Abstract
Background:
Systemic lupus erythematosus (SLE) is a chronic, multi phenotypic, autoimmune inflammatory disease and renal involvement significantly worsens its prognosis. Apoptosis dysregulation plays a key pathogenic role. Survivin, a protein from the apoptosis inhibitors family, has been considered a promising strategy in cancer therapy and evaluated as one of the regulatory pathways in the scenario of immune-mediated disorders.
Objective:
This study aims to explore survivin behaviour in SLE patients with lupus nephritis (LN), assessing its potential as a therapeutic and prognostic biomarker.
Methods:
297 SLE patients were classified based on the American College of Rheumatology (ACR) 1997 criteria, from 2000 to 2015. In a cross-sectional study, the serum level of survivin was measured by an ELISA test and compared between 200 SLE individuals and healthy controls. In a longitudinal cohort, 97 patients with active LN had the concentration of survinin measured, before and after treatment with cyclophosphamide pulse therapy.
Results:
The serum concentration of survivin was significantly lower in the SLE group than in healthy controls, regardless of concomitant NL or disease activity. The longitudinal evaluation revealed a significant reduction in survivin serum level after treatment. However, survivin rates were not able to discriminate groups that achieved remission from those that maintained nephritis activity.
Conclusion:
Our study suggests that survivin levels in SLE patients are lower than in the general population. Even so, its use as a biomarker in SLE seems limited, not reflecting disease activity or response to LN treatment, as in other contexts.
Introduction
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease with a broad range of clinical manifestations and variable morbidity and mortality.1,2 Its treatment is mainly guided by the organ or system involvement, whose evaluation is useful for prognosis too. 3 A set of clinical manifestations and laboratory tests grouped as the revised American College of Rheumatology (ACR) classification criteria is useful for diagnosis and research purposes. 4
Renal involvement occurs in about 60% of SLE patients and may be asymptomatic or present as lupus nephritis (LN). LN significantly worsens the prognosis.5,6 To minimize cumulative damage and preserve glomerular function, therapy should be initiated as soon as the diagnosis is made. Corticosteroids associated with cyclophosphamide or mycophenolate are commonly the first line of therapy, aiming to suppress glomerular inflammation due to the deposition of immune complexes. 7 Kidney biopsy remains the most accurate approach for LN, allowing classification, differential diagnosis and decision on the best therapy along with information about long-term prognosis. 8 The glomerular filtration rate, urinary sediment activity and proteinuria are parameters for the evaluation of renal injury that add to the histopathological study information. 1 Complete remission of nephritis, the main goal of therapy, is related to long-term renal function, despite being achieved in less than 50% of treated patients. 1
Current methods of assessing disease activity in SLE, including acute phase reactants, dsDNA antibodies and serum complement, are of limited sensitivity and specificity, driving the search for better biomarkers of disease activity and prognosis.9-11 Improved blood and urine markers can help early diagnosis, distinguishing exacerbation from chronic damage and monitoring therapeutic response.
Several questions about the pathogenesis of SLE remain uncertain. It is relatively recent the knowledge that neutrophils and other apoptotic cells are important in the development of the disease. 2 Patients with SLE carry many abnormalities in programmed cell death, leading to the presentation of toxic products and potential autoantigens for an overactive immune system.12-14 Increased apoptosis is a key process in the pathogenesis of SLE,15,16 resulting from a disbalanced relation between pro and anti-apoptotic pathways, such as Fas-FasL-caspases and the inhibitors of apoptosis proteins (IAPs), respectively.17,18
Survivin, a member of IAPs, is a multifunctional protein expressed in the nucleus, cytoplasm and mitochondria. Survivin synthesis, expression and degradation are cell cycle-dependent in normal tissues; they are abundantly expressed during the G2/M phase and dramatically drop during the G1 phase. 19 It inhibits apoptosis in both caspase-dependent (external) and mitochondrial (internal) cascades.20,21 During the G2/M phase of mitosis, survivin aids cell division and exerts an anti-apoptotic effect, the reason why it has been studied as a promising strategy in cancer therapy. 22 Survivin also has an important role in multiple immune system processes. 23 By controlling the density of the MHC II on dendritic cells (DCs), survivin is essential for the maturation of these antigen-presenting cells. 24 In acquired immunity, survivin is upregulated in B cells of the germinal centres, triggering the expansion of immune cells and hypermutation of B cell receptors. 23 It also takes part in the regulation of T lymphocytes development, differentiating CD4+ and maintaining CD8+ memory cells.
Recent evidence indicates that dysregulated survivin expression is involved in the pathogenesis of several autoimmune diseases, contributing to tolerance breakdown.25,26 Previous studies revealed an increased expression of survivin in autoreactive lymphocytes and neutrophils under chronic inflammatory conditions, suggesting its interference in reducing apoptosis rate.14,23,27 It is supposed that survivin may be positively regulated in B cells leading to increased production of autoantibodies in SLE. 23 Despite the important function of apoptosis in the pathogenesis of SLE, we do not know whether survivin participates in the process nor if its participation has clinical relevance.
