Abstract
Keywords
Introduction
Systemic lupus erythematosus (SLE),a chronic autoimmune disease characterised by the production of auto-antibodies against nuclear and cytoplasmic antigens, shows diverse clinical manifestations, such as neuropsychiatric SLE, diffuse alveolar haemorrhage, mesenteric vasculitis, and lupus nephritis.1,2,3 The incidence and prevalence of SLE are affected by several factors and tend to vary worldwide, and the prevalence of SLE has been increasing in recent years.4,5
Following advances in laboratory testing methods and medical technology, the therapeutic approach to SLE has improved from simply administering hydroxychloroquine, systemic glucocorticosteroids and conventional immunosuppressants to the use of biologic agents as an option .6,7 However, many patients with SLE are unresponsive to standard therapy or show relapse after initial clinical remission, and management of such cases is therapeutically challenging.8,9 SLE disease activity index score (SLEDAI) is an effective model for the global assessment of SLE disease activity by experienced clinicians. It represents a consensus of experts in the field of lupus research. 10 Reportedly, bortezomib, a proteasome inhibitor, may reduce the SLEDAI score and may be useful in selected patients with SLE.11,12 A Segarra et al. have reported the possible role of bortezomib in refractory SLE, but they all included a small number of patients, and none of them was large-scale randomised controlled trials.18,19,20 To our knowledge, no meta-analysis has reported the efficacy of bortezomib in SLE. In this meta-analysis, we investigated the efficacy of bortezomib in patients with SLE.
Materials and methods
Search strategy and study selection
We searched several databases, including PubMed, Embase, Cochrane Library, China National Knowledge Infrastructure, and the Chinese BioMedical database, for articles published before 18 August 2021 to identify all studies relevant to SLE. We used the following keywords (we scrutinised the Mesh, Title, Abstract, and Keywords of all articles): ‘lupus erythematosus, systemic’,‘Systemic Lupus Erythematosus’,‘Lupus Erythematosus Disseminatus’,‘Libman-Sacks Disease’,‘Disease, Libman-Sacks’,‘Libman Sacks Disease’and bortezomib. The detailed search strategy is described in the Appendix.
Inclusion and exclusion criteria
Based on the existing literature suggesting that bortezomib may reduce SLEDAI scores in patients with SLE, the following were our inclusion criteria: (1) written in all languages, (2) bortezomib administration in patients with SLE and, (3) a clear description of SLE Disease Activity Index (SLEDAI) scores.
Following were the exclusion criteria: (1) duplicate publications (the most recent article was included for analysis)and, (2) animal experiments, case reports, meeting reports, and reviews.13,14
Two investigators (Huan He and Man Luo) independently searched the databases and reviewed all articles to analyse the data for methodological assessment. Disagreements between investigators were resolved through discussion and consultation with a third investigator (Jin-He Xiong).
Methodological assessment
We used the Delphi list for randomised controlled trials (RCTs) and the Quality Assessment of Diagnostic Accuracy Studies tool for methodological assessment of all included studies. 15 The following study characteristics were assessed: (1) nature of study (cohort vs RCT), (2) single versus multicentre study, (3) enrolment of consecutive patients versus random patient sample, (4) description of the inclusion/exclusion criteria and, (5) confirmation regarding representativeness of the study sample in real-life clinical practice, (6) the country, sample size, sample sex, sample age.
