Abstract
Objective:
To analyze drug utilization (DU) pattern of antidiabetes drug (ADD) prescription in elderly type 2 diabetes mellitus (T2DM) in rural West Bengal based on 2016 World Health Organization (WHO) Anatomical Therapeutic Chemical/Defined Daily Dose (ATC/DDD) Index.
Methods:
This was a prospective observational study. Prescription data of 600 elderly patients (age > 60 years) attending outpatient clinic were screened over 12 months (January 2015 to January 2016) from 5 different rural hospitals in West Bengal. Pooled data were sorted and classified in accordance with 2016 ATC/DDD WHO Index. Direct cost associated and consumption of ADD were measured as DDD/1000 patients/day. The adverse drug reactions (ADRs) related to antidiabetic medicines were monitored.
Results:
During the study period, mean age of patients recorded was 66.4 ± 5.0 years, with 66.6% (n = 396) having history of T2DM > 5 years. Follow-up encounters (n = 2328) revealed metformin (94.67%), sulfonylureas (SUs) (50.54%), pioglitazone (24.22%), voglibose (22.50%), insulin (9.75%), and acarbose (6.82%) to be more prevalent, constituting DU 90% (92.01%). Combination of metformin plus SU was recorded in most of the patients (56%). Insulin, however, was found to be an underutilized class (P < .005). The DDD/1000 patients/day of metformin (2.918), glimepiride (1.577), and gliclazide (0.069) conformed to 2016 WHO ATC/DDD Index. The total ADD consumption during study period was 5.03 DDD/1000 patients/day. The average drug cost per encounter per day was Rs 11.24 ± 2.01. Nineteen ADRs were reported and their descriptions were found to be of hypoglycemia (n = 9), pedal edema (n = 2), and gastrointestinal upsets (n = 8). Target glycemic status was achieved in 40% monthly follow-up encounters. Low-store drug availability and poor compliance to treatment (>60%) were major determinants. Lack of regular aerobic exercises (>85%) and proper knowledge regarding medical nutrition therapy (MNT) (>80%) and low average consultation time (3.5 ± 0.6 minutes) were important contributing factors.
Conclusions:
The study exhibited increased utilization of 2 drug combinations of oral ADD and lower utilization of insulin during study period. Such inferences merit further exploration.
Introduction
The global prevalence of type 2 diabetes mellitus (T2DM) in adults aged ⩾20 years was estimated to be 171 million in 2000 and is expected to be 366 million by 2030. 1 World Health Organization (WHO) projects number of patients with T2DM in India to be 79.4 million by 2030. 2 Presently, 26% of elderly population in India have T2DM. 2 Increasing urbanization, aging population, and physical inactivity are all contributing to the rise of T2DM worldwide in the 21st century. 3 In general, the chances and incidence of complications are higher in elderly as bodily function (eg, hepatorenal) decline progressively with age. 4 Several studies have demonstrated that good glycemic control and mitigation of complications are the cornerstones of T2DM management.5-8
Oral hypoglycemic agents are the first-line therapy for T2DM in India. 9 The choice antidiabetes drug (ADD) largely depends on target Glycated Hemoglobin (A1C), drug safety and efficacy profile, and therapy cost-effectiveness. However, a significant proportion of elderly patients with T2DM are on concomitant drugs for various comorbidities (eg, hypertension, ischemic heart disease, and neuropathy). This principal of “polypharmacy” is sometimes irrational and even detrimental, hence requires strict therapeutic vigilance. Moreover, for a developing country like India, the economic burden of T2DM is concerning, mainly because of the complexity involved in identifying the direct and indirect costs of the disease therapy. Considering the practice of “polypharmacy” to be fairly common, for an estimate of approximately 20 million patients with T2DM in India, the annual estimated cost for health care could be Rs 90 200 million (US $2.2 billion). 10 Assuming an average Indian earning less than Rs 33.0 (US $0.48) per day in rural areas, 11 these costs are disproportionately high.
Study rationale
The principal aim of drug utilization (DU) research studies is to facilitate “rational drug prescribing.” For individual patients, “rational” use of a drug implies the “prescription of a well-documented drug in an optimal dose on the right indication, with the correct information and at an affordable price.” 12 Without knowledge on how drugs are being prescribed and used, it is difficult to initiate rational drug prescribing practices and suggest measures to alter prescribing habits for the better.
This study tries to assess pattern of utilization of ADD in rural eastern India and assess how close they are to optimal care, determine the different variables that can affect this pattern, and evaluate the effect of this practice on health outcome by assessing the metabolic parameters. Although several studies have been undertaken in the urban setup, very few data exist depicting the trend in rural setups, where >60% of Indian population reside and access to specialty health care is limited. This study addresses this need and draws important insights for health care policymakers.
Objective
To analyze DU pattern of ADD prescription in elderly T2DM in rural West Bengal based on 2016 WHO Anatomical Therapeutic Chemical/Defined Daily Dose (ATC/DDD) Index, 13 which “serves as a tool for drug utilization research in order to improve quality of drug use and compare drug consumption statistics at international levels.”
Materials and Methods
This study was a prospective, longitudinal study conducted over 12 months after obtaining approval from Institutional Ethics Committee at R. G. Kar Medical College and Hospital, Kolkata (R. G. Kar MCH). Prescription data of elderly patients with T2DM (age > 60 years) attending outpatient clinic for follow-up were collected from 5 different rural hospitals adjoining R. G. Kar MCH. Informed consent was obtained from all the patients before conducting the study. Prescriptions of patients with gestational diabetes were excluded. Data once obtained were sorted and classified in accordance with 2016 ATC/DDD WHO Index. Parameters depicting patient care and prescribing patterns were also assessed. Assessment of metabolic parameters was based on 2015 American Diabetes Association (ADA) guidelines. 14 Any adverse event arising was notified to the Institutional Pharmacovigilance Committee at R. G. Kar MCH. Table 1 describes parameters which were assessed after pooling of data based on WHO Guidelines on How to Investigate Drug Use in Health Facilities. 15
Assessment of drug use indicators.

Measurement of drug consumption in DDD/1000 patients/day
Drug consumptions were calculated with respect to DDD. The drug consumption has been according to DDD concept, to overcome objection against traditional units of measurement of drug consumption. Defined Daily Usage is defined as the assumed average maintenance dose per day for a drug used for its main indication in adults. It provides a fixed unit of measurement independent of the price and formulations. 16 The DDD/1000 patients/day is therefore calculated by the following formula:

Adverse drug reaction monitoring
The adverse drug reactions (ADRs) related to ADD were monitored and documented in suitably designed ADR documentation form after initial notification of the suspected ADRs by physicians. Additional details were collected by review of the patient case records and interview with patients. Severity and causality of the ADRs were assessed using the Modified Hartwig and Seigel 17 scale and the Naranjo algorithm, 18 respectively.
