial that randomized TNBC patients to receive atezolizumab/placebo 1200 mg IV every 3 weeks concurrently with both sequential regimens of weekly paclitaxel 80 mg/m2 for 12 doses with every 3 weeks carboplatin AUC5 (4 cycles maximum) followed by AC or EC every 2 to 3 weeks for 4 cycles. Adjuvant therapy resumes atezolizumab/placebo 1200 mg each 21 days for 6 months. 79
Tarantino et al. performed a systematic review and meta-analysis evaluating the main clinical trials randomizing TNBC patients to neoadjuvant chemo-immunotherapy regimens. Results confirmed that combination of PD-L1 blockade plus neoadjuvant chemotherapy improves pCR rates. Moreover, in line with NeoTRIPaPDL1 trial, anthracycline-based regimens appeared to be the most efficient strategy to enhance tumour immunogenicity. TONIC is an adaptative, noncomparative, phase II trial that randomized metastatic TNBC patients with less than 4 lines of palliative chemotherapy to 1 of 5 arms: (1) 24(3 × 8) Gy irradiation of one metastatic lesion; (2) doxorubicin 15 mg weekly for 2 weeks; (3) cyclophosphamide 50 mg daily orally; (4) cisplatin 40 mg/m2 weekly for 2 weeks; (5) no induction. Following this induction period, all received nivolumab 3 mg/kg until RECIST 1.1 progression. 80 Main results showed that short-term chemotherapy or irradiation followed by nivolumab lead to clinical benefit with higher durable response rates. Chemotherapy regimens based on doxorubicin or cisplatin had higher response rate to nivolumab. 81
Conclusion
KEYNOTE-522 results led to a fast change in clinical practice, supporting US Food and Drug Administration (FDA) approval for the combination of pembrolizumab plus standard chemotherapy for neoadjuvant treatment of high-risk, early-stage TNBC patients with CPS ⩾ 1. Despite this approval, evidence is lacking for the selection of responders to upfront pembrolizumab. KEYNOTE-522 subgroup analysis showed a consistent benefit regardless age, performance status, chemotherapy schedule, tumour size, PD-L1, or nodal status.
Regarding the results of KEYNOTE-522, combination of pembrolizumab plus standard chemotherapy followed by adjuvant pembrolizumab should be considered for high-risk, early-stage TNBC fit for the combination. Future investigation on predictive biomarkers to better tailor the use of chemo-immunotherapy and the role of neoadjuvant immunotherapy for oestrogen receptor low patients is needed.
Emerging Therapies for TNBC
New molecules aiming to provide new therapeutic options and tailored approaches are under investigation in the neoadjuvant setting for TNBC patients.
AR targeting in TNBC
Androgen receptor expression occurs in 10% to 40% of TNBC, and its targeting is an interesting therapeutic option because LAR subtype has been associated with better prognosis, less chemotherapy sensitivity, and lower pCR after neoadjuvant treatment. 82 Inhibition of AR was first studied in the metastatic setting for AR-positive TNBC on a phase II clinical trial where patients received enzalutamide 160 mg id until disease progression with clinical benefit. 83 Also in the metastatic setting, abiraterone demonstrated a 6.7% response rate with 2.8 months of PFS and a clinical benefit rate of 20% for AR-positive TNBC over 6 months. 84 Despite not approved, bicalutamide, enzalutamide and abiraterone might offer a chemo-free alternative for chemo-resistant patients with TNBC LAR subtype.
Identifying chemotherapy-resistant TNBC patients during neoadjuvant treatment is being assessed by the phase II ARTEMIS trial, where patients were randomized to 4 cycles of doxorubicin-based neoadjuvant chemotherapy. For chemotherapy-insensitive TNBC, patients were offered the possibility to be randomized to one of the single-arm molecular profile defined clinical trials. From the 15 patients with TNBC LAR subtype offered enzalutamide (ZT) 160 or 120 mg daily plus paclitaxel 80 mg/m2 weekly (maximum 12 cycles), 5 showed response (NCT02276443).
