Abstract
In the past decade, immune checkpoint inhibitors (ICI) have been approved for treatment of genitourinary malignancies and have revolutionized the treatment landscape of these tumors. However, despite the remarkable success of these therapies in some GU malignancies, many patients’ tumors do not respond to these therapies, and others may experience significant side effects, such as immune-related adverse events (iRAEs). Accordingly, biomarkers and improved prognostic tools are critically needed to help predict which patients will respond to ICI, predict and mitigate risk of developing immune-related adverse events, and inform personalized choice of therapy for each patient. Ongoing clinical and preclinical studies continue to provide an increasingly robust understanding of the mechanisms of the response to immunotherapy, which continue to inform biomarker development and validation. Herein, we provide a comprehensive review of biomarkers of the response to immunotherapy in GU tumors and their role in selection of therapy and disease monitoring.
Introduction
Genitourinary (GU) cancers include cancers along the male and female urinary tracts and the male reproductive tract, encompassing the adrenal glands, kidneys, bladder, ureters, urethra, prostate, penis, and testicles. In the United States alone, prostate, bladder, and kidney cancers are the first, fourth, and sixth most common cancers in men respectively, and kidney cancer is the ninth most common in women. 1 While the arsenal of treatments available for GU cancers has expanded in recent years, there remains significant clinical need for development of new tools for predicting patient prognosis and optimizing patient selection, especially when administering immunotherapy. For example, while immune checkpoint inhibitors (ICI) have seen impressive response rates in some GU malignancies, the therapeutic activity has been lacking in others, and administration of these regimens is not without risk, such as of iRAE development, underscoring the importance of investigating clinical indicators, such as biomarkers, of ICI activity and response.2,3 These biomarkers, which are laboratory or tissue-based values that reflect activity or change in disease processes in malignancy, may be either prognostic, providing information about patient outcomes, or predictive, providing indication about which patients may most benefit or be at risk of adverse events.4,5 Here, we review the current landscape of biomarkers for immunotherapy in GU cancers, as well as discuss the emerging data that continue to shape the evolution of urgently needed clinical biomarkers for immunotherapy in GU malignancies.
Renal Cell Carcinoma Biomarkers
While immune checkpoint blockade has seen considerable success in renal cell carcinoma (RCC), not all patients derive clinical benefit, and there are no approved clinical biomarkers that reliably predict treatment response. 6 This is, however, not for lack of investigation into potential such biomarkers.
In RCC, the clinical prognostic tool, IMDC risk score (International Metastatic RCC Database Consortium) has been the most widely used prognostic tool for therapy selection. This tool classifies patients into favorable, intermediate, and poor risk groups and incorporates time to systemic therapy, performance status, neutrophilia, anemia, thrombocytosis, and hypercalcemia. 7 Historically, patients IMDC favorable risk score have been considered to be preferentially driven by angiogenesis, when compared intermediate risk or poor risk disease, leading to the suggestion that intermediate/poor risk patients may gain more benefit from ICI.6-8 This was utilized for patient stratification in all first-line immunotherapy trials for mRCC and may continue to be a consideration for clinicians administering ICI to patients with RCC as evidenced by guidance from the European Association of Urology, which recommends dual ICI/VEGF therapy with pembrolizumab+axitinib, pembrolizumab- + envatinib, and avelumab + axitinib or nivolumab+cabozantinib for all risk groups with dual ICI with nivolumab + ipilimumab in intermediate to poor risk groups.7,9-11
Another biomarker of consideration in RCC is the presence of sarcomatoid features on pathologic analysis (Table 1). Despite the aggressive behavior of tumors with these features and associated poor prognostic features, sarcomatoid features have been associated with improved response to ICI, in several phase 3 trial cohorts, such as in Checkmate-214, where tumors with sarcomatoid features demonstrated improved outcomes when treated with nivolumab + ipilimumab when compared to sunitinib alone.12,24 Similar results were also seen in IMmotion 151, where patients with sarcomatoid features enjoyed improved progression free survival (PFS) when treated with atezolizumab+bevacizumab when compared to sunitinib alone. 13 Similar results have also been observed in non-trial cohorts, such as the IMDC dataset. 13 However, it is important to note that clinical use of this as a factor for patient selection still requires tissue-based pathological examination, which may present a challenge in certain cases or clinical settings.
RCC biomarkers table summary.

