Abstract
Dengue disease is an emerging mosquito-borne viral infection transmitted between humans by Aedes spp. that are distributed mainly in the tropical and subtropical region along with chikungunya and zika diseases. The distribution of dengue disease is influenced by local variation, such as geography, rainfall, temperature, and rapid urbanization or migration. The epidemy of mosquito-borne infection significantly led to increased number of cases and hyperendemicity which induce a more severe form of dengue accompanied by cocirculation of chikungunya and zika. The rapid global spreading of dengue disease created public health burdens that are presently unfulfilled by the absence of specific therapy, simple diagnosis tool for the early phase, and effective and efficient vector control system. This review highlights the current situation of dengue distribution, epidemiology, and new strategies for early dengue diagnosis and risk prediction of severity that can be used to improve oversight and alleviate the heavy burden of the disease.
Introduction
In the past decade, the escalation of dengue as a threat to health, finance, and health services has increased substantially. 1 This mosquito-borne viral infection has grown 30-fold since it was first reported and then expanded and diversified globally. 2 A recent distribution of dengue virus (DENV) and other mosquito-borne viruses, such as chikungunya and zika, has similar epidemiology and transmission cycle in urban areas. 3 Here, we present an overview of the current situation of dengue distribution and epidemiology, especially in Southeast Asia and Pacific. To this end, the review also addresses new strategies for early dengue diagnosis and risk prediction of severity that can be used to improve oversight and alleviate the heavy burden of the disease.
Current Burden of Dengue
Dengue is the most important acute systemic arthropod-borne viral infection in humans. 2 This disease is becoming a global public health concern, spreading from tropical regions to most subtropical regions of the world, causing human suffering and massive socioeconomic losses.1–6 It is estimated globally that 50 to 100 million dengue cases occur each year across approximately half of the world’s population, especially in areas with cocirculation of multiple virus serotypes, known as hyperendemic regions in Southeast Asia and Pacific.1,7–8 It is also estimated that dengue is responsible for 20 000 deaths anually.7–9 A study by Bhatt et al 10 estimated that dengue infection cases have increased more than 3 times per year with 67 to 136 million cases annually which manifest clinically at any level of severity.
Dengue disease in humans produces wide spectrum of clinical features ranging from atypical nonsevere or nonspecific febrile syndrome to potentially fatal dengue hemorrhagic fever (DHF) or dengue shock syndrome (DSS), conditioned by age, secondary infection, immunologic status, dengue serotype, and genotype.11–13 Severe dengue known as DHF or DSS has become a major public health problem with a burden of 372 disability-adjusted life years per million populations, higher than other viral diseases.1,9 The average cost for dengue outpatient case was $514 and for hospitalized case was $1394. 6 A study in Latin America 14 showed that the total annual cost of dengue infection was $46.45 million and even higher in Southeast Asia of $950 million. 9 Approximately 52% of the amount was due to the loss of productivity, excluding the budget spent on basic precautions and vector control measures.9,10
The primary vector for dengue disease transmission is Aedes aegypti, 15 whereas another less effective vector Aedes albopictus is also surprisingly responsible for the spread of this disease in recent years. 4 Global situations, such as migration to urban area, traveling, and lack of environmental management, stimulate the rise and rapid spread of these vectors. As a consequence, Aedes spp. and all 4 dengue serotypes they carry are circulating in almost every tropical and subtropical regions of the world, thereby creating the pattern of endemic and hyperendemic regions. Data in 2014 pointed that there were significant increase in mosquito-borne viral infection in Pacific region with concurrent circulation of DENV, chikungunya virus (CHIKV), and zika virus (ZIKV). 2
Dengue Virus
The 4 dengue serotypes (DENV 1-4) belong to the genus Flavivirus of the family Flaviviridae. Characteristic features of dengue genome are as follows: a spherical particle of small size (diameter 40-50 nm) covered by glycoprotein envelope and single-strand messenger (positive) sense RNA of approximately 11 kb in length. The order of protein-encoded open reading frames is shown in Figure 1. 16
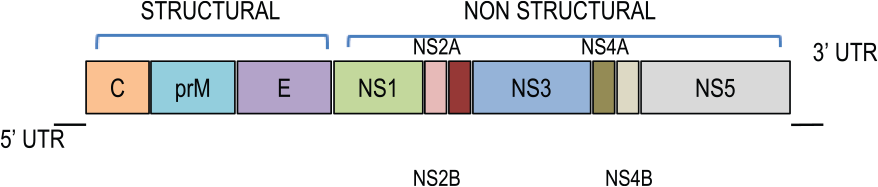
Full-length DENV genome and protein organization. The dengue virus open reading frames encode 3 structural proteins: capsid (C), precursor membrane (prM), and envelope (E), with 7 nonstructural (NS) proteins. UTR indicates untranslated region.
