Abstract
A novel melatonin, estrogen, and progesterone hormone therapy was developed as a safe bio-identical alternative hormone therapy for menopausal women based on the Women’s Health Initiative findings that PremPro™ increased breast cancer risk and mortality of all types of breast cancer in postmenopausal women. For HER2 breast cancer, melatonin, estrogen, and progesterone delayed tumor onset and reduced tumor incidence in neu female mice. For other breast cancers, its actions are unknown. In this study, melatonin, estrogen, and progesterone hormone therapy were assessed in human ER+ (MCF-7) and triple negative breast cancer (MDA-MB-231) cells, and found to decrease proliferation and migration of both breast cancer lines. Inhibition of MEK1/2 and 5 using PD98059 and BIX02189, respectively, inhibited proliferation and migration in MDA-MB-231 cells and proliferation in MCF-7 cells; however, when combined with melatonin, estrogen, and progesterone, BIX02189 blocked melatonin, estrogen, and progesterone–mediated inhibition of migration in MCF-7 cells and induced Elf-5. For MDA-MB-231 cells, BIX02189 combined with melatonin, estrogen, and progesterone inhibited proliferation and increased pERK1/2 and β1-INTEGRIN; levels of pERK5 remained low/nearly absent in both breast cancer lines. These findings demonstrate novel anti-cancer actions of melatonin, estrogen, and progesterone in ER+ and triple negative breast cancer cells through intricate MEK1/2- and MEK5-associated signaling cascades that favor anti-proliferation and anti-migration.
Keywords
Introduction
A novel melatonin, estrogen, and progesterone hormone therapy (MEMP HT) was developed as an alternative bio-identical hormone therapy (HT) for menopausal women to relieve menopausal symptoms without increasing the risk of breast cancer (BC) shown to occur with conjugated equine estrogens (CEE) and medroxyprogesterone acetate (MPA).1,2 The MEMP HT, which contains 5 mg melatonin (M), 0.5 mg estradiol (E/E2), and half the recommended dose of progesterone (50 vs 100 mg; P/P4), decreased tumor incidence, delayed tumor onset, and protected the uterus in HER2/neu mice. 3 The MEMP’s anti-cancer actions against ER+ and triple negative breast cancer (TNBC) are not known but highly relevant based on the fact that PremPro increased myriad BCs that included ER+, HER2, and TNBC. 1
Melatonin has been incorporated in MEMP HT due to its multiple effects in organisms, including immunomodulatory, metabolic, geno-protective, anti-estrogenic, or direct antineoplastic activities. 4 Melatonin demonstrated pro-apoptotic, 5 anti-proliferative, 6 anti-metastatic, 7 anti-angiogenic, 8 or anti-oxidant effects 9 in different cancer models. The mechanisms underlying MEMP’s anti-cancer actions in neu mice demonstrate actions on the mitogen activated protein kinases (MAPKs), Mek1/2, and Mek5 in mammary (young and old) and tumor tissue; however, these actions are tissue-dependent (non-tumor mammary vs tumor) and time-dependent (young, 3 months vs old, 1 year).3,10 Mek1/2/5 and proteins that lay downstream from them (i.e., Runx2, NF-κB, Rankl, Elf-5, and β1-integrin) have been shown in past studies to be involved in BC by regulating proliferative and/or metastatic properties.11-20 Although MEMP HT’s anti-cancer actions in neu female mouse mammary were associated with Mek1/2- and Mek5-dependent pathways, 10 definitive studies regarding their involvement can only be achieved by knocking out, knocking down, or inhibiting Mek1/2 and 5. The involvement of MEK1/2 and 5 (and downstream proteins) in MEMP-mediated anti-cancer actions in MCF-7 and MDA-MB-231 cells was addressed by use of small molecule inhibitors selective for MEK1/2 or MEK5 and by using therapeutically relevant and equivalent concentrations of melatonin, 17β-estradiol (E2), and progesterone (P4)3,21 to determine whether and how MEMP HT affects ER+ and TNBC.
Materials and Methods
Radioligand binding
Melatonin receptor expression was assessed by total 2-[125I]-iodomelatonin binding described previously,22,23 and estrogen receptor (ER) expression was assessed by total [3H]-estradiol binding. Saturation and total binding analyses were conducted on whole cell lysates prepared from MCF-7 or MDA-MB-231 grown to confluence on 10 cm plates, then washed with 5 mL of phosphate-buffered saline (PBS), lifted into buffer (10 mM KPO4, 1 mM ethylenediaminetetraacetic acid [EDTA], pH 7.4), pelleted by centrifugation (277g, 5 minutes), and then resuspended in Tris-HCl buffer (1 mL; 50 mM, pH 7.4). To assess melatonin receptor expression, cells were added to tubes containing 2-[125I]-iodomelatonin (2200 Ci/mmol, Perkin Elmer, Waltham, MA, USA; 0-500 pM for saturation analysis and 500 pM for total binding assays) in the absence (total binding) or presence (non-specific binding) of 1 µM melatonin (Sigma, St. Louis, MO, USA). To assess ER expression, cells were added to tubes containing 3 nM [3H]-estradiol (94 Ci/mmol; Perkin Elmer) in the absence (total binding) or presence (non-specific binding) of 10 µM tamoxifen (Millipore Sigma, Burlington, MA, USA). Cells were incubated for 1 hour at room temperature and then terminated by the addition of ice-cold Tris-HCl solution (50 mM) and rapid filtration over glass-fiber filters (0.22 µm; Schleicher & Schuell, Keene, NH, USA) presoaked in 0.5% polyethyleneimine solution (v/v; Sigma). Each filter was washed twice with 5 mL of cold buffer. Filters were placed in glass vials (2-[125I]-iodomelatonin) or vials containing scintillation fluid ([3H]-estradiol) and then counted in a gamma counter or scintillation counter, respectively. Total protein measurements were made on cell lysates using the Pierce BCA protein assay. Saturation binding analysis was already performed for [3H]-estradiol binding in MCF-7 cells as described 24 and not performed in MDA-MB-231 cells due to a lack of [3H]-estradiol expression (Figure 1A).