Measurement of PD-L1 expression in tumors has also been trialed as a strategy for predicting which patients will benefit from ICI, but fails to comprehensively identify those patient populations who will most benefit from ICI in several tumor types, despite its relative success in other tumor types, such as in lung cancers and in melanoma.14,39-46 Notably, PD-L1 expression is not a clinical criterion in the IMDC risk stratification, as it has been not established as a definitive marker of ICI benefit in RCC. While early trials suggested increased activity of ICI in patients with PD-L1+ disease, others have shown no significant increase in response in patients with PD-L1+ disease. For example, a phase II trial of nivolumab in mRCC showed increased response in patients with >5% PD-L1 expression, in KEYNOTE-427 increased response rates to single agent pembrolizumab were seen in patients with >1% PD-L1 expression, and in the Checkmate 214 trial, combination nivolumab/ipilimumab demonstrated improved PFS (when compared to sunitinib) only in patients with PD-L1+ disease.7,47,48 On the other hand, Checkmate 025 showed similar responses in patients with >1% or <1% PD-L1 expression, and the JAVELIN 101 trial also reported failure of PD-L1-based selection to achieve a statistically significant improvement in overall survival with axitinib+avelumab treatment.49-51 Updated findings from KEYNOTE-564 demonstrated a significant survival benefit in patients with treatment-naive ccRCC receiving adjuvant pembrolizumab after nephrectomy and/or metastasectomy. Patients were randomly allocated 1:1 to receive pembrolizumab or placebo with a median follow up of 57.2 months. The primary endpoint was DFS and secondary endpoints being OS and safety. DFS rate in the pembrolizumab group was 64.9% compared to placebo which was 56.6% (HR: 0.72, 95%, CI: 0.59-0.87). Approximately 75% of patients in each study arm had PD-L1 CPS ⩾ 1. OS benefit was sustained in patients with PD-L1 CPS ⩾ 1 with OS rate 91.2% with pembrolizumab versus 86.0% with placebo (HR 0.62, 95%, CI: 0.44, 0.87, P = .002). 15
The inconsistency seen across trials has been attributed to potential heterogeneity in PD-L1 expression in resected primary tumors (where PD-L1 expression is most often measured) and metastases (where therapeutic activity is desired), changes in PD-L1 expression over time, or intrinsic challenges in the assay itself, as currently utilized PD-L1 assays are not standardized or comparable across assays.14-16,52 Each assay is designed only for each specific ICI agent and has different thresholds for scoring PD-L1 positivity, and cross-trial comparisons can have significant limitations. Accordingly, significant challenges with the interpretation of these trials persist, given the lack of uniform testing strategies alongside the highly variable results across trials. Additionally, use of PD-L1 as a biomarker may be less reliable as tumors may also express PD-L2, which is also be targeted by anti-PD-1 agents but would not be detected by PD-L1 measurement assays. 17
Another widely studied biomarker is tumor mutational burden (TMB). TMB encompasses the total number of mutations present per coding sequence of the tumor gene and has been proposed as a possible biomarker of response to ICI in other tumor types.17,53,54 While TMB ⩾ 10 has been approved as an immunotherapy-related biomarker in TMB-H unresectable or metastatic advanced solid tumor malignancies generally, this biomarker has not been approved in RCC specifically. 18 Indeed, number of coding somatic mutations per megabase and objective response rate are strongly correlated in many cancer types, but notably, renal cell tumors depart from this general trend, having relatively modest TMB. 25 Amongst mutations present in RCC, insertions and deletions are the most frequently observed mutations in RCC, which have been associated with presence of neoantigens and with CD8+ T cell activation. RCC tumors have also been demonstrated to display endogenous retrovirus activation, which is also associated with T cell cytolytic activity and with neoantigen availability in RCC.18-21 Notably, TMB has failed to be a useful tool for selecting patients who will respond favorably to ICI therapies, as in the Checkmate 214, IMmotion150, and JAVELIN renal 101 trials, stratifying by TMB did not show any association with improvements in PFS or OS to ICI.22-24,51
RCC is known to be a tumor type with relatively high T cell infiltration, and in other tumors, such as melanoma, the level of CD8 T cell infiltration has been associated with the response to ICI.27-31 In RCC specifically, the role of infiltrating CD8 T cells in patient prognosis and in the response to immunotherapy has been debated, though translational studies have showed that some of these discrepancies may be owed to differences in measurement techniques, which subpopulation of CD8 T cells is considered, or which part of a patient’s tumor is sampled.55-59 Indeed, in JAVELIN Renal 101, patients with higher levels of CD8 T cells at the invasive margin of the tumor have improved PFS when treated with axitinib+andavelumab. 26 This is consistent with retrospective analyses demonstrating the correlation between intratumoral CD8 T cell infiltration and clinical benefit following ICI.32,33 Further study regarding measurement of infiltrating CD8 T cells as a biomarker for ICI response, with particular focus on standardizable methods of measurement, are needed to provide definitive determination of the clinical utility of this biomarker.