Statistical analysis
We calculated odds ratios (ORs) based on 2 × 2 tables to determine the efficacy of bortezomib in patients with SLE. We used the fixed- and random-effects models in cases with I2 < 25% and >25%, respectively. The results are presented as forest plots. A
Results
Search strategy and study selection
Our search of the aforementioned databases yielded 160 articles, of which only 42 articles were left for inclusion in the study after excluding duplicates, animal experiments, case reports, meeting reports and reviews. Following a thorough scrutiny of the abstract, nine articles were included, and full texts were retrieved for further selection. Only three articles matched our inclusion criteria because we could not extract data from the remaining six. Figure 1 shows the relevant data. The results of the search strategy, study selection and inclusion.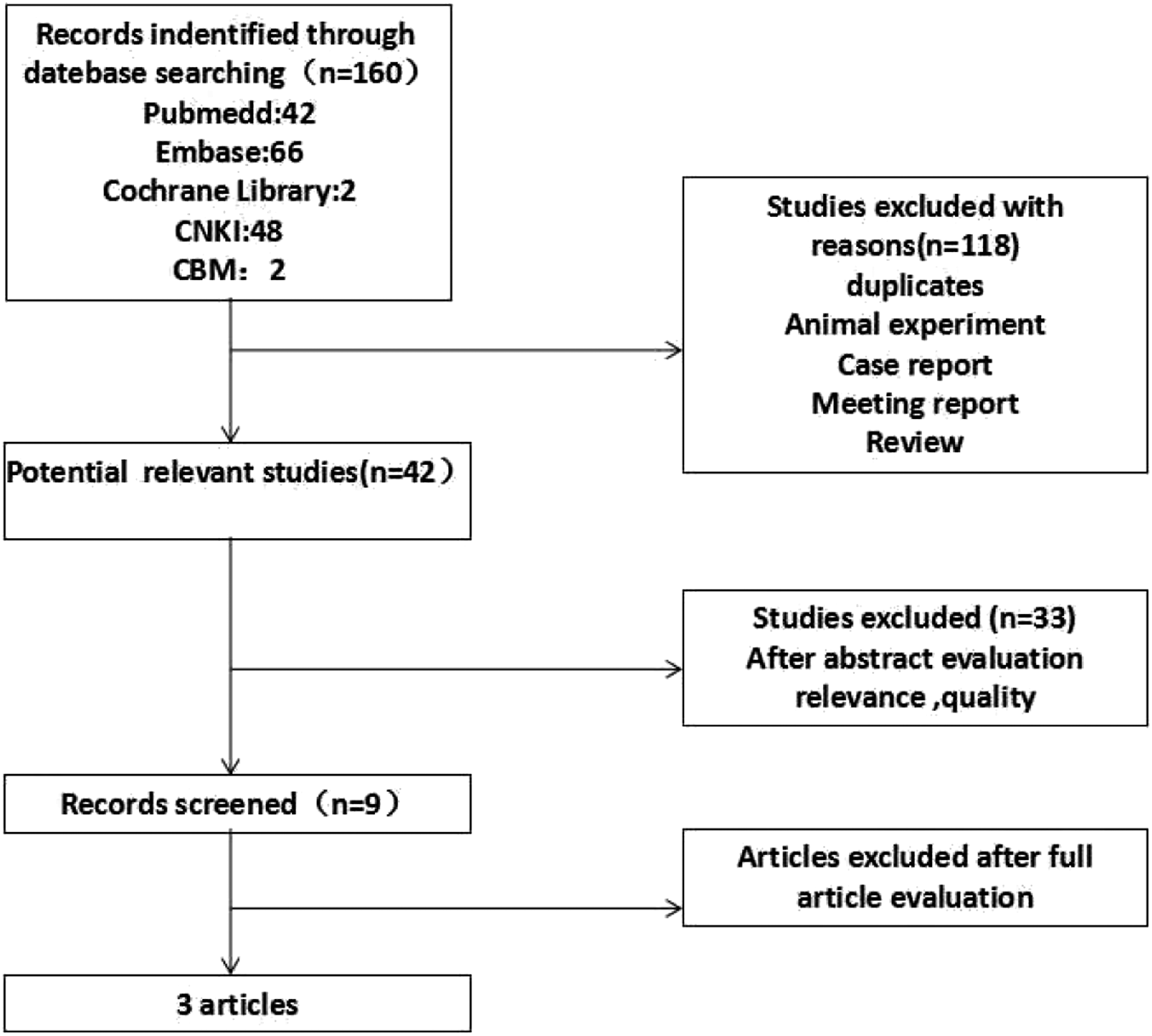
Methodological assessment

aThe study was a cohort or randomized controlled trial (RCT).
bThe study was a single centre or multicentre study.
cA consecutive or random sample of patients was enrolled.
dInclusion/exclusion criteria were specified.
eThe time period after bortezomib application.
fThe whole (or random sample) underwent follow-up for bortezomib application.
