Abstract
Background:
Immune checkpoint inhibitors (ICIs) hold a great promise in treatment of non-small cell lung cancer (NSCLC), while only a portion of patients benefited from the treatment, and others could not achieve optimal therapeutic effects from initial immunotherapy, even for those patients with PD-L1 (Programed cell death ligand 1) tested positive. However, the clinical markers for the selection of patients who will benefit from ICIs combination treatment beforehand are largely unknown.
Objectives:
The purpose of this study was to explore the non-invasive biomarkers that can predict the efficacy of immune combination therapy in advanced/metastatic NSCLC patients.
Design:
This study employed a retrospective cohort design to analyze dual predictive biomarkers in advanced non-small cell lung cancer (NSCLC) patients with immune combination therapy.
Method:
An analysis was conducted on baseline information of 144 patients with advanced/metastatic NSCLC who received ICIs treatment from the November of 2018 to the January of 2023 in Beijing Chest Hospital. We established a scoring group chart to make quantitative prediction for overall survival (OS) and progression-free survival (PFS) based on 4 variables, and set up the nomogram model as well as Decision curve analysis (DCA) to assess clinical benefits of ICIs combination in treatment of patients with advanced/metastatic NSCLC.
Results:
We found that serum globulin (GLB) >26.6 (g/L) (HR = 1.865, P = .002), absolute neutrophil counts (ANC) (109/L) > 5 (HR = 2.146, P < .001), and bone metastasis (HR = 2.148, P < .001) were independent factors affecting the PFS of NSCLC patients. GLB > 26.6 (g/L) (HR = 1.741, P = .018), ANC (109/L) >5 (HR = 1.807, P = .008), bone metastasis (HR = 1.651, P = .002), and PD-L1 Negative (HR = 2.432, P = .032) were independent factors affecting the OS of NSCLC patients. Same variables and cut-off value have good predictive efficacy in both PFS and OS.
Conclusion:
In patients with advanced/metastatic NSCLC receiving ICIs combination treatment, the GLB, ANC, bone metastasis, and PD-L1 may serve as useful predictive markers for the prognosis of NSCLC patients with ICIs combination treatment.
Introduction
Lung cancer takes the leading position in human cancers in terms of incidence and mortality worldwide.1,2 Non-small cell lung cancer (NSCLC) accounts for 80% to 85% of all lung cancer cases in the Word. 3 The application of immune checkpoint inhibitors (ICIs) to immunotherapy in lung cancer led to striking improvement in progression-free survival (PFS) and overall survival (OS) of NSCLC patients.4-6 Despite the increasing role of immunotherapy in lung cancer, a minority of patients still could not benefit from these therapies. Furthermore, the occurrence of immunotherapy-related toxic effects in some patients diminishes the quality of life.7-10 Thus, it becomes imperative to determine clinical biomarkers, by which the patients who will benefit from ICIs could be hopefully differentiated from those who will not, to avoid unnecessary treatment. PD-L1 was the first FDA-approved predictive biomarker for (NSCLC) in 2015, and series of studies have also obtained this outcome.11,12 Moreover, tumor mutational burden (TMB) and microsatellite instability-high (MSI-H) can also serve as effective predictive indicators of prognosis in these patients, and other indicators such as circulating tumor DNA (ctDNA) and circulating tumor cells (CTC) have shown promise in prognostic prediction.13,14 However, there are some limitations and disadvantage of using these markers, for example, examination of these markers is expensive, and inconvenient to perform. An easily predictive model is urgently required in clinical practice to accurately guide clinical treatment for NSCLC.
Currently, the predictive model of ICIs for NSCLC treatment has much room for improvement, single predictive markers for PFS and OS are affected by drug toxicities and crossover therapy, and no efficient marker has been seen to predict both PFS and OS in patients treated with immune-combination therapy. Some investigators suggested the utilization of the Lung Immune Prognostic Index (LIPI), a composite of lactate dehydrogenase (LDH) and neutrophil-associated ratio (dNLR), to predict prognostic outcome.15,16 T cell-associated immunoglobulins may be related to the phenomenon of antibody neutralization, where immunoglobulins may bind to immune checkpoint inhibitors (ICIs) or other immunotherapeutic drugs, thus inhibiting their activity and reducing treatment effectiveness. 17 However, the predictive model of ICIs combined therapy was not so effective as expected, and the clinical value of the predictive model that integrates the clinical characteristics and laboratory indicators of patients before treatment deserves further validation.
