Abstract
Background:
Circadian rhythms are physiological and behavioral cycles with a period of approximately 24 hours that control various functions including gene expression. Circadian disruption is associated with a variety of diseases, especially cancer. Although some of the oncogenes and tumor suppressor genes (TSGs) are known as clock-controlled genes (CCGs), the analysis and annotation of circadian expression of most human oncogenes and TSGs are still lacking. This study aims to investigate the circadian expression of a list of human oncogenes and TSGs.
Methods:
A bioinformatic analysis was conducted on a gene library comprising 120 genes to investigate the circadian expression of human oncogenes and TSGs. To achieve this purpose, the genotranscriptomic data were retrieved from COSMIC and analyzed by R statistical software. Furthermore, the acquired data were analyzed at the transcriptomic and proteomic levels using several publicly available databases. Also, the significance of all analyses was confirmed statistically.
Results:
Altogether, our results indicated that 7 human oncogenes/TSGs may be expressed and function in a circadian manner. These oncogenes/TSGs showed a circadian expression pattern at CircaDB database and associated with at least one of the circadian genes/CCGs based on both genotranscriptomic and correlation analyses.
Conclusions:
Although 4 of 7 finally outputted genes have been previously reported to be clock controlled, heretofore there is no report about the circadian expression of 3 other genes. Considering the importance of oncogenes/TSGs in the initiation and progression of cancer, further studies are suggested for the identification of exact circadian expression patterns of these 3 human oncogenes/TSGs.
Introduction
The circadian system refers to an internal time-keeping system that has been shaped by evolution over millions of years and exists in almost every organism studied from archaea to human.1,2 Circadian rhythms are physiological and behavioral cycles synchronized by circadian clocks in a period of approximately 24 hours.3,4 The suprachiasmatic nucleus, the master circadian pacemaker, is located in the anterior part of the hypothalamus of mammals; however, the same oscillating system exists in peripheral tissues and even all body cells including immortalized cells.5,6 The timing of circadian rhythms can be notably variable among different individuals, and twin studies have indicated that nearly half of this variability is environmentally controlled. 7 Circadian rhythms are orchestrated by the master, subsidiary, and peripheral pacemakers through transcriptional and translational feedback loops involving several clock genes. 8 Numerous microarray studies investigating the circadian control of gene expression have reported that a considerable proportion of genomes of many organisms are clock controlled. 4 Sengupta et al 9 have demonstrated that transcriptional rhythms are regulated in a circadian manner in Caenorhabditis elegans. The mammalian transcriptome is also circadian regulated through rhythmic recruitment of key modifiers of chromatin structure and transcriptional/translational processes. The circadian transcriptional regulation, together with posttranslational modifications, establishes the central and peripheral circadian oscillators. 10 The circadian regulation covers not only clock genes (circadian genes) but also various clock-controlled genes (CCGs) including several cell cycle genes. 11
The integrity of the circadian timing system (CTS) is interconnected with the quality of life and can predict the survival rate of patients with cancer. 12 Abundant evidence indicates that circadian disruption, which is the disruption of circadian rhythms, is associated with a wide variety of long-term diseases including cancer. 13 Several environmental factors such as night shift work, exposure to artificial light, and irregular diet result in circadian disruption and finally can increase the risk of cancer. 14 Cell cycle, cell proliferation and metabolism, DNA damage repair, and apoptosis are strictly correlated with the circadian clock in peripheral tissues.15–17 As restriction of cell proliferation to defined time windows is exerted by circadian clocks, dysfunction of molecular and cellular clockworks is involved in uncontrolled cell proliferation and tumorigenesis.18–20 Circadian disruption has been implicated in the development of various human cancers such as breast, 21 ovarian, 22 prostate, 23 gastric, 24 colorectal, 25 and hematopoietic26,27 cancers. Frequent studies have indicated that defects in circadian genes resulted by single-nucleotide polymorphism, deletion, etc, are closely associated with increased risk of cancer. 15 In addition, aberrant epigenetic modifications (eg, hypermethylation) due to circadian disruption can lead to a variety of cancers even in the absence of circadian gene mutations. 28 Koike et al in a study on core circadian clock in mammals showed that almost 22% of the circadian regulation of messenger RNA (mRNA) transcripts is exerted at the transcriptional level and the rest is posttranscriptionally regulated. Thus, different posttranscriptional regulations including various epigenetic modifications (eg, H3K9ac, H3K27ac, and H3K4me3) play a significant role in the circadian cycling of mRNA transcripts. 29
Considering the potential roles of oncogenes and tumor suppressor genes (TSGs) in the initiation and progression of all types of cancer, characterization of their unknown features can significantly help cancer diagnosis, prognosis, and treatment. Several oncogenes and TSGs have been reported to be clock controlled in model organisms, 30 but the circadian expression of most human oncogenes and TSGs remains yet to be investigated. In this study, we conducted a bioinformatic analysis to explore the circadian expression of a list of human oncogenes and TSGs. Moreover, to achieve more valuable results and reduce false positives, we analyzed the obtained data at the transcriptomic and proteomic levels. An outline of the procedure is given in Figure 1.

