Abstract
Aim:
To study the prognostic value of immunohistochemically detected low Claudin3 expression in breast cancers.
Methods:
This retrospective study included patients with breast cancer who were investigated at our unit from 2006 to 2015. Tissue microarrays were constructed, and immunohistochemical staining was done to assess the Claudin3 expression and to classify breast cancers according to the immunohistochemical surrogates for molecular classification. Kaplan-Meier model and log-rank test were used for recurrence-free survival and breast cancer–specific survival analysis.
Results:
Of the 853 patients, overall low expression of Claudin3 was seen in 18.4%. Recurrence-free survival of patients with overall low Claudin3 breast cancers was poor in luminal A (P = .006) and luminal B (Her2−) (P = .009) subtypes compared with those who had Claudin3 expression in each group.
Conclusions:
Assessment of Claudin3 expression by immunohistochemistry is suggested for luminal A and luminal B (Her2−) subtypes to identify patients with poor prognosis.
Background
Breast cancer is the most common cancer among women world over. It is recognized as a heterogeneous disease, both at phenotypic and genetic levels. Up to the present decade, classification and prognostication of breast cancer was based mainly on the histopathologic features, and the treatment too was based on clinical and histopathologic parameters. In the year 2000, Perou et al 1 revealed that breast cancers could be classified by the differences in their gene expression patterns. Based on molecular genetic analysis, 5 molecular subtypes of breast cancer were described: luminal A and B, Her2 positive, basal like, and normal like. 1 As gene expression studies evolved, further subclassifications were suggested, and in the year 2007, the Claudin-low subtype was described. 2
Many investigators found that prognosis and response to adjuvant therapy are significantly different among the molecular subtypes. 3 Due to the cost and the time taken for the analysis, gene profiling in subtyping of breast cancer in the routine setup is of limited value. Therefore, many research studies came up proposing surrogates for the identification of the molecular subtypes. Blows et al 4 described 6 such subtypes of breast cancer defined by expression of 5 immunohistochemical (IHC) markers and showed that they have distinct survival differences. Molecular subtyping by genetic profiling has not been incorporated in guidelines yet, neither there is a consensus on IHC surrogates for all molecular subtypes. 5 The Claudin-low subtype has been identified at genetic level, but its demonstration by robust histologic methods has not as yet been defined. Therefore, this study was designed to identify the Claudin-low subtype of breast cancer using IHC and to correlate it with the conventional prognostic parameters, breast cancer–specific survival (BCSS), and recurrence-free survival (RFS).
Claudins are important components of tight junctions and involved in the regulation of paracellular permeability and maintenance of epithelial cell polarity.6,7 Expression of Claudin is altered in various cancers and Claudin-1, 3, 4, and 7 are among the most commonly dysregulated members of the 24 member family of Claudins. 8 The downregulation of Claudin is consistent with disruption of membrane tight junctions during tumorigenesis. However, overexpression and mislocalization of Claudin within the cell also have been reported in various cancers. This study therefore investigated on the cellular component of Claudin expression in breast cancer cells which should be evaluated in identifying Claudin-low breast cancers.
Tissue microarray (TMA) is now an established and valuable tool that is particularly important in translational research both in the investigation of routine and novel biomarkers in large series. Pinder et al 9 claim that TMAs can also be used in the optimization of IHC biomarkers. Therefore, TMA technique was used for this study.
Materials and Methods
The study population included all patients with breast cancer who sought the IHC laboratory service of our unit from 2006 to 2015. All breast cancer tissue blocks were retrieved from the department files and used to construct TMAs for the IHC analysis of Claudin expression. Breast cancer blocks with perished tissue were excluded. Claudin3 was selected as the antibody to identify Claudin-low breast cancer as it is found to have altered expression in many cancers and has been used previously for 3 studies on prostatic, breast, and renal carcinoma.8,10,11 Estrogen receptor (ER) and progesterone receptor (PgR) expressions were scored using the Allred score, and HER2 (human epidermal growth factor receptor 2) expression was assessed based on the UK recommendations, 12 on the original whole sections used for routine assessment of prognostic parameters. All breast cancers were classified into the molecular subtypes using the IHC surrogates. 13 Clinicopathologic data were retrieved from the records available in our laboratory. The study was commenced following approval from the Ethical Review Committee of our institution.