To improve the prognostic survival of patients treated with ICIs and select suitable groups of patients by predictive biomarkers for ICIs treatment, we conducted this study to develop a non-invasive and cost-effective multivariable prognostic model. This predictive model predicts both PFS and OS and can provide precise clinical value. We speculated that a model that integrated clinicopathological characteristics and laboratory indicators could simultaneously predict the survivals of NSCLC patients that received ICIs combined therapy.
Materials and Methods
Study design and patients
Total 144 patients with advanced NSCLC (III/IV) who received immune checkpoint inhibitor therapy between November 2018 and January 2023 were retrospectively analyzed. Data were collected from the records of hospitalized. This study was approved by the Beijing Chest Hospital Medical Ethics Committee (YJS-2023-09), according to the local regulations and the Helsinki declaration. A waiver for patients’ notice consent was obtained by the Beijing Chest Hospital Medical Ethics Committee due to the retrospective nature of the study.
Inclusion and Exclusion Criteria are as follows:
Inclusion criteria
(1) Adult patients aged 18-80 years; (2) Patients with NSCLC were diagnosed by imaging and pathology; (3) Diagnosis of advanced or metastatic NSCLC (IIIB/IV); (4) Patients did not receive surgical operation and radical synchronous or sequential radiotherapy and were treated with immune checkpoint inhibitors at least 4 cycles of immunotherapy.
Exclusion criteria
(1) Combined with other tumors and infectious diseases; (2) Steroid hormone systemic therapy was used 1 month before treatment; (3) Patients with hematological or autoimmune diseases.
Treatment regimen
All included patients received PD-1 inhibitor and combine platinum-based chemotherapy, including taxanes, pemetrexed, and gemcitabine. All Immunotherapy-based combinations were approved by National Medical Products Administration (NMPA) in China. The dosage and frequency of immune checkpoint inhibitors used in clinical trials are recommended according to the manufacturer’s instructions. Eighteen cases (12.5%) were treated with PD-1 inhibitor monotherapy, and 126 cases (87.5%) with immunotherapy combined chemotherapy.
The dosage of Pembrolizumab, Nivolumab, Sintilimab, Toripalimab, Tislelizumab, Camrelizumab, Atezolizumab, and Penpulimab were 200 mg every 3 weeks. Based on these immune drugs, there are combined chemotherapy drugs or anti-angiogenic drugs:Bevacizumab (400 mg), vinorelbine (100 mg), arotinib (12 mg), pemetrexed (800 mg), nedaplatin (120 mg) every 3 weeks, etc.
Primary endpoint and follow-ups
The time of the first ICIs treatment was recorded as the starting point of observation, and each patient was followed up with interviews by telephone, outpatient review, and other forms of visit. Follow-ups were performed until disease progression or decease, and the follow-up study ended on December 25, 2023. Overall survival is the time from the start of immunotherapy to decease or the last follow-up visit. Progression-free survival (PFS) was defined as the time from the start of immunotherapy to clinical or imaging progression.
Observation indicators
The clinical indicators collected by electronic medical record system included: Age, gender, smoking history, clinical stage, Eastern Cooperative Oncology Group Performance Status scale (ECOG PS) score, histology, therapeutic regimen, PD-L1 expression, Actionable mutation, and metastasis for tumor progression. The baselines were defined as the measurements taken within 1 week prior to the treatment with ICIs. The baseline data of peripheral blood included WBC (white blood cell count), ANC (Absolute neutrophil count), LY (Lymphocyte count), NLR (Neutrophil to lymphocyte ratio), PLR (Platelet to lymphocyte ratio), CRP (C-Reactive Protein), SII (Systemic Immune Inflammation Index), MLR (Monocyte to lymphocyte ratio), LDH (lactic dehydrogenase), GLB (serum globulin), A/G (serum albumin to globulin ratio). A flow chart of sample screening and model construction was shown in Figure 1.

Flow chart of sample and predicted indexes screening.
Assessment of response and toxicity
Evaluation was performed routinely every 2 treatment courses after starting treatment with the PD-1 inhibitor. The response to therapy (complete response (CR), partial response (PR), stable disease (SD), progressive disease (PD) was assessed according to the Response Evaluation Criteria in Solid Tumors (RECIST) (version 1.1). ORR was the percentage of patients who achieve CR or PR according to RECIST criteria. Adverse events (AEs) were assessed according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 4.0.3. AEs that occurred during hospitalization were registered and graded by their attending doctor timely.
