Abstract
Background:
Threat to blood transfusion–transmitted dengue virus (DENV) and its antibodies has recently emerged worldwide. Dengue fever is an endemic disease in Saudi Arabia, particularly in its Western region. The aim of this study was to estimate the seroprevalence of asymptomatic DENV infection and its antibodies among eligible Saudi blood donors.
Methods:
Serum samples from 910 healthy/eligible adult male Saudi blood donors, who reside in Holy Makkah City of Saudi Arabia, were collected between March 2015 and August 2016 and screened for the detection of DENV nonstructural protein 1 (NS1) antigen and anti-DENV IgM and IgG antibodies using commercial enzyme-linked immunosorbent assay kits (Panbio, Brisbane, QLD, Australia).
Results:
Among the tested donors, 48 (5.3%) were seropositive for DENV-NS1 antigen, whereas 50 (5.5%) and 354 (38.9%) were seropositive for anti-DENV IgM and IgG antibodies, respectively. Seropositivity for DENV-NS1 antigen and/or anti-DENV IgM antibody among the tested donors reflects their ongoing asymptomatic viremic infectious stage with DENV during their donation time, whereas high prevalence of anti-DENV IgG seropositivity reflects the high endemicity of dengue disease in this region of Saudi Arabia.
Conclusions:
These results show high prevalence of asymptomatic DENV infection and its antibodies among Saudi blood donors, raising the importance of establishing blood screening for dengue disease at different blood donation services and units in Saudi Arabia to improve the guarantee of blood transfusions and to control DENV dissemination.
Introduction
Dengue is the most prevalent arthropod-borne viral disease in the world. It is caused by a single-stranded RNA arbovirus of the genus Flavivirus with 5 distinct antigen serotypes (DENV1-5) that are transmitted to humans through the bites of infected mosquitoes, namely, Aedes aegypti and Aedes albopictus.1,2 According to the World Health Organization (WHO), the current worldwide burden of dengue is about 2.5 billion infected people in more than 100 countries with approximately 20 000 fatal cases per year, and its global incidence is predicted to grow dramatically to affect about half of the world’s populations.3,4 The transmitted DENV replicates in humans within 3 to 14 days after biting, 5 and its positive cases have a wider spectrum of clinical manifestations, ranging from asymptomatic infection, which is the most common case, to potentially fatal cases of dengue hemorrhagic fever (DHF) and dengue shock syndrome (DSS) due to plasma leakage, increased vascular permeability, and homeostasis disorders.6,7 Until now, there is no licensed DENV vaccine in Saudi Arabia, and similarly, there are no approved specific treatments available against DHF or DSS, and the mortality rate varies and is dependent on the DENV virulence, human host factors, and immune response to previous dengue infection.4,7 Recovery from infection of 1 DENV serotype only provides homotypic lifelong immunity against that particular serotype without long crossprotective immunity against other DENV serotypes. Thus, individuals can acquire a second or a subsequent dengue infection from different DENV serotypes. 6 In addition, the severe clinical forms of dengue disease are more likely to occur during a second infection with a different DENV serotype from that which caused the primary infection,7,8 and this phenomenon is attributed to the enhancement effect of preexisting heterogeneous antibodies on maturation, virulence, and replication of the second heterotopic DENV serotype.9–11
Since 1994, multiple outbreaks of different DENV serotypes have been reported in the Kingdom of Saudi Arabia, particularly in its Western region, including Jeddah and Holy Makkah.1,12–15 Now, dengue disease has become an endemic disease in these regions, wherein the main mosquito vector is the Aedes genus.12–15 Another particular concern is attributed to the potential role of foreign visitors who arrive at these regions from the neighboring Asian and African countries with known dengue endemicity. In that respect, visitors who arrive during their asymptomatic or subclinical viremic stage of acute dengue infection can expedite the dissemination of DENV.1,12–15
In addition to mosquito vectors, DENV is currently facing a specific global attention as a potential transfusion-transmitted virus that endangers blood transfusion safety.3,16–20 Dengue virus has been widely detected in the blood and blood products of asymptomatic or subclinical acute infected donors around the world, and transmission of DENV from such donors to their corresponding recipients with subsequent disease development has been evidenced.17–31 Most importantly, transmission of heterogeneous anti-DENV antibodies from donors has also been suggested to enhance the viral infectivity and virulence in recipients who are later exposed to a heterotopic DENV serotype infection, 23 and this in turn represents an additional risk of dengue disease to global blood transfusion safety and availability as more blood donors are deferred. 24 These collective facts serve as evidence that both DENV and its non-neutralizing antibodies are indeed transmissible by blood transfusion and imperil blood safety. Nevertheless, at present, there are no available efficient measures and diagnostic services for such events among blood donors in several countries with well-known endemicity with dengue disease, including Saudi Arabia. 1 Therefore, establishment of routine blood screening of DENV and its antibodies at the blood donation services became a paramount global demand to secure blood transfusion safety and prevent disease dissemination.3,6,24 Toward this goal, we have recently conducted a pilot study on a narrow scale (n = 100) of asymptomatic Saudi blood donors, and its results have highlighted the prevalence of both DENV (represented by its nonstructural protein 1 [NS1] antigen) and its IgM and IgG antibodies among the tested donors. 1 To verify and confirm our previous findings, we conducted this study on a large scale (n = 910) of asymptomatic/eligible blood donors who live in Holy Makkah City. Surprisingly, our results showed the presence of high seropositivity of DENV-NS1 (5.3%) and anti-DENV IgM (5.5%) and anti-DENV IgG (38.9%) antibodies among the enrolled donors, which in turn increase the importance of approving and establishing routine blood screening for dengue at different Saudi blood donation services and units to improve blood transfusion safety and enforce the control measures against the spreading and endemicity of dengue infections in Saudi Arabia.