The envelope functions in hemagglutination phenomenon, neutralization, and interaction between virus and host cell during the early phase of infection. 4 Dengue virus and its soluble membrane–associated NS1 activate human complement system and express in the blood during the early phase of infection. Viral load, terminal SC5b-9 complement complex, and NS1 antigenemia have been shown to be associated with dengue severity.2,17
Epidemiological Pattern of Dengue Serotypes
Four different serotypes of DENV (DENV 1-4) have been identified as causes of dengue infection since first recognized in 1943, later accompanied by the fifth serotype (DENV-5) discovered in 2013.18,19 The spread of all DENV serotypes was being reported worldwide, extending to South America and Europe.20–22 Global climate change, unsuccessful vector eradication, precipitation, and human density variables closely related to mosquito density were crucial factors of DENV pattern in these regions.21–23 Consequently, there were different patterns of DENV serotypes in many countries. Recent studies showed that DENV-1 was the predominant serotype in Indonesia, China, Malaysia, Pacific region, Portugal, and South America.18,22,24–26 At the same time, there was a discrepancy in the spatial pattern of endemic areas; for example, in one area in Indonesia DENV-2 predominates, whereas in other areas DENV-1 predominates. Despite different predominant serotypes between these areas, genotype distribution was similar, explicating the association of viruses that circulated earlier and the virulence itself.27,28
The inconsistent DENV type–specific global map was reported especially in endemic and hyperendemic areas in Southeast Asia and Pacific regions. After its first discovery in 1943, DENV-1 distribution constantly increased in Asia and South America regions over the next few decades. 18 DENV-1 has been documented as the dominant serotype during 1-year (2012-2013) outbreak in New Caledonia with 10 978 confirmed cases and 5 deaths. 2 Similar occurrences were also reported in South American countries, such as Columbia, Costa Rica, Paraguay, Cuba, Brazil, and Venezuela.18,29
The occurrence of DENV-2 serotype has been reported in Southeast Asia since 1944, first in Papua New Guinea and Indonesia, later in Malaysia and Thailand in the early 1960s, and then in China, India, and Singapore in 1970s and Central America in 1980s.18,30 In 1997, the American-Asian DENV-2 genotype, known to be the more virulent genotype, was identified and was responsible for severe dengue cases since that time. 20 DENV-2 was the primary cause for the severity of dengue infection in Cuba, Thailand, and Malaysia.5,31 Many studies reviewed the shift of DENV-2 as the important serotype in fatal dengue disease, especially in children.13,32,33
The DENV-3 serotype has been circulating worldwide since first reported in 1953 in Southeast Asia. DENV-3 was the primary cause of severe dengue cases in Indonesia. The distribution of this serotype in Africa has been recognized since 1984 with sporadic occurrence. 18 The DENV-4 serotype is almost yearly seen in Southeast Asia and Pacific endemic regions since first reported in Philippines and Thailand in 1953. An outbreak in Boa Vista, Brazil, in 2010 marked the reappearance of DENV-4 after a 28-year absence. 29 A cohort work in Jakarta, Indonesia, found that clinical manifestation of DENV-4 infection was milder than other serotypes. However, concurrent infection with other serotypes yielded clinical manifestation of severe dengue, DHF grades I and II. 34
Coinfection by multiple DENV serotypes has been widely recognized in many endemic countries in China, Southeast Asia, Brazil, and India.24–29,32,35–37 In 2012, for the first time, the Pacific region registered a high prevalence of all 4 serotypes of DENV.2 The present work in Malaysia found the presence of concurrent DENV-2 and DENV-3 infection, with all 4 serotypes detected. 26 It was proposed that the increasing hyperendemic transmission has a role in the concurrent circulation of multiple DENV. The concurrent infection was estimated as one of the factors that influenced the high frequency of dengue case. Most studies pointed that concurrent infection by any DENV serotype did not provoke severe manifestation of dengue disease.13,35,37 In contrast, other studies reported that many concurrent infection cases had more severe illness, particularly in secondary infection.29,32–35 The DENV cocirculation around the world in the 2000-2013 period is shown in Figure 2.