Construction of TMAs
Archival tissue blocks were first examined for its physical suitability to be included in TMA. Suitable blocks were assembled and a histopathologist reviewed the hematoxylin-eosin–stained slides of each case and then selected the area of the tumor with minimum fixation artifacts and the invasive tumor front. From each of these donor blocks, a core of 2 mm diameter was extracted using TMA Builder (Thermo Fisher). The diameter of a core was determined to be 2 mm considering the surface area it covers which is almost similar to 4 cores of 1 mm diameter. 9 The fact that our IHC laboratory was a referral center for IHC in the region meant that sufficient representative material would remain for diagnostic record and this was the other reason to decide on a 2 mm diameter without using triplicates of 1-mm-diameter cores. The cores were transposed into the recipient TMA paraffin wax mould prepared previously which contained 24 pits. A core of brain tissue from a pre-prepared paraffin wax block, which was easy to be punched by the plunger, was transposed into the 24th pit as a guide to identify the rows and the columns of the TMA. A map for each TMA block was designed to link the biomarker score to clinicopathologic data of each case.
IHC staining and assessment
Estrogen receptor α clone 1D5 (Dako-M7047), PR (Dako-M3569), and Her2 (Dako-A0485) had been used with the secondary antibody (Dako Real EnVision) for IHC staining of all breast cancers to assess the ER, PgR, and HER2 expression. Anti-Claudin3 antibody (Abcam-ab15102) was used in 1/150 dilution, with the same secondary antibody. Different dilutions were also tried before embarking on the staining of TMAs, and we found 1 in 150 to be the best as per the manufacturer’s instructions. Ki67 (Dako M7240) in 1/75 dilution, CK 5/6 (Dako M7237) in 1/50 dilution, and epidermal growth factor receptor (EGFR) (Dako M3563) in 1/100 dilution were used for the corresponding markers. Epidermal growth factor receptor antigen retrieval was done using proteinase. For Claudin3 and CK 5/6 staining, microwave antigen retrieval with Tris-buffered saline (pH 9) was done at 1100 W for 20 minutes following preheating. Citrate buffer at pH 6 was used for antigen retrieval by pressure cooking (for 7 minutes after preheating) for the rest of the antibodies. Tissue microarray sections were incubated with the primary antibody for 30 minutes. Phosphate-buffered saline was used for washing in between the steps. Positive controls were included for each staining run.
Allred score of ≤3 for IHC staining for ER and PgR and a score of 0 or +1 for HER2 were considered the criterion for categorizing as triple-negative breast cancer (TNBC). The ER and PgR were considered positive when the Allred score for each was ≥3. HER2 was considered positive when the score was 3+. Patients with HER2 equivocal expression (2+) were excluded when in situ hybridization results were not available.
Claudin3 expression was scored as follows: no staining = 0, weak staining = 1, moderate staining = 2, and strong staining = 3, separately for membrane, cytoplasmic, and nuclear staining. The scoring was done in comparison with the normal breast epithelial staining in the 3 cellular locations with Claudin3 which was considered score 2 (Figure 1). A score <2 was considered Claudin3 low. 14 Overall Claudin3-low expression was defined as Claudin3 expression of <2 at all 3 cellular locations.

Microscopic appearance of immunohistochemical staining of normal breast acini and breast cancers with Claudin3 (original magnification ×400): (A and B) normal breast acini (score 2 for membrane, nuclear, and cytoplasmic staining); (C) membrane and cytoplasmic staining (score 3) with no nuclear staining (score 0); and (D) membrane, nuclear, and cytoplasmic staining (score 3 at all 3 cellular levels).
Follow-up and outcomes
Patients whose breast cancer tissue could be included in TMA construction were enrolled for the study. The follow-up details of all patients were retrieved from the clinic files. The mean follow-up time of the cohort was 120 months. The actual minimum follow-up period was 12 months (65% for 24 months, 40% for 36 months, 40% for 48 months, and 27% for 5 or more years).