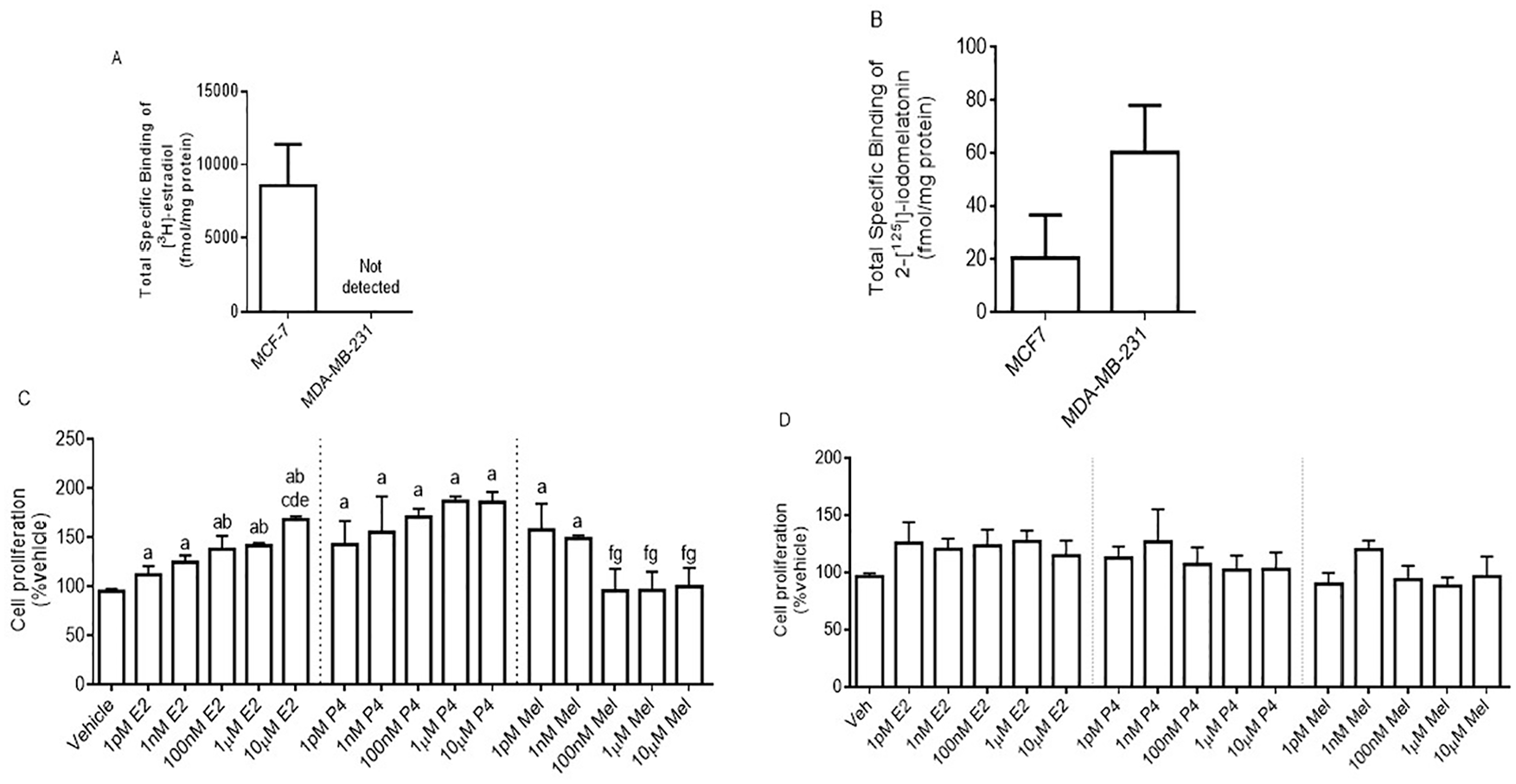
Melatonin and estrogen receptor binding in MCF-7 and MDA-MB-231 cells, and the effect of E2, P4, and melatonin on cell proliferation and migration. Total 2-[125I]-iodomelatonin and [3H]-estradiol binding to ERs (A) and melatonin receptors (B), respectively, expressed in MCF-7 and MDA-MB-231 cells. Cells were exposed to E2 (1 pM, 1 nM, 100 nM, 1 µM, and 10 µM), P4 (1 pM, 1 nM, 100 nM, 1 µM, and 10 µM), or Mel (1 pM, 1 nM, 100 nM, 1 µM, and 10 µM) for 24 hours and then counted using trypan blue in MCF-7 (C) and MDA-MB-231 (D) cells. Data represent the mean ± SEM from 3 independent experiments. Data were analyzed by one-way ANOVA followed by Newman-Keuls’ post hoc t-test, where a = P < .05 vs vehicle; b = P < .05 vs 1 pM E2; c = P < .05 vs 1 nM E2; d = P < .05 vs 100 nM E2; e = P < .05 vs 1 µM E2; f = P < .05 vs 1 pM Mel; g = P < .05 vs 1 nM Mel. ANOVA indicates analysis of variance; SEM, standard error of the mean.