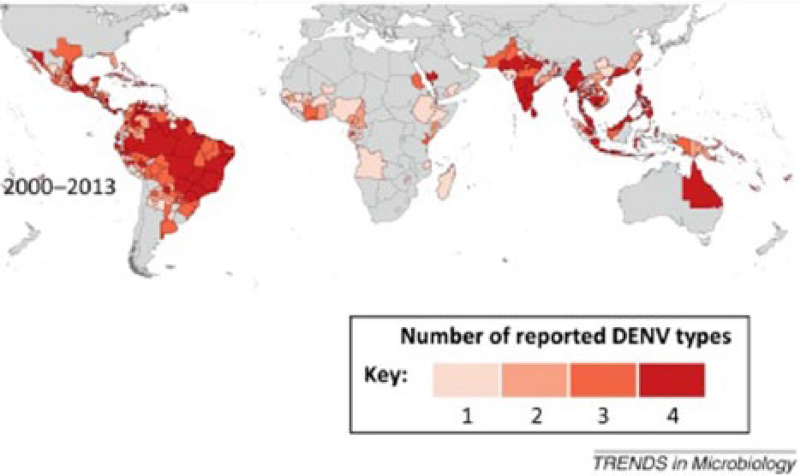
Dengue virus cocirculation in the world in the 2000–2013 period. 18
Concurrent Spread of Dengue, Chikungunya, and Zika
Zika virus is a mosquito-borne virus that has been spreading globally recently and has similarities to DENV and CHIKV.3,22 An outbreak of fever and rash associated with ZIKV was detected in Yap Island, Micronesia, in 2007, 38 followed by a large outbreak in Polynesia in 2013-2014. 2 Subsequently, this virus rapidly spread to several countries, such as Cook Island and New Caledonia in 2014 and Brazil in late 2014. 2 In recent years, concurrent circulation of multiple DENV serotypes, CHIKV, and ZIKV was documented in the Pacific, Brazil, and Columbia hyperendemic regions. 39
Zika virus was first isolated in 1950s and has been recognized to circulate in almost every region of Africa and Asia, but little evidence is remarkable about genetic relationships among diverse geographic virus strain. This explains why human zika infection emerges outside of Asia and Africa sporadically. An important finding related to dengue is that zika antibodies have the capability to enhance DENV infection. It was the reason that in many zika-affected regions dengue allegedly remains endemic.2,40
New Approach to Dengue Diagnosis
Epidemiology of dengue in most endemic region has shifted to older-age cases, possibly due to extreme climate or environmental events, prolonged period of vector control, migration, and urbanization that lead to escalation of mosquito borne-vector susceptibility.41–43 Atypical characteristics not attributable to plasma leakage resulting from increased vascular fragility and permeability, such as gastrointestinal problem, pleural effusion, and neurological complication, have been reported.44–46 Previous studies showed that the frequency of gastrointestinal problems was 10.2% to 48.9%.44,47,48 Neurological manifestation, such as encephalitis, encephalopathy, and Guillain-Barré syndrome, was increasingly encountered in severe dengue.49,50
The clinical features of dengue infection were often similar to other febrile illnesses.44,51,52 The World Health Organization (WHO) 1997 classification would miss some cases due to these common features along with the absence of main characteristics, such as signs of bleeding or plasma leakage and retro-orbital pain in many patients. The WHO 2009 classification was able to determine the severe dengue cases not classified as DHF or DSS in WHO 1997 classification.53,54 Although the WHO dengue classification is clinically helpful, definitive diagnosis needs sophisticated examinations based on viral or antigen-antibody detection that would be difficult to implement in many endemic regions.4,55 Limited resources such as laboratories and infrastructure supports in many rural endemic areas necessitate new approach for the early detection of dengue infection.