Outline of bioinformatic analysis of circadian expression of oncogenes and tumor suppressor genes (TSGs). The correlation and association of all genes under investigation were analyzed using a combination of mutation distribution and over/underexpression data in different cancers which were extracted from the COSMIC database. These analyses resulted in 64 outputted genes. Then, the coexpression and other associations of all 64 outputted genes were analyzed using different online tools and databases. Also, searching the CircaDB database illustrated a robust circadian expression pattern in 12 oncogenes/TSGs. At last, the oncogenes and TSGs which had associations with at least one of the circadian genes/clock-controlled genes (CCGs) and showed a circadian expression pattern at CircaDB database were outputted and named as Circadian Oncogenes/TSGs.
Methods
Gene library preparation
A list of most known human oncogenes (25 genes) and TSGs (52 genes) was obtained from the Cell Signaling Technology (http://www.cellsignal.com). Also, most known circadian genes (13 genes) and CCGs (30 genes) were mined from the literature17,31,32 and included in the gene library. These gene lists are available in Supplementary file 1.
Genotranscriptomic analysis
Mutation distribution and overall overexpression and underexpression of all desired genes (120 genes) were extracted from 61 498 tumor samples, covering 39 tissue types, using COSMIC database v72 (a comprehensive resource of somatic mutations in human cancer) (http://cancer.sanger.ac.uk). 33 These data are available in Supplementary file 2.
Cluster analysis
All the genotranscriptomic data were measured by log2 to increase the sensitivity and accuracy of our next analyses. Then, a heatmap was achieved by R statistical software using genotranscriptomic data obtained from the COSMIC database, separately for oncogenes-circadian genes, oncogenes-CCGs, TSGs-circadian genes, and TSGs-CCGs (Supplementary file 3). The heatmaps were generated using “pheatmap” package and its default clustering method (complete) and distance function (Euclidean). Then, the statistical significance of clustering of all heatmaps was tested using SigClust R package. 34 The oncogenes and TSGs with the most proximity (lowest Euclidean distance) to any of circadian genes/CCGs were outputted (altogether 37 genes). Hereinafter, these outputted oncogenes and TSGs are referred to as Notable Oncogenes and Notable TSGs, respectively.
Correlation analysis
COXPRESdb database (http://coxpresdb.jp) 35 was used to analyze the coexpression between Notable Oncogenes/TSGs, selected by cluster analysis, and their corresponding circadian genes/CCGs. The COXPRESdb database provides online tools for coexpression analysis of 2 or more genes based on microarray data sets. Also, cross-validated coexpression of each Notable Oncogene/TSG and its corresponding circadian gene/CCG was investigated using SEEK database (an online computational tool for gene coexpression analysis) (http://seek.princeton.edu) 36 with its default settings. Furthermore, GeneMANIA (http://www.genemania.org) 37 and iRefWeb v13.0 (http://wodaklab.org/iRefWeb/) were used to analyze the association of each Notable Oncogene/TSG and its corresponding circadian gene/CCG. GeneMANIA and iRefWeb databases assist in the investigation of the associations such as physical interaction and common pathway between desired genes.
Circadian expression analysis
CircaDB database (last version, update—June 9, 2014) (http://circadb.hogeneschlab.org/) 38 was used to investigate the circadian expression of Notable Oncogenes/TSGs (altogether 37 genes) in mouse models. CircaDB database is a circadian expression search engine based on microarray and statistical analyses. The oncogenes and TSGs that showed robust circadian expression were outputted.
Statistical analysis
All the heatmaps were achieved by R statistical software using pheatmap package (https://cran.r-project.org/web/packages/pheatmap/index.html) and its default clustering method (complete) and distance function (Euclidean). The statistical significance of clustering of all heatmaps was tested using SigClust R package (https://cran.r-project.org/web/packages/sigclust/index.html). Also, all other statistical and mathematical analyses were done by R statistical software (R Development Core Team (2014), freely available at http://www.r-project.org). In all the analyses, the P < .05 was used as the significance threshold.