Phosphatidylinositol-3-kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR)-targeted therapies
PI3K/AKT/mTOR signalling pathway represents the most activated cancer driver with PI3KCA, PTEN inactivating, and AKT1 activating mutations occurring in 25% of TNBC.85 -87
One arm in the I-SPY 2 trial assessed the addition of AKT inhibitor (MK2206) to weekly paclitaxel followed by AC. In the TNBC subgroup, estimated pCR rate was 40% for the AKT inhibitor and 22% for chemotherapy alone.62,88 Addition of Everolimus 30 mg to paclitaxel was compared to paclitaxel, 5-fluorouracil, epirubicin and cyclophosphamide every 3 weeks for 4 cycles in a phase II clinical trial, not statistically improving pCR rate. 87
The FAIRLANE clinical trial aimed to evaluate the addition of ipatasertib to taxane-based regimens, with ipatasertib achieving 17% of pCR rate versus 13% in the placebo arm for the ITT population. Subgroup analysis showed that pCR rate was 16% in the PTEN-low group with ipatasertib (versus 13% without) and 18% (versus 12% without) in the PIK3CA/AKT1/PTEN-altered subgroup. Addition of ipatasertib to paclitaxel did not statistically significantly improve pCR but was higher for the biomarker-selected subgroup. 89
Epidermal growth factor (EGFR) inhibitors
Epidermal growth factor receptor is expressed in 45% to 70% of TNBC, playing an important role in TNBC and being associated with poor prognosis.90 -92 Epidermal growth factor receptor mutations are rarely observed in this subgroup, and its inhibition appears as an interesting option with several anti-EGFR monoclonal antibodies and tyrosine kinase inhibitors (TKIs) being developed. 93
Epidermal growth factor receptor inhibitors were first explored in the metastatic setting, with phase II clinical trials assessing the addition of gefitinib to standard treatment for hormone receptor positive or HER2-positive metastatic breast cancer (MBC). Arteaga et al. 94 designed a phase I-II trial assessing the addition of gefitinib to weekly trastuzumab in HER2 positive MBC, without clinical benefit comparing to monotherapy with trastuzumab. Ciardiello et al., assessed addition of gefitinib with docetaxel as first-line therapy for MBC in a phase II clinical trial. Final analysis suggested that the combination could be potentially active in untreated MBC patients. 95
The addition of cetuximab to docetaxel in the neoadjuvant setting was evaluated in a multicentric phase II trial for operable stage II–III TNBC patients, achieving a pCR of 22%. 96
Panitumumab was first considered in a 2014 multicentric trial. This multicentric phase II pilot study combined panitumumab with anthracycline-taxane-based chemotherapy in stage II-III TNBC patients with a 46.8% of pCR. 97 A more recent single-arm phase II trial reviewed the impact of combining panitumumab plus neoadjuvant chemotherapy achieving a pCR of 42% (95% CI: [20%, 66%]). 98
Women’s triple-negative first-line study, an ongoing phase II clinical trial, assessed the role of panitumumab in patients with chemo-insensitive TNBC (NCT02593175).