The Modified Hartwig and Siegel scale grades ADRs as mild (level 1 and level 2), moderate (level 3, level 4 (a), and level 4 (b)), and severe (level 5, level 6, and level 7). The Naranjo algorithm scale grades causality of ADRs as definite, probable, possible, and unlikely.
Statistical analysis
To evaluate the pattern of drugs used in this study, data were subjected to χ2 test and percentage value. The level of significance (P value) was set at .05. Patient’s demographic data were presented as mean ± SD. The data were analyzed using SPSS version 12.0.
Results
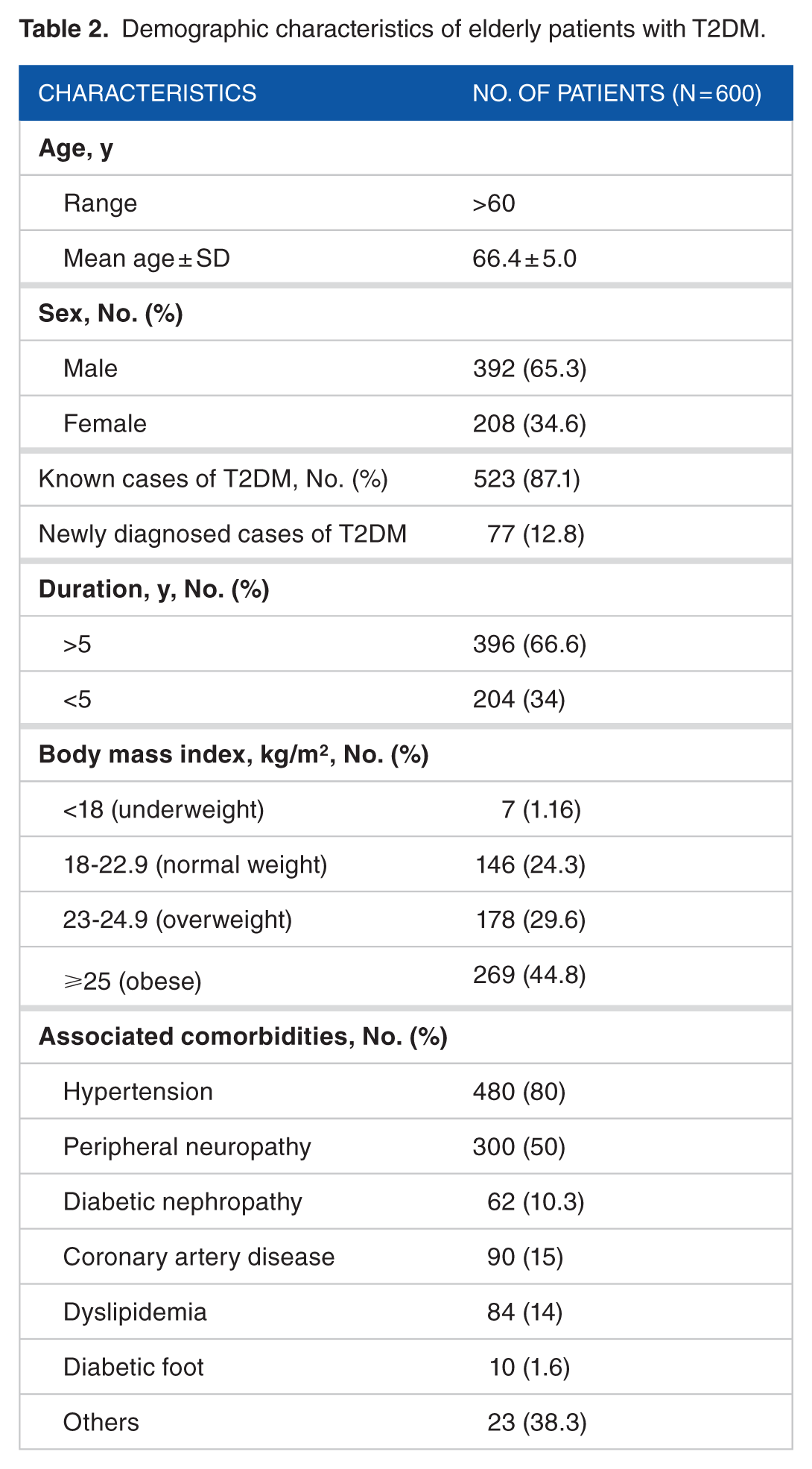
Overall, prescription data of 600 elderly patients with T2DM (men: 392; women: 208) were collected with 2328 follow-up encounters. Most of the patients (52.36%) belonged to 60 to 70 years (Table 2). Four subsets of population were noted in drug use:
Those receiving the cheapest available brand of antidiabetic medication.
Those receiving drug entirely from hospital pharmacy.
Those receiving drug from Central Government Health Scheme (CGHS) (receiving comparatively costlier brands of drug).
Those receiving some drugs from hospital pharmacy and some drugs from local retail pharmacy shops.
Demographic characteristics of elderly patients with T2DM.
Pattern of drug use
Analysis of collected data revealed the following ADDs to be most commonly prescribed:
Biguanides: metformin;
Second-generation sulfonylureas (SUs): glimepiride, glibenclamide, and gliclazide;
Thiazolidinediones: pioglitazone;
Alpha-glucosidase inhibitors (AGIs): acarbose and voglibose;
Uncommon: insulin-human insulin preparations of regular insulin, Neutral Protamine Hagedorn (NPH) insulin, premixed human insulin (regular/NPH) and human analogue insulin glargine and DPP4 inhibitors such as sitagliptin.
In total, 90% of encounters (n = 2104) had multidrug regimens. Utilization pattern of commonly encountered multidrug regimens have been shown in Table 3. The 92.01% DU comprised of metformin, glimepiride, pioglitazone, voglibose, and regular insulin (Table 4). Dual oral drug combinations (metformin and glimepiride) were found to be more prevalent. In single drug prescriptions, usage of metformin (44.6%; n = 100) and insulin (44.6%; n = 100) was similar.
Utilization pattern of most commonly encountered multidrug ADD regimens (n = 238).