Results
Common mutation distribution pattern and over/underexpression
Analysis of the statistical significance of clustering of all heatmaps using SigClust approved the validity of our clustering approach, which means that clusters are well established and organized within heatmaps (P < .02) (Figure 2). Based on the cluster analysis using genotranscriptomic data, 19 oncogenes (Notable Oncogenes) and 18 TSGs (Notable TSGs) had a correlation (vicinity) with at least one of the circadian genes/CCGs (Table 1). This vicinity was indicative of the correlation in mutation distribution and over/underexpression between gene neighbors.
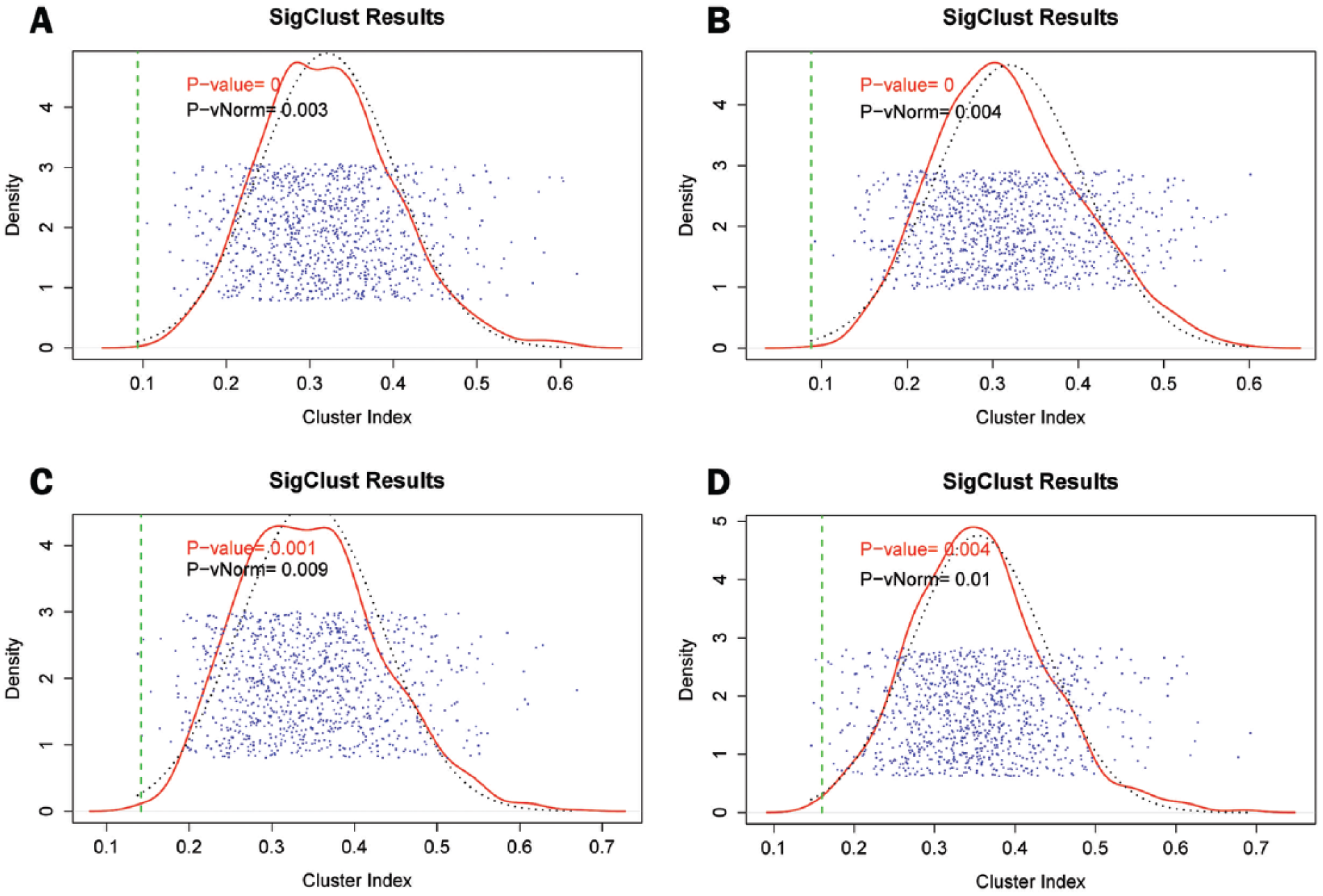
Statistical significance of clustering of heatmaps. This plot consists of 4 parts, including A (Oncogenes-CCGs), B (Oncogenes-Circadian genes), C (TSGs-CCGs), and D (TSGs-Circadian genes), which represent the statistical significance of clustering of heatmaps. The abbreviations CCGs and TSGs are representative of clock-controlled genes and tumor suppressor genes, respectively. These analyses were done using SigClust package of R statistical software.