Molecular subtyping and transcriptional analysis of tumors has been another area of investigation in efforts to predict the response to ICI in RCC, though notably has not been validated for widespread clinical use. In the IMmotion 150 trial comparing atezolizumab with or without bevacizumab, angiogenic, T effector cell, and myeloid inflammatory gene expression signatures were established, and the T effector cell signature was associated with increased PD-L1 expression, CD8 T cell infiltration, and improved response to PD-L1 blockade. 23 Although not consistently validated and clinically utilized, patients have demonstrated varying responses to therapy depending on the tumor expression signature. For example, patients with high expression of T-effector signature demonstrated benefit from atezolizumab+bevacizumab. 60 Subsequently, the IMmotion 151 trial confirmed this, demonstrating a PFS benefit in patients with tumors with T effector cell signatures.61,62 Similarly, in the JAVELIN Renal 101 trial, a gene expression signature of 26 genes related to T cell activity, NK cell cytotoxicity, and chemokine expression was defined and associated with improved PFS in patients receiving axitinib+avelumab.51,63 Validation of the JAVELIN Renal 100 cohort confirmed this association as well. 64 Additionally, in Checkmate 214, a signature of high myeloid-related gene expression trended toward PFS improvement in dual ICI. 23 Notably, the association of single cell profiling with clinical outcomes in metastatic ccRCC is currently being investigated. 65 Importantly, as with measurement of PD-L1 expression and CD8 T cell density, these gene signatures may vary with the portion of a tumor sampled due to intratumoral heterogeneity or may evolve over time as a patient’s disease progresses. Furthermore, the analysis of sequencing data and determination of these signatures requires bioinformatic expertise, which may limit or affect the clinical application of these signatures as bona fide, usable biomarkers, especially in lower resource settings.
In addition to gene signatures, presence of specific gene mutations has also been interrogated as potential biomarkers in the response to ICI. In RCC specifically, loss of function mutation in PRBM1, a polybromo protein associated with the PBAF SWI/SNF chromatin remodeling complex, is common in ccRCC. 36 Mutations in PBRM1 have been associated with clinical response to ICI in RCC in multiple studies, including in Checkmate 009 and Checkmate 025.24,36,37 This was further validated in pre-clinical murine models, where inactivation of PBRM1 increased tumor cell sensitivity to IFN-γ and T cell killing.38,66 Similarly, analysis of ccRCC cell lines lacking PBAF function demonstrated altered transcriptional readouts of immune signaling. 37 Further study validating the role of PBRM1 mutation in ICI response will help delineate its clinical role as a predictive or prognostic biomarker in this setting.24,38
In addition to these, several additional biomarkers or adjuncts have been investigated in RCC, such as the role of the microbiome or of germline genetic variation. For example, certain bacterial species’ presence in the gut microbiota was proposed to potentially predict response to ICI, and temporary erasure of the gut microbiota through antibiotic use has been associated with impaired response to ICI in RCC. 67 A recent phase 1 trial demonstrated improved PFS in treatment-naïve patients with intermediate to poor IMDC risk after receiving dual ICI with CBM588, a live fecal microbiota, compared to receiving only dual ICI (12.7 months vs 2.5 months, HR 0.15, 95% CI: 0.05-0.47, P = .001). Based on these results, it has been suggested that addition of fecal microbiota may potentiate immunogenic response, however future larger-scale, prospective trials are needed to further validate this.
Additionally, heritable/germline genetic variation has been investigated as potential biomarkers, with 1 study reporting improved OS following lenvatinib+pembrolizumab in patients with high HLA-I evolutionary divergence and another reporting decreased OS after ICI in patients with a certain HLA type (HLA-A*03).68,69 Further study is warranted to define the potential impact of HLA variation on the response to immunotherapy, especially as these genes encode parts of the major histocompatibility complex (MHC), which is integral to antigen presentation to T cells.
Finally, some germline variations have also been investigated as biomarkers of increased risk of irAE after ICI therapy, with 1 study reporting IL7 gene germline variation to be associated with increased lymphocyte stability after ICI therapy and predicting downstream irAE, in addition to improved survival following therapy. 70 Further validation is needed to confirm this association, and further study should hope to uncover novel predictive markers of clinical response and of irAE, for both ccRCC and non-clear cell RCC. Investigating biomarkers for non-clear cell RCC is a particular need, as the vast majority of biomarker studies have exclusively focused on ccRCC, though some studies do report on chromophobe RCC, which faces particularly poor ICI response rates.9,47
Urothelial Cell Carcinoma Biomarkers
Urothelial cell carcinoma (UCC), the most common subtype of bladder cancer, has long been regarded as an immunogenic tumor type, and immunotherapy has been particularly effective for UCC patients with disease ineligible for or refractory to platinum-based therapy. 71 As with RCC, despite these successes, there is need for more definitive delineation of biomarkers with high clinical yield for prediction and prognostication in UCC patients receiving ICI.