Breast cancer–specific survival time was calculated from the date of diagnosis of the disease to the date of death. Patients who died of breast cancer or who died with breast cancer (progression/metastasis) were included. 15 Patients died of other causes or from unknown causes were censored to the date of death. The cause of death of the patient was obtained from the death certificate issued by the Department of Registrar General. Recurrence-free survival time was calculated from the date of surgery/first therapeutic intervention to first locoregional and/or first distant recurrence. 15 Radiologic and histopathologic evidence were used to confirm the recurrence. The date on which the said investigation done was considered the date of recurrence. Patients who did not experience the relevant end point were censored at the last follow-up in analyzing RFS. 15
Statistical analysis
The Pearson χ2 test was used to determine the association between the expressions of Claudin3 at each of the 3 cellular locations with the clinicopathologic features. Kaplan-Meier model was used to estimate the BCSS and RFS. The log-rank test was used to compare the survival of different groups. A P value <.05 was considered significant in all analyses. We adhered to the reporting recommendations for tumor marker prognostic studies (REMARK) in analyzing and presenting data. 16
Results
A total of 1124 patients with breast cancer were included. Tissue cores of 11.5% of breast cancers were lost during staining of TMA. Another 4.3% did not have tumor in the TMA core. Breast cancer tissue of 88 breast cancers could not be included in TMA due to physical unsuitability. Therefore, the total number of breast cancers with Claudin3 staining was 853. Claudin3 was first tested on whole sections of breast cancers and normal breast tissue. Normal breast acini expressed Claudin3 in the nucleus, cytoplasm, and in the membrane, mostly in the apical region of the acini (Figure 1A and B).
The study cohort included female patients except for one. Invasive ductal carcinoma was present in 97.4%, whereas invasive lobular carcinoma comprised 1.6% of the cohort. Mucinous carcinoma was found in 0.5%, and other types (papillary, metaplastic, and juvenile carcinomas) accounted for the rest. Risk stratification of the cohort was done using St. Gallen criteria. 5 The study cohort was composed mainly of patients in intermediate-risk category (55.2%), but a large percentage belonged to the high-risk category (41.5%), whereas the low-risk group comprised only 3.3%. 5 These 3 clinical risk categories had a significantly different BCSS (P < .001). No deaths were reported in the low-risk group. The details of the other clinicopathologic features of the study subjects are given in Table 1.
Clinicopathologic features of the study cohort and the subgroups according to the Claudin3 expression at the 3 cellular locations.

Abbreviations: EGFR, epidermal growth factor receptor; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; PgR, progesterone receptor.
Statistically significant P values are in bold.
The study subjects have been treated for breast cancer according to the current guidelines. There were 315 (60.2%) who received hormone therapy of the 523 patients who had hormone receptor-positive breast cancers. About 91.8% (783/853) received adjuvant chemotherapy. This included 83.3% of luminal A, 90.2% of luminal B (Her2−), 96.9% of luminal B (Her2+), 94.6% of Her2 enriched, 94.5% of TNBC, and 94.1% of basal-like breast cancers. Trastuzumab has been started for 31% of those who were positive for Her2 (68/219). Mastectomy with level II axillary clearance had been done for 91.7% (1031/1124) of patients. Postmastectomy radiotherapy has been given to 69.6% (592/851) patients.
Low Claudin3 expression
Immunohistochemical staining for Claudin3 in the 3 cellular locations were seen in different combinations. Cytoplasmic staining was often seen with nuclear as well as membrane staining but it was not seen alone (Figure 1C and D). Claudin3 membrane expression (Claudin3M) was low (a score of 0 or 1) in 50.2% of breast cancers, and low nuclear expression (Claudin3N) was seen in 36.9%. Cytoplasmic Claudin3 expression (Claudin3C) was low in 70.2% of breast cancers included in the study. Overall low expression of Claudin3 was seen in 18.4% of breast cancers (Claudin3 low). Most of the Claudin3-low breast cancers were invasive ductal carcinomas (99.4%). One metaplastic carcinoma was also present, but no medullary carcinoma was present in the cohort. This distribution of histologic subtypes did not change significantly within the subgroups defined according to the Claudin3 expression (P > .05).
Distribution of Claudin3-low breast cancer within each molecular subtype was assessed following classification of all breast cancers using the IHC surrogates. Triple-negative breast cancers with no basal marker expression (CK 5/6 and EGFR) were included in the triple-negative molecular subtype, and TNBCs with expression of either of the 2 basal markers (CK 5/6 or EGFR) were considered basal-like breast cancers. The study cohort comprised luminal A—24.6%, luminal B (Her2−)—12.1%, luminal B (Her2+)—6.42%, Her2 enriched—15.4%, TNBC—32.0%, and basal like—9.8% (of 788 breast cancers for which results of all IHC markers used for molecular classification were available). There was a significant difference in the prevalence of low Claudin3 expression among the molecular subtypes of breast cancer at each of the 3 cellular locations. Low Claudin3C (76.8%) and Claudin3M (62.4%) expressions were most prevalent in the luminal A breast cancers. Low Claudin3N was most prevalent in the TNBC (43.4%) and basal-like (40.2%) molecular subtypes. Overall low Claudin3 expression was most prevalent in the TNBC (28.6%) and basal-like (19.5%) breast cancers. In the luminal A subtype, 16.5% (32/194) were overall low Claudin3 breast cancers.