Statistical analysis
Data processing was conducted using SPSS 27.0 software. For descriptive analysis, the normal and non-normal distributions of continuous variables were expressed by mean ± standard deviation (SD) and median [interquartile range (IQR)], respectively, while categorical variables were presented as percentages. X-tile software was utilized to determine the optimal cut-off value and divides continuous data into categorical variables. Lasso regression was used to screen for the risk factors. Prediction models including parameters screened by Lasso regression were established based on Cox regression. The predictive abilities of models were compared by C-index. Graphpad prism10.0 for Kaplan Meier method was used to calculate PFS and OS curves, and log-rank test to evaluate differences.
The survival, rms, glmnet packages, etc. from Rstudio (version 4.3.2) were used to draw clinical decision curves, column charts for prediction of survivals, and Lasso regression plots. A P value < .05 was considered statistically significant. A chart of model construction was shown in Figure 2.

Flow-chart of model construction.
Results
This study included a total of 144 patients (109 males, and 35 females) with advanced/metastastic NSCLC (Table 1). Among these patients, 47 were ⩽60 years old, and 97 > 60 years old. According to clinical stages, 38 patients in stage III and 106 in stage IV. There were 82 cases of adenocarcinoma and 62 cases of squamous cell carcinoma. 100 patients had a smoking history. All patients were treated with PD-1 or PD-L1 immune checkpoint inhibitors. Most people have an ECOG score of 0 to 1 (137/144, 95.1%). Most patients had no or undetected sensitive gene mutations (136/144, 94.4%). Patients with the distant metastases suffered from bone metastases in most cases (38/144, 26.3%).
Basics of 144 patients with advanced NSCLC.

Abbreviations: ALK, anaplastic lymphoma kinase; CT, chemotherapy including Cisplatin, Carboplatin, Pemetrexed, Gemcitabine, Vinorelbine, Paclitaxel; EGFR, epidermal growth factor receptor; KRAS, Kirsten rats arcomaviral oncogene homolog; ROS1, c-ros oncogene 1.
Immunotherapy-based combinations regimen
All included patients received PD-1 inhibitor combined with platinum-based chemotherapy, including pemetrexed, gemcitabine, and taxanes. Most patients (n = 126, 87.5%) received immunotherapy-based combinations. Patients were treated with PD-1 inhibitors combined with 1 of the following therapy: sintilimab, nivolumab, camrelizumab, toripalimab, tislelizumab, or atezolizumab .
Most of the patients (n = 129, 89.59%) received 4 to 6 cycles of immune combination therapy, followed by immune maintenance monotherapy. The median number of immune maintenance therapy cycle was 2 (ranging, 0-35); 15 patients (n = 15, 10.42%) had adverse events during the 4 cycles, changed to change to later line therapy.
Overall, the median PFS and median OS were 10.7 and 30.0 months, respectively. Survival curves of immunotherapy for advanced NSCLC were shown in Figure 3a and b.

Survival curves of immunotherapy for advanced NSCLC.
Basic clinically pathological characteristics
There were 82 cases of adenocarcinoma and 62 cases of squamous cell carcinoma. As shown in Figure 4a, the median PFS of the 2 groups of patients were 10.8 and 10.7 months respectively, with the HR = 0.878 (95% C1:0.667-1.156) and P = .354, suggesting that the difference between the 2 groups of patents were not significant. As shown in Figure 4b, the median OS were 33.7 and 25.3 months respectively, with the HR = 1.146 (95% CI: 0.906-1.449) and P = .257, and there was no difference between the 2 groups of patients. These data indicate the survivals of patients with adenocarcinoma and squamous cell carcinoma is similar.

Survival curves of patients according to histological examination and PD-L1 expression. (a and b) PFS and OS curves of patients with Adenocarcinoma and Squamous cancers as examined by histology. (c and d) PFS and OS curves of patients without (negative) or with (positive) PD-L1 expression.
Response
Among all 144 patients, the objective response rate (ORR) took 31.9%, and the disease control rate (DCR) 80.56%. The ORR was 27.78% in the monotherapy group and 32.5% in the combination therapy group (P = .792). The DCR was 77.78% in the monotherapy group and 80.95% in the combination therapy group (P = .753). The difference in the response rate between these 2 groups was not significant (Table 2).