Perhaps the foremost biomarker that has been considered in in UCC is measurement of PD-L1 expression (Table 2). Higher levels of PD-L1 expression in UCC tumors are associated with increased pathologic stage at resection, worse mortality outcomes, and higher rates of recurrence, in addition to impaired responses to standard therapy, making targeted immunotherapy a particularly appealing option for these patients.72-76 However, use of PD-L1 as a biomarker for response has been similarly fraught as in RCC, potentially due to the discrepancies in the 4 different clinical assays used and varying cell types (tumor cells vs infiltrating immune cells) for PD-L1 scoring.77-79 For example, PD-L1 positivity was associated with improved OS in a phase I/II trial of durvalumab and with improved DFS in patients with NIMBC in Checkmate 274, 18 but no such association was noted in IMVigor Cohort 2, Checkmate-275, or Keynote-045.80-84 A subgroup analysis of the JAVELIN Bladder 100 trial, comparing avelumab+best supportive care (BSC) compared to BSC alone, demonstrated a potential association of avelumab benefit in patients with high PD-L1 expression and TMB > median status (median = 7.66 SNVs/Mb). 85 The PURE-01 trial also provided promising results as an open label single arm phase II study evaluating the role of pembrolizumab efficacy in muscle invasive bladder cancer (MIBC) along with biomarker analysis. This trial resulted a 42% complete pathological response significantly enriched in patients with PD-L1 combined positive score (CPS) of ⩾10%).85,86,87 Similarly, PD-L1 positivity, CPS > 10, TMB-H, and dMMR were associated with higher pCR with nivolumab/ipilimumab in the NABUCCO trial. 88 The ABACUS trial, another single arm phase II trial, examined pathologic response in muscle invasive bladder cancer in patients who received 2 cycles of atezolizumab prior to cystectomy. Results showed a pathological complete response rate of 31% (95% CI: 21-41). 88 The study showed that baseline biomarkers such as activated T cells were correlated with outcome, while other biomarkers like tumor mutational burden, did not predict outcome, differentiating this from the metastatic setting.
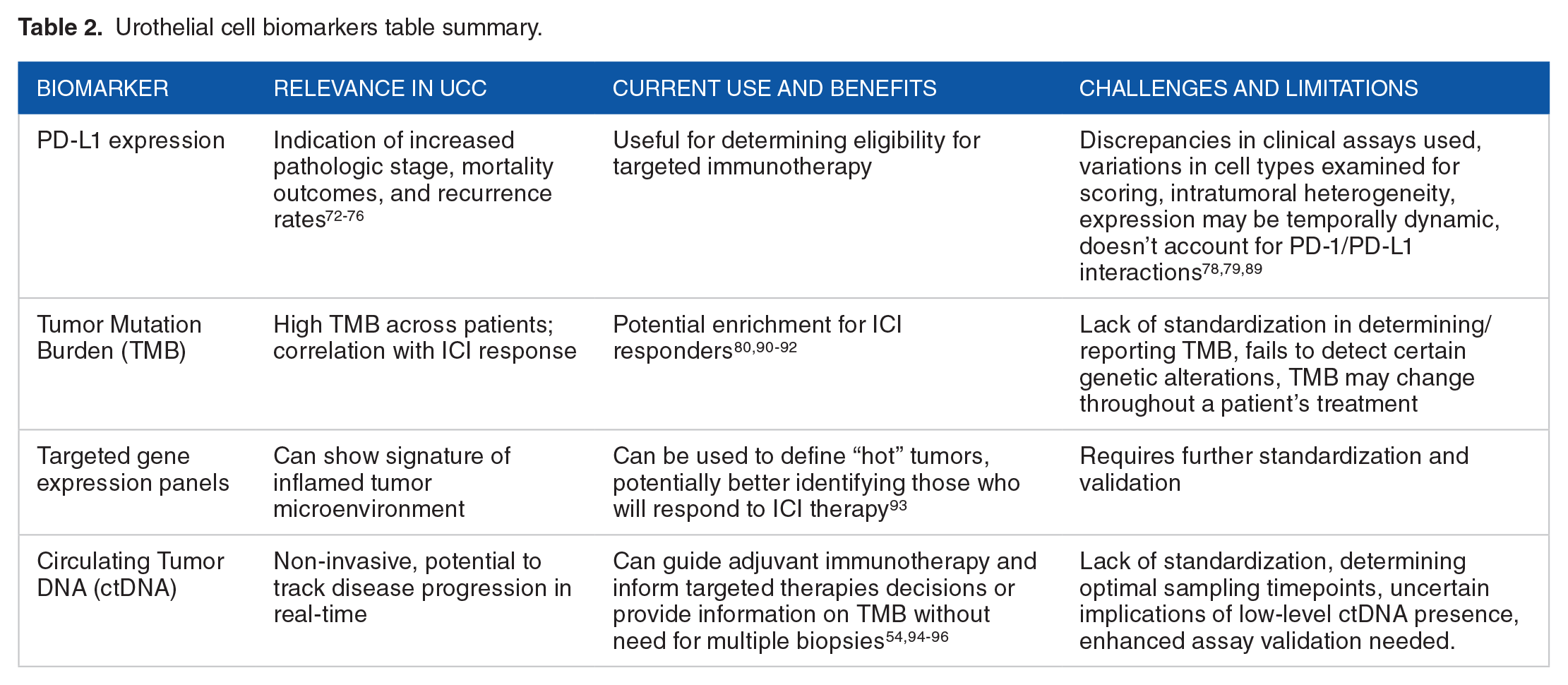
Urothelial cell biomarkers table summary.