Clinicopathologic features of low Claudin3 expression were analyzed according to the 3 cellular locations and then again compared with the overall Claudin3-low breast cancers (Table 1). Low Claudin3N expression had a significant association with many established prognostic features. Most of the low Claudin3N were ER and PR negative and were of higher Nottingham grade and larger tumors compared with the breast cancers with positive Claudin3N expression (scores 2 and 3). However, they mostly had low proliferation fraction indicated by ≤Ki67 expression. Low Claudin3N expression was more prevalent among 36- to 60-year age group. Low Claudin3M expression was unexpectedly associated with good prognostic features: low grade, PR-positive, and Her2-negative tumors, no expression of basal markers, and low proliferation fraction. Low Claudin3C expression was associated with Her2 negative and lower prevalence of lympho-vascular invasion. Proliferation fraction indicated by Ki67 was also mostly lower. Overall low Claudin3 expression in breast cancer cells had a significant association with only negative ER expression, negative Her2, and low Ki67 expression.
Claudin3 expression and survival
Of the total number of subjects, follow-up details were available for 649 patients. The median follow-up period of the molecular subtypes ranged from 24 to 40 months and it was 31 months for the total cohort. There were 98 deaths due to breast cancer and 27 of them were Claudin3 low. Data on tumor recurrence were available for 609 patients, and 127 patients had tumor recurrence where 22.8% of them were Claudin3 low.
Breast cancer–specific survival of the cohort according to the TNM stage and the molecular subtypes are given in Figure 2. Survival of patients in different stages and molecular subtypes of the whole cohort tallies well with their expected survival.