This study aims to evaluate the serum levels of survivin in patients with LN and its potential as a biomarker of therapeutic response and prognosis.
Patients and Methods
Study design and patients
This is an observational study with data collected retrospectively from medical records. We conducted both cross-sectional and cohort studies. Firstly, for the cross-sectional study, SLE patients were divided into 4 subgroups according to the disease activity and the evidence of LN, current or previously. Subgroups were compared among them and with a group of healthy subjects. Secondly, a different group of SLE patients with active LN took part in the cohort study. These patients were enrolled just before the beginning of therapy with methylprednisolone and cyclophosphamide and followed up for 6 months.
The general inclusion criteria were age ⩾18 years, regular attendance to rheumatology outpatient clinic and diagnosis of SLE made from 2000 to 2015; diagnosis of SLE according to ACR-1997 classification criteria. 4 The clinical and laboratory parameters listed as SLE classification criteria (ACR, 1997) in the groups were those present at any point of the disease, cumulatively, until enrolment in the study. All suspected cases of LN underwent renal biopsy (HE and immunofluorescence staining) and were classified according to the International Society of Nephrology/Renal Pathology Society (ISN/RPS).28-30 Patients with additional kidney injuries, such as diabetic nephropathy, renal replacement therapy, hepatic diseases, malignancy, other autoimmune conditions, pregnancy or lactation and incomplete recorded data were excluded.
Clinical and laboratory variables
Demographic, clinical aspects and results of laboratory tests were obtained from medical records. We evaluated complete blood count, serum creatinine (normal value: 0.4-1.1 mg/dL), serum complement levels of C3 (normal value: 0.9-1.8 g/L) and C4 (0.1-0.4 g/L), urinalysis and the presence of antinuclear (ANA), anti-double-stranded DNA (anti-dsDNA), anti-phospholipid (anti-APL) and anti-Smith (anti-SM) antibodies. Survivin level measurements were carried out by analysis of the samples previously collected and stored at −80°C in the hospital’s bio reservoir.
Study groups
Patients with SLE were allocated into 4 categories based on disease activity and evidence of LN and compared with each other and with healthy volunteers, in the cross-sectional study. Among them, there are cases of active lupus nephritis (aLN), inactive lupus nephritis (iLN), active SLE without nephritis (aSLE) and cases of inactive SLE without nephritis (iSLE). We considered patients with SLEDAI > 4 as active disease and patients with active urinary sediment and proteinuria >0.5 g/24 hours as active LN. The group of healthy volunteers was composed of adult subjects, matched by sex and age to the SLE patients and recruited among employees of the university hospital.
A different selection of SLE patients with active lupus nephritis was compared pre- and post-treatment. In this cohort study, only patients with active lupus nephritis who required cyclophosphamide as a remission-inducing treatment were enrolled. Clinical and laboratory data and blood samples for measurement of survivin concentration were obtained immediately before and after the treatment. These patients received monthly i.v. cyclophosphamide (0.5 g/m2 body surface area) and methylprednisolone (1 g) pulse therapy for 6 consecutive months.
Measurement of SLE disease activity
Disease activity was evaluated by a rheumatologist using the systemic lupus erythematosus disease activity index (SLEDAI) and classified as an active disease if SLEDAI > 4 (5-9: mild, 10-14: moderate, ⩾15: severe) and inactive disease if SLEDAI ⩽ 4, for patients on a stable dose of immunosuppressive and/or antimalarial medications and on prednisone dose ⩽5 mg/day.31,32
Assessment of remission of lupus nephritis
Complete remission (CR) of nephritis after treatment was considered in patients who have achieved normal glomerular filtration rate, proteinuria lower than 0.5 g/24 hours and inactive urinary sediment. Partial remission (PR) was considered in patients who presented proteinuria lower than 3 g/24 hours if baseline proteinuria was higher than 3 g/24 hours or 50% reduction if initial proteinuria was lower than 3 g/24 hours along with a 25% improvement in glomerular filtration rate. Therapeutic remission failure (RF) was considered in patients who did not reach minimal improvement of the parameters described. 1
Histologic pattern of lupus nephritis
A kidney biopsy was performed in all patients with LN included in this study. Based on the International Society of Nephrology/Renal Pathology Society 2003 classification of lupus nephritis, the patients were classified as having mesangial proliferative nephritis (class II), focal lupus nephritis (class III), diffuse segmental or global nephritis (class IV), membranous lupus nephritis (class V), combinations of membranous and proliferative glomerulonephritis (class III-V or IV-V) and advanced sclerosing nephritis (class VI). The histologic classification and stage of renal disease (activity and chronicity) helped determine the treatment. However, once the histological diagnosis was established, the transition from one type of glomerular lesion to another was not followed by serial biopsies. Therefore, a detailed clinical and laboratory evaluation was also used to predict the histologic pattern of lupus nephritis and even though it is not accurate enough, it was applied as assistance in choosing more specific immunosuppressive management.