Tumor response in patients with advanced/metastatic NSCLC receiving PD-1inhibitors therapy in the monotherapy and combination therapy groups.

Abbreviations: DCR, disease control rate; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease.
Toxicities and adverse events
One hundred thirteen patients (78.4%) displayed treatment-related adverse events, most of which were classified as grades 1 to 2. Grades 3 to 4 treatment-related adverse events were experienced by 42 patients (20.4%), with the most common being myelosuppression after chemotherapy leads to granulocytopenia in 11 patients (7.6%).
The immune-related adverse effects (irAE) occurred in 65 (45.14%) patients, the most commonly-seen adverse event was fatigue (48/144), all of which were graded 1 to 2, followed closely by nausea (45/144) and rash (22/144). Grades 3 to 4 irAE were reported with pneumonia in 10 patients (6.9%) and rash in 9 patients (6.3%). In this cohort, 1 patient in the combination group died of immune-related pneumonia.
Interestingly, when analyzing 102 cases with negative PD-L1 expression and 42 cases with positive PD-L1 expression via PFS and OS, we found that the median PFS of the 2 groups of patients were 9.0 and 14.2 months respectively, with HR = 1.732 (95% C1:1.108-2.708) and P = .016 (Figure 4c), suggesting that the PFS of patients with PD-L1 expression was greater than that of patients without PD-L1 expression. Consistently, the median OS of the 2 groups of patients were 23.4 and 38.5 months respectively, with HR = 2.432 (95% C1:1.483-3.988) and P < .001 (Figure 4d), indicating that patients with PD-L1 expression survived much longer than the ones without PD-L1 expression. These data suggest that PD-L1 expression in patients affects the efficacy of ICIs treatment.
X-tile software was used to calculate the optimal cut-off value for PFS and OS, dividing peripheral blood markers into high and low groups. Their cut-off values were: NLR>3, LMR>2.4, SII>979, GLB (g/L)>26.6, A/G>1.4, WBC (109/L)>6.4, LY (10 9/L)>1.5, and ANC (10 9/L)>5. No optimal cut-off values were determined for PLR, CRP (mg/L), and LDH (U/L). These cut-off values can be used to predict the survivals of the NSCLC patients with ICIs treatment.
Determination of OS and PFS by univariant, lasso, and multivariate analysis of biomarkers
The results of univariate Cox regression analysis show that a total of 10 univariate factors, including clinical stage, ECOG score, bone metastasis, LMR, SII, GLB, A/G, WBC, LY, and ANC, had statistical significance in PFS (P < .05), and that a total of 10 single factors, including Histology, ECOG score, treatment methods, bone metastasis, PD-L1 expression in tissues, NLR, SII, GLB, A/G, WBC, and ANC, were statistically significant in OS (P < .05). Lasso regression was also used to screen parameters related to OS and PFS. As shown in Figure 5a, 10 variables related to PFS were included in Lasso regression, and when the model fitted well, 7 to 10 independent variables were contained in between (Figure 5b). For OS, 12 variables were included in Lasso regression (Figure 5c), and when the model fitted well, 2 to 10 independent variables were contained in between (Figure 5d). After Lasso regression, these variables would subject to, Cox multivariate analysis.

Screening of variables based on Lasso regression. (a and b) The variation characteristics of the coefficient of variables for PFS (a) and the selection process of the optimum value of the parameter λ in the Lasso regression model by cross-validation method (b). (c and d) The variation characteristics of the coefficient of variables for OS (c) and the selection process of the optimum value of the parameter λ in the Lasso regression model by cross-validation method (d).
To select significant variables and Identify factors of statistical significance to both PFS and OS, we performed Cox multivariate regression, and found that GLB (g/L)> 26.6, ANC>5(109/L), and bone metastasis were statistically significant in the multiple regression of PFS and OS (P < .05, Table 3).
Univariate and multivariate analyses of PFS and OS.

P < .05; Category contains risk factors (Risk factor = 1, Protective factors = 0); Age was used as a continuous variable.