Notable Oncogenes/TSGs and their corresponding circadian genes/CCGs.

Abbreviations: ATR, ATR serine/threonine kinase; AURKA, aurora kinase A; BAX, BCL2-associated X protein; BCL6, B-cell CLL/lymphoma 6; BLM, Bloom syndrome, RecQ helicase-like; BMPR2, bone morphogenetic protein receptor type II (serine/threonine kinase); BRAF, B-Raf proto-oncogene, serine/threonine kinase; CCND1, cyclin D1; CCNE1, cyclin E1; CDK1, cyclin-dependent kinase 1; CDK2, cyclin-dependent kinase 2; CDK4, cyclin-dependent kinase 4; CDKN1A, cyclin-dependent kinase inhibitor 1A (p21, Cip1); CDKN2A, cyclin-dependent kinase inhibitor 2A; CHEK1, checkpoint kinase 1; CHEK2, checkpoint kinase 2; CRY1, cryptochrome circadian clock 1; CSNK1D, casein kinase 1 delta; CSNK1E, casein kinase 1 epsilon; CTNNB1, catenin (cadherin-associated protein), beta 1, 88 kDa; ESRRB, estrogen-related receptor beta; FANCA, Fanconi anemia complementation group A; FH, fumarate hydratase; FOXO1, forkhead box O1; GLI1, GLI family zinc finger 1; HMGCR3, hydroxy-3-methylglutaryl-CoA reductase; INPP4B, inositol polyphosphate-4-phosphatase type II B; KITLG, KIT ligand; KMT2A, lysine (K)-specific methyltransferase 2A; KRAS, Kirsten rat sarcoma viral oncogene homolog; LEF1, lymphoid enhancer-binding factor 1; MITF, microphthalmia-associated transcription factor; MUTYH, mutY DNA glycosylase; NBN, nibrin; NR1D1, nuclear receptor subfamily 1, group D, member 1; PARP1, poly (ADP-ribose) polymerase 1; NPAS2, neuronal PAS domain protein 2; PDGFRA, platelet-derived growth factor receptor, α-polypeptide; PER3, period circadian clock 3; PIK3CA, phosphatidylinositol-4,5-bisphosphate 3-kinase, catalytic subunit alpha; PPARA, peroxisome proliferator-activated receptor alpha; PPARD, peroxisome proliferator-activated receptor delta; PRKDC, protein kinase, DNA-activated, catalytic polypeptide; RTKN, rhotekin; RARA, retinoic acid receptor alpha; RORA, RAR-related orphan receptor A; SIRT1, sirtuin 1; SMAD3, SMAD family member 3; SMO, smoothened, frizzled class receptor; SREBF1, sterol regulatory element–binding transcription factor 1; TAL1, T-cell acute lymphocytic leukemia 1; TCF3, transcription factor 3; VEGFA, vascular endothelial growth factor A; WEE1, WEE1 G2 checkpoint kinase; WNT1, wingless-type MMTV integration site family member 1; WNT5A, wingless-type MMTV integration site family member 5A; WRN, Werner syndrome, RecQ helicase-like; XPA, xeroderma pigmentosum complementation group A; XPC, xeroderma pigmentosum complementation group C.
Corresponding gene: The gene with the most proximity (lowest Euclidean distance) based on cluster analysis.
Notable Oncogene.
Notable TSG (tumor suppressor gene).
Circadian gene.
CCG (clock-controlled gene).
Association of Notable Oncogenes/TSGs and their corresponding genes
The coexpression analysis using COXPRESdb database demonstrated that 21 Notable Oncogenes/TSGs had coexpression with their corresponding circadian genes/CCGs in different tissues (Table 2 and Supplementary file 4). Also, analysis of 50 first ranked data sets of the results of SEEK database indicated that most Notable Oncogenes/TSGs (except PIK3CA, PRKDC, FANCA, MUTYH, XPA, BRAF, CTNNB1, and WNT1) had significant cross-validated coexpression (P < 0.05, difference of cross-validation scores <1) with their corresponding circadian genes/CCGs in various tissues and diseases (Supplementary file 5). Furthermore, we found several associations, such as coexpression and physical interaction, between 9 Notable Oncogenes/TSGs and their corresponding circadian genes/CCGs using GeneMANIA and iRefWeb v13.0 (Figure 3).
Coexpression of Notable Oncogenes/TSGs and their corresponding circadian genes/CCGs.