Materials and Methods
Ethical approval
For this study and its entire protocol, an ethical approval was obtained from the Institutional Review Board of the Faculty of Applied Medical Sciences, Umm Al-Qura University, Holy Makkah, Saudi Arabia. In addition, an official governmental approval was secured by writing to the Deputy Director of Health of the Holy Makkah Municipality. All tubal blood samples were collected after obtaining informed written consent from all the participant donors. This study meets the standards of the Declaration of Helsinki.
Participants, specimen collection, and testing
During March 2015 to August 2016, and at the blood transfusion service in Hira General Hospital and The Regional Laboratory, Holy Makkah, Saudi Arabia, a total of 910 eligible healthy male blood donors of Saudi nationality (age ranged between 25 and 55 years; mean age: 37.13 ± 7.45 years) were randomly included in this cross-sectional seroprevalence study. At the time of donation, all participant donors were negative for infection with human immunodeficiency virus (HIV), hepatitis B virus (HBV), and hepatitis C virus (HCV); did not suffer from fever or any sign of dengue infection as per WHO guidelines; and were accepted for blood donation according to the policy set up by the Kingdom of Saudi Arabia Health Ministry. From each participant donor, 10 mL of whole venous blood was collected in sterile plain tubes without anticoagulant. Blood was allowed to clot at room temperature for 30 minutes, followed by centrifugation at 3000 rpm for 15 minutes. Serum was aseptically collected and transferred to sterile 5-mL screw cap tubes and stored at −20°C until use. The frozen sera samples were shipped in ice boxes to the Central Lab of Faculty of Applied Medical Sciences, Umm Al-Qura University, Holy Makkah, Saudi Arabia, for serodetection of DENV-NS1 antigen as a rapid, sensitive, and cost-effective test to detect asymptomatic acute DENV-infected donors 24 and anti-DENV IgM and IgG antibodies. The tests were conducted using commercially available DENV Pan-E NS1 early enzyme-linked immunosorbent assay (ELISA), DENV IgM capture ELISA, and DENV IgG capture ELISA, respectively (all from Panbio, Brisbane, QLD, Australia). In each assay, all samples were processed in duplicate on a fully automated ELISA system (Human Diagnostics, Wiesbaden, Germany), according to the manufacturer’s recommendations and were repeated again if the results from the duplicate testing were not concordant.
Data entry, interpretation, and analysis
As reported by the manufacturer, a positive ELISA result for either DENV-NS1 antigen or anti-DENV IgM antibody was defined as having an index value >11 Panbio Units (PU), whereas a positive ELISA result for anti-DENV IgG antibody was defined as having an index value > 22 PU. Furthermore, index values of 9 to 11 PU for either DENV-NS1 antigen or anti-DENV IgM antibody and index values of 18 to 22 PU for anti-DENV IgG antibody were considered as equivocal reactions in accordance with the manufacturer’s recommendations. Data analysis was conducted using SPSS version 16, and χ2 test was used.