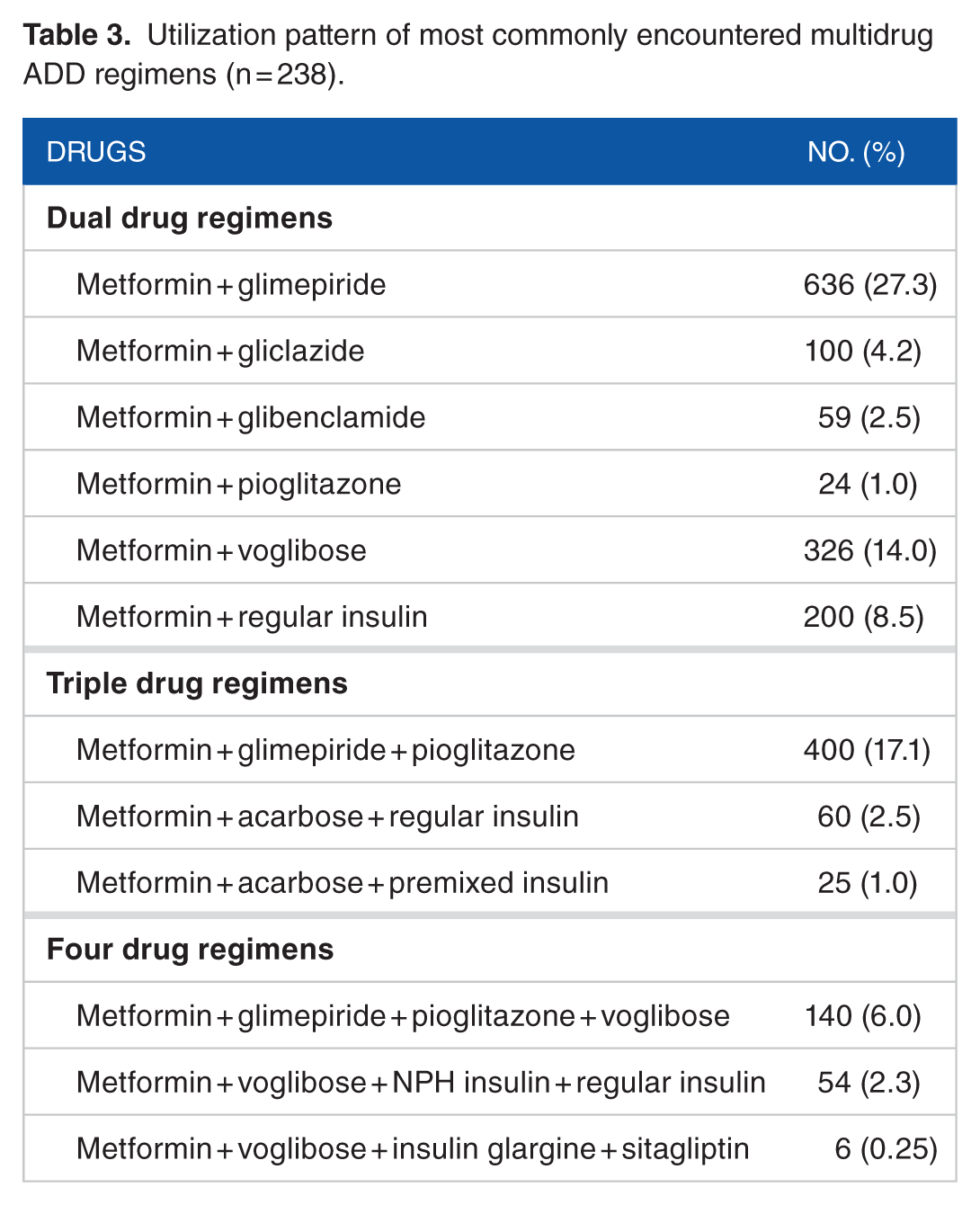
Pattern of drug use.
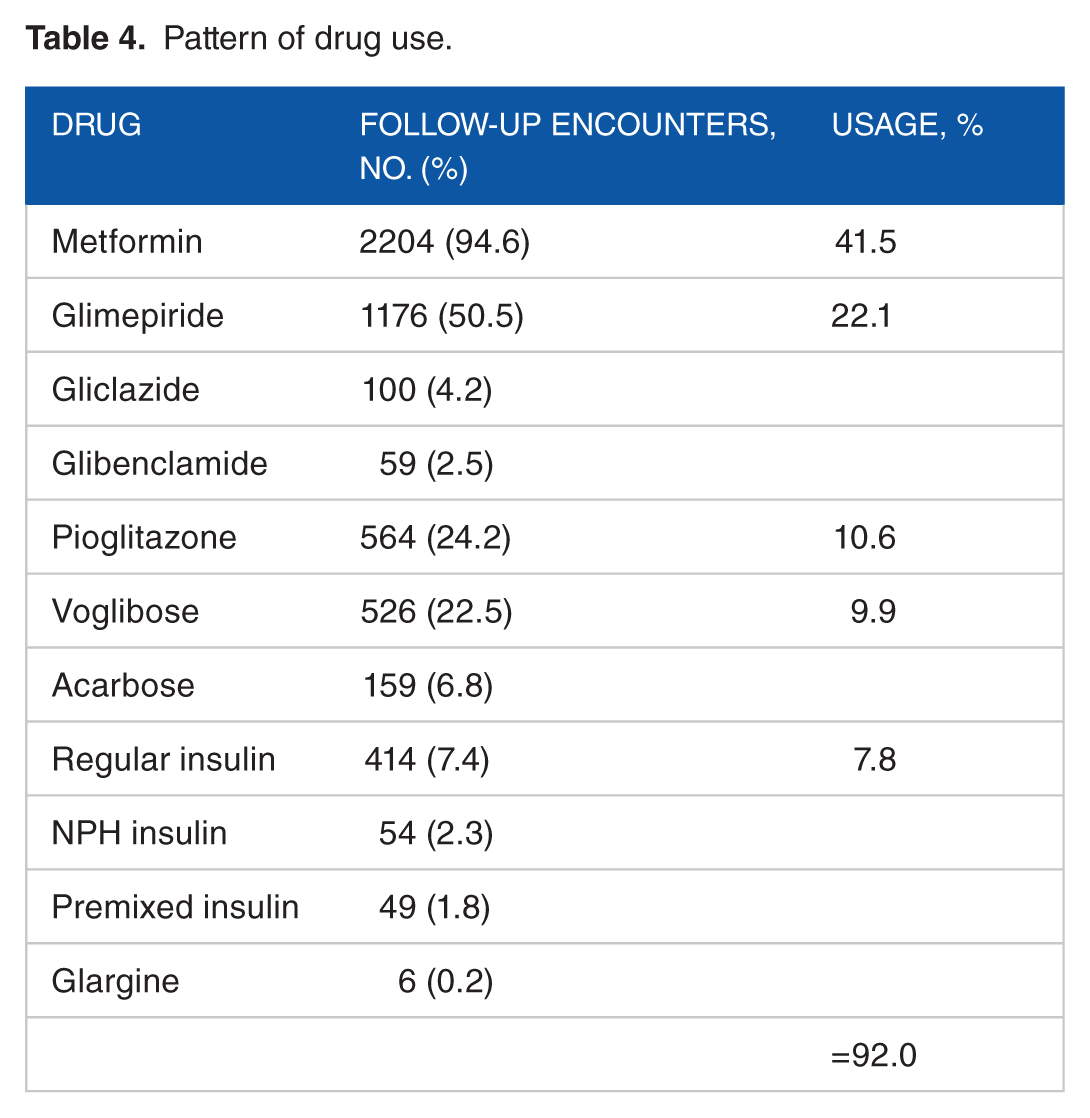
Pattern of daily dosing of various drugs constituting 90% drug utilization.