A scoring system would be a beneficial tool to diagnose dengue disease in the early phase and predict its severity.56–59 The prerequisites for the scoring system are only clinical and simple laboratory features that would be easily obtained. The proposed dengue scoring model is shown in Tables 1 and 2. Studies found that dengue scoring system had 79.7% to 88.1% sensitivity and 68.0% to 94.9% specificity to estimate dengue illness,56,57 whereas its ability to determine DSS from dengue fever and DHF was approximately 75% (95% confidence interval: [74.18-77.57]).58,59
The proposed dengue diagnosis scoring by Chang et al. 56
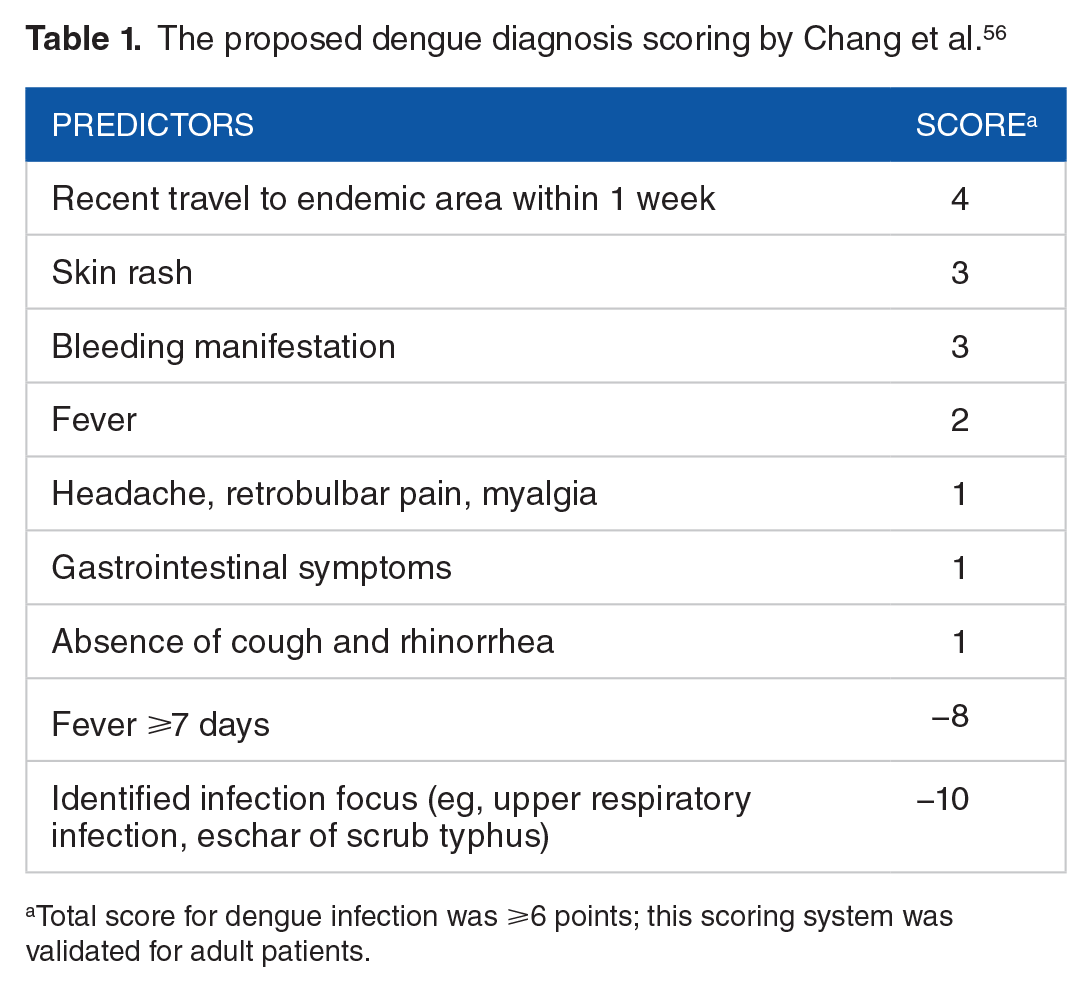
Total score for dengue infection was ⩾6 points; this scoring system was validated for adult patients.