Univariate and multivariate analyses of biomarkers for OS and PFS
For the overall population, the medians of OS and PFS demonstrate distinct outcomes based on specific biomarkers. According to the univariate analysis, the factors of statistical significance were as follows:
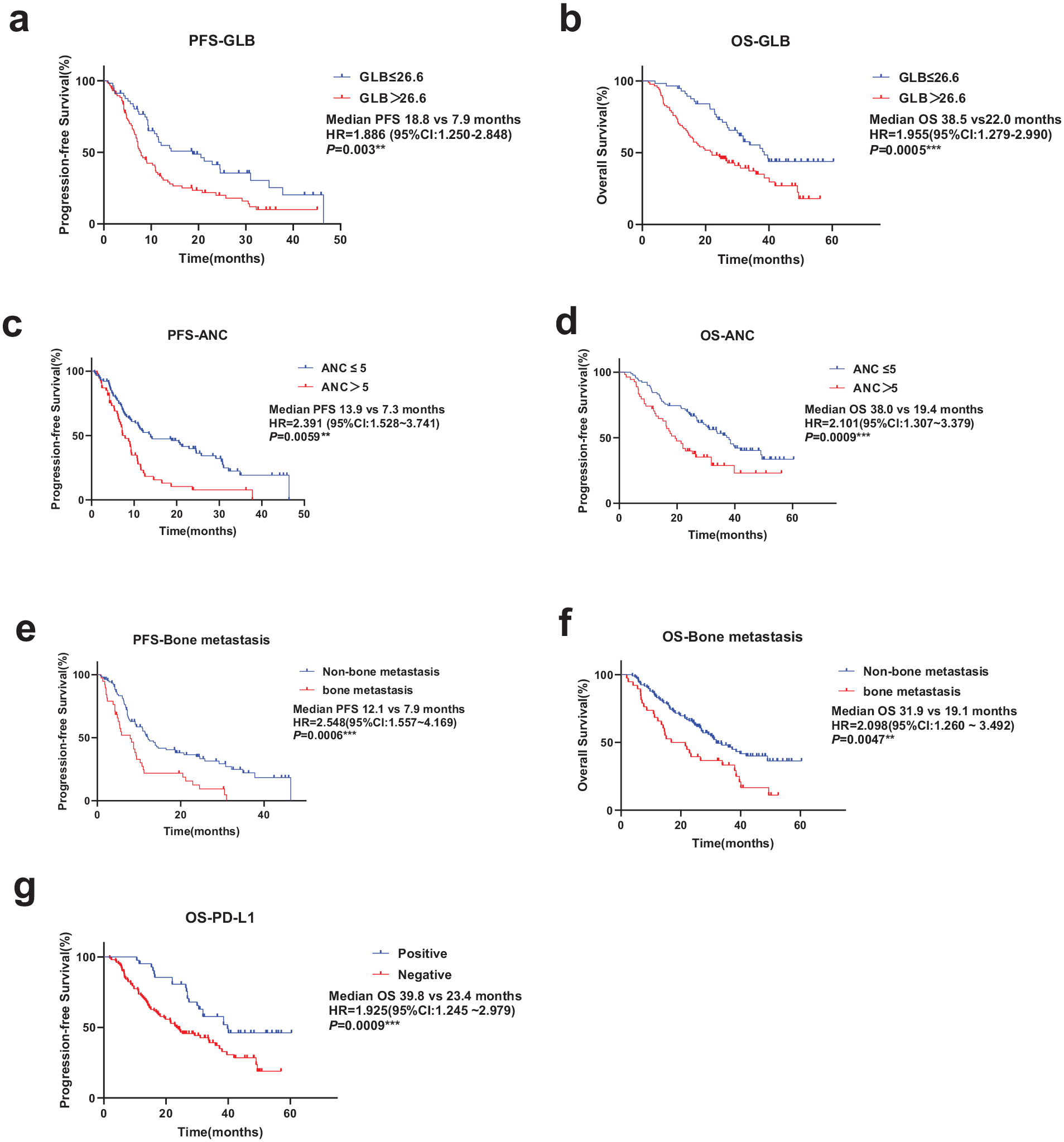
PFS (a, c and e) and OS (b, d, f and g) curves of patients as analyzed by GLB, ANC, bone metastasis and PD-L1 markers.
According to the univariate analysis, the levels of GLB, ANC, bone metastasis and PD-L1 expression are highly correlated to the survival of patients, and could be used as a useful biomarkers of prognosis for patients.