Serum quantification of survivin
Serum samples were stored at −80°C. The concentration of survivin was determined using a human quantitative enzyme immunoassay, Quantikine® Human Survivin Immunoassay by R&D Systems, in an ELISA plate reader. The sensitivity of the assay was 1.58 to 9.96 pg/mL, a mean of 4.44 pg/mL. The intra-assay precision for serum was 3.4% to 5.5% and inter-assay precision was 5.6% to 9.5%.
Statistical analysis
The sample size calculated was 200 patients (50 patients in each study group), to obtain a 95% confidence interval with a margin of error of 5% around a prevalence estimate under 1%. The formula used to calculate the sample size for a reliable estimate of the average population was n = (Zα/2 × σ/E) 2 , (n = sample size, Zα/2 = degree of confidence, σ = standard deviation, E = margen of error).
Estimates of effect size and standard deviation were based on previous experiences of our research group and on the existing literature. To calculate the power of analysis we assumed α = .05 and β = .2. The normal distribution of data was verified with the Kolmogorov-Smirnov test. Normally and non-normally distributed continuous variables were reported as mean and standard deviation and median and interquartile range. Qualitative variables were reported as frequency and percentage. Differences between groups were compared using t-test and Mann-Whitney U test, as appropriate. Kruskal-Wallis ANOVA tests were applied in the cross-sectional analysis. Correlations between variables were analysed by Spearman rank correlation analysis. P values lower than .05 were adopted for statistical significance. All analyses were performed using IBM SPSS version 21.0 (IBM Corp., Armonk, NY).
Ethical considerations
Linked to this and other previous research projects in autoimmune diseases of the rheumatology department, a bio repository was created to store serum from the SLE patients and healthy subjects. Before any procedure, all patients and healthy volunteers signed an informed consent. The study was approved by the institutional review board, HC-FMRP Research Ethics Committee (No. 146/2011 and Opinion No. 2789973) and followed the Conference on Harmonization of Good Clinical Practice (ICH-GCP).
Results
Patients
A total number of 810 medical records of SLE patients followed in the Department of Rheumatology from 2000 to 2015 were retrospectively analysed, resulting in 297 cases enrolled. Among these patients, 191 (64.3%) were diagnosed with SLE and lupus nephritis (LN) and 106 (35.7%) were SLE without lupus nephritis. In the whole study population (n = 297), 91.6% were women and the mean age was 33.5 ± 12.6 (18-69) years.
The main clinical and demographic characteristics of SLE patients were described (Table 1). Besides LN, the most common classification criteria were cutaneous (68.6%) and articular (62.6%) involvement, followed by autoimmune cytopenias (48.4%) and serositis (28.9%). The frequency of positive ANA was 97%, while positivity for anti-dsDNA, anti-APL and anti-SM antibodies were 69%, 36% and 28%, respectively. The kidney biopsy rate was 64.3% (n = 191/297) in all SLE patients and 100% in the SLE lupus nephritis subgroup, with a predominance of the proliferative and/or membranoproliferative classes. A few individuals were treated with pulse therapy despite findings of mesangial proliferative nephritis (class IIb) on biopsy. Management was preferable to conventional treatment for class IIb due to clinical and laboratory findings of a severe nephritic-nephrotic syndrome in these cases.
Demographic and clinical aspects, laboratory tests and survivin serum levels of all SLE patients (n = 297). Active and nonactive systemic lupus erythematosus patients, with or without lupus nephritis, those who underwent pulse therapy or not.