Gene pairs whose correlation value was more than 0.1 were outputted.
Samples whose contribution score was more than 10 and the difference of Z score expression of query genes less than 1 were outputted.
Notable Oncogene.
Notable TSG (tumor suppressor gene).
CCG (clock-controlled gene).
Circadian gene.
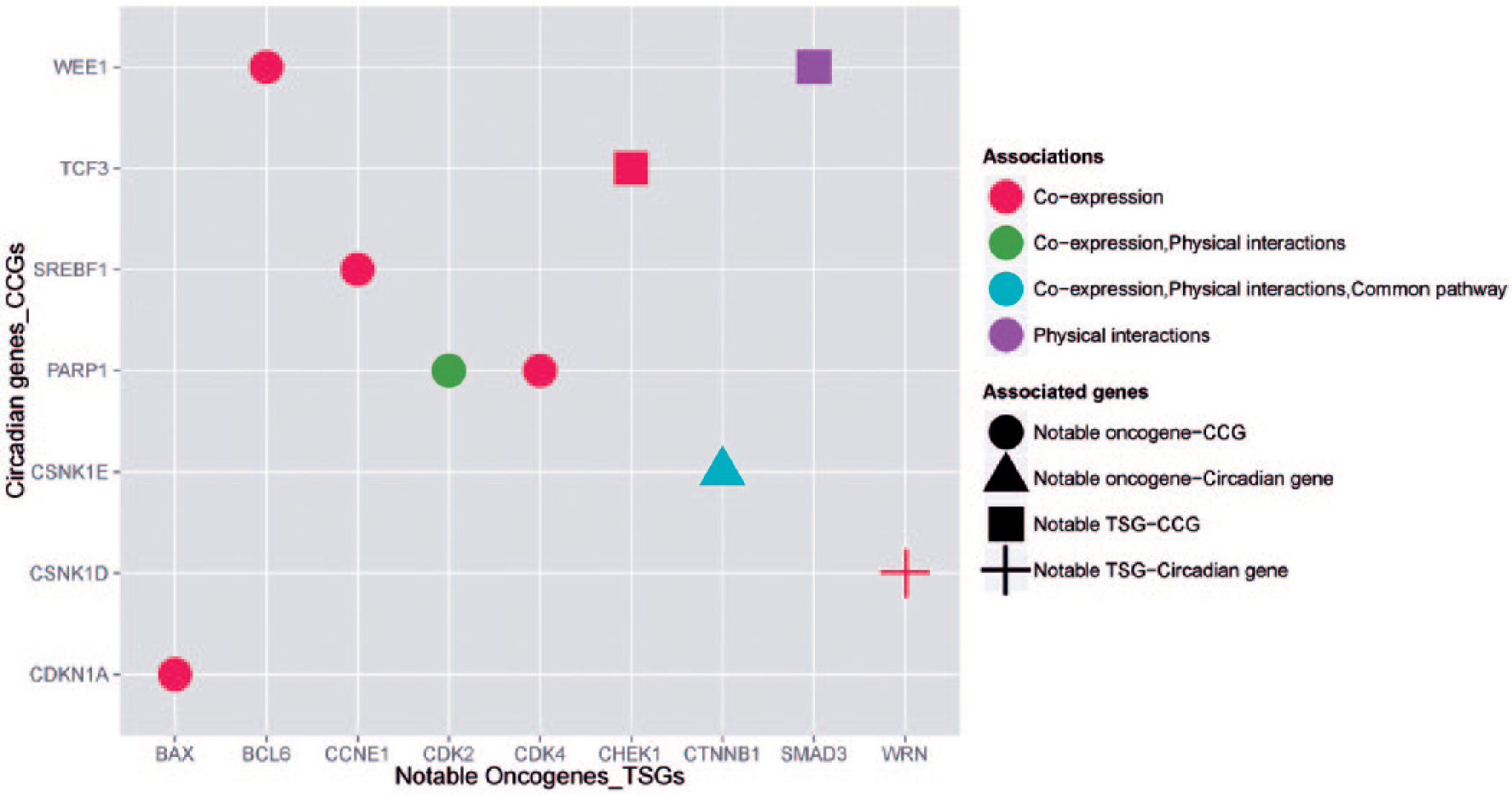
The association between Notable Oncogenes/TSGs and their corresponding circadian genes/CCGs. This plot indicates that 9 Notable Oncogenes/TSGs have various associations with their corresponding circadian genes/CCGs. The abbreviations CCGs and TSGs are representative of clock-controlled genes and tumor suppressor genes, respectively. The data were drawn from iRefWeb v13.0 and GeneMANIA databases. The scatter plot was achieved using R statistical software.