Anti-vascular endothelial growth factor therapies
Bevacizumab and apatinib are the most relevant anti-VEGF in the neoadjuvant setting for TNBC. Five trials evaluated the role of bevacizumab in the neoadjuvant setting, with a meta-analysis confirming the benefit of combining bevacizumab with standard chemotherapy. Improve in pCR rate was not reflected in DFS or definitive OS. 99
In 2020, a phase II multicentric trial randomized 17 stage IIB-IIIC TNBC patients to apatinib 500 mg daily or placebo added to standard chemotherapy for 6 cycles every 21 days. An increase of 22% in pCR rate was reported (72.7% versus 50%; p = .600). 100
Other agents and drug conjugates
Eribulin, a microtubule inhibitor, has been studied as a potential alternative to taxane-based regimens. As a microtubule inhibitor, eribulin inhibits microtubule growth and disrupts mitosis. A multicenter randomized phase II trial stratified by HRD status assessed the impact of adding eribulin to neoadjuvant chemotherapy. For high HRD score or germline BRCA-mutated TNBC, addition of eribulin to weekly paclitaxel plus carboplatin followed by anthracycline regimen resulted in a pCR rate of more than 40%. 101
Sacituzumab govitecan (SG), an antibody-drug conjugate, was first evaluated in the metastatic setting for second and subsequent lines. Results from the ASCENT Trial showed an increase in OS from 6.7 months for the single-agent chemotherapy group to 12.1 months with SG (HR: 0.48; 95% CI: [0.38, 0.59]; p < .001). 102 Based on this evidence, the role of SG in the early setting is being assessed by the NeoSTAR trial for neoadjuvant treatment and SASCIA trial in patients with residual disease after neoadjuvant therapy. The NeoSTAR clinical trial evaluates SG 10 mg/kg (days 1, 8 of each 21 day cycle) in the neoadjuvant setting. After 4 cycles, if biopsy-proven residual disease exist, patients will be allowed to receive standard neoadjuvant therapy at physician’s discretion (NCT04230109). 103
Post-neoadjuvant strategies
Despite lack of solid evidence on the impact of neoadjuvant chemotherapy on disease-free or OS, this strategy enables a reduction on tumour burden and in vivo assessment of chemosensitivity. Moreover, rate of pathological response after neoadjuvant treatment has been consolidated as a surrogate for oncological outcomes. Assessment of pathological response allows the stratification of patients regarding the need of treatment intensification or de-intensification in the adjuvant setting. Currently, 3 major strategies have been assessed – chemotherapy, immunotherapy, and PARPis.
CREATE-X, a randomized phase III clinical trial, proved the significant improvement on both DFS and OS with post-neoadjuvant capecitabine for TNBC patients versus control group at 5 years follow-up (DFS: 74.1% vs. 67.6%; OS: 89.2% vs. 83.6%; p = .001). 15 The benefit of platinum agents in the adjuvant setting for stage II/III TNBC patients with residual disease was evaluated in the ECOG-ACRIN EA1131 phase III clinical trial, without improvement on DFS for cisplatin or carboplatin versus adjuvant capecitabine at 3 years follow-up (HR: 1.09; 95% CI: [0.62, 1.90]). 104
Increase on EFS at 36 months stated on the KEYNOTE-522 for the arm of pembrolizumab plus standard chemotherapy and adjuvant pembrolizumab, adds evidence to the potential benefit of ICIs in the adjuvant setting.
The multicentre phase III OlympiA trial demonstrated the benefit in mortality rate, disease, and distant-free survival (p = .02; p < .001, and p < .001) for 1 year of adjuvant olaparib for germline TNBC-mutated patients with residual disease after neoadjuvant treatment. 105
Results from these trials reinforce the value of neoadjuvant systemic treatment for TNBC, allowing for a personalized treatment strategy.
Conclusion and Future Directions
The growing clinical evidence on TNBC led to significant changes on management for both metastatic and neoadjuvant settings. Better definition and stratification of TNBC patients according to molecular characteristics allows for therapeutic tailoring and consequent improvements in response rates and survival outcomes. Recent clinical trials showing the benefit of adding ICI to neoadjuvant chemotherapy regimens led to clinical practice changes, reflecting the need to bring molecular classification of TNBC to the core of therapeutic decision. Stratification of TNBC patients according to molecular features, such as HRD, germline BRCA mutation or EGFR expression, will help to achieve better response rates to neoadjuvant treatment and improve oncological outcomes.
Future clinical trials designed to better define which subgroup of TNBC patients are best candidates to treatment with conventional cytotoxic agents or combination of cytotoxic agents plus targeted therapies are needed.
Footnotes
Author Contributions
DS: Literature review, manuscript writing, proof editing and submission.
AM: Conceptualization, supervision, manuscript review.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