Breast cancer–specific survival of the cohort according to the (A) TNM stage (P < .001) and (B) molecular subtype (P = .001).
Patients with low Claudin3C expression had a better RFS (P = .028), but Claudin3N or Claudin3M or overall Claudin3 low expression had no RFS difference (P > .05). There was no significant effect on the BCSS due to low Claudin3N or Claudin3M or Claudin3C or overall low expression of Claudin3 (Figure 3). However, the BCSS curves of overall Claudin3-low expression and the rest of the patients overlap and diverged after 24 months but without reaching a statistical significance. Figure 3D shows that the BCSS of overall Claudin3-low expression has a poor survival.
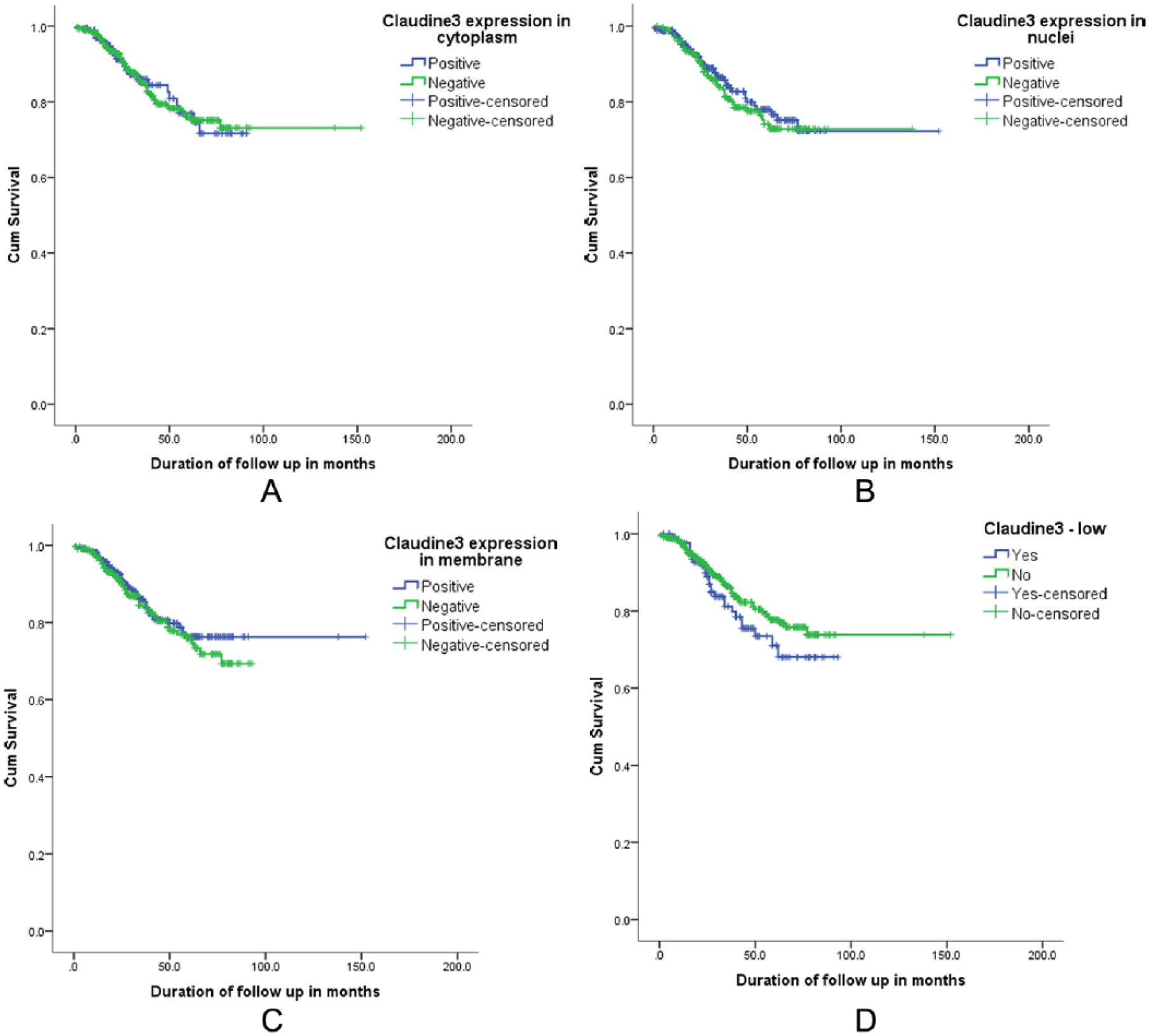
Breast cancer–specific survival of the cohort according to the (A) Claudin3 expression in the cytoplasm (P = .974), (B) Claudin3 expression in the nucleus (P = .542), (C) Claudin3 expression in the membrane (P = .487), and (D) overall low Claudin3 expression (P = .193).
Breast cancer–specific survival and RFS of overall patients with Claudin3-low breast cancer in each of the molecular subtype against the rest in the same subtype were compared. Only the luminal A breast cancers had a significantly poor BCSS due to overall low Claudin3 expression (P = .02). Within the luminal A (P = .006) and luminal B (Her2−) (P = .009) subtypes, RFS of patients with Claudin3-low breast cancer was significantly poor (Figure 4). Other molecular types did not have a statistically significant difference (P > .05), although the RFS of overall Claudin3-low breast cancer was comparatively poor within these groups too. Patients with ER-positive breast cancers had a significant RFS disadvantage (P = .029) when there was overall Claudin3-low expression (Figure 4). There was no effect of Claudin3-low expression on the RFS of the 2 subsets of patients who received endocrine therapy and chemotherapy (P > .05).