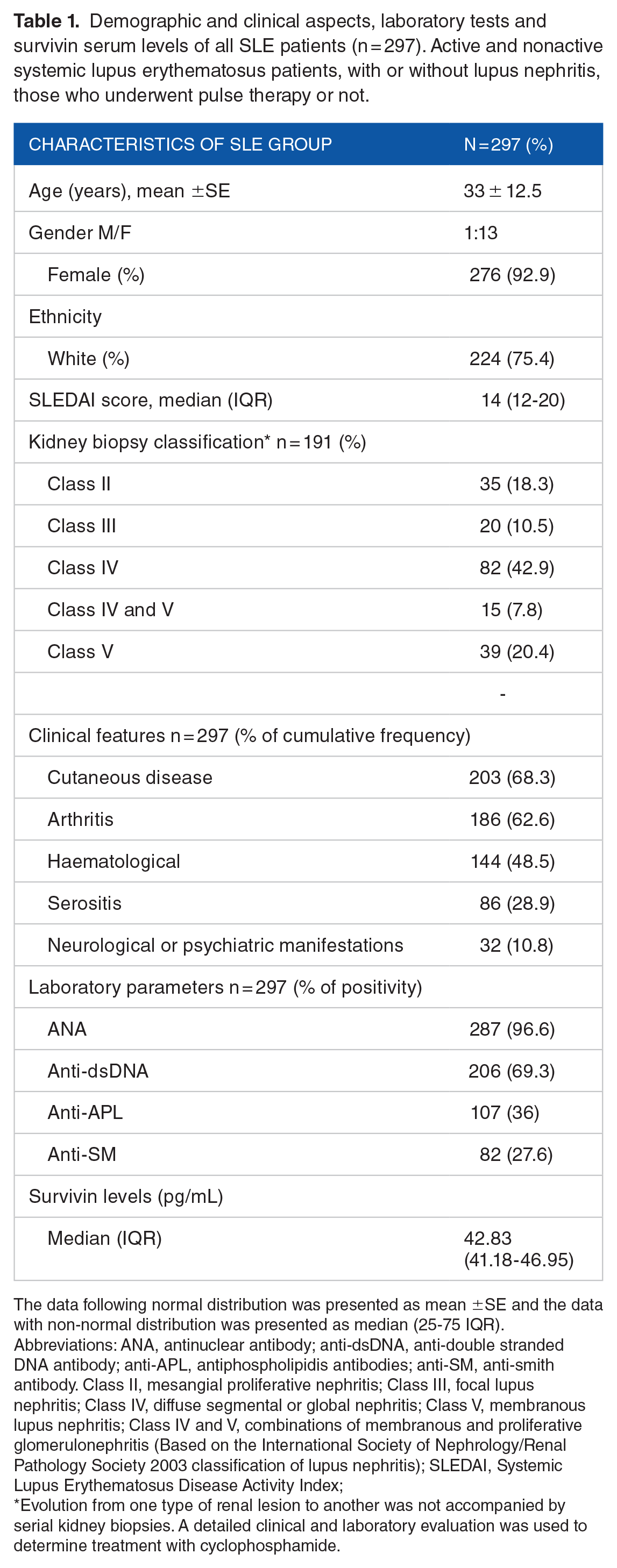
The data following normal distribution was presented as mean ±SE and the data with non-normal distribution was presented as median (25-75 IQR).
Abbreviations: ANA, antinuclear antibody; anti-dsDNA, anti-double stranded DNA antibody; anti-APL, antiphospholipidis antibodies; anti-SM, anti-smith antibody. Class II, mesangial proliferative nephritis; Class III, focal lupus nephritis; Class IV, diffuse segmental or global nephritis; Class V, membranous lupus nephritis; Class IV and V, combinations of membranous and proliferative glomerulonephritis (Based on the International Society of Nephrology/Renal Pathology Society 2003 classification of lupus nephritis); SLEDAI, Systemic Lupus Erythematosus Disease Activity Index;
Evolution from one type of renal lesion to another was not accompanied by serial kidney biopsies. A detailed clinical and laboratory evaluation was used to determine treatment with cyclophosphamide.
Two hundred SLE patients established 4 subgroups. The disease activity of each subgroup was scored by SLEDAI (median, IQR) at the time of sample collection. Patients were divided into active-LN (n = 50; SLEDAI = 16.0, 12.0-18.0), inactive-LN (n = 44; SLEDAI = 0, 0-2.0), active-SLE without nephritis (n = 52; SLEDAI = 7.5, 6.0-10.0) and inactive-SLE (n = 54; SLEDAI = 0, 0-2.0). Another independent group of 97 SLE patients with active lupus nephritis was compared before and after monthly i.v. cyclophosphamide and methylprednisolone (1 g) pulse therapy (Figure 1).

Flowchart of the allocation of patients diagnosed with systemic lupus erythematosus (SLE), among those followed by the rheumatology outpatient clinic. Considering the 1999 American College of Rheumatology classification criteria for SLE, the 2003 International Society of Nephrology/Renal Pathology Society classification criteria for lupus nephritis and the data obtained by the investigators, patients were classified as active SLE or non-active SLE, SLE with active nephritis or non-active lupus nephritis (LN). The figure shows a group of active-LN patients (n = 97) allocated in the longitudinal study to receive pulse therapy with cyclophosphamide for 6 months (dotted boxes). Another 4 subgroups of SLE patients were included in the cross-sectional study and compared with each other (n = 200) and with healthy volunteers (n = 45).