Nonpharmacologic interventions
Dietary charts with calorie estimations were not found in any of the cases. Approximately, 37.8% prescriptions had dietary advices (n = 880) and 64.4% had aerobic exercises (n = 1500) mentioned, especially if they were newly diagnosed. But, overall compliance to these interventions was poor. Only 20% (n = 120) patients did pursue regular exercises, and 15% (n = 90) had some idea about proper medical nutrition therapy (MNT) in T2DM.
Pattern of antidiabetic regimens prescribed
In total, 5059 ADDs were prescribed over 18 months in 2328 follow-up encounters. In 90% follow-up encounters (n = 2104), multiple antidiabetes agents were used, and only 10% (n1 = 224) follow-up encounters had single drug agent. Split analysis reveals that 67.44% (n2 = 1419) follow-up encounters had 2 ADDs, 23% (n3 = 485) follow-up encounters had 3 drugs, and 9.5% (n4 = 200) follow-up encounters had 4 ADDs.
Single drug prescriptions mostly had metformin (44.64% of n1 = 100), regular insulin (44.64% of n1 = 100), or premixed insulin (10.71% of n1 = 24). Triple drug regimens were fairly common (n3 = 485), which included mostly glimepiride, metformin, and pioglitazone combination regimens (82.4% of n3 = 400) (Table 3). Interestingly, many prescriptions had combination of metformin plus acarbose with regular insulin (12.37% of n3 = 60) or premixed insulin (5.15% of n3 = 25). Among 200 follow-up encounters with 4 drug combinations, glimepiride, metformin, pioglitazone, and voglibose were majority (70% of n4 = 140). Few cases with sitagliptin was encountered (ns = 6).
Figure 1 shows that metformin plus SU (56% of n2 = 795) and metformin plus AGI (23% of n2 = 326) were the most common prescribed dual antidiabetes regimens. Among the SUs, glimepiride (80%; n = 636) and voglibose (81.5%; n = 326) were the most commonly encountered SU and AGI, respectively, in combination with metformin (Figure 2).
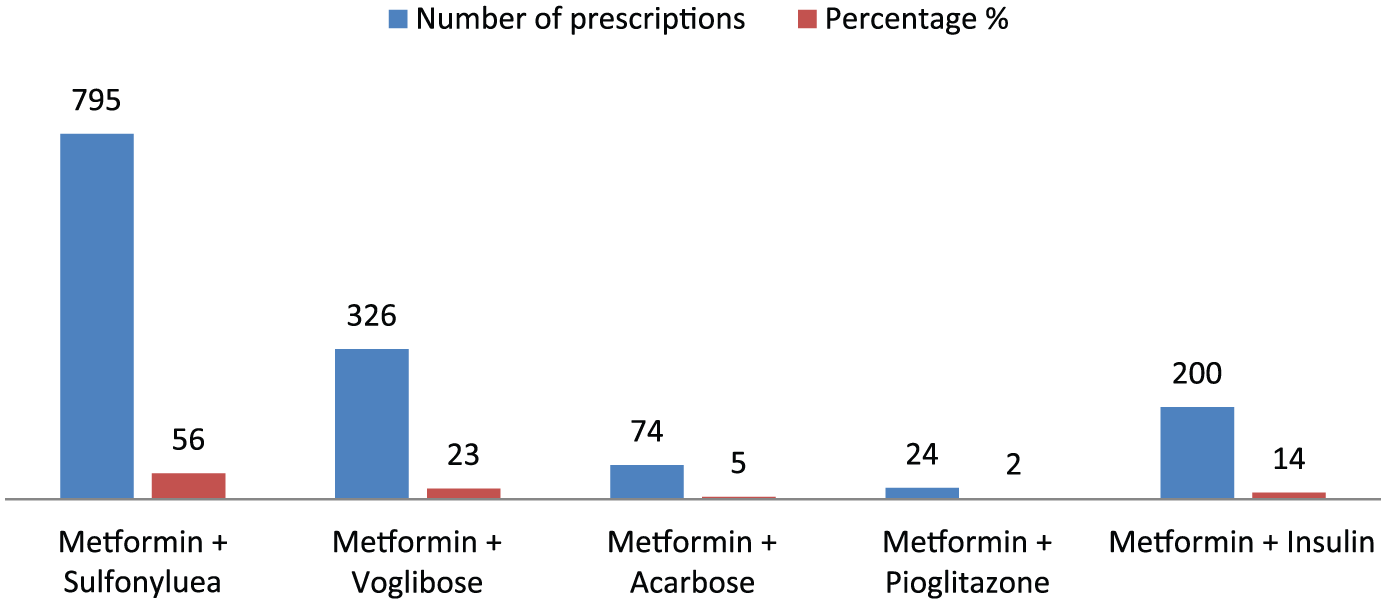
Pattern of dual antidiabetes regimens prescribed.