Cell proliferation
MCF-7 and MDA-MB-231 cell lines, obtained from American Type Culture Collection (Manassas, VA, USA), were cultured in DMEM:F12 medium (Thermo Scientific, Pittsburgh, PA, USA) containing 10% fetal bovine serum (FBS; Atlanta Biologicals, Lawrenceville, GA, USA) and 1% penicillin/streptomycin (Sigma-Aldrich, St. Louis, MO, USA) at 37°C in a humidified atmosphere with 5% CO2. Cells were plated in 24-well plates (3000 cells/well) and cultured in DMEM:F12 supplemented with 10% FBS and 1% penicillin-streptomycin for 24 hours prior to the addition of the treatments (vehicle, 50 pM E2 + 10 nM P4, or 50 pM E2 + 10 nM P4 + 1 nM Mel). After 24 hours, cells were counted by trypan blue exclusion. Concentration effects of E2, P4, and melatonin individually (1 pM, 1 nM, 100 nM, 1 µM, 10 µM) were also tested to determine how each component of MEMP HT affected MCF-7 and MDA-MB-231 cell proliferation. For the inhibitor studies, cells were exposed to 10 µM BIX02189 (Santa Cruz Biotechnology, Dallas, TX, USA) or 10 µM PD98059 (Santa Cruz Biotechnology) alone or in combination with 50 pM E2 (Sigma), 10 nM P4 (Sigma), and 1 nM Mel (Sigma). Inhibitors were added 15 minutes before the addition of MEMP HT. The in vitro concentrations of E2, P4, and melatonin used in this study were based on serum levels of MEMP HT achieved in neu mice following consumption of 0.5 mg E2 and 50 mg P4 in the diet and 15 mg/L melatonin in water at night.3,21 These doses of melatonin, E2, and P4 demonstrated anti-cancer actions in neu female mice. 3
Migration or “scratch” assay
To investigate the effect of the various treatments on cell migration, the wound healing, or “scratch,” assay was employed as described. 24 Confluent cells from one 10 cm2 plate were lifted into 12 mL medium and were seeded into 24-well plates (0.5 mL/well) and grown overnight. A cell border was created using a 10 µL pipette tip that was dragged across the bottom of each well. Next, the medium from each well was removed, cells were washed once with 1× PBS, and then cells were re-fed with fresh medium (0.5 mL) containing the treatments described previously. Images of cell borders (Figures 2D and 3D) were taken at 0 hour and after 24 hours exposure to the treatments with an EVOS digital inverted fluorescence microscope (magnification 10×) under transmittance light. The wound area was quantified by measuring the bandwidth of the scratch at 0 hour (baseline) and following 24 hours of treatments as described. 24 A more negative number, indicated by a decrease in border width, would be indicative of a more invasive phenotype, whereas a more positive number, indicated by a wider border width, would be indicative of a less invasive phenotype.

Effect of MEMP HT, MEK1/2, and MEK5 on MCF-7 cell proliferation and migration. The MEMP HT and inhibitors were used on MCF-7 cell viability (A, B) and migration (C). The representative images of the scratches were given for each treatment 0 and 24 hours (D). The MEK1/2 inhibitor (10 µM PD98059) or the MEK5 inhibitor (10 µM BIX02189) was added to the treatments. For migration assays, % border width ([24 hours – 0 hours] / 0 hours) is represented on the y-axis, and for the proliferation assays, % of living cells relative to the total number of cells counted is represented on the y-axis. Data were analyzed by one-way ANOVA followed by Newman-Keuls’ post hoc t-test where significance was defined as P < .05. Each bar represents the mean ± SEM from 3 independent experiments; a = P < .05 vs vehicle; b = P < .05 vs 50 pM E2 + 10 nM P4. ANOVA indicates analysis of variance; MEMP HT, melatonin, estrogen, and progesterone hormone therapy; SEM, standard error of the mean.

Effect of MEMP HT, MEK1/2, and MEK5 on MDA-MB-231 cell proliferation and migration. The MEMP HT and inhibitors were used on MDA-MB-231 cell viability (A, B) and migration (C). The MEK1/2 inhibitor (10 µM PD98059) or the MEK5 inhibitor (10 µM BIX02189) was added to the treatments. The representative images of the scratches were given for each treatment 0 and 24 hours (D). For migration assays, % border width ([24 hours – 0 hours] / 0 hours) is represented on the y-axis, and for the proliferation assays, % of living cells relative to the total number of cells counted is represented on the y-axis. Data were analyzed by one-way ANOVA followed by Newman-Keuls’ post hoc t-test where significance was defined as P < .05. Each bar represents the mean ± SEM from 3 independent experiments; a = P < .05 vs vehicle; b = P < .05 vs 10 µM BIX02189; d = P < .05 vs MEMP HT. ANOVA indicates analysis of variance; MEMP HT, melatonin, estrogen, and progesterone hormone therapy; SEM, standard error of the mean.