Circadian expression of Notable Oncogenes/TSGs
Investigation of the circadian expression of Notable Oncogenes/TSGs using CircaDB database illustrated a robust circadian expression in 7 oncogenes and 5 TSGs (P < .05) (Table 3 and Supplementary file 6). However, these Notable Oncogenes/TSGs showed diverse circadian expression patterns. In the end, the oncogenes and TSGs that showed a circadian expression pattern at CircaDB database and had associations with at least one of the circadian genes/CCGs based on both genotranscriptomic and correlation analyses were outputted (7 genes) and named as Circadian Oncogenes/TSGs (Table 4). Of these 7 genes, 3 genes including SMO, RTKN, and WRN have neither been previously reported to be circadianly expressed nor to be clock controlled in human.
Notable Oncogenes and TSGs with a circadian expression pattern.

Oncogene.
TSG (tumor suppressor gene).
P value of circadian expression of mouse homologs of Oncogenes/TSGs, which were extracted from CircaDB database.
Circadian Oncogenes/TSGs and their corresponding circadian genes/CCGs.
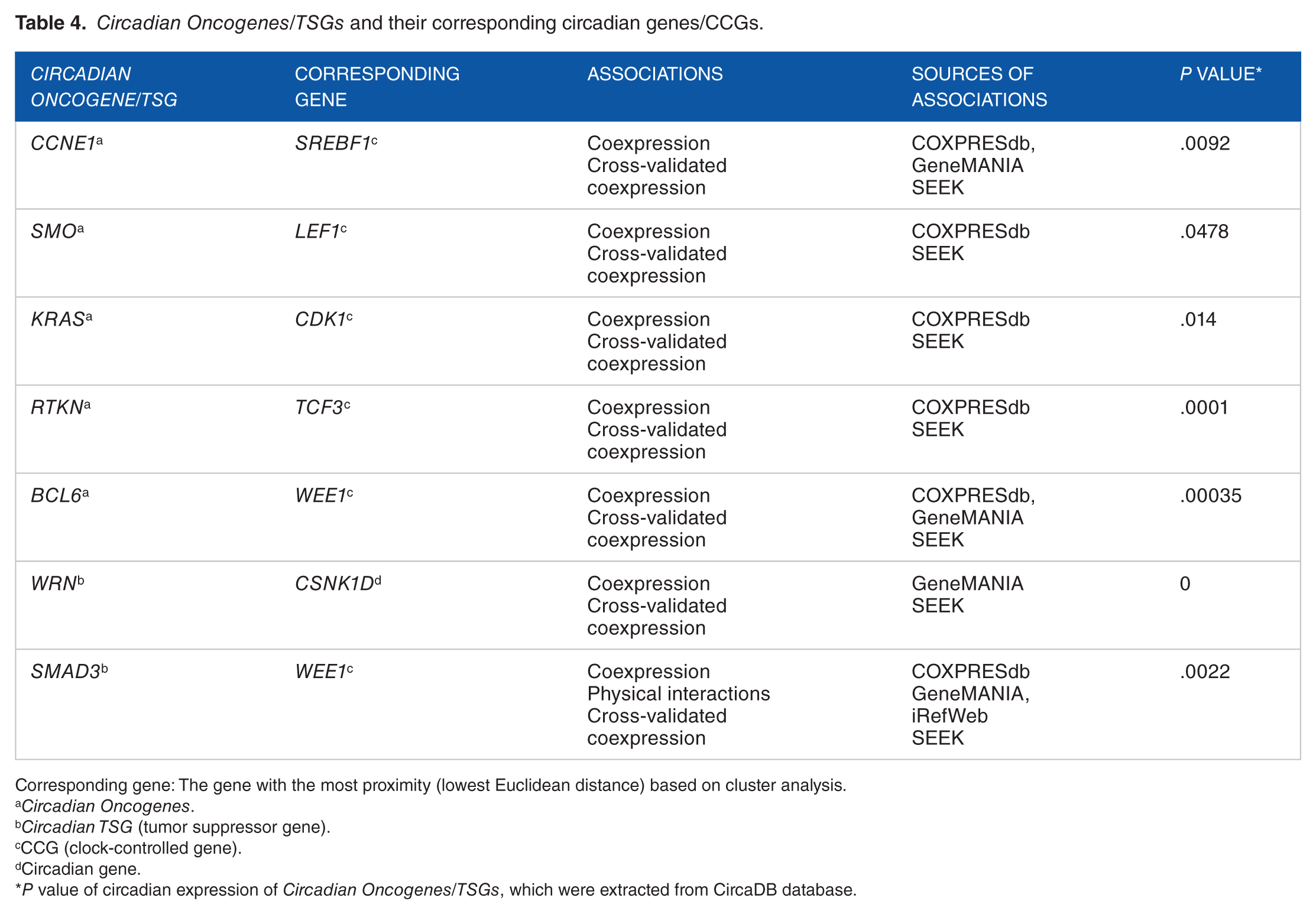
Corresponding gene: The gene with the most proximity (lowest Euclidean distance) based on cluster analysis.