Establishment of a prognostic scoring system
Based on the number of advantaged factors (ie, GLB ⩽ 26.6 g/L, ANC⩽5 * 109/L, no bone metastasis, and PD-L1 expression positive), We explored the OS and PFS (Figure 7a and b). It was found that there was statistical differences among the groups, and that the OS and PFS of Groups A and B significantly increased compared to that of the other groups (C, D, E) (P < .0001), suggesting that the survivals of patients increased depending on the points of the prognostic scoring system.

PFS (a) and OS (b) curves of the multifactor model based on the number of advantage factors at baseline (GLB ⩽ 26.6 g/L, ANC ⩽ 5 (109/L), non-bone metabolism, PDL1-Positive). Abbreviation: NR, not reached; Group A: 0 point; Group B: 1 point; Group C: 2 points; Group D, 3 points; Group E: 4 points.
Nomogram diagnostic model construction
To further evaluate the predictive model and the contribution of each variable to survival, we established a 1-year PFS rate and 1-year OS rate nomogram based on multivariate Cox regression (Figure 8a and b). The C index of 1-year PFS rate was 0.731 (0.648-0.814) (P < .001), and the C index of 1-year OS rate was 0.733 (0.635-0.832) (P < .001). The results of Decision Curve Analysis (DCA) demonstrate that the nomogram model performed well in clinical practice (Figure 8c and d).

Data analysis by Nomogram diagnostic model. (a and b) Nomogram prediction of 1-year PFS rate and 1-year OS rate in patients with advanced NSCLC. (c and d) DCA curves for predictive prognostic modeling of Nomogram. Abbreviations: ANC, absolute count of neutrophils; Bone, bone metastasis; DCA, decision curve analysis; GLB, serum globulin; OS, overall survival; PFS, progression-free survival.
Internal model validation
After internal cross-validation calibration, tested with 5-fold cross-validation, and repeating the procedure 10 times, the C statistic (AUC under the ROC curve of 1-year PFS rate) was 0.808 (95% CI: 0.655-0.961), and the C statistic (AUC under the ROC curve area of 1-year OS rate) was 0.783 (0.633-0.933) (Figure 9a and b). These data suggest that the models were reliable.

The ROC curve after five-fold cross validation: (a) 1-year PFS rate and (b) 1-year OS rate.
Discussion
Although it was reported that noninvasive peripheral blood markers and clinical factors had predictive values in the treatment of advanced NSCLC, such as NLR, SII, there is no universal optimal cut-off value determined in different studies in the treatment of NSCLC patients with ICIs, and application of combined clinical markers is rarely seen.18,19 In this study, 144 patients with advanced/metastatic NSCLC were treated with ICIs combination therapy and analyzed by combined clinical features, organ metastasis, peripheral blood and biochemical indicators. A prognostic score including GLB, ANC, bone metastasis, and PD-L1 expression was determined and validated. The final cumulative score by grouping the 4 factors can predict survival prognosis of patients. In both OS and PFS, the higher the score, the worse the prognosis, and the difference between groups with scores by this subgrouping was highly significant (P < .001). Compared with the commonly-used continuous peripheral blood variables without generally-recognized optimal cut-off value in most studies, this scoring system would be a much more valuable alternative to clinical guidance.
This predictive model uniquely forecasts both PFS and OS for NSCLC patients treated with ICIs combination therapy. Predicting OS with one model has limitations due to non-neoplastic deaths and the impact of subsequent or cross-over treatments. PFS predictions might be biased by medication-related adverse effects, leading to a “spurious” benefit in short-term follow-ups. 20 Simultaneous PFS and OS markers offer more precise prognostic insights, enhance clinical study efficiency, and provide a comprehensive patient outcome assessment.
In order to further understand the contribution of each factor to the survival of patients and the clinical application value, we constructed 1-year PFS and OS rate nomogram for individual survival prognosis prediction. The DCA curves of 1-year PFS and OS rate are effective in clinical practice. The whole area of DCA curve of 1-year OS rate indicate that this model is of high clinical relevance (Figure 8d).
It was reported that serum globulin had prognostic relevance in patients with cancer,21,22 for example, a retrospective analysis of 186 patients with gastric cancer indicated that a baseline GLB of less than 33.4 (g/L) in patients was a favorable prognostic factor. 21 In addition, a nomogram prediction model established using the baseline peripheral blood data of 336 patients with oral cancer set the optimal cut-off values of GLB as 26.35 g/L, which could predict the prognosis of patients, 22 and the multivariate result OR = 1.093 (1.014-1.177), P = .019 was close to the GLB cut-off value in the present study (GLB > 26.6). According to our results, abnormal serum is a poor prognostic factor in patients with tumors. More efforts will be required to elucidate the mechanism of GLB underlying ICIs-mediated survivals of NSCLC patients.