Western blot
To assess potential intracellular targets underlying MEMP HT action in MCF-7 and MDA-MB-231 cells, western blot analysis was performed analyzing pERK1/2, pERK5, NF-κB, RUNX2, β1-INTEGRIN, Elf-5, and RANKL levels. MCF-7 and MDA-MB-231 cells were exposed to the treatments described previously for 24 hours and then scraped into Laemmli sample buffer containing β-mercaptoethanol and stored at −20°C until use. Equal amounts of each sample (25 µL) and 8 µL of molecular weight marker (Precision Plus ProteinTM, CAT# 161-0373, BioRad, Hercules, CA, USA) were loaded onto 10% SDS-PAGE (sodium dodecyl sulfate–polyacrylamide gel electrophoresis) gels. Proteins were transferred to nitrocellulose membranes and then incubated in blocking buffer (LI-COR Biosciences, Lincoln, NE, USA) for 30 minutes at room temperature. Next, membranes were incubated with primary antibodies at 4°C overnight followed by washing (4×) and incubation with secondary antibodies for 15 minutes at room temperature. Primary antibodies included rabbit anti-phospho-ERK1/2 (1:1000, Cell Signaling, Danvers, MA, USA), rabbit anti-phospho-ERK5 (T218/Y220; 1:1000, Cell Signaling), rabbit anti-NF-κB (p52; sc-298; 1:1000; Santa Cruz Biotechnology), rabbit anti-Runx2 (sc10758; 1:1000, Santa Cruz Biotechnology), rabbit anti-integrin β1 (M-106; sc-8978; 1:1000: Santa Cruz Biotechnology), rabbit anti-RANKL (sc-9073; 1:1000; Santa Cruz Biotechnology), rabbit anti-Elf-5 (ab104410; 1:500; Abcam, Cambridge, MA, USA), and mouse anti-β-actin (926-42212, LI-COR Biosciences). Secondary antibodies included goat anti-mouse (1:20 000; LI-COR Biosciences; IR680LT dye) or goat anti-rabbit (1:20 000, LI-COR Biosciences; IR800CW dye). Protein bands were analyzed using fluorescent microscopy detection at 700 (β-actin) and 800 (all other proteins) channels on an Odyssey Infrared Imaging System. Proteins were normalized by β-actin to control for protein loading.
Statistical analysis
All statistical analyses were performed using GraphPad Prism (version 6; GraphPad Software, La Jolla, CA, USA). Each bar graph represents the mean ± SEM (standard error of the mean) from 3 independent studies. Data were analyzed by one-way analysis of variance (ANOVA) followed by Newman-Keuls’ multiple comparisons test and significance was defined as P < .05.
Results
Effect of E2, P4, and melatonin on cell proliferation and migration
MCF-7 cells, ER+/PR+ epithelial BC cells that also contain melatonin receptors, were exposed to each of the hormones (E2, P4, melatonin) to determine their effects on cell proliferation. As shown in Figure 1, MCF-7 cells expressed ERs at a level of 8.6 pmol/mg protein (Figure 1A) with an affinity (Kd) of 2.2 nM, 24 and melatonin receptors at a level of 20.55 fmol/mg protein (Figure 1B) and affinity (Kd) of 13 pM (data not shown). A concentration-dependent stimulatory effect of E2 occurred on MCF-7 cell proliferation, whereas a concentration-dependent inhibitory effect of melatonin was observed (Figure 1C). P4 also increased MCF-7 cell proliferation compared with the vehicle (Figure 1C). MDA-MB-231, ER-/PR-/HER2-epithelial BC cells, expressed melatonin receptors at a level of 60.09 fmol/mg protein (Figure 1B) and an affinity (Kd) of 1.7 nM (data not shown). No [3H]-estradiol binding sites were detected in MDA-MB-231 cells (Figure 1A) and no concentration-dependent actions of E2, P4, or melatonin on proliferation in MDA-MB-231 cells were observed (Figure 1D).
Effect of MEMP HT, MEK1/2, and MEK5 on MCF-7 cell proliferation and migration
MEMP HT decreased MCF-7 cell proliferation (Figure 2A) and migration (Figure 2C and D) compared with the vehicle. To further clarify the role of MEK1/2 and MEK5 on MEMP HT-mediated breast cancer protection, MCF-7 cells were exposed to MEMP HT in the presence or absence of MEK1/2 or 5 inhibitors using PD98059 or BIX02189, respectively. Exposure to PD98059 or BIX02189 alone decreased MCF-7 cell proliferation, which was without further effect when combined with MEMP HT (Figure 2B). For migration, both PD98059 and BIX02189 alone were without effect and similar to the vehicle; however, when combined with MEMP HT (Figure 2C and D), BIX02189 but not PD98059 blocked MEMP HT’s effect on cell migration.
Effect of MEMP HT, MEK1/2, and MEK5 on MDA-MB-231 cell proliferation and migration
MEMP HT decreased MDA-MB-231 cell proliferation (Figure 3A) and migration (Figure 3C and D) compared with the vehicle. PD98059 and BIX02189 alone decreased cell proliferation compared with the vehicle (Figure 3B). The addition of PD98059, but not BIX02189, with MEMP HT, further decreased MDA-MB-231 proliferation (Figure 3B). With respect to migration, MEMP HT alone, PD98059 alone, and BIX02189 alone decreased cell migration compared with the vehicle; however, when combined with MEMP, no further decreases or blockade occurred (Figure 3C and D).
Effect of MEMP HT on pERK1/2, pERK5, NF-κB, β1-INTEGRIN, Elf-5, and RANKL in MCF-7 cells
As shown in Figure 4, MEMP HT alone increased pERK5 (Figure 4B) compared with the vehicle with no other effects observed for the other proteins. Acute (1 hour) or chronic (24 hours) exposure to PD98059 decreased pErk1/2 levels as expected (Figure 4A and inset). PD98059 combined with MEMP HT decreased pERK5 and increased β1-INTEGRIN vs MEMP HT alone; β1-INTEGRIN levels also increased vs all other groups tested (Figure 4D). BIX022189 given alone for 1 hour (Figure 4B inset) or 24 hours (Figure 4B) trended toward a decrease in pERK5 and increased NF-κB (Figure 4C) with no other effects occurring on the other proteins. When BIX02189 was combined with MEMP HT, pERK5 levels decreased compared with vehicle (Figure 4B) and BIX02189-mediated increases in NF-κB were attenuated (Figure 4C). No effects occurred for Elf-5 (Figure 4E) and RANKL (Figure 4F) regarding any treatments. Representative band images were shown in Table 1. RUNX2 was not tested because this protein is not expressed in MCF-7 cells. 11

Treatment effects on pERK1/2, pERK5, NF-κB, β1-INTEGRIN, Elf-5, and RANKL expression in MCF-7 cells. MCF-7 cells were exposed to vehicle or MEMP HT for 24 hours and their effects on pERK1/2 (A), pERK5 (B), NF-κB (C), β1-INTEGRIN (D), Elf-5 (E), or RANKL (F) expression in MCF-7 cells by western blot analysis. The MEK1/2 inhibitor (10 µM PD98059) or the MEK5 inhibitor (10 µM BIX02189) was added to the treatments. Treatment groups are denoted on the x-axis and protein expression (normalized by β-actin) on the y-axis. Data were analyzed by one-way ANOVA followed by Newman-Keuls’ post hoc t-test where significance was defined as P < .05; a = P < .05 vs vehicle; b = P < .05 vs 10 µM BIX02189; c = P < .05 vs 10 µM PD98059; d = P < .05 vs MEMP HT; e = P < .05 vs MEMP HT + 10 µM BIX02189. Each bar represents the mean ± SEM from 3 independent experiments (please see Table 1 for band images). ANOVA indicates analysis of variance; MEMP HT, melatonin, estrogen, and progesterone hormone therapy; SEM, standard error of the mean.