Circadian Oncogenes.
Circadian TSG (tumor suppressor gene).
CCG (clock-controlled gene).
Circadian gene.
P value of circadian expression of Circadian Oncogenes/TSGs, which were extracted from CircaDB database.
Discussion
Circadian rhythms and circadian gene expression are key factors in the regulation of physiology and behavior at the molecular, cellular, and tissue levels of almost all organisms. Aberrant expression of circadian genes may result in genomic instability and enhanced cell proliferation through transactivation of targets that control cell cycle and apoptosis. 11 It has been reported that activation of Per2 (a circadian gene) leads to c-myc overexpression and an increased tumor incidence. 39 Also, it has been frequently reported that carcinogenic dysfunctions within the cells including disrupted cell cycle, DNA damage response, and apoptosis are significantly interconnected with circadian disruption. Relógio et al 40 demonstrated that perturbations induced by even a single oncogene can disrupt the mammalian circadian clock. In this context, discovering underlying processes and pathways that are clock controlled might have an important impact on our understanding of cancer etiology, which requires identification of circadian-regulated (clock-controlled) genes and their circadian expression pattern. 41 This circadian regulation is done by the circadian machinery which exists in all body cells. 39 Also, there are lots of known CCGs which play different roles within the cells. In addition, some of the circadian genes have been known to have an oncogenic or tumor suppressive function. The PER2 circadian gene, for example, has been reported to have a tumor suppressive activity. 42 However, some of the oncogenes and TSGs may be expressed and function in a circadian manner. Although some of the oncogenes and TSGs such as MYC, 43 TP53, 31 CDKN1A, 17 JUN, 31 and FOS 31 are known as CCGs, investigation of the circadian expression and function of all human oncogenes and TSGs can potentially help prognosis/diagnosis and treatment of cancer in a variety of ways. Furthermore, identification of the signaling pathways activated and controlled by circadian-regulated factors may potentially help the development of novel strategies for cancer prevention and treatment. 20 Sharma et al 44 discovered a circadian gene expression within C6 glioma tumorspheres enriched in cancer stem cells. This observation can be suggestive of a hypothesis that circadian disruption may result in changing the circadian pattern of gene expression into a new pattern that leads to desynchronization of intra/intercellular networks and consequently causes cancer. These data elucidate the potential roles of circadian rhythms in our health and how their disruption or re-entrainment can lead to cancer development or survival, respectively.
Identification of groups of genes that show similar expression patterns can be achieved by coexpression analysis that compares gene expression under hundreds of different conditions. 45 Furthermore, protein coexpression analysis can help in the identification of proteins in the same network. 46 Our results demonstrate that 7 human oncogenes and TSGs are distinct from other cancer-related genes under investigation in that they successfully passed all the bioinformatic analyses. These bioinformatic tests include the analysis of genotranscriptomic association (ie, common mutation distribution pattern and over/underexpression in cancer) and significant correlations (ie, coexpression and/or physical interaction/common pathway) of cancer genes with their corresponding circadian genes/CCGs and the analysis of their circadian expression in mouse models. Thus, we hypothesized that these 7 genes might have a robust circadian expression in human. Although literature review shows that the CCNE1, 47 SMAD3, 48 BCL6, 49 and KRAS 50 genes have been previously reported to be clock controlled, there is no report about the circadian expression of 3 other genes including SMO, RTKN, and WRN. The point that 4 of 7 genes have been previously experimentally approved to be clock controlled confirms the validity and integrity of our methodology. Nevertheless, the circadian or clock-controlled expression of the 3 aforementioned genes has to be experimentally approved. It is also worth mentioning that these 7 genes are filtrated according to our methodology, and other cancer-related genes that have not yet been reported to be circadianly expressed might be clock-controlled as well, which requires further studies and experiments to be identified. Considering the importance of cancer-associated genes and also circadian rhythms in human health, further studies and experiments are suggested regarding the identification of exact circadian expression patterns of these 3 oncogenes/TSGs and their associations with circadian genes/CCGs, which may significantly help cancer prognosis, diagnosis, and treatment.