Serum survivin concentration: A cross-sectional evaluation of SLE patients and healthy controls
The SLE patients’ clinical and laboratory data, of which 94 (47%) from individuals with lupus nephritis and 106 (53%) from individuals without lupus nephritis, were described (Table 2). Two hundred previously collected SLE serum samples were examined for survivin levels and compared to healthy controls (n = 45).
Demographic and clinical aspects, laboratory tests and survivin serum levels of the 4 SLE subgroups (active and nonactive systemic lupus erythematosus patients, with or without lupus nephritis) compared with healthy controls. Results of a cross-sectional analysis using Kruskal-Wallis ANOVA test.
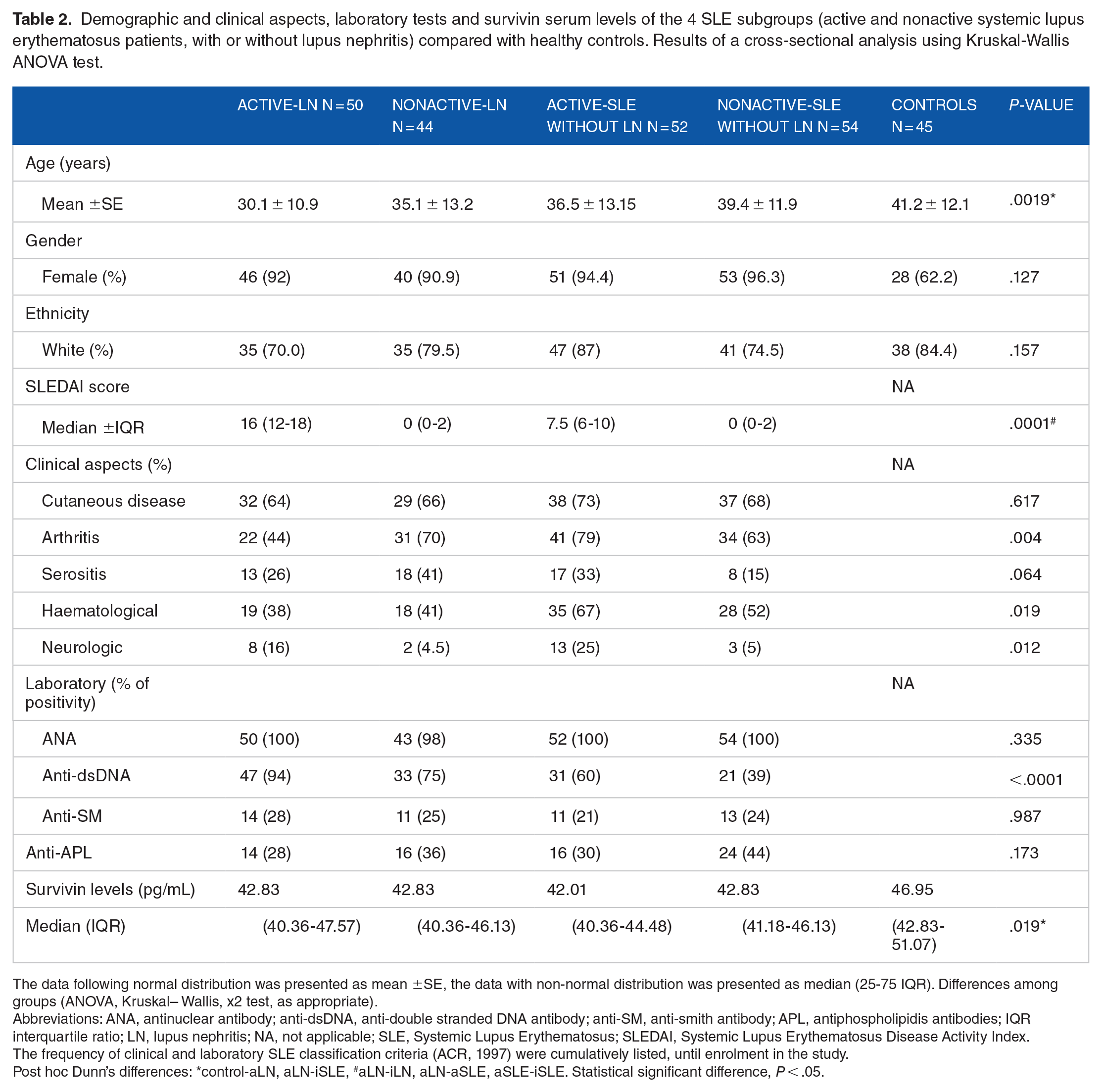
The data following normal distribution was presented as mean ±SE, the data with non-normal distribution was presented as median (25-75 IQR). Differences among groups (ANOVA, Kruskal– Wallis, x2 test, as appropriate).