Representative protein band images of pERK1/2, pERK5, nuclear factor NF-kappa-B, beta 1 integrin, Elf-5, and RANKL in MCF-7 cells.

Abbreviation: MEMP HT, melatonin, estrogen, and progesterone hormone therapy.
Each protein band is representative of 3 independent experiments. Beta-actin bands were used as the loading control.
Effect of MEMP HT on pERK1/2, pERK5, NF-κB, RUNX2, β1-INTEGRIN, and Elf-5 expression in MDA-MB-231 cells
As shown in Figure 5, MEMP was without effect on any of the proteins analyzed when compared with the vehicle; however, effects were observed when combined with MEK1/2 or MEK5 inhibitors. Acute (1 hour) exposure to PD98059 reduced pERK1/2 vs vehicle (Figure 5A inset); however, when given chronically for 24 hours, levels of pERK1/2 increased and were similar to vehicle (Figure 5), and levels of pERK5 (Figure 5B) and RUNX2 (Figure 5G) also increased vs vehicle. BIX029189 alone (1 hour exposure) decreased pERK5 (Figure 5B inset); however, levels increased and were similar to the vehicle when given chronically for 24 hours (Figure 5B). PD98059 combined with MEMP increased pERK1/2 vs BIX02189 alone and MEMP alone (Figure 5A) and increased β1-INTEGRIN vs vehicle, PD98059 alone, and MEMP alone (Figure 5D). MEMP combined with PD98059 also decreased pERK5 (Figure 5B) and RUNX2 (Figure 5G) induced by PD98059 alone. BIX02189 alone (24 hours) was without effect on any of the proteins examined; however, when BIX02189 was combined with MEMP, levels of pERK1/2 (Figure 5A), β1-INTEGRIN (Figure 5D), and Elf-5 (Figure 5E) increased vs vehicle, BIX02189 alone, and MEMP alone. No effects occurred for RANKL regarding any treatments (Figure 5F), and representative band images can be found in Table 2.

Treatment effects on pERK1/2, pERK5, NF-κB, β1-INTEGRIN, Elf-5, RANKL, and RUNX2 expression in MDA-MB-231 cells. MDA-MB-231 cells were exposed to vehicle or MEMP HT for 24 hours and its effects on pERK1/2 (A), pERK5 (B), NF-κB (C), β1-INTEGRIN (D), Elf-5 (E), RANKL (F), or RUNX2 (G) expression in MDA-MB-231 cells by western blot analysis. The MEK1/2 inhibitor (10 µM PD98059) or the MEK5 inhibitor (10 µM BIX02189) was added to the treatments. Treatment groups are denoted on the x-axis and protein expression (normalized by β-actin) on the y-axis. Data were analyzed by one-way ANOVA followed by Newman-Keuls’ post hoc t-test where significance was defined as P < .05; a = P < .05 vs vehicle; b = P < .05 vs 10 µM BIX02189; c = P < .05 vs 10 µM PD98059; d = P < .05 vs MEMP HT; e = P < .05 vs MEMP HT + 10 µM BIX02189. Each bar represents the mean ± SEM from 3 independent experiments (please see Table 2 for band images). ANOVA indicates analysis of variance; MEMP HT, melatonin, estrogen, and progesterone hormone therapy; SEM, standard error of the mean.
Representative protein band images of pERK1/2, pERK5, nuclear factor NF-kappa-B, beta 1 integrin, Elf-5, RANKL, and RUNX2 in MDA-MB-231 cells.

Abbreviation: MEMP HT, melatonin, estrogen, and progesterone hormone therapy.
Each protein band is representative of 3 independent experiments. Beta-actin bands were used as the loading control.
Discussion
The Women’s Health Initiative study on PremPro in postmenopausal women demonstrated that BC risk and mortality increased for all types of BC.1,2,25 The BC risk did not increase with the estrogen-only HT, Premarin, which suggested that the progestin, MPA, in PremPro, was involved.26,27 In response to these findings, a novel MEMP HT containing bio-identical hormones (5 mg melatonin, 0.5 mg E2, 50 mg P4) was developed with the dose of progesterone halved to decrease the development of BC that was potentially associated with progestin exposure. The MEMP HT demonstrated anti-cancer and uteroprotective actions in HER2/neu female mice, 3 which was associated with a modulation of progesterone receptor A (PRA) (producing a higher PrA to PrB ratios) as well as Mek1/2- and Mek5-associated proteins like Runx2, NF-κB, Rankl, Elf-5, and β1-integrin. These effects of MEMP HT in neu female mice were dependent upon age (young, 3 months vs old, 1 year) and tissue (non-tumor mammary vs tumor).3,10 Although MEMP HT demonstrated anti-cancer actions against HER2 BC, the effect of and mechanisms underlying MEMP HT actions on ER+ and TNBC are not known and formed the basis for the present study.