Accumulating studies have revealed that high ANC will promote tumor progression and deterioration23-25 A retrospective study on the analysis of 88 patients with NSCLC treated with PD-1 inhibitors indicated that the lower the patient’s ANC at baseline, the better the response to treatment (P = .017). 26 In addition, it was found that high ANC before treatment was associated with poor prognosis in lung cancer patients treated with Anlotinib. 27 A descriptive review of immunotherapy for advanced renal cancer also stated that low ANC and NLR were associated with good prognosis of patients. 28 A predictive model for ICIs treatment of NSCLC patients has been established, including the impact of ANC and LDH and some clinical characteristics, ECOG PS score, BMI, etc. on prognosis. 29 Several reports indicated that the prognosis of tumor immunotherapy was related to NLR,30-32 Although both ANC and NLR are related to immune status and the degree of inflammation, their biological significance may vary. In some cases, ANC may directly indicate immunosuppressive or inflammatory status, while NLR may suggest the balance between neutrophils and lymphocytes, and the prognostic impact of immunotherapy may be relatively complex.
In the present study, we took into account the impact of distant organ metastasis on prognosis, and found that only bone metastasis was of statistical significance and predictive value in PFS and OS. We proposed that bone metastasis may affect the immune status of patients and the response to immunotherapy, which may lead to immunosuppression, inflammatory response, and bone marrow fibrosis, thus affecting the effectiveness of immunotherapy. In addition, bone metastasis may also affect the prognosis of immunotherapy through other mechanisms, such as the change of tumor microenvironment. 33 Thus, our result supported the previous study that bone metastasis may serve as a reliable indicator of prognosis for the patients with advanced NSCLC treated with ICIs. 34
In the present study, we found that GLB, ANC, bone metastasis and PD-L1 were closely associated with the survival of NSCLC patients, and could be used as potential biomarkers for diagnosis of NSCLC patients with ICIs combination therapy. We look forward to following up this study to further investigate the different treatment markers for NSCLC patients, providing a basis for individualized treatment. It would be interesting to conduct further animal experiments to verify the therapeutic effect of reducing neutrophils combined with PD-1 inhibitors on lung cancer. It was reported that anti-Ly6G removal of neutrophils associated with PD-1 inhibitor therapy have synergistic effect in the treatment of liver cancer. 35 However, there are some limitations to this study, including the number of patients, most of the 126 patients treated with ICIs combination drugs, not ICIs monotherapy. Some of these drugs are not used for first-line treatment, and relevant information needs to be supplemented in the follow-up study to continue to analyze the predictive value for the same first-line treatment. Of note, as the patients in this study did not have surgical operation, pathological samples obtained through biopsy may have certain diagnostic errors, which may render the study unavoidable limitations. In addition, as this study covers a larger time span, including the transition of immunotherapy from second-line to first-line treatment, which may introduce potential research bias.
Conclusion
In conclusion, our data show that the examination of ANC, GLB and bone metastasis can predict the PFS and OS of advanced/metastatic NSCLC patients treated with ICIs combination therapy, and the optimal cut-off value can provide valuable reference for clinical practice.
Footnotes
Acknowledgements
None.
Abbreviations
NSCLC, non-small cell lung cancer; OS, overall survival; PFS, progression-free survival; DCA, Decision curve analysis; GLB, serum globulin; ANC, absolute neutrophil counts; PD-L1, Programed cell death ligand 1; TMB, tumor mutational burden; MSI-H, microsatellite instability-high; ctDNA, circulating tumor DNA; CTC, circulating tumor cells; LIPI, Lung Immune Prognostic Index; LDH, dehydrogenase; dNLR, neutrophil-associated ratio; WBC, white blood cell count; LY, Lymphocyte count; NLR, Neutrophil to lymphocyte ratio; PLR, Platelet to lymphocyte ratio; CRP, C-Reactive Protein; SII, Systemic Immune Inflammation Index; MLR, Monocyte to lymphocyte ratio; LDH, lactic dehydrogenase; A/G, serum albumin to globulin ratio; CT, chemotherapy; Anti-Angio, Anti-angiogenic drugs; ECOG PS, Eastern Cooperative Oncology Group Performance Status scale.