Abbreviations: ANA, antinuclear antibody; anti-dsDNA, anti-double stranded DNA antibody; anti-SM, anti-smith antibody; APL, antiphospholipidis antibodies; IQR interquartile ratio; LN, lupus nephritis; NA, not applicable; SLE, Systemic Lupus Erythematosus; SLEDAI, Systemic Lupus Erythematosus Disease Activity Index.
The frequency of clinical and laboratory SLE classification criteria (ACR, 1997) were cumulatively listed, until enrolment in the study.
Post hoc Dunn’s differences: *control-aLN, aLN-iSLE, #aLN-iLN, aLN-aSLE, aSLE-iSLE. Statistical significant difference, P < .05.
We observed that serum levels of survivin were lower in SLE patients compared to the healthy subjects (P = .019). However, there was no difference in survivin levels among the subgroups of patients with SLE, regardless of the renal involvement or the disease activity (Table 2). Even without statistical difference, a lower proportion of female patients was observed in the control group.
Serum survivin concentration: Evaluation of active-LN patients pre- and post-treatment
Ninety-seven SLE patients underwent a 6-month cyclophosphamide pulse therapy for active-LN treatment (Table 3). Serum samples for survivin measurements were collected before and after the treatment. There was a statistically significant reduction in survivin levels after the treatment, when compared to the initial pre-treatment values, despite nephritis outcomes (P < .0001).
Demographic aspects, clinical characteristics, laboratory tests and histological parameters of the nephritis-active systemic lupus erythematosus group (n = 97), that underwent pulse therapy with cyclophosphamide and methylprednisolone (longitudinal analysis group).

The frequency of clinical and laboratory SLE classification criteria (ACR, 1997) were cumulatively listed, until enrolment in the study. Class II: mesangial proliferative nephritis, Class III: focal lupus nephritis, Class IV: diffuse segmental or global nephritis, Class V: membranous lupus nephritis, Class IV and V: combinations of membranous and proliferative glomerulonephritis, Class VI: advanced sclerosing nephritis (Based on the International Society of Nephrology/Renal Pathology Society 2003 classification of lupus nephritis).
Abbreviations: ANA =, antinuclear antibody; anti-dsDNA , anti-double stranded DNA antibody; anti-SM , anti-smith antibody; anti-APL , antiphospholipidis antibodies.
All cases of lupus nephritis were confirmed by renal biopsy (HE and immunofluorescence stains). Evolution from one type of lesion to another was not accompanied by serial renal biopsies. A detailed clinical and laboratory evaluation was used to determine treatment with cyclophosphamide.
In agreement, changes in other typical parameters used to evaluate lupus activity and remission of nephritis have also been demonstrated. Significant reduction in SLEDAI score (P < .0001), decreased 24 hours proteinuria (P < .0001), increased complement C3 and C4 fractions (P < .0001), improvement in glomerular filtration rate assessed through creatinine clearance (P = .003) and lower frequency of positive dsDNA antibody (P < .0001) were achieved after treatment (Table 4).