The efficacy of bortezomib
We indirectly investigated the efficacy of bortezomib in patients with SLE based on changes in SLEDAI scores before and after bortezomib administration. Unfortunately, not all nine papers provided complete data on changes in the SLEDAI score. We attempted to communicate with the authors of the aforementioned studies to obtain the relevant data; however, we were unsuccessful. Complete data for meta-analysis were available in only three papers. Therefore, we performed data analyses only if 2 × 2 data could be extracted from the studies.
We observed that the post-treatment SLEDAI scores were significantly lower than pre-treatment scores in patients with refractory SLE (OR 11.30, 95% confidence interval [CI]7.03–15.56, Changes of SLEDAI scores before and after bortezomib treatment.
Discussion
Our study showed statistically differences between the pre- and post-treatment SLEDAI scores in patients with SLE treated with bortezomib, which suggests that bortezomib may be a useful therapeutic option for refractory SLE.
Proteasomes are protein complexes that degrade proteins in prokaryotes and eukaryotes and are named based on their Svedberg. 21 As key players in the targeted degradation of cellular proteins, proteasomes serve as a therapeutic target for various diseases, including but not limited to several haematological malignancies. Proteasome-induced protein degradation requires ubiquitination; however, a portion of the protein may be independently degraded following direct interaction with the subunits at the core of the 20S proteasome.22,23,24 Bortezomib, also referred to as PS-341, is a dipeptidyl boronic acid proteasome inhibitor that reversibly binds to the CT-L subunit of the proteasome 25
Long-lived plasma cells (PCs) are resistant to conventional and B-cell-depleting therapies, which constitute a therapeutic challenge in cases of refractory SLE. 26 Bortezomib shows therapeutically relevant targeting of autoreactive PCs in patients with refractory SLE. Therefore, it is reasonable to conclude that bortezomib may serve as a promising therapeutic option in SLE refractory to conventional therapy. 27
Following are the limitations of our meta-analysis: (a) Owing to the small number of articles and patients included in the meta-analysis, the final results should be interpreted cautiously. The three articles analysed in our study only included patients with refractory SLE; therefore, the results of our meta-analysis may not be generalisable across patients with all types of SLE. (b) Owing to the small number of articles analysed, we did not investigate the efficacy of bortezomib in SLE based on changes in other indicators such as serum protein, serum creatinine, associated auto-antibody, and complement factor levels. (c) We did not consider the duration or dose of bortezomib administered because of unavailability of data. We did not perform subgroup analysis based on the administration of other drugs pre- and post-bortezomib treatment. (d) As only three articles were included in the final meta-analysis, we did not use a funnel plot to evaluate publication bias. The pooling of heterogeneous studies with regard to patient selection is another well-known limitation. For example, the definition of refractory lupus nephritis varies across articles.
Conclusion
Bortezomib may reduce SLEDAI scores in patients with refractory SLE, which suggests that this drug may serve as an effective therapeutic option in such cases. However, our meta-analysis included a small number of articles and patients. Therefore, large-scale RCTs are warranted to validate the results of our meta-analysis.
Supplemental Material
Supplemental Material - Efficacy of bortezomib in reduction of systemic lupus erythematosus disease activity index score: A meta-analysis
Supplemental Material for Efficacy of bortezomib in reduction of systemic lupus erythematosus disease activity index score: A meta-analysis by Huan He, Man Luo and Jin-He Xiong in European Journal of Inflammation
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