The proposed dengue diagnosis scoring by Cucunawangsih et al. 57
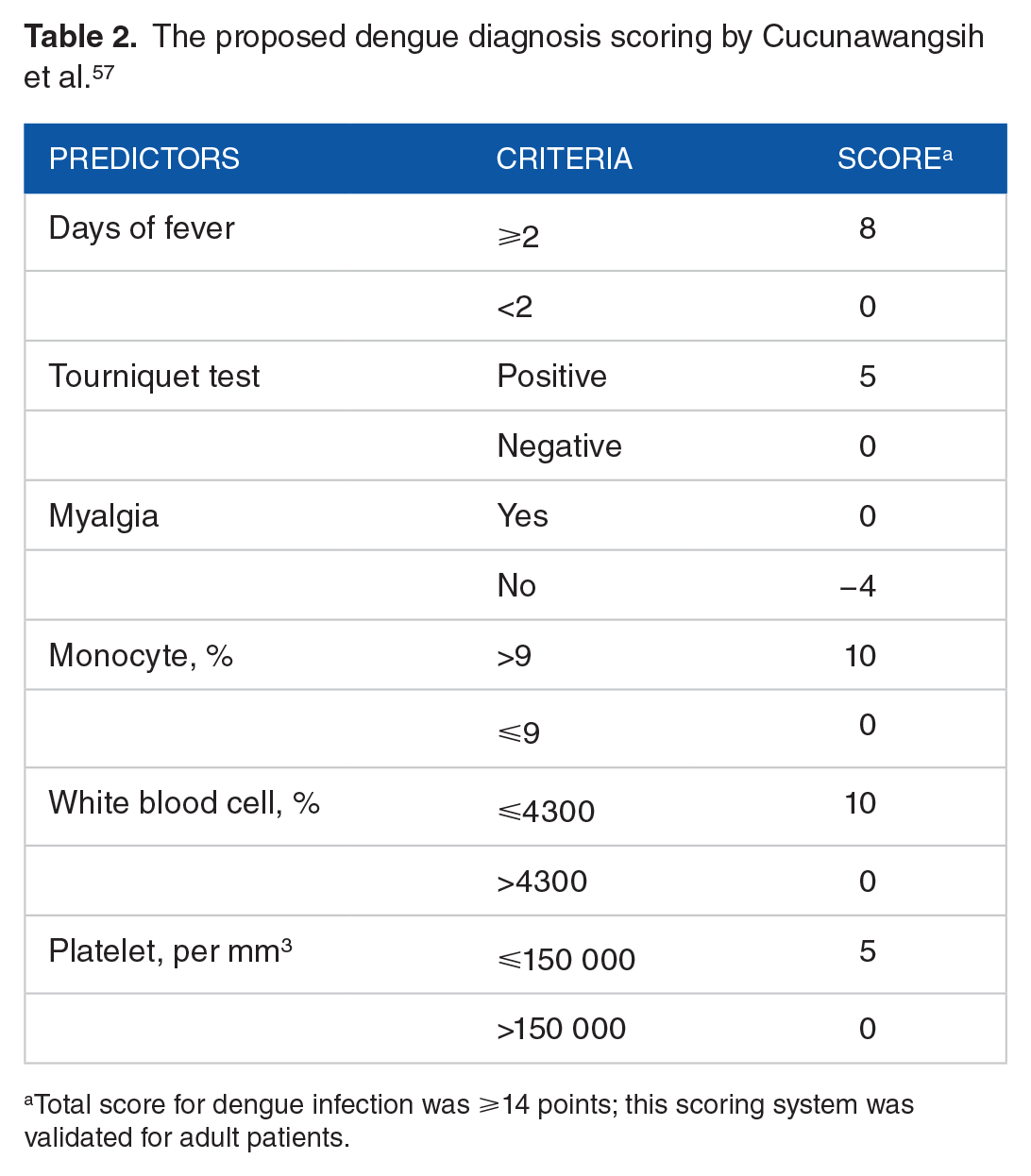
Total score for dengue infection was ⩾14 points; this scoring system was validated for adult patients.
Application of the newly developed strategic model of dengue scoring in clinical settings is to distinguish acute dengue illness from other febrile illness with or without simple laboratory data. It could assist the clinicians and public health workers to detect dengue cases immediately, thus initiating prompt treatment strategies and also specific and efficient control measures for prevention. Parallel with this, dengue risk index enables public health workers to promote effective and efficient early prevention program.