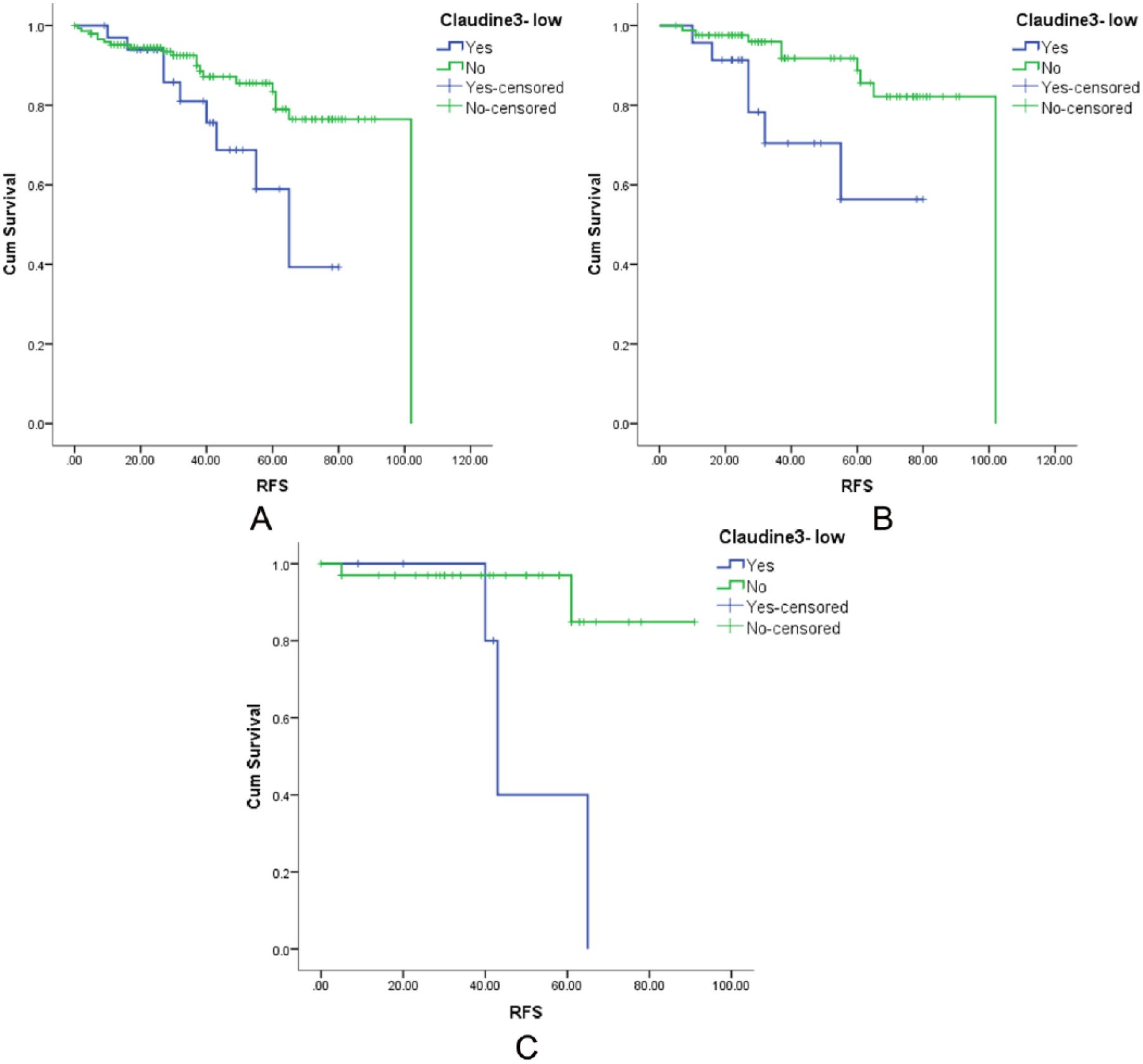
The effect of overall low Claudin3 expression on the recurrence-free survival of patients (A) who had ER-positive breast cancers (P = .029), (B) who had luminal A breast cancers (P = .006), and (C) who had luminal B (Her2 negative) breast cancers (P = .009). ER indicates estrogen receptor.
The effects of low Claudin3 expression on BCSS and RFS were analyzed within the St. Gallen risk categories. We did not observe a significant BCSS or RFS difference in the intermediate-risk or high-risk categories imparted by low claudin3 expression (P > .05). This effect was not analyzed within the low-risk category as it comprised only 3.3% of the cohort and there were no deaths during the follow-up period.
Univariate and multivariate analyses were done including Claudin3 expression to find out the factors with an independent effect on the BCSS (Table 2). All prognostic parameters included in the univariate analysis except low Claudin3 expression had an effect on the BCSS of the whole cohort. However, only the lymph node stage and PgR expression had an independent effect (Table 2). Breast cancer–specific survival of patients with luminal A breast cancers was influenced only by the lymph node stage and low Claudin3 expression. In the multivariate analysis, only the lymph node stage 3 had an independent effect on the BCSS of luminal A breast cancers.
Univariate and multivariate analyses of factors affecting breast cancer–specific survival.

Abbreviations: CI, confidence interval; Claudin3 low, low expression of Claudin3 in membrane, cytoplasm, and nucleus; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; HR, hazard ratio; PgR, progesterone receptor.
Multivariate analysis was not performed for luminal B.
Statistically significant P values are in bold.