Results
In this study, a total of 910 eligible/healthy adult male Saudi blood donors, negative for HIV, HCV, and HBV infections and accepted for blood donation according to the policy set up by the Kingdom of Saudi Arabia Health Ministry, were separately screened for the seroprevalence of DENV-NS1 antigen and anti-DENV IgM and IgG antibodies using commercially available ELISA kits (Panbio). As shown in Figure 1, of the 910 donors tested, 48 donors (5.3%) were seropositive for DENV-NS1 antigen, whereas 50 (5.5%) and 354 (38.9%) donors were seropositive for anti-DENV IgM and IgG antibodies, respectively (Figure 1). Noticeably, and as demonstrated in Figure 2, the donors who were seropositive for DENV-NS1 antigen (48 of 910; 5.3%) were subdivided as follows: 7 donors (0.8%) showed monopositivity to DENV-NS1 alone; 2 (0.2%) and 26 (2.9%) donors showed dual positivity to DENV-NS1 plus anti-DENV IgM or IgG antibodies, respectively; and 13 donors (1.4%) showed triple positivity to DENV-NS1 plus anti-DENV IgM and IgG antibodies (Figure 2).
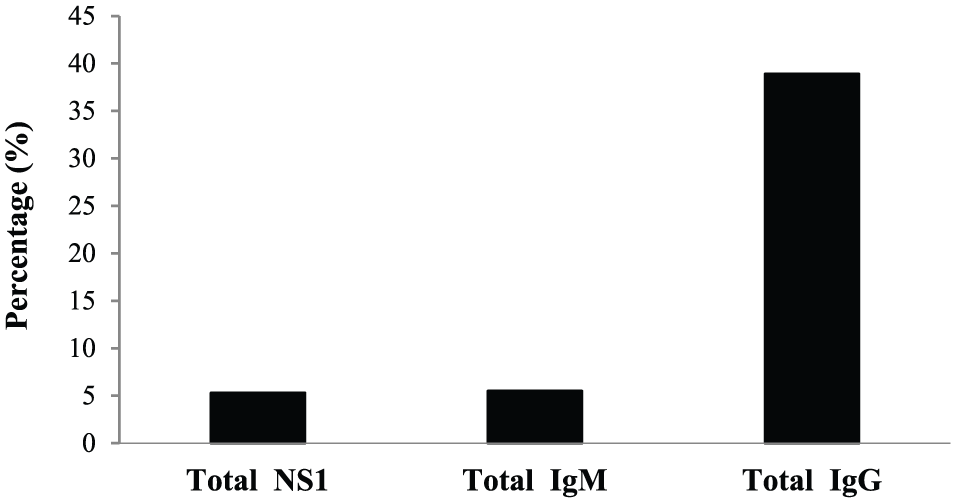
Seroprevalence of Dengue virus (DENV) nonstructural protein 1 (NS1) antigen and anti-DENV IgM and IgG antibodies among the asymptomatic/healthy/eligible Saudi blood donors (n = 910) tested. Values of DENV-NS1 antigen or anti-DENV IgM antibodies are considered positive when >11 Panbio Units (PU), whereas values of anti-DENV IgG antibodies are considered positive when >22 PU, according to the manufacturer’s recommendations (Panbio, Brisbane, QLD, Australia).
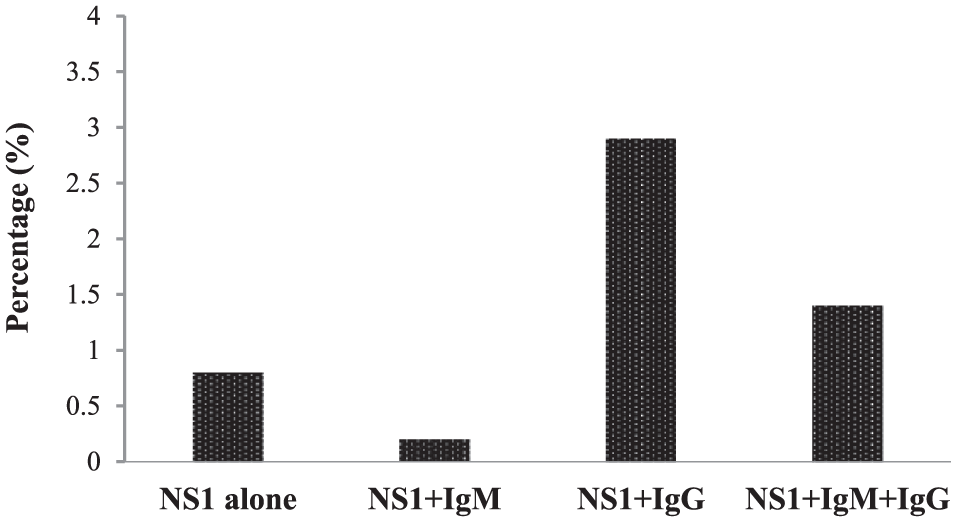
Subdivision of the total donors with seropositivity for Dengue virus (DENV) NS1 antigen (n = 48 of 910).