An interim analysis of the AMBASSADOR study, an open-label randomized phase III trial, evaluated efficacy of pembrolizumab as adjuvant therapy in patients with high-risk muscle-invasive urothelial carcinoma who were ineligible or declined cis-based therapy following resection.97,98 Although adjuvant pembrolizumab demonstrated statistically significant median DFS improvement of 29 months over 14 months in patients who were observed [HR 0.69 (95% CI: 0.55-0.87, P = .0013)], preliminary findings indicate a nonsignificant benefit with respect to median OS of 50.9 months versus 55.8 months in pembrolizumab versus observation respectively [HR 0.98 (95% CI: 0.76-1.26), P = .88)]. Of note, the study allowed for crossover with 22% of patients in the observation arm later receiving pembrolizumab. In addition to the aforementioned technical challenges, as with RCC, intratumoral heterogeneity or temporal dynamics of PD-L1 expression may also limit the utility of PD-L1 expression as an individual biomarker, and measuring PD-L1 only in the tumor itself may fail to consider PD-1/PD-L1 interactions that may be occurring in the tumor draining lymph nodes, restraining the anti-tumor immune response in that setting, before even reaching the tumor tissue itself.
The distinction between platinum-sensitive and platinum-refractory urothelial cancer is critical when utilizing PD-L1 expression as a prognostic biomarker for immunotherapy response. Conventionally, PD-L1 expression is a more accurate predictor of response in patients with platinum-sensitive urothelial cancer compared to their platinum-refractory disease.98,99 This is considered to be primarily due to the heightened immune activity associated with PD-L1 expression in platinum-sensitive cases, which may predispose these patients to a superior immunotherapeutic response. Nevertheless, some platinum-refractory urothelial cancer patients may still benefit from checkpoint-based therapy, as recent research has shown that platinum-refractory patients exhibiting high PD-L1 expression had a significantly extended overall survival following immunotherapy compared to those with lower PD-L1 expression. 99
CheckMate-901 is a phase 3, randomized, international, open-label trial that compared chemotherapy and ICI against chemotherapy solely in untreated, unresectable metastatic urothelial carcinoma. 100 A subgroup analysis demonstrated improvement in OS and PFS with the combination regimen of cisplatin, gemcitabine, and nivolumab followed by nivolumab compared to cisplatin and gemcitabine only. Nivolumab and chemotherapy resulted in longer OS compared to chemotherapy alone (HR 0.78, 95% CI: 0.63-0.96, P = .02), and in patients with higher PD-L1 expression, a trend toward favoring nivolumab+chemotherapy over chemotherapy alone was observed, though did not reach significance. 101 Checkmate-274 is a phase 3 double blind trial in which 353 patients with MIUC were randomized 1:1 to adjuvant nivolumab versus placebo demonstrating sustained DFS of 52.6 months with nivolumab versus 8.4 months with placebo in PD-L1+ patients and 22.0 months with nivolumab versus 10.9 months with placebo in the ITT population. 102 The utility of PD-L1 based stratification, however, is also influenced by a myriad of other factors. These can include the specific type of PD-L1 assay employed, the degree of PD-L1 expression, and the activity of other mechanisms of adaptive immune resistance. 89 For example, in KEYNOTE-057, PD-L1 staining did not predict outcomes in high-risk NMIBC refractory to BCG. 103 As such, further research is essential to fully understand the role of PD-L1 expression in urothelial cancer, and more importantly, to accurately identify the patients most likely to benefit from immunotherapy.
As with RCC, TMB has also been investigated as a biomarker which may potentially enrich for favorable responses to ICI, given the relatively high TMB across all patients and an estimated 1% incidence of mismatch repair deficiency (dMMR) in UCC, especially in upper tract disease where incidence is higher and dMMR may due to Lynch Syndrome.104-106 Indeed, in an exploratory analysis of the IMVigor210 Cohort II, responding patients demonstrated an increased mutational load when compared to non-responding patients, and in IMVigor210 Cohort 1, patients with TMB in the highest quartile had improved OS. These findings are congruent additional trials’ exploratory analyses, demonstrating the utility of TMB in enriching for ICI responders in UCC. However, clinical application of TMB as a biomarker can be challenging, due to lack of standardization in methods of determining/mode of reporting TMB, failure to detect certain kinds of genetic alterations in some technical assays, and the dynamics of changing mutation burden as patients receive 1 or more lines of therapy.80,82,90,91 However, given the positive evidence supporting TMB correlation with ICI in UCC, standardization efforts may prove fruitful in progressing toward use of TMB as a surrogate marker of predicting ICI response.
Subclassifying UCC tumors based on gene expression patterns has also been trialed as a biomarker for ICI in UCC. These efforts have predominantly focused on utilizing The Cancer Genome Atlas (TCGA) to identify urothelial cancer subtypes. For example, in IMVigor210 Cohort II, TCGA subtypes were used to group patients into luminal or basal type disease and subclustered within those types. 92 While responses to ICI were reported in all subtypes, patients with luminal cluster II subtype had improved response rates when compared to luminal I, basal I, or basal II subtypes. 82 This trend held in the IMVigor Cohort I, where luminal cluster II patients demonstrated the highest response rates.77,107 On the other hand, Checkmate 275 reported basal I subtype tumors to have the highest response rate with nivolumab and report TMB being associated with OS, PFS, and ORR, irrespective of PD-L1 expression.84,108 However, it is proposed that the tissue source (as both trials allowed for biopsy specimens from primary tumor, lymph node, or metastatic lesions) or differential methods used for determining subclassification, may account for these discordant reports. 107 As such, definitive utilization of TCGA subtyping may prove challenging as a reliable biomarker for ICI in UCC. Definitive utilization of TCGA subtyping may prove challenging as a reliable biomarker for ICI in UCC.