Prediction of dengue severity using scoring system has significant usefulness in daily practice because it uses simple clinical and laboratory data routinely obtained in all levels of health care facility. 58 These prognostic indicators could prevent over- or underestimation of the severity of patients with dengue. They minimize hospitalization for those who do not require and prioritize care for those who need in accordance with severity scores.
One limitation of the studies on new strategy for dengue diagnosis and prognostic indicators was that it was derived only from adult dengue cases. Thus, the validity and performance would differ when applied to general populations. Moreover, different types of dengue infection, ie, primary or secondary infection, would also confound the results of the study. Further studies involving large samples from different settings are needed to generate representative data. This resourceful and impactful scoring system can be applied to detect dengue in patients presenting to primary care facilities with acute undifferentiated fever in the absence of rapid diagnostic tests and predict its severity.
Dengue Treatment
Dengue infection is a disease with complex clinical manifestations; thus, therapeutic management should be simple, low cost yet effective in saving lives through correctly performed and timely institutionalized interventions. 12
Options for treatment are limited as there are no effective antiviral drugs for this infection accessible to date.12,60 Therefore, clinical management for dengue infection focuses on supportive care, with particular emphasis on careful fluid management.4,12
Patients in the early febrile phase with no complications may recover without requiring hospital admission with intensive daily monitoring for any bleeding manifestation or warning sign suggestive of severe dengue by health care providers. Early stage of plasma leakage or critical phase indicated the need of hospitalization.4,12 Sensible and optimal fluid resuscitation is essential to maintain organ perfusion during the critical phase which simultaneously encourages favorable outcomes.61,62 Isotonic crystalloid solutions, such as 0.9% normal saline, Ringer’s lactate, or Hartmann’s solution, could be used for initial resuscitation of those with shock according to the latest WHO guidelines on fluid management algorithm.4,12,63
The goal of fluid resuscitation is to prevent complications of vascular leakage and hypovolemic shock. 60 Despite fluid therapy intervention, it was estimated that 30% of DSS cases experienced recurrent episodes of shock. 62 Dengue infection covers diverse epidemiology, with most of the cases involving adults. Previous studies on fluid therapy were mostly conducted in children; thus, it is important to enclose adult patients in the future studies.
Transfusion of blood product, such as packed red cell, platelets, or fresh-frozen plasma, can be lifesaving for patients with severe bleeding. However, the use of prophylactic platelet transfusion in severely thrombocytopenic dengue patients without any sign of bleeding manifestation is not supported by evidence.4,60
The rationale of dengue antivirals to enhance effective clinical management is still unmet; therefore, the role of these drugs in the treatment of dengue infection was limited.62,64 There are currently ongoing clinical research works to find specific antidengue drugs.64,65 Direct dengue antivirals would be useful to reduce the severity of the disease, although these drugs need to inhibit all 4 viral serotypes.
Conclusions
Dengue poses a health threat in almost all countries located in tropical and subtropical territorials. There is an increasing new trend of concurrent multiple DENV, CHIKV, and ZIKV in hyperendemic dengue areas. The social and economic burden of mosquito-borne viral infection is widely alarming, and the proven global impact of these diseases is discredited. The observation of dengue epidemiology highlights the needs to strengthen control of the emerging virus and management in an outbreak. Dengue and other mosquito-borne viral infection morbidity and mortality can be reduced by performing early identification of high-risk patients and appropriate management for severe cases. Dengue virus infection morbidity can be diminished by strengthening case prediction and finding through epidemiological surveillance based on local data. New strategy for early dengue infection and severity risk prediction early in the course of illness is indispensable so that management strategies can be promptly implemented. Routine clinical indicators may be used to predict dengue infection in high-burden areas where suitable allotment of restricted resources is crucial for the outcome.
Footnotes
Acknowledgements
The authors express their gratitude to Faculty of Medicine, Pelita Harapan University in Tangerang, Indonesia, who made this work possible.
Peer review:
Seven peer reviewers contributed to the peer review report. Reviewers’ reports totaled 608 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
C conceived the idea of the study, analyzed, and writing the manuscript; NPHL participated in data gathering and revised the manuscript. All authors contributed and agreed upon the final manuscript.