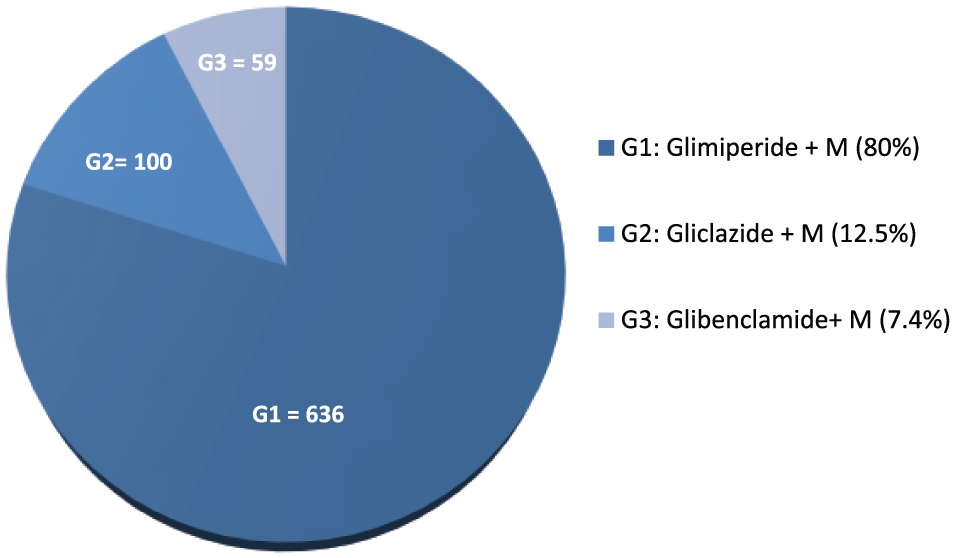
Pattern of sulfonylurea-metformin (M) combinations used.
Patient care indicators
The average consultation time per encounter was found to be 3.5 ± 0.6 minutes. Only 60% of the patients had correct knowledge regarding drug dosage. The average drug cost per encounter per day was Rs 11.24 ± 2.01 (Table 6).
Details of drug use indicators.
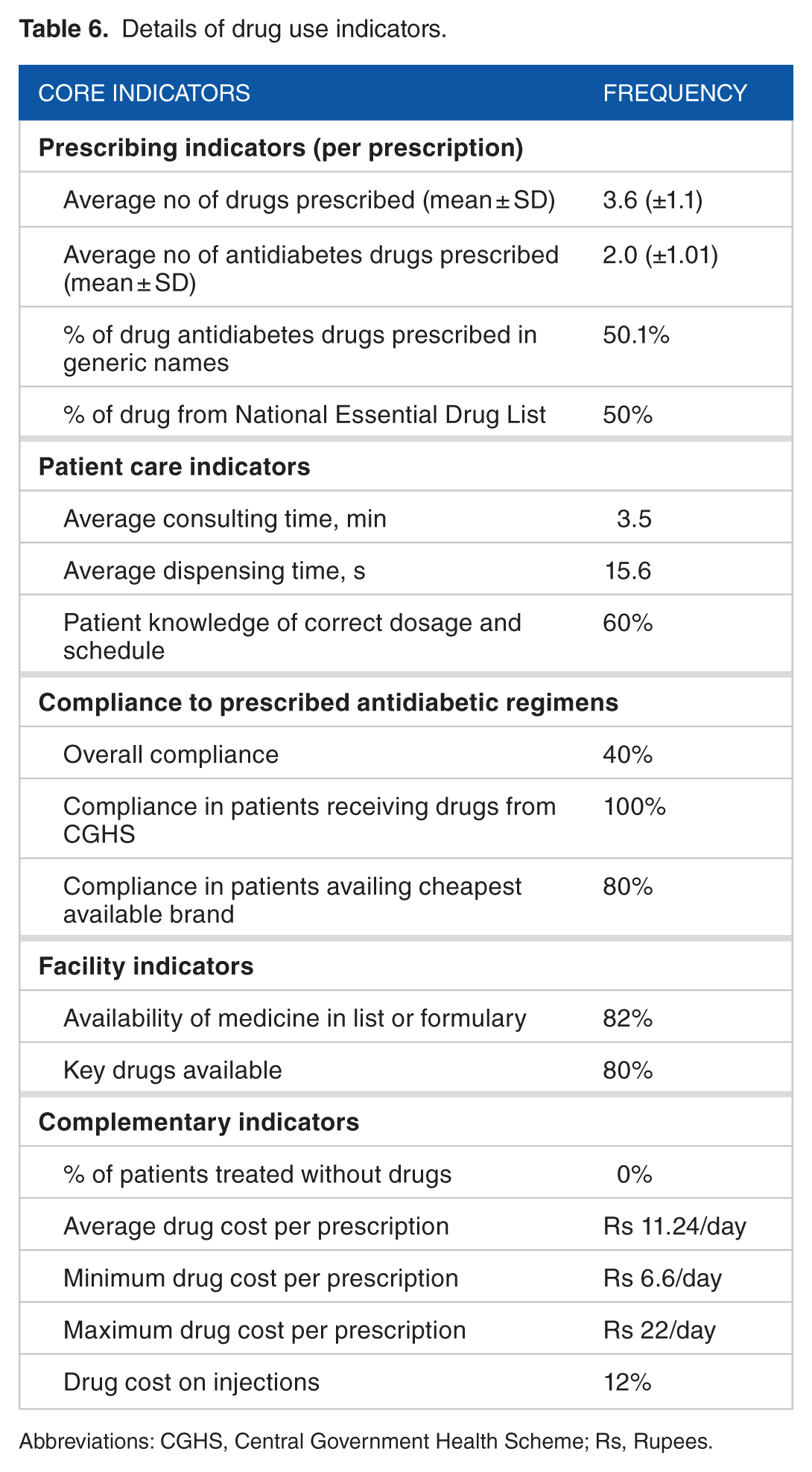
Abbreviations: CGHS, Central Government Health Scheme; Rs, Rupees.
Assessment of metabolic parameters
Target glycemic status was achieved in 40% follow-up encounters at monthly follow-up visits.