From a diagnostic standpoint, it can be speculated that donors who were seropositive to DENV-NS1 antigen and/or anti-DENV IgM antibodies were in their ongoing asymptomatic acute DENV infection phase at the time of their donation25,26,31 and could act as silent carriers to transmit DENV to their corresponding recipients,6,24–27 whereas the high prevalence of anti-DENV IgG seropositivity, the presence of which may indicate previous exposure, 26 could reflect the high endemicity of dengue disease in this region of Saudi Arabia.12-15
Discussion
Securing the safety of blood transfusion and blood products for the recipients is a mandatory medical demand. In that respect, the risk of blood transfusion-transmission of DENV and/or its antibodies from donors to recipients has recently emerged and become an important clinical fact.6,17–31 This is because more than 70% of DENV can be present for about 7 days in the blood of acute infected individuals without appearance of any symptoms of dengue during this period, and such asymptomatic DENV carriers may offer to donate blood. 22 Therefore, screening of DENV and its antibodies among blood donors and blood products at blood donation services may be a global demand to improve blood transfusion safety and control disease spreading and severity around the world.3,29–31 With this concept, the Kingdom of Saudi Arabia, particularly its Western region, is endemic with different serotypes of DENV since 1994.1,12–15 Nevertheless, blood screening tests for dengue among the donors have not been approved or established yet in this country. 1 We therefore performed this cross-sectional study to highlight the seroprevalence of DENV and its antibodies among eligible Saudi blood donors. Toward this goal, we enrolled a total of 910 healthy and eligible adult male Saudi blood donors who live in Holy Makkah City, Saudi Arabia, and none of them were viremic or suspected to be suffering from any sign of dengue infection as per WHO guidelines, such as fever, headache, myalgia, retro-orbital pain, rash, and/or hemorrhagic manifestations, at the time of donation, and all of them were accepted for blood donation according to the policy set up by the Kingdom of Saudi Arabia Health Ministry at the time of donation. All donors were individually screened for seropositivity of DENV-NS1 antigen and anti-DENV IgM and IgG antibodies using commercially available ELISA kits (Panbio). Importantly, the results of this study show the presence of high seropositivity for DENV-NS1 (5.3%), as well as for anti-DENV IgM (5.5%) and anti-DENV IgG (38.9%) antibodies, among the tested donors, which in turn makes it mandatory to establish routine blood screening for DENV at the Saudi blood donation services and units.
From a diagnostic standpoint, there are different laboratory approaches that can be used to detect the acute stages of dengue infection. 31 In that regard, using direct virus isolation is a time-consuming, complex, and fastidious process that demands specialized viral isolation units, cells, and highly experienced staff. Similarly, application of polymerase chain reaction (PCR) for quantitative detection of DENV-RNA and for diagnosis of early DENV infections is relatively expensive and also requires specialized equipment and difficult technical skills that make it an unaffordable routine diagnostic tool for large-scale blood banks or may not be available in resource-poor settings and in countries with limited financial resources.1,24 However, the serological assay of DENV-NS1 antigen capture ELISA was developed as a surrogate diagnostic marker for serodetection of early DENV infections and viremia. 32 At that point, circulating NS1 antigen is secreted from DENV-infected cells as early as 1 day after infection to play an essential role in viral replication, and ELISA-based serodetection of this antigen has been proposed as a potential marker for early diagnosis of asymptomatic, subclinical, or clinical cases of acute dengue disease, and such diagnostic procedures provide many advantages, including more flexibility, lower cost, and global availability of required reagents and equipment.31-33 Furthermore, DENV-NS1 antigen has been detected in the blood of DENV-infected patients as early as PCR detection of DENV-RNA,32-36 and importantly, there are some reports indicating that NS1 assay may be more superior than reverse transcription polymerase chain reaction (RT-PCR) assay based on negative results on PCR and positive results on NS1,37,38 and NS1 antigen can be detected up to 9 days in serum even after PCR detection of viral RNA has become negative.34,35,39,40 Taken together, serodetection of DENV-NS1 antigen may be a very useful, easy, rapid, feasible, and cost-effective diagnostic tool for early diagnosis of asymptomatic DENV viremic donors 24 and could be used as an alternative to RT-PCR, particularly in developing countries. 33 Given all these advantages, we herein measured the DENV-NS1 antigen among our enrolled blood donors, and their results showed that among the tested donors, 5.3% had true positivity for serum DENV-NS1 antigen, suggesting that these donors were actively infected with DENV and had ongoing asymptomatic viremia or were carriers of the virus at the time of donation.5,23,41 In agreement, it has been postulated that stake holders in blood transfusion practices should consider the crucial role of asymptomatic/subclinical DENV-infected donors as active carriers silently transmit the virus to prospective recipients, and these recipients can develop fever, neutropenia, severe thrombocytopenia, hypotension, and other severe clinical signs of dengue disease.17,25,26,41-43