Comparative analysis of survivin levels, disease activity index (SLEDAI), 24-hour proteinuria and frequency of laboratory parameters in patients with lupus nephritis, before and after 6-monthly pulse therapy treatment (n = 97). Results of a longitudinal study using paired t test and Wilcoxon matched-pairs non-parametric test.
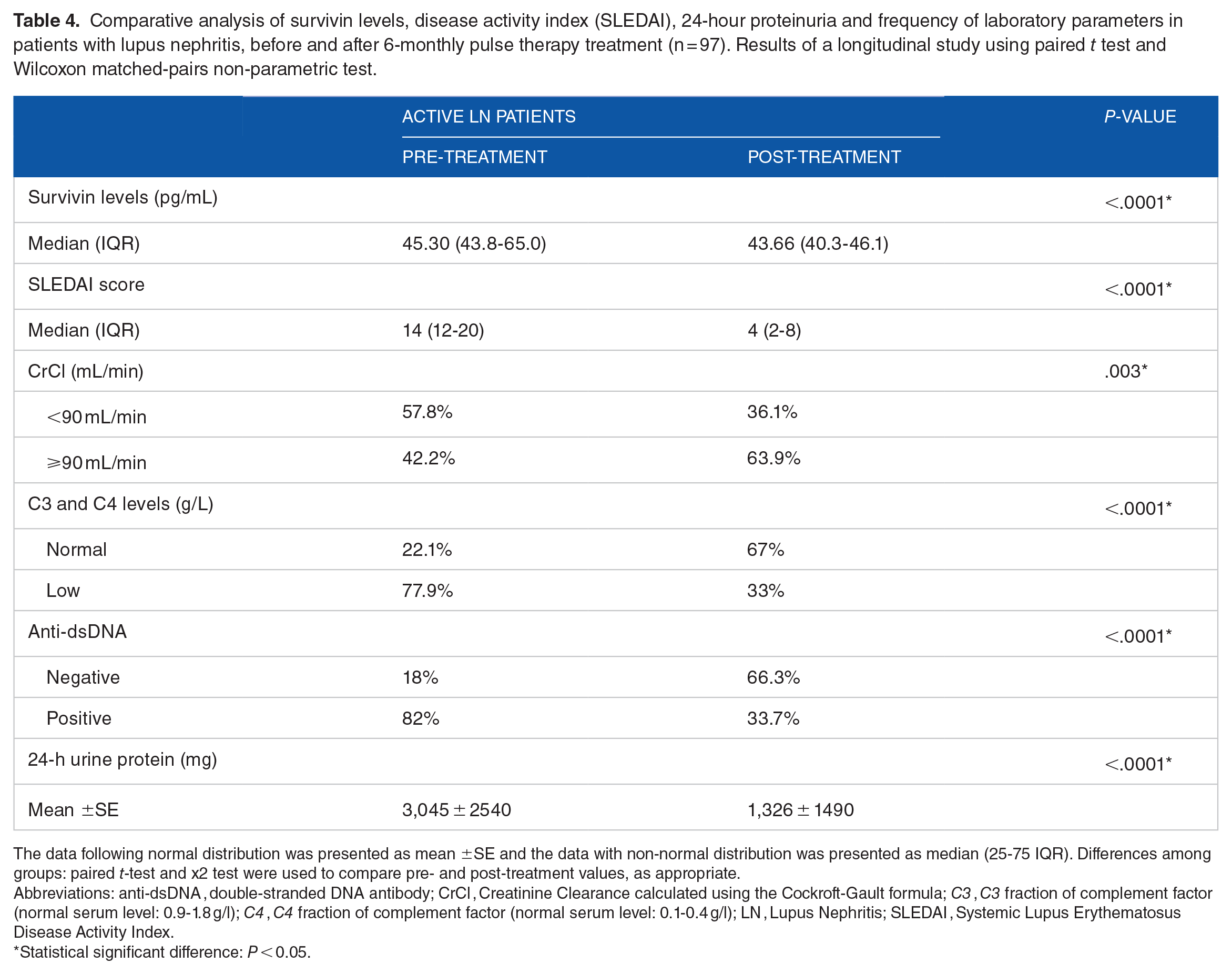
The data following normal distribution was presented as mean ±SE and the data with non-normal distribution was presented as median (25-75 IQR). Differences among groups: paired t-test and x2 test were used to compare pre- and post-treatment values, as appropriate.
Abbreviations: anti-dsDNA , double-stranded DNA antibody; CrCl , Creatinine Clearance calculated using the Cockroft-Gault formula; C3 , C3 fraction of complement factor (normal serum level: 0.9-1.8 g/l); C4 , C4 fraction of complement factor (normal serum level: 0.1-0.4 g/l); LN , Lupus Nephritis; SLEDAI , Systemic Lupus Erythematosus Disease Activity Index.
Statistical significant difference: P < 0.05.
Nevertheless, among all 97 patients treated for nephritis, only 62% (n = 60/97) achieved remission after 6 months, of which 29% for complete remission (CR) and 33% for partial remission (PR) according to previously established criteria. The remission failure (RF) group was attributed to individuals who maintained disease activity (38%, n = 37/97) after the 6-months cyclophosphamide pulse therapy.
To assess whether survivin would be able to predict or discriminate patients responding to nephritis treatment, comparative analysis in subgroups with CR or PR and with RF to cyclophosphamide pulses was performed. We observed a significant reduction in survivin levels after treatment in both the remission and FR groups (P = .0014 and P = .0008, respectively). There was no statistical difference between the remission (CR and PR grouped) and the FR groups neither before nor after cyclophosphamide treatment (P = .158 and P = .431, respectively) (Figure 2). No correlation was found between survivin levels and the traditional biomarkers (serum albumin, proteinuria, serum complement and serum creatinine) or SLEDAI after treatment.

Behaviour of survivin serum levels before and after treatment of lupus nephritis with cyclophosphamide. Overall and subgroups analysis of patients who achieved remission of nephritis (CR or PR) and those who failed treatment (FR). Wilcoxon matched-pairs non-parametric test; values presented as median (IQR).
Discussion
Lupus nephritis usually occurs within 5 years of the onset of SLE and the early diagnosis of the involvement of the kidneys can greatly improve renal outcomes and patients’ survival rates. In this study, we tested survivin as a potential biomarker of disease activity and treatment response.