In MCF-7 and MDA-MB-231 BC cells, MEMP HT decreased proliferation and migration, which is consistent with what was found for HER2/neu mammary cancer in female mice. 3 Melatonin decreased the invasiveness of HER2-positive SKBR-3 cells and inhibited lung metastasis in orthotopic HER2-positive BC xenografts. 28 The melatonin component of MEMP HT is more than likely contributing to these anti-proliferative and anti-migratory actions because melatonin is widely known for its anti-cancer actions in numerous studies as reported in BC research.4,20 Also, melatonin given to athymic “nude” mice transplanted with MDA-MB-231 cells demonstrated a reduction in tumor cell proliferation, tumor growth, and angiogenesis compared with animals given vehicle. 29 Melatonin also has been shown to inhibit proliferation and metastatic properties of tumor cells grown in culture30-33 and counteract estrogen-induced invasiveness of MCF-7 cells34,35 possibly through melatonin-induced ERα down-regulation.35,36 Melatonin decreased viability and migration in MDA-MB-231 cells. 32 These findings suggest that melatonin may be working to decrease proliferation and migration through its own signaling cascades (eg, MAPKs) but may also be countering the pro-stimulatory actions of E2 and P4 on MAPK pathways as shown.37-40
These anti-cancer actions of MEMP HT may be attributed to the melatonin component working through melatonin receptors in both BC lines, particularly MT1, as demonstrated in MCF-7 cells.35,36,41 Melatonin, acting through melatonin receptors, also prevented tumor growth in an MCF-7 xenograft model. 42 Melatonin receptor expression also demonstrates a strong correlation with TNBC metastases and mortality where tumors that did not express MT1 melatonin receptors had a significantly higher hazard ratio for disease progression, shorter progression-free survival, and disease-related death compared with those that expressed MT1 melatonin receptors. 43 Although melatonin receptor expression was 3 times higher for MDA-MB-231 cells compared with MCF-7 cells, melatonin inhibited MCF-7 proliferation but was without effect in MDA-MB-231 cells. These variable effects of melatonin in MDA-MB-231 cells have been demonstrated previously29,44 and may be due to other variables within the BC cells like intracellular signaling networks (eg, MAPKs) that influence cell proliferation and migration. 20 For example, MCF-7 cells are luminal type A and so they express ERs and PRs but not HER2,45-47 whereas MDA-MB-231 cells are triple negative devoid of ERs, PRs, and HER2. 47 MDA-MB-231 cells also express higher basal levels of pERK1/2 compared with MCF-7 cells. 24 These intracellular differences may underlie, in part, their BC characteristics where MCF-7 cells are less proliferative and invasive than MDA-MB-231 cells indicated by low Ki67 levels in MCF-7 compared with MDA-MB-231. 46
Melatonin combined with E2 and P4 as in MEMP HT inhibited MDA-MB-231 cell proliferation and migration suggesting that melatonin may be working in concert with E2 and P4 through melatonin receptor–independent pathways (ie, anti-oxidant, free-radical scavenging) and/or through melatonin-mediated anti-oxidant actions on the mitochondria as shown previously.23,48,49 In MCF-7 cells, the presence of ERs and melatonin receptors may have contributed to the anti-proliferative and anti-migratory actions of MEMP HT through reciprocal modulation to enhance melatonin receptor function and decrease ER function. The melatonin contained within MEMP HT may have decreased the pro-stimulatory actions of E2 (via down-regulation of ERα) as shown,35,36 while the E2 contained within MEMP HT may have increased melatonin receptor activity in MCF-7 cells increasing its response to melatonin 50 and, therefore, MEMP HT’s anti-cancer actions in MCF-7 cells.
Elucidation of the mechanisms underlying MEMP HT effects in MCF-7 and MDA-MB-231 cell proliferation and migration is complicated and far from clear; however, most of the protein expression changes are induced by MEMP HT support anti-cancer actions and involvement of MAPK pathways. In MCF-7 cells, MEMP HT-mediated decreases in proliferation and migration (regardless of treatment) were associated with low pERK1/2; however, treatment-dependent effects were observed for pERK5, β1-INTEGRIN, and NF-κB. For MDA-MB-231 cells, decreases in proliferation and migration (regardless of treatment) were associated with increases in pERK1/2, pERK5, β1-INTEGRIN, Elf-5, and RUNX2. High ERK1/2 levels are shown to be linked with good BC disease outcomes.51,52 Low pERK5 is associated with anti-proliferative properties because high ERK5 is associated with pro-survival properties of cells.13,14 High Elf-5 is involved with epithelial-to-mesenchymal transition decreasing invasiveness 12 and motility. 53 High β1-INTEGRIN is associated with increased cell-to-cell contact, less transformation, and less aggressive invasive phenotype. High RUNX2 in MDA-MB-231 cells is not clear because high RUNX2 is associated with invasiveness 11 and poor prognosis 54 ; however, given the findings that RUNX2 lay downstream of MEK5 and that pERK5 is low following MEMP HT exposure suggests that RUNX2 effects are negligible.