Assessment of DDD
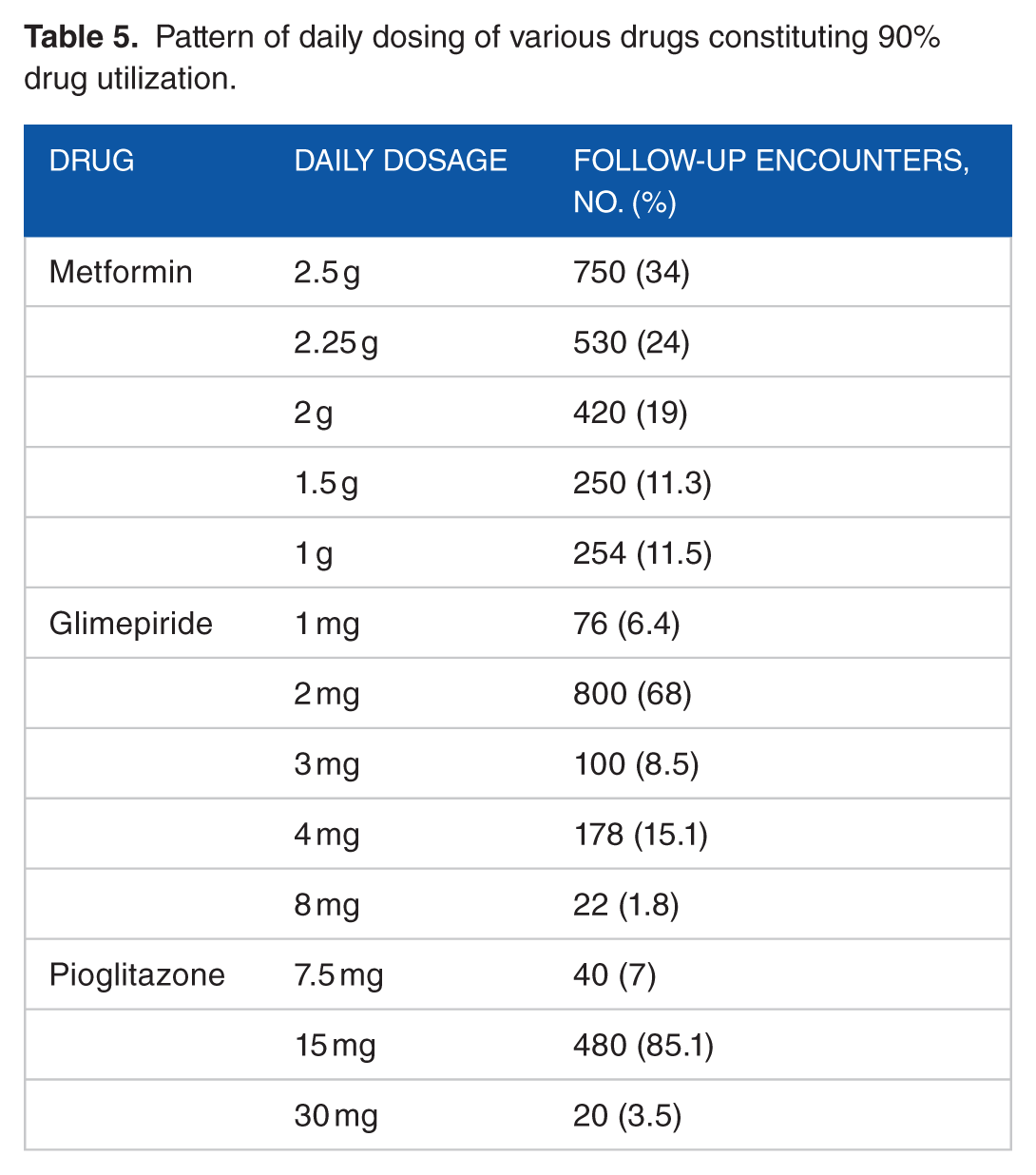
Table 6 shows that apart from metformin, glimepiride, and gliclazide, most other drugs have been prescribed in lower doses in comparison with WHO-recommended DDD.
Incidence of ADRs
Overall, 9 episodes of hypoglycemia were noted and registered with the Institutional Pharmacovigilance Committee. Of these, 3 were due to oral glucose–lowering agent (2 ADRs of mild L1 and 1 ADR of mild L2), among which 2 episodes due to combined triple drug regimen (glimepiride, metformin, and pioglitazone) and others probably due to glimepiride and metformin. In all these cases, complications were averted by changing the existing regimens; for example, in glimepiride-metformin combination–induced hypoglycemia, glimepiride was withdrawn or substituted.
There were 6 episodes of hypoglycemia (4 ADRs of moderate L3 and 2 ADRs of moderate L4 (a)) induced by insulin (mostly premixed insulin), which was averted by decreasing the daily dosage. The dose of insulin was decreased accordingly in the follow-up visit.
Two episodes of pedal edema were noted in the patients receiving pioglitazone for which the drug was stopped.
There were discrete 8 episodes of gastrointestinal discomfort (6 ADRs of mild L1 and 2 ADRs of mild L2) of on intake of metformin (>2 g/day). Dose was reduced to 1.5 to 2 g as necessary.
None of the ADRs were severe. Among all the ADRs (n = 19), 17 ADRs were of possible category, followed by 2 ADRs which were of probable category on the causality assessment scale.
Conclusions
In total, 92.01% DU consisted of metformin, glimepiride, pioglitazone, voglibose, and regular insulin. This was partly in accordance with 2016 ADA/American Association of Clinical Endocrinologists (AACE) guideline because there was not enough use of long-acting basal insulin as recommended by ADA or AACE. The average doses of metformin, glimepiride, and glibenclamide used were comparable with recommended WHO ATC/DDD Index, whereas pioglitazone and insulin were prescribed in dosage less than recommended values (Table 7).
Calculated average DDD for various drugs in this study population with respect to WHO-recommended DDD.