Targeted gene expression panels have also been explored as a biomarker in UCC, namely by utilizing these to define a signature of an inflamed, and thus presumed more ICI-responsive, tumor microenvironment. As this approach encompasses more than a single marker of immune-activity (ie, in contrast to measuring PD-L1 as a stand-alone biomarker), it may hold an advantage in reliably identifying “hot” tumors and enriching for patients who will respond favorably to ICI therapy. For example, Checkmate 275 employed a 25-gene IFN-γ signature as an exploratory biomarker. This study reported higher IFN-γ gene signature expression in patients with favorable clinical responses to ICI, when compared to patients with low or medium expression of these genes. 93 While such a gene signature requires further standardization and validation, the multi-input nature of measuring multiple markers at once may present a particularly promising avenue for biomarker development.
In the era of genomic medicine, circulating tumor DNA (ctDNA) has also emerged as a potential biomarker for UCC, offering advantages in its non-invasive assessment and potential to track disease progression and response to therapies in real-time. The role of ctDNA in guiding adjuvant immunotherapy in UCC has become a topic of both clinical and research interest. 109 ctDNA, fragments of DNA in the blood stream that are released from dying tumor cells, carry genetic information about the tumor, such as specific mutations or changes in DNA methylation patterns. By analyzing this ctDNA, insights can be gained into the tumor’s genetic architecture, instrumental in guiding therapeutic decisions, particularly with regard to use of immunotherapies.110-112 In UCC, 1 study of UCC patients receiving adjuvant immunotherapy post-resection found that ctDNA was detectable in the majority of patients with residual disease following surgery, and ctDNA clearance after adjuvant therapy was strongly associated with disease-free survival (DFS). ctDNA also offers potential to reveal actionable genomic alterations and immune-related biomarkers, such as providing information on TMB. 113 By analyzing ctDNA, estimates of the TMB across the entire tumor burden can be made without the need for multiple biopsies, and the ability to sample ctDNA multiple times over the course of a patient’s disease addresses the challenge of intratumoral and temporal heterogeneity in measuring TMB as a potential biomarker for ICI response.54,94-96
IMVigor010 is a global, open-label, randomized phase 3 intention-to-treat trial that evaluated ctDNA status in 581 patients receiving atezolizumab versus observation in MIUC. 114 With a median follow up of 46.8 months, ctDNA positivity was associated with shorter OS in the observation group. Patients with ctDNA positivity receiving atezolizumab compared to those in observation had greater OS benefit [HR 0.59 (95% CI: 0.42-0.83)]. Greater reduction in ctDNA levels with atezolizumab at cycle 3 timepoint was associated with longer OS, while persistent ctDNA positivity was associated with relapse. However, while the role ctDNA in UCC management may be promising, its routine clinical application is not yet standardized and necessitates further standardization and validation. Key areas that require additional investigation include determining optimal sampling timepoints, understanding the implications of low-level ctDNA presence, and the need for assay validation. As the UCC biomarker landscape evolves, the integration of ctDNA analysis, alongside other biomarkers PD-L1 expression, may provide a more comprehensive understanding of each patient’s disease state, complementing and obviating the challenges of relying on any one of these biomarkers alone and allowing for a more personalized approach to immunotherapy in UCC. 115 In sum, as each of these potential biomarkers has shown some promise in UCC, but also each presents particular challenges, considering a hybrid biomarker, such as considering TMB or ctDNA and PD-L1 expression together or measuring multiple gene signatures simultaneously, may be an especially fruitful effort.