The melatonin component within MEMP HT may be producing anti-proliferative and anti-migratory actions via melatonin’s actions on MAPK pathways, which has been demonstrated in myriad studies, including in cancer cells.31,55-60 Although it is widely reported that MAPKs function in cancer, how they function in cancer is not clear because the studies are mixed with respect to ERK1/2 expression and their association with BC.14,51,52,61 This may be due to the fact that intracellular localization of pERK1/2 (ie, cytosolic vs nuclear) was not accounted for in these studies, which is important considering that pERK1/2 (nuclear) is pro-proliferative while pERK1/2 (cytosolic) is pro-differentiation. 62 Because it has been demonstrated that estrogen increases nuclear localization of ERK1/2 in MCF-7 cells, 37 then, perhaps, melatonin is counteracting the E2-mediated increases in pERK1/2 translocation to the nucleus decreasing cell proliferation. Melatonin in other cell lines has been shown to retain pERK1/2 in the cytoplasm and regulate differentiation processes.63,64 Studies examining pERK1/2 (nuclear) vs (cytosolic) following MEMP HT exposure should be tested in future studies in vivo and in vitro.
In MCF-7 cells, PD98059 decreased MEMP-mediated increases in pERK5 and increased β1-INTEGRIN. These findings suggest that MEMP HT may be working through MEK1/2, MEK5, and β1-INTEGRIN to decrease the epithelial-to-mesenchymal transition 12 reducing BC cell motility 53 and also through β1-INTEGRIN to increase cell-to-cell contact and lessen migration in MCF-7 cells.40,65 Although reports on β1-INTEGRIN’s role in cancer are mixed with respect to how adhesion proteins function in tumor transformation, aggressiveness, and metastases,15,40,65-67 estrogens have been demonstrated to down-regulate 68 and melatonin up-regulate 68 adhesion proteins like E-cadherin and β1-integrin. Perhaps an interplay between E2 and melatonin in the MEMP HT occurred where melatonin counteracted E2-mediated decreases in β1-INTEGRIN in MCF-7 cells increasing β1-INTEGRIN levels resulting in greater cell-to-cell contact and less migration.
Although MEMP HT alone did not affect NF-κB, MEMP HT combined with BIX02189 decreased levels induced by BIX02190 alone. This suggests that MEMP HT produces inhibitory actions on NF-κB in MCF-7 cells through MEK5, which is consistent with other reports. For example, low NF-κB levels are associated with BC protection,69,70 high levels with increases in BC,18,70 tumor resistance to chemotherapy,69,71 conversion of ER(+) tumors to ER(–), 72 and MEK1/2/5-mediated modulation of its expression.18,19
The MEMP HT combined with PD98059 produced greater effects in MDA-MB-231 cells compared with MCF-7 cells, which may be attributed to differences in intracellular signaling mechanisms (Figure 6) especially in MEK1/2-associated coupling to downstream proteins, to MEK5, or to having lower endogenous basal levels of pERK1/2 compared with MCF-7. 24 For example, acute (1 hour) exposure to PD98059 inhibited pERK1/2 levels in both BC lines; however, how they responded to chronic (24 hours) inhibition by PD98059 or BIX02189 differed. For example, acute (1 hour) and chronic (24 hours) exposure to PD98059 decreased pERK1/2 levels in MCF-7. Interestingly, this same 24 hours exposure to PD08059 also decreased pERK5 levels suggesting that some form of MEK1/2-mediated cross-modulation on MEK5 occurred in MCF-7 cells (Figure 6A). For MDA-MB-231 cells, acute (1 hour) exposure to PD9059 decreased pERK1/2 levels, but following 24 hours exposure, levels of pERK1/2 and pERK5 increased. These findings suggest that homologous up-regulation of MEK1/2-associated pathways occurred affecting MEK5-associated signaling proteins like pERK5 (Figure 6B). These cross-modulatory pathways between MEK1/2 and 5, unique to each BC cell line, may also explain why BIX02189 but not PD98059 blocked MEMP-mediated inhibition of migration in MCF-7 and not in MDA-MB-231 cells. Future studies exploring these relationships should be examined in vivo.