Laboratory diagnosis of dengue disease is also done using antibody screening in the serum samples. The diagnostic window period is about 3 to 5 days for anti-DENV-IgM antibodies, and anti-DENV IgG antibodies are detectable after approximately 9 to 10 days. 31 In this study, both anti-DENV IgM and/or IgG antibodies were predominately detected in the serum of the tested donors. Noticeably, the seropositivity for IgM that we reported here (5.5%) points to an ongoing recent infection, and such donors are in a carrier stage of infection,25,26,28,31 whereas high prevalence of anti-DENV IgG antibody (38.9%) in the circulation of tested donors indicates the high endemicity of dengue disease.15,25,26,28 On the overview of dengue immunity and pathogenesis, it is well known that the severe clinical forms of dengue disease commonly occur during second or sequential infection with heterogeneous DENV serotypes from those which caused the primary or previous infection.3,4,7,8 Similarly, lifelong immunity is mediated by IgG neutralizing antibodies against the infecting DENV serotype (so-called homotypic immunity), and these antibodies do not mediate long crossprotective immunity (heterotypic immunity) against other DENV serotypes. 11 Thus, individuals can acquire second or subsequent infection with another serotype of DENVs during their lifetime. 6 Most importantly, such antibodies have been proposed to not only fail to neutralize the second or sequential infections with different (heterotypic) DENV serotypes but instead opsonize and help the immature DENV particles to infect more human cells and increase the number of infected cells and viral virulence/titers, and it is thought to lead to more pronounced inflammatory response and disease severity.9,11,44-46 Moreover, such pheromone has also been observed in infants who are born to dengue-immune mothers and who therefore have maternally derived dengue-neutralizing IgGs, whereby the titers of these IgGs wane and such infants will have an increased risk of DHF during their primary heterotopic dengue infection. 11 In a constant line, this phenomenon and its association of enhancement of viral infectivity and virulence have been suggested to occur in blood recipients who receive anti-DENV IgG antibodies from donors and who are later (within 6 months) exposed to a second infection with a heterotopic DENV serotype after transfusion 23 and could represent an additional threat and risk to blood transfusion safety and availability as more blood donors are deferred. 24 Nevertheless, prospective studies are still needed to confirm the possible importance of this phenomenon in blood-transfused subjects who receive crossreactive heterogeneous non-neutralizing anti-DENV antibodies from DENV-infected donors and are later exposed to another type of DENV infection.23,24
Conclusions
Data from this study are the first to indicate the high seroprevalence for DENV and its antibodies among asymptomatic/healthy/eligible Saudi blood donors and, in turn, increase the importance of establishing routine diagnostic methods for screening for DENV and its antibodies at different Saudi blood donation services to improve the safety quality of blood transfusions and also to improve the control measures against the dissemination and endemicity of DENV in this country. Future studies are still required to provide further estimation of asymptomatic DENV and its antibodies among eligible Saudi and non-Saudi blood donors. In these future studies, detection of DENV-RNA via RT-PCR should be added with DENV-NS1 ELISA assay to detect all cases of early active nonsymptomatic DENV infections. In addition, determination of the possible importance of different seasons, including pilgrimage and Umrah seasons, and participants’ age and job on the prevalence of DENV infection and the seropositivity spectrum of DENV and its antibodies among asymptomatic blood donors should be considered.
Footnotes
Peer review:
Six peer reviewers contributed to the peer review report. Reviewers’ reports totaled 1651 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research work was financially supported by a grant (43409045) from the Institute of Scientific Research (ISRRIH), Umm Al-Qura University, Holy Makkah, The Kingdom of Saudi Arabia.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AMA and AGE conceived and designed the experiments. All authors have contributed to sample collection and processing, laboratory measurements, analyzing of the data, writing and drafting the manuscript, and reviewing and approving the final manuscript.