Increased expression of survivin was previously demonstrated in several autoimmune diseases like rheumatoid arthritis, juvenile idiopathic arthritis, myasthenia gravis, multiple sclerosis, psoriasis, systemic sclerosis,25,33-38 and its regulation has served as a marker of immune cells apoptosis inhibition and response to treatment.23,25,37,39 Although oestrogen regulates various facet of autoimmune diseases, the aspects of oestrogen-mediated effects on survivin are not clarified in each of them as they are in cancer. Apoptosis is an essential mechanism in the deletion of autoreactive lymphocytes during the negative selection process in the thymus and bone marrow, as well as in removing the chronically activated cells and controlling maturation of dendritic cells and expression of MHC II molecules. Due to previous knowledge about the anarchic process of apoptosis involved in the pathogenesis of lupus, we assumed that survivin levels would be altered in SLE patients.23-27 Thus, a proliferation of autoreactive cells could explain the role of survivin in the disease. 37
In fact, we found lower serum survivin in SLE patients than in healthy volunteers, irrespective of the presence of glomerulonephritis or active disease. It seems dissimilar to other T cell-mediated autoimmune conditions, where protein expression is increased.23,25,33,35,36,38,39 Consistent with our results, Ebrahimian et al described lower serum survivin levels in SLE, regardless of disease activity, clinical presentation or response to treatment.40,41 Evidence of a distinct effector mechanism of survivin in the pathogenesis of SLE. 40 Just recently, several microRNAs have been implicated in survivin regulation which may downregulate anti-apoptotic survivin expression in apoptotic cells or, in opposite direction, contribute to survivin upregulation, enhancing their sustained activation and autoreactivity in autoreactive immune cells. 42
The usefulness of survivin as a marker of tumour progression, drug resistance and aggressiveness in neoplastic diseases is currently well-established.22,43 In rheumatoid arthritis, this protein may comprise a prognostic effect, signalling worse radiological evolution and predicting therapeutic response in the early stage of the disease. 37 This idea emphasizes another relevant finding of the present study, which was the reduction in serum survivin levels after the treatment with cyclophosphamide. Nevertheless, the decrease occurred independently of the treatment outcome (complete or partial remission of nephritis or therapeutic failure). When comparing survivin levels of both remission and failure groups, no significant difference was established and survivin was not able to differentiate them. Our data suggest that survivin might not distinguish active from inactive SLE groups, nor reflect therapeutic response or prognosis of nephritis. It stands as a possible limitation of survivin utility as a biomarker in LN.
Some drugs used in the treatment of autoimmune diseases affect survivin expression with indirect inhibition. 44 The reduction in survivin levels after treatment, compared with values before pulse therapy, seems to involve cyclophosphamide’s mechanism of action. This drug binds to the DNA of high turnover cells and its metabolite phosphoramide acts as a DNA alkylating agent, limiting replication and assisting lymphocyte apoptosis, with the consequent decrease in the number of these cells.45,46 We assumed that this could trigger negative feedback on survivin’s regulation, reducing its serum levels. The effect would predominate in lymphocytes, the main target cell of the treatment, due to its relatively low aldehyde dehydrogenase activity, the key enzyme that confers resistance to cyclophosphamide. 46
Signal transducer and activator transcription 3 (STAT3) is known to be overexpressed and hyperactivated in lymphocytes of SLE patients. 47 STAT3 pathways are activated by inflammatory cytokines receptors like IL-6 R and IFN-gamma and upregulate survivin expression. Immunosuppressive therapy such as cyclophosphamide, mycophenolate mofetil and calcineurin inhibitors, usually recommended for LN treatment, can suppress the IL-6-induced STAT3 phosphorylation and lead to apoptosis by reducing the transcription of survivin as well.48,49
It was noteworthy the slight quantitative difference between the serum values of survivin in active and inactive lupus groups. Nevertheless, the variation in the pre- and post-cyclophosphamide serum levels was significant. The sample sizes, aspects of the control group and the retrospective design expose some limitations of the current study. Future attempts to assess the role of survivin, its transcriptional variants in lymphocyte subgroups and the survivin-targeting miRNAs axis may clarify its correlation with the pathogenesis of SLE.
Conclusion
In this study, patients with systemic lupus erythematosus exhibited lower serum levels of survivin compared with healthy subjects, regardless of the renal involvement or the disease activity. The overall reduction of survivin after treatment, despite the clinical response, makes remote the possibility of survivin as a biomarker of response to cyclophosphamide in LN.
Supplemental Material
sj-pdf-1-bmi-10.1177_11772719221131470 – Supplemental material for The Behaviour of Serum Survivin in Patients With Lupus Nephritis
Supplemental material, sj-pdf-1-bmi-10.1177_11772719221131470 for The Behaviour of Serum Survivin in Patients With Lupus Nephritis by Renata Valente Lisboa, Fabiola Reis de Oliveira, Thaise Oliveira Quaresma, Rafael Moura de Almeida, Rene Donizeti Ribeiro Oliveira and Paulo Louzada Junior in Biomarker Insights
Footnotes
Acknowledgements
None.
Declarations
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