Discussion
Claudins are integral components of tight junctions of cell membranes. They are involved in the regulation of paracellular permeability and maintenance of epithelial cell polarity.6,7 Downregulation of Claudin expression has been linked to carcinogenesis. However, there are many reports on overexpression of Claudins, particularly Claudin3 and Claudin4 in cancer 17 and their mislocalizations as well. These aberrant expressions are claimed to be associated with tumorigenesis and probably the progression of various cancers including breast cancer. 11 Although Claudin is expected to be expressed in the membrane, as the normal location of Claudins being the tight junctions of epithelial cells, there are a few reports on its expression in the cytoplasm and the nucleus. 11 A number of studies have shown that overexpression of Claudin may promote tumor progression through its positive effect on cell migration, invasion, and metastasis. The mislocalization of Claudin proteins also may contribute to their role in tumorigenesis.18,19 Todd et al 11 stated that overexpression and delocalization of Claudin3 may contribute to the tumor progression, and therefore, Claudin3 expression is a plausible target for a therapeutic approach.
Our study supports the concept published by Todd et al 11 as we observed Claudin3 expression in the membrane, cytoplasm, and nucleus of breast cancer cells. However, we found that the normal glandular tissue in the breast also shows staining of all 3 cellular components. The term Claudin membrane localization probably can be used when breast cancer cells show membrane staining right around the cell unlike in the apical region in normal breast epithelial cells (Figure 1A and B). Contrastingly, low Claudin3M was associated with good prognostic features in our study cohort but it did not reach statistical significance in the survival analysis.
There is limited data on the identification of Claudin-low subtype by IHC in the literature. The most recent publication recommends using Claudin3, Claudin4, Claudin7, and E-cadherin as IHC markers to identify Claudin-low subtype, yet it does not describe the localization of staining with any of the Claudins they used. 10 A study done in 2013 to identify Claudin-low breast cancer has used Claudin3 as one of the IHC markers and it describes membrane staining but does not describe any nuclear or cytoplasmic staining, neither in normal breast tissue nor in breast cancer tissue. 20 They had used a different brand of Claudin3 primary antibody in 1/1000 dilution. For this study, a dilution of 1/150 was used, as per the manufacturer’s instructions. It could be assumed that the high concentration of primary antibody may have nonspecifically stained the cytoplasm and nucleus as well. However, we observed very specific localization of Claudin3 staining in membrane, nuclei, and in the cytoplasm when they were positive, eliminating the support for an assumption that incubation with too much primary antibody may have given rise to nonspecific background staining precluding the assessment.
Our data reveal that low Claudin3 expression in the 3 cellular components is associated with a mixture of good and poor prognostic features and finally overall low Claudin3 expression is associated only with ER and Her2 negativity and low Ki67 expression. This may be one of the reasons why there is no significant BCSS difference between overall low Claudin3–expressing breast cancers and the rest of the cohort, according to this study. Breast cancer–specific survival curve of overall low Claudin3–expressing breast cancers against the rest shows that the 2 curves overlap displaying a similar survival, until 24 months. Thereafter, the 2 curves tend to move apart and patients with overall low Claudin3–expressing breast cancer show a comparatively poor prognosis. Probably, the long-term survival of low Claudin3–expressing breast cancers in this cohort is poor compared with the rest. Log rank test does not identify a significant survival difference between these 2 groups most probably due to the almost similar survival encountered in the early years of the disease. The similar survival during the first 2 years may be contributed to by the relatively radical surgery done for over 90% of patients. Further follow-up of this cohort may demonstrate a long-term survival difference imparted by overall low Claudin3 expression. The univariate analysis revealed that overall low Claudin3 expression affects the BCSS of luminal A group adversely but its significance is lost as an independent factor, and only lymph node stage 3 becomes the single independent factor affecting BCSS of the luminal A group. The prognostic effect of low Claudin3 expression may be dependent on the other associated poor prognostic factors to influence survival. Breast cancer–specific survival of luminal B subtype is independently influenced only by the presence of lympho-vascular invasion.
Claudin3 and Claudin4 functions have been found to sustain an epithelial phenotype and that their loss promotes epithelial-mesenchymal transition (EMT). 21 Therefore, when Claudin is low, EMT is promoted causing tumor progression. Epithelial-mesenchymal transition may be one of the mechanisms playing a key role in poor prognosis in some luminal breast cancers. Our study reveals that low Claudin3 expression is associated with low Ki67 expression, raising the possibility of poor response to chemotherapy in this subset of patients.