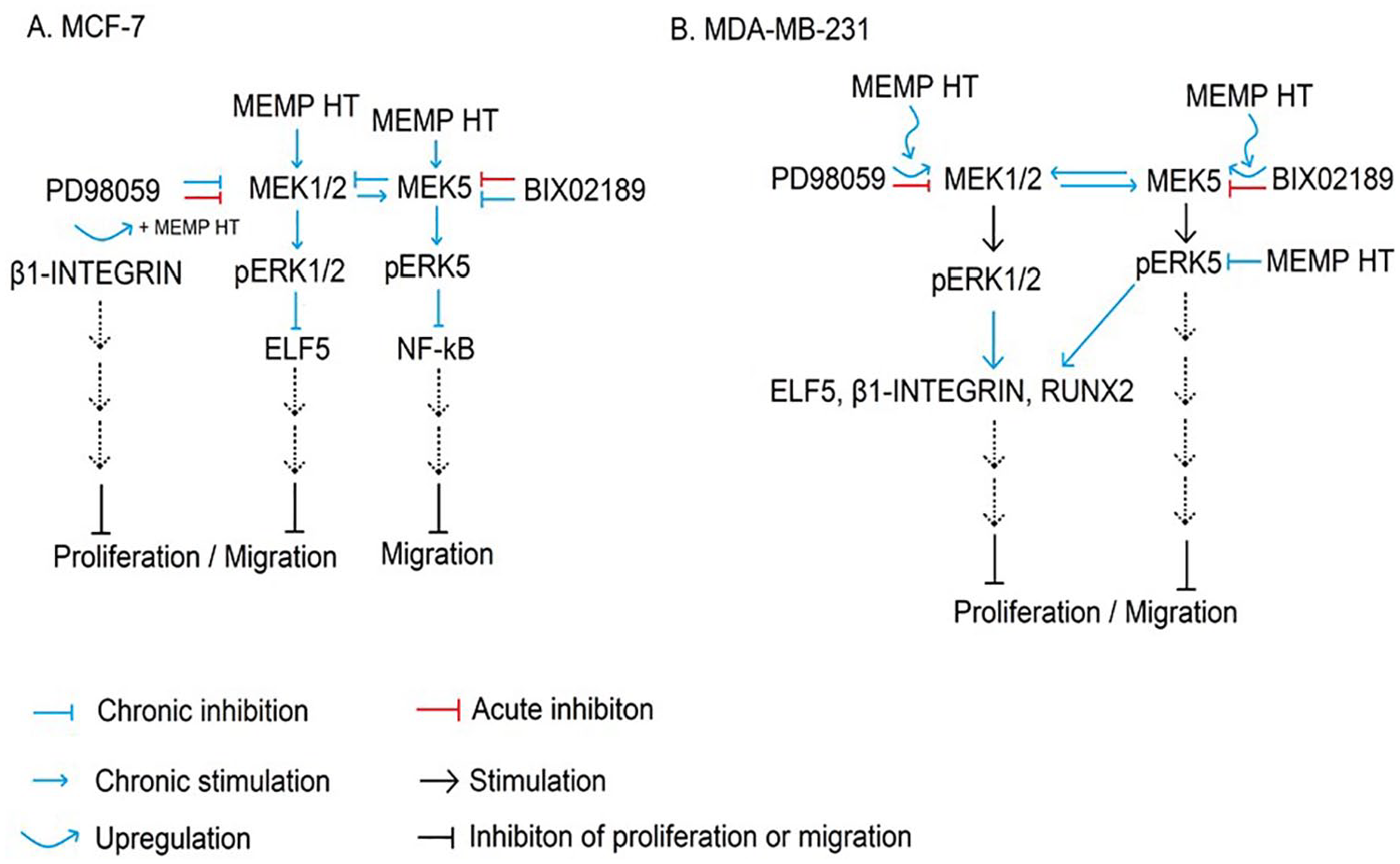
Summary of mechanisms of action underlying MEMP HT in BC cells. As depicted, MEMP HT inhibits proliferation and migration in MCF-7 cells (A) and MDA-MB-231 BC cells (B). Acute (1 hour) or chronic (24 hours) exposure to the MEK1/2 (PD98059) or MEK5 (BIX02189) inhibitors in MCF-7 cells decreased pERK1/2 and pERK5 (trends), respectively, leading to decreases in NF-κB and increases in Elf-5. In MCF-7 cells, MEMP HT alone increased pERK5 in MCF-7 cells, which was blocked in the presence of 02189 and PD98059, suggesting that both MEK1/2 and 5 modulate MEMP HT’s actions on pERK5. β1-INTEGRIN levels increased only when MEMP HT was combined with PD98059 suggesting that a co-(up)-regulation of β1-INTEGRIN occurred between MEMP HT and MEK1/2. For NF-κB, the BIX02189-mediated up-regulation of NF-κB was blocked in the presence of MEMP HT, suggesting that the MEK5 pathway inhibits NF-κB and MEMP HT reverses this. For MDA-MB-231 cells, acute (1 hour) exposure to the MEK1/2 (PD98059) or MEK5 (02189) inhibitors decreased pERK1/2 and pERK5, respectively; however, chronic (24 hours) exposure to PD98059 alone produced a compensatory up-regulation of MEK1/2 and MEK5 pathways leading to increases in pERK1/2, pERK5, and RUNX2; RUNX2 was blocked when MEMP HT was combined with PD98059. Although MEMP HT alone was without effect on the proteins examined, when it was combined with PD98059 a compensatory up-regulation of pERK1/2 and β1-INTEGRIN occurred and when combined with 02189 a compensatory up-regulation of pERK1/2, β1-INTEGRIN, and Elf-5 occurred suggesting cross-modulation of these signaling pathways is occurring in MDA-MB-231 cells distinct from that of MCF-7 cells. BC indicates breast cancer; MEMP HT, melatonin, estrogen, and progesterone hormone therapy.
Conclusions
The findings from this study demonstrate that novel MEMP HT produced anti-cancer actions in ER(+) and TNBC. This study also demonstrates that MEMP HT works through MEK1/2- and MEK-5-dependent intracellular signaling cascades uniquely in each BC line resulting in the modulation of key intracellular signaling proteins (ie, pERK1/2, pERK5, NF-κB, β1-integrin) in MCF-7 cells (Figure 6A) and pERK1/2, pERK5, RUNX2, Elf-5, and β1-INTEGRIN in MDA-MB-231 cells (Figure 6B). The MEMP HT continues to demonstrate anti-cancer actions against myriad BCs, including HER2, 3 ER(+), and TNBC (present study) providing further support for its use in menopausal women to relieve them of their menopausal symptoms without increasing their risk of BC. Future studies assessing MEMP HT’s actions against ER(+) and TNBC in vivo are warranted.
Footnotes
Acknowledgements
We would like to acknowledge Rebecca Kramer, Brianna Peckich, and Spencer Yingling for their contributions to this work.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Marie-Clement Rodier, C.S.Sp. Endowed Chair fund awarded to P.A.W.-E.
Declaration of Conflicting Interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: P.A.W.-E., inventor, Duquesne University, assignee. Combination hormone replacement therapy (HRT) and melatonin to prevent and treat mammary cancer. US Patent 8618083 (2013) and 9370526 (2016). The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Author Contributions
M.H. and P.A.W.-E. participated in research design; M.H., K.H., L.G., T.D., E.B., and P.A.W.-E. conducted experiments; M.H., K.H., L.G., T.D., E.B., and P.A.W.-E. performed data analysis; and M.H. and P.A.W.-E. wrote or contributed to the writing of the manuscript.