All the present data about the CTS and circadian gene expression can potentially be used to devise new therapeutic approaches for cancer treatment. In this context, human peripheral blood cells can be used to investigate the circadian rhythms and their associated disorders. 8 Circadian realignment (ie, adherence to scheduled sleep-wake times, exposure to light, and avoidance of exposure to light at inappropriate times) can be used for the treatment of circadian-associated diseases. 51 In addition, mealtime schedule and pharmaceuticals can be used for chronotherapeutic treatments.1,28 The action, disposition, therapeutic efficacy, and toxicity of various drugs are regulated by circadian gene expression and based on dosing time. Hence, identification of circadian gene expression can importantly help optimization of dosing time for existing drugs and the development of new pharmaceuticals. 52 Thus, further studies are suggested regarding the identification of the best schedule for shift workers, which can significantly help prevention of circadian-associated diseases, especially cancer.
Supplemental Material
sj-pdf-3-bbi-10.1177_1177932217746991 – Supplemental material for Bioinformatic Analysis of Circadian Expression of Oncogenes and Tumor Suppressor Genes
Supplemental material, sj-pdf-3-bbi-10.1177_1177932217746991 for Bioinformatic Analysis of Circadian Expression of Oncogenes and Tumor Suppressor Genes by Adrian Salavaty, Niloufar Mohammadi, Mozhdeh Shahmoradi and Maryam Naderi Soorki in Bioinformatics and Biology Insights
Supplemental Material
sj-pdf-4-bbi-10.1177_1177932217746991 – Supplemental material for Bioinformatic Analysis of Circadian Expression of Oncogenes and Tumor Suppressor Genes
Supplemental material, sj-pdf-4-bbi-10.1177_1177932217746991 for Bioinformatic Analysis of Circadian Expression of Oncogenes and Tumor Suppressor Genes by Adrian Salavaty, Niloufar Mohammadi, Mozhdeh Shahmoradi and Maryam Naderi Soorki in Bioinformatics and Biology Insights
Supplemental Material
sj-xlsx-1-bbi-10.1177_1177932217746991 – Supplemental material for Bioinformatic Analysis of Circadian Expression of Oncogenes and Tumor Suppressor Genes
Supplemental material, sj-xlsx-1-bbi-10.1177_1177932217746991 for Bioinformatic Analysis of Circadian Expression of Oncogenes and Tumor Suppressor Genes by Adrian Salavaty, Niloufar Mohammadi, Mozhdeh Shahmoradi and Maryam Naderi Soorki in Bioinformatics and Biology Insights
Supplemental Material
sj-xlsx-2-bbi-10.1177_1177932217746991 – Supplemental material for Bioinformatic Analysis of Circadian Expression of Oncogenes and Tumor Suppressor Genes
Supplemental material, sj-xlsx-2-bbi-10.1177_1177932217746991 for Bioinformatic Analysis of Circadian Expression of Oncogenes and Tumor Suppressor Genes by Adrian Salavaty, Niloufar Mohammadi, Mozhdeh Shahmoradi and Maryam Naderi Soorki in Bioinformatics and Biology Insights
Supplemental Material
sj-xlsx-5-bbi-10.1177_1177932217746991 – Supplemental material for Bioinformatic Analysis of Circadian Expression of Oncogenes and Tumor Suppressor Genes
Supplemental material, sj-xlsx-5-bbi-10.1177_1177932217746991 for Bioinformatic Analysis of Circadian Expression of Oncogenes and Tumor Suppressor Genes by Adrian Salavaty, Niloufar Mohammadi, Mozhdeh Shahmoradi and Maryam Naderi Soorki in Bioinformatics and Biology Insights
Footnotes
Acknowledgements
The authors would like to thank Dr Ali Mohammad Foroughmand and Dr Mohammadreza Hajjari for their valuable scientific consultations and Ms Marzie Samimifar for the proof reading of English language.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Kashan and the Shahid Chamran University of Ahvaz in Iran.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AS: Prepared the gene library; Conducted the statistical, association, bioinformatic, and meta-analyses; Prepared the figures and tables; Wrote the first manuscript draft; Involved in critical revisions of the manuscript draft. NM: Made substantial contributions to acquisition of data; Analyzed the heatmaps; Conducted the proteomic analyses; Involved in critical revisions of the manuscript draft. MS: Made substantial contributions to acquisition of data; Conducted the genotranscriptomic and circadian expression analyses; Involved in critical revisions of the manuscript draft. MNS: Made substantial contributions to conception and design of the study; Made substantial contributions to interpretation of data; Revised and proof read the final version of the manuscript draft. All authors read and approved the final manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