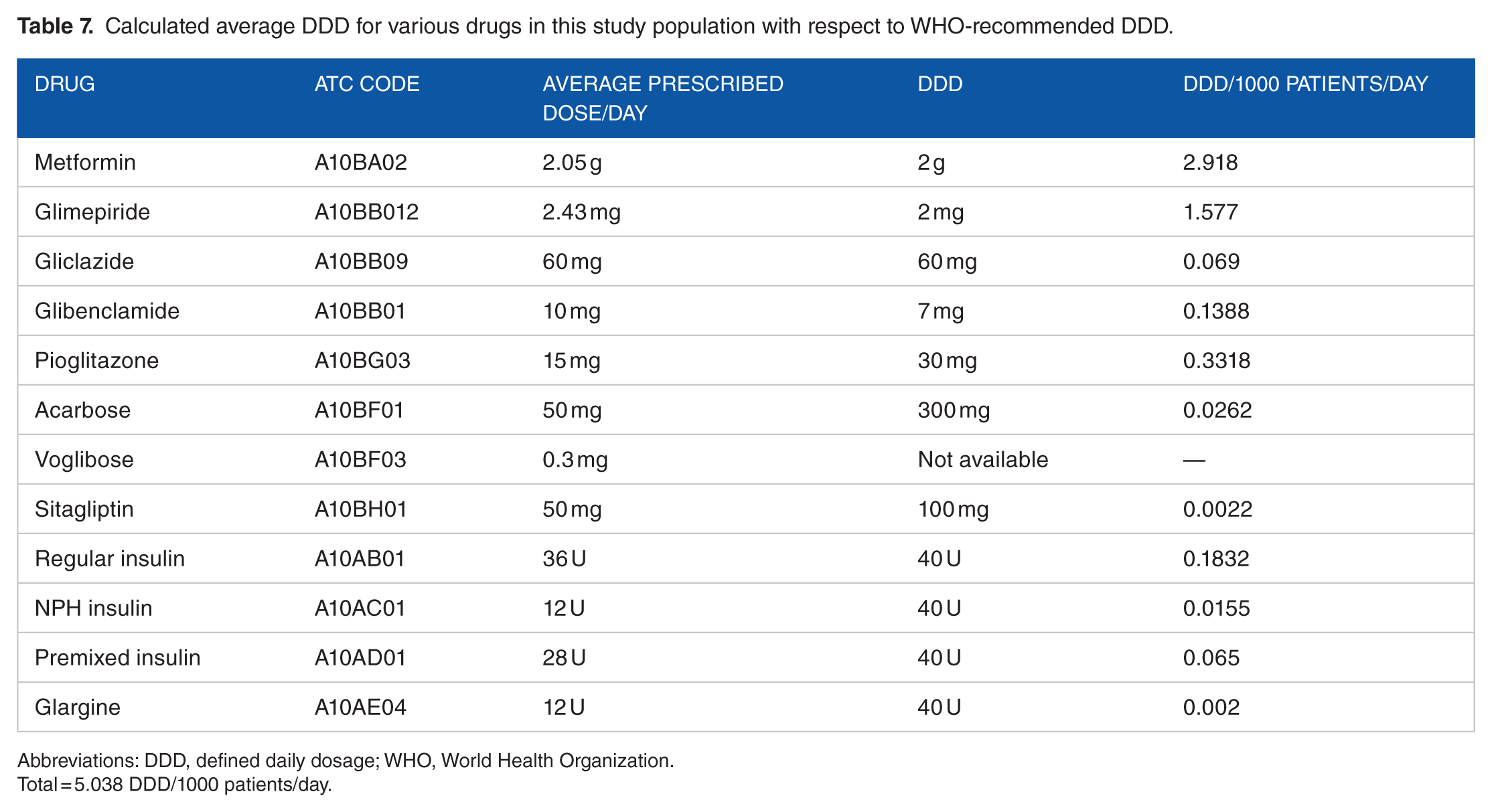
Abbreviations: DDD, defined daily dosage; WHO, World Health Organization.
Total = 5.038 DDD/1000 patients/day.
Discussion
In rural India, where affordability is a prime issue, clinicians are often faced with a problem of having limited access to sensitive investigations like A1C. In these settings, clinical judgment is mainly guided on the basis of fasting plasma glucose (FPG) and 2-hour plasma glucose levels, which may be spurious at times. As was the case in our setting, choice of ADDs was possible solely on monthly FPG and postprandial plasma glucose values in few follow-up encounters (37%). A1C values were available for only a subset of population, especially on CGHS (n = 12; 2.0%) out of 600 patients.
Assessment of ADD prescribing pattern
Metformin is the most commonly prescribed drug in T2DM. 19 In our study, 92.75% encounter metformin has been used either as monotherapy or as a part of polytherapy. The average DDD for metformin was 2.05 g/day. In 2204 follow-up encounters with metformin, 71.12% (n = 1700) encounters had the prescribed dose initiated at ⩾2 g/day. Polymorphism of organic cationic transporter (OCT1) protein in this subset of rural elderly population necessitating more amount of metformin could be a possibility. 20 In total, 36% follow-up encounters had concomitant vitamin B complex prescribed along with metformin, based on the rationale that metformin is known to be associated with 20% to 30% lower blood levels of vitamin B12. 21
Dual antidiabetic drug prescriptions were common, mostly consisting of SU-metformin combination (52%; n = 795). The combination of SU-metformin was cheaper and widely available, with better patient acceptability in comparison with addition of basal insulin to metformin as an add-on therapy. In 80% metformin + SU combinations (n = 636), glimepiride has been used. Moreover, glimepiride and metformin were available in the hospital pharmacy. Of these, in 62.89% (n = 400), glimepiride and metformin were prescribed separately and in rest various fixed dose combinations of glimepiride and metformin.
Glimepiride has emerged as a front liner among the SU because of its long t1/2, stronger extrapancreatic action, less hyperinsulinaemia, and lower incidence of hypoglycemia. 22 This justified 80% (metformin and SU) follow-up encounters with glimepiride in comparison with other SUs in the study. The calculated average DDD for glimepiride was 2.43 mg in comparison with 2 mg accredited by WHO. Although glimepiride is a long-acting drug and is used once in a day, 22 in about 31.44% (n = 200) follow-up encounters, it had been used twice a day especially when the dose has exceeded 4 mg/day. Although there is no data supporting the twice daily regimens over once daily, the trend was noted; probably for better diurnal blood glucose control and lesser chances of hypoglycemia.
Glibenclamide was used mostly as a replacement when glimepiride was out of supply from hospital pharmacy. The calculated average DDD of glibenclamide was 10 mg in comparison with 5 mg accredited by WHO. Gliclazide was reserved in settings of ischemic heart disease, based on its purported property of cardiovascular protection due to antioxidant action and noninterference with ischemic preconditioning. 23 The DDD of gliclazide has been 60 mg same as that of WHO index values.
Alpha-glucosidase inhibitor was used as an add-on therapy in 400 prescriptions with metformin (voglibose: n = 326; acarbose: n = 74) because in most of these follow-up encounters, there was significant postprandial hyperglycemia with controlled FPG. This was in accordance with International Diabetes Federation 2016 guidelines. 24 Overall, 520 prescriptions had voglibose and acarbose in only 134 prescriptions. It was contrary to data supporting the use of acarbose as a first-line adjuvant in comparison with voglibose for reducing cardiovascular mortality in T2DM.25,26 Probably, the lower cost of voglibose in comparison with acarbose could have been the reason behind this change in prescription pattern. Average prescribed dose of acarbose in this study has been 50 mg, whereas the recommended DDD of WHO has been 300 mg which reflects an underutilization of acarbose.