Prostate Cancer Biomarkers
In contrast to RCC and UCC, use of ICI in prostate cancer (PCa) has not seen the same level of success, perhaps in part to 1 or a combination of factors about the disease including its comparatively low mutational burden and relative “immunologic coldness” of prostate cancer.17,104,116 Especially given the great success of non-immune based therapies, such as androgen deprivation, novel hormonal agents, and, recently, PARP inhibition, immune checkpoint blockade has had limited role in treating prostate cancer. Additionally, a key consideration in use of immunotherapy in this population are potentially significant irAEs and proper risk-benefit must be discussed, given the limited success of these agents thus far. Nonetheless, pembrolizumab is used in metastatic hormone-resistant prostate cancer that has progressed after docetaxel and novel hormonal therapies and has TMB ⩾ 10 mut/Mb, dMMR, or MSI-high status. This is based on results from Keynote-028, which included several prostate cancer patients and led to FDA approval of pembrolizumab for tumors with MSI-H disease or dMMR that have progressed on prior treatment or have no alternative treatment options. 117 In this and subsequent reports of use of pembrolizumab in mCRPC (ie, Keynote 199 and Keynote 158), response to ICI in PCa remains limited, but has been suggested to be relatively increased among PCa patients in those patients with MSI-H disease, dMMR, high TMB, and POLE mutations (which may lead to increased TMB).117-125 Furthermore, as “immunogenic” subsets of prostate cancer have been reported (in addition to those with MSI-H or dMMR), further investigation may be warranted into identifying subsets of patients who may see significant clinical benefit from ICI therapy and what biomarkers (such as level of CD8 T cell infiltration or genomic loss of RB1, BRCA2, and CHD1) may identify these patients.111,126-131 A single center retrospective study evaluated 23 patients with metastatic hormone-resistant prostate cancer who received pembrolizumab. Notably, PSA response (defined as 50% reduction) were achieved only in the TMB-high group, in contrast to none in the TMB-low/medium group. While OS was not associated with TMB status, PFS favored the TMB-high group (19.34 months vs 2.53 months, HR 0.08, 95% CI: 0.02-0.40, P < .001). 132 While important to acknowledge a limited sample size and historically no survival benefit with ICIs in mCRPC patients, these studies suggest TMB status may hold some potential as a biomarker in aiding selection of patients who would benefit most from ICI therapy.
Finally, CONTACT-2 is a randomized phase 3 intention-to-treat (ITT) study of cabozantinib + atezolizumab in patients with hormone-refractory prostate cancer who had previously received novel hormonal agents. 133 The control group included patients receiving abiraterone and prednisone or enzalutamide. Median rPFS was the primary endpoint assessed per blinded independent review committee and was significantly improved in the cabozantinib/atezolizumab group versus control (6.3 months vs 4.2 months HR 0.65, 95% CI: 0.50-0.84, P = .0007). ORR was the secondary endpoint and was greater in the experimental versus control arm (14% vs 4%) in those with follow up greater than 6 months. Notably, there were 48% of patients who experienced grade 3 or 4 adverse events in the experimental arm compared to 23% in the control arm resulting in discontinuation of treatment in 16% in the cabozantinib/atezolizumab versus 15% in the control arm. This was an interim analysis awaiting OS data with median OS of 16.7 months (95% CI: 15.1-20.9) in the cabozantinib/atezolizumab arm versus 14.6 months (95% CI: 11.6-22.1, P = .13) in the NHT arm, and the field eagerly awaits final results from the study, including whether any biomarker based subgroup analyses shed light on predicting which patients may most benefit from this therapeutic regimen.
Rare GU Malignancies
Briefly, for rare GU malignancies, ICIs may be a potential therapy in some subsets of patients. A multi-center international cohort examined survival outcomes in patients with advanced penile squamous cell carcinoma reporting OS 9.8 months and PFS 3.2 months, with ORR of 13%. Of the 27 patients, 20 (77%) were PD-L1+ and, of patients with MSI data available, all were MSI-stable. There was no association between survival and PD-L1 status, though the study sample size was limited. However, some studies postulate that patients with HPV-related disease may benefit most from immunotherapy. A large query of a real-world database for advanced adrenal cortical carcinoma (ACC) demonstrated low overall prevalence of biomarkers associated with ICI response in other tumors, as well as monocyte and endothelial cell enrichment in metastatic tumors, suggesting an overall immunosuppressive environment in ACC.134-139 CABATEN is a phase 2 prospective, multicenter, open label study evaluating efficacy of cabozantinib/atezolizumab in locally advanced/metastatic ACC demonstrating median OS 13.5 months (95% CI: 8.8-NR) and PFS 2.9 months (95% CI: 2.8-5.7 months), demonstrating modest activity in a rare malignancy with a poor prognosis. 140 Much additional study is needed to clearly define the role of immunotherapy in treating rare GU malignancies, and in turn, establishing biomarkers of the response to immunotherapy in these cancers.
Discussion
In summary, this review article explores the current landscape of available immune checkpoint inhibitor associated biomarkers. In contrast to other solid tumor malignancies where some prognostic biomarkers have seen great success, RCC and UCC have been more challenging to identify reliable biomarkers for therapeutic response to immunotherapy. Although data support the notion that ICIs may be a particularly useful therapeutic regimen in some GU tumors (namely, RCC and UCC), response rates continue to leave room for improvement, which will require continued investigation of and validation of biomarkers for response. At the current juncture, definitive biomarkers are lacking, there also remains significant heterogeneity among biomarker measurement and application, and consensus regarding their role in prognostication or selection of treatment therapies has not been reached. For RCC, IMDC criteria continues to remain the standard to determine front-line therapy and prognosis. In contrast to intermediate to high-risk disease, nivolumab/ipilimumab did not demonstrate significant OS advantage in the favorable risk subset. 141 Thus, PD-L1 status should not be solely utilized to determine if patients should receive ICI in RCC. Tumor mutational burden is also of low utility in contrast to other malignancies such as lung cancer). 142 Although investigations are ongoing regarding use of genetic sequencing to identify clinically actionable mutations in RCC, current clinical landscapes may not be able to integrate this technology, limiting its utility in informing frontline therapy. 142 Moreover, the value of ctDNA is uncertain in the current landscape but may represent an attractive method and opportunity for further study for understanding disease temporality and dynamics in conjunction with prognostic tools.