The first study to report on the local recurrence rates for Claudin-low tumors reported that this subtype had the lowest rate of local recurrence (1.3% at 5 and 10 years) of any molecular subtype in their study sample. 10 We found that there is no significant RFS difference between overall low Claudin3–expressing breast cancers and the rest of the patients with breast cancer in the study. However, overall low Claudin3 expression resulted in a significant RFS disadvantage within the luminal A subgroup which generally has a good survival and within the Luminal B (Her2−) subgroup. The poor prognostic effect of overall low Claudin3 expression becomes significant in ER-expressing patients with breast cancer but not with the other types which do not express ER: Her2-enriched, TNBC, and basal-like breast cancers. Triple-negative breast cancer and basal marker expression themselves are poor prognostic features, and overall low Claudin3 expression may not be adding sufficient survival disadvantage to show a significant difference.
The overall low Claudin3 expression in breast cancer cell is the most useful in identifying Claudin-low breast cancer by immunohistochemistry without restricting the assessment to a single cellular localization. However, if one cellular component is to be selected, nuclear staining should be investigated further as it is the one which fits the clinicopathologic profile the most.
The subtype Claudin-low breast cancer is one of the least characterized molecular subtypes of breast cancers which was originally described in 2007. This Claudin-low subtype is found to be related to metaplastic cancer, EMT, and poor prognosis. 20 However, our study cohort did not contain a significant number of metaplastic cancers, and the presence or absence of EMT was not evaluated to highlight a difference in histology.
In 2014, Sabatier et al 22 published the largest series of Claudin-low breast cancer describing its clinical, pathologic, molecular, and prognostic characterization. They found that the Claudin-low subtype contains a significant proportion of hormone receptor–expressing tumors (36%) compared with the study of Prat et al. 2 Our results also show that 33.4% of the overall low Claudin3–expressing tumors were ER positive and support the findings of Sabatier et al. 22
Triple-negative breast cancer and basal-like breast cancers show low expression of Claudin3 in all 3 cellular components more often and became the most common molecular subtypes in our cohort to show Claudin3-low expression, in keeping with the previous studies.10,14 Immunohistochemistry detected low Claudin3–expressing breast cancers accounted for 18.4% of our study cohort. This is comparable with the proportion discovered by the previous studies by genetic profiling which ranged from 7% to 14%. 2 Our study too shows that a significant percentage of both TNBC and basal-like breast cancers are included in low Claudin3–expressing breast cancers. The basal-like breast cancer and TNBC molecular types comprised 19.5% and 28.6% of the Claudin3-low breast cancer in our cohort. Hence, our study demonstrates the heterogeneity of Claudin-low breast cancers. Therefore, the clinicopathologic profile and survival function of Claudin3-low breast cancer identified by immunohistochemistry tally well with the previous gene profiling studies.
Conclusions
Overall low Claudin3–expressing breast cancers identified by IHC have similar clinicopathologic features described by the studies based on gene profiling. The survival of overall low Claudin3–expressing breast cancers is poor in line with the other studies. We also identified that subtype low Claudin3–expressing breast cancer is as heterogeneous as breast cancer itself and imparts significant survival disadvantage to some molecular subtypes. Therefore, the prognostic value of Claudin-low expression is further confirmed by this study.
In addition, we described the survival disadvantage it adds to the molecular subtypes which is most significant in the luminal A (both BCSS and RFS) and luminal B (Her2−) (RFS) subtypes. This study highlights the value of IHC in identifying low Claudin3–expressing breast cancer and suggests IHC staining for Claudin3 to be used as an additional marker to identify patients with breast cancer with poor prognosis especially within the molecular type with generally good prognosis: the luminal A subtype.
Footnotes
Acknowledgements
The authors wish to acknowledge the Ministry of Science, Technology and Research, Sri Lanka for funding the project under the Indo-Sri Lanka Joint Research Programme 2012 and the technical support provided by Mrs GGDD Gunawardhane and the staff of the Department of Pathology, Faculty of Medicine, University of Ruhuna and the staff of the Oncology Unit of the Teaching Hospital Karapitiya, Galle, Sri Lanka, for the support extended in retrieval of follow-up details.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science, Technology and Research, Sri Lanka (Grant MTR/TRD/AGR/3/1/9—Indo-Sri Lanka Joint Research Programme 2012).
Declaration of conflicting interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: S.G. and D.A. received a monthly stipend from the funding agency as Research Assistant’s allowance. Other authors declare that they have no conflicts of interests.
Author Contributions
LM conceived and designed the experiments. HP analyzed the data. LM wrote the first draft of the manuscript and developed the structure and arguments for the paper with contribution from HP. SG, DA, and TL contributed to the data collection and interpretation. All authors agree with manuscript results and conclusions, made critical revisions and approved final version, and reviewed and approved the final manuscript.