The use of pioglitazone as add-on therapy to metformin was encountered in only 24 prescriptions (2% of n2). Overall, pioglitazone was seen in 24.22% follow-up encounters. It could probably be because of pioglitazone being dispensed from hospital pharmacy. Although metformin-pioglitazone combination has been shown to reduce insulin resistance and cardiovascular morbidity [5-8], the combination was found to be an underutilized class in our study, probably due to concerns of side-effects. 27 Pioglitazone is associated with weight gain, a small reduction in the hematocrit, and a mild increase in plasma volume. Peripheral edema and congestive heart failure (CHF) are more common in individuals treated with these agents. These agents are contraindicated in patients with liver disease or CHF (class III or IV), which are more common in elderly. 28 There is concern of worsening of diabetic macular edema related to its use. 29 An increased risk of fractures has also been noted in women taking these agents. 30
Studies have demonstrated increased association of bladder cancer with pioglitazone.31,32 One such retrospective cohort study in United Kingdom had also demonstrated increased association of bladder cancer with pioglitazone (Hazard Ratio [HR] 1.83, 95% confidence interval: 1.10-3.05). The rate increased as a function of duration of use, with the highest rate observed in patients exposed for more than 24 months (HR 1.99, 1.14-3.45) and in those with a cumulative dosage greater than 2.8 g (2.54, 1.05-6.14). 32 In our study, only 2 episodes of pedal edema were noted for which pioglitazone had been stopped. The average DDD of pioglitazone in this study has been 15 mg, which is at 50% of WHO recommended 30 mg.
Overall, in 469 follow-up encounters, insulin was used. Insulin constituted 9.75% of total ADDs prescribed which is significantly lower in comparison with a previous Nigerian study (2008), 33 in which insulin comprised 16.7% of antidiabetic drug load. The possible reasons for underutilization of insulin in this study are poor patient compliance, arising mainly due to the following issues:
A phobia for injections.
Inability to use insulin subcutaneously and thereby depending on paramedical staffs for daily injections.
Notion of lifelong use, making it unpopular among patients and relatives.
The higher average daily expenditure for insulin compared with oral ADDs.
Interrupted availability in hospital pharmacy.
There has also been a shift in trends, particularly in the type of insulin used over the years. In comparison with the Nigerian (2008) study, 33 use of human short-acting and intermediate-acting insulin was seen. In our study, human regular insulin, NPH insulin and premixed insulin, and long acting human analogue insulin glargine have been used. DU 90% comprised of metformin, glimepiride, voglibose, and regular insulin. The prescribing frequency of SU was more compared with metformin. Among the SUs, there was higher use of glibenclamide. In neither of the earlier studies, extensive AGI usage has been noted, unlike in our study, where voglibose comprised about 10% of antidiabetic drug load and was included in DU 90%.
In our study, only 50% of the drugs were prescribed from National Essential Drug List 2015, 34 which was significantly low. The obvious reason was the change in the pattern of use of SU. In earlier studies, glibenclamide had been widely used, but in our study, it was glimepiride that has emerged as the first choice SU. Gliptins were found to be rarely prescribed, probably because of the cost factor and nonavailability in the hospital pharmacy. Also, no prescriptions of sodium/glucose cotransporter 2 (SGLT-2) inhibitors were documented.
The percentage of generic ADD drug prescriptions at 50% was lower as compared with previous studies (where it was 70%).35,36 The probable reason was nonavailability of generic drugs in hospital pharmacy, encountered on numerous occasions. Moreover, at times, drugs with various trade names had been prescribed with concomitant oral ADDs.
Drug cost analysis
The average drug cost per encounter per day in this study was Rs 11.24 ± 2.06 which was higher as compared with previous studies.35,36 Moreover, the drug cost spent on injection was 14% of the total antidiabetic drug cost, in comparison with 36% to 64% from previous study data, depicting that insulin utilization was still much less in these parts of India, even after many years of these studies. The utilization of oral ADD and insulin was 4.78 and 0.26 DDD/100 patients/day, respectively. Total DDD was 5.03 DDD/100 patients/day.
Problem areas and recommendations
The study demonstrates many grey areas that need attention. Most important problem identified during this study is of poor compliance (>60%). Proper knowledge on drug usage was only seen in 60% population. This got reflected in the fact that only in 40% cases, follow-up encounters’ target glycemic status could be achieved. Compliance was 100% for patients receiving drugs from CGHS. Poor compliance trends were noted in those receiving drugs from hospital pharmacy and local pharmacy retail shops. Those availing cheapest available brands had >80% compliance. Those receiving drugs exclusively from hospital pharmacy had compliance below 10% because drugs were being supplied only for a fortnight. Due to financial constraints, most patients were found to discontinue treatment after 2 weeks, hence presenting with deranged glycemic status on follow-up visits. This problem could be addressed to some extent by supplying drugs for at least 1 month on each encounter, besides the basic need of education regarding the importance of proper glycemic control and prevention of microvascular and macrovascular complications. Moreover, newer drugs such as gliptins and SGLT-2 inhibitors should be made available in the hospital pharmacy.
Poor average consultation time (3.5 ± 0.6 minutes) was another limiting factor. Despite huge patient burden in the outpatient setting, an improvement in this area could mitigate the situation to some extent. This can be sorted out either by running diabetic clinic thrice a week or in 2 shifts biweekly so that the average consultation time increases.
Lack of proper knowledge regarding MNT and noncompliance to regular aerobic exercises was seen in 80% to 85% cases. In most cases, there was no advice on diet or no diet charts were given, which is an important cornerstone in the management of T2DM. This necessitates recruitment of dietitians and patient care educators for patient benefit.
Footnotes
Acknowledgements
The authors thank Department of Pharmacology, R. G. Kar MCH, Kolkata and Institute of Post Graduate Medical Education and Research, Kolkata, in helping us conducting this work.
Peer review:
Four peer reviewers contributed to the peer review report. Reviewers’ reports totaled 604 words, excluding any confidential comments to the academic editor.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
AK conceived and designed the experiments. SL and AK analyzed the data, made critical revisions, and approved final version. SC wrote the first draft of the manuscript. RS and AM contributed to the writing of the manuscript. RS, AM, AK, and SC agree with manuscript results and conclusions. AK, RS, and SL jointly developed the structure and arguments for the paper. All authors reviewed and approved the final manuscript.
Disclosures and Ethics
The authors have read and confirmed the agreement with the ICMJE authorship and conflict of interest criteria. They also confirm that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. The external blind peer reviewers have no conflicts of interest.