As discussed urothelial malignancies face similar challenges in utilizing currently available biomarkers to inform treatment. In metastatic UCC, IMVigor130 suggests cisplatin-ineligible patients with PD-L1+ tumors may benefit from single agent immunotherapy. 143 Notably, a subgroup analysis of gemcitabine/cisplatin and atezolizumab demonstrated a larger effect on a statistically significant PFS compared to gemcitabine/carbotaxol and atezolizumab. 143 Patients receiving cisplatin backbone therapy may have transcriptional changes induced in immune cells, which could enhance T cell activation and antigen presentation. For example, Galsky et al postulates that gemcitabine/cisplatin may have favorable immunomodulatory effects on the tumor microenvironment by promoting dendritic cell activation and antigen-specific T cell attack. This study demonstrates that in patients receiving platinum-based chemotherapy + atezolizumab, patients with higher PD-L1 levels receiving gemcitabine + cisplatin + atezolizumab experienced longer OS than those with gemcitabine + carbotaxol + atezolizumab.144,145 On a similar note, Keynote-361 demonstrated that the first-line combination of pembrolizumab and platinum-based chemotherapy in metastatic UCC did not significantly improve OS in the overall population or in the PD-L1+ group. 146 Studies are ongoing in determining the utility of ctDNA in measuring residual disease and of TMB in treatment stratification in UCC.
Patients with metastatic prostate cancer have not achieved the same success with immunotherapy as have patients with RCC or UCC, and the role of biomarkers in informing use of these treatments is uncertain given their limited success in this setting. Studies are ongoing in assessing efficacy of ICIs in metastatic hormone-resistant prostate cancer. Notably, a study evaluating the immune landscape and evasion of prostate cancer in 498 patients revealed that nearly 90% of samples were non-immunogenic in their gene expression patterns, suggesting that this avenue of selecting patients may be a challenge in prostate cancer.147,148
IrAEs may represent a clinical biomarker of response for patients treated with immunotherapy.149,150 These adverse events create distress for the patient but may also be a surrogate for treatment response. A large retrospective study comparing patients with mRCC exhibiting irAEs to those who did not after receiving ICIs in first- or second-line demonstrated that patients with irAE had prolonged OS. An improved OS was also associated in cisplatin-refractory or cisplatin-ineligible patients with mUC receiving ICIs.35,151,152 Accordingly, continued investigation into understanding the relationship between the response to immunotherapy and ICI administration is warranted, particularly in hopes of optimizing a therapeutic regimen which may maximize therapeutic response while minimizing irAE and harm to patients.
Finally, we discuss the potential of using multiple biomarkers together to improve their predictive power. Mancini et al 153 highlights this notion of the importance of utilizing combination approaches while all of the FDA-approved biomarkers have value, none of them alone provide sufficient predictive power. No single biomarker or definitive set of biomarkers have risen to the surface as a reliably predictive marker in ICI response or prognostication in GU tumors. Thus, while emerging data and technology generate much excitement, considerable work remains in the future of GU biomarkers for immunotherapy.
Inherently, this review article has some notable limitations. This review focuses on immune checkpoint blockade, but the therapeutic landscape in GU malignancies has vastly grown beyond this in recent years with incorporation of antibody drug conjugates such as enfortumab-vedotin, which targets nectin-4 in UCC. It was recently approved for second line treatment after patients progress with cisplatin-based therapies and ICI and for cisplatin-ineligible patients. with locally advanced or metastatic urothelial carcinoma. Other notable targeted treatments include erdafitinib for UCC with fibroblast growth factor receptor 3 (FGFR3) alterations, disitamab-vedotin and trastuzumab-deruxtecan for HER-2/ERBB2 overexpressed UCC, and sacituzumab-govitecan for Trop-2 enriched locally advanced unresectable/metastatic UCC refractory to chemotherapy and ICIs.154,155 The field anticipates the emergence of more studies regarding these important emerging treatments. Other topics not discussed are mechanisms of resistance to ICI blockade, sequencing/combination of therapies involving ICIs, and molecular tumor profiling to inform selection of antibody drug conjugates and other T-cell engager therapies.
Conclusions
In summary, we present a comprehensive overview of ICI-related biomarkers in GU malignancies. ICIs have cemented itself as an important backbone of therapy in several GU malignancies, such as RCC and UCC. While immunotherapy has significantly improved outcomes in these patients, a significant subset of patients invariably develops relapsed or refractory disease. Although ICI-related biomarkers have seen some success with prognostication and therapy selection in other solid tumor malignancies, many have not been validated in RCC and UC. Utility of biomarkers of the response to ICI result with a binary result or predetermined thresholds may be better enhanced with data from molecular tumor profiling markers. Further efforts are required in conducting robust, multicenter, prospective studies to evaluate their feasibility.
Footnotes
Acknowledgements
None.
