Abstract
Introduction
Lupus nephritis (LN), a severe manifestation of systemic lupus erythematosus (SLE), is associated with high fatality rate in patients. The pathogenesis of lupus nephritis is complex and has not been fully elucidated. Kidney inflammation, renal cell damage, and accumulation of immune complexes in the glomerular basement membrane often occur in patients with lupus nephritis. Spermatid nuclear transition protein 1 (TNP1) might be a potentially interesting autoantigen in exploring the pathogenesis and therapy of lupus nephritis.
Objective
This study aimed to explore the effect of TNP1 and its complexes with anti-double-stranded DNA antibodies on the levels of interleukin-6 (IL-6) and interferon-α (IFN-α) in vitro.
Methods
We studied the effect of the synthetic peptide of the autoantigen on the pathogenic characteristics of the G2-6 and G5-8 antibodies in mouse macrophages, using enzyme-linked immunosorbent assay, quantitative RT-PCR, western blotting, and flow cytometry.
Results
The antibodies exhibited cross-reactivity to spermatid TNP1 in direct-binding and competitive enzyme-linked immunosorbent assay. Results of quantitative RT-PCR and western blotting revealed that the antibodies alone enhanced the levels of IL-6 and IFN-α transcripts and proteins, respectively. Flow cytometry revealed that treatment with the autoantigen enhanced the cell-penetrating activities of G2-6 and G5-8 and remarkably increased the cytokine levels.
Conclusion
TNP1 enhanced the cell-penetrating activities of anti-dsDNA auto-Abs, G2-6 and G5-8, and remarkably increased the levels of IL-6 and IFN-α in macrophages, suggesting that TNP1 and cell-penetrating pathogenic anti-dsDNA auto-Abs is potential targets for future therapeutic approaches to treat LN/SLE.
Keywords
Introduction
Lupus nephritis (LN) is a severe manifestation of systemic lupus erythematosus (SLE) affecting multiple organs, including the kidney. Production of auto-antibodies (auto-Abs) against double-stranded DNA (dsDNA) is a typical characteristic of LN/SLE; therefore, the auto-Abs are potential biomarkers of the disease.1–4
Several pathogenic anti-dsDNA auto-Abs possess cell-penetrating and mouse macrophage-binding as well as human mesangial cell-binding abilities.5–7 Anti-dsDNA auto-monoclonal Abs (auto-mAbs) such as G2-6, but not G5-8, exhibit cell-penetrating activities in mesangial cells 6 and the activities are characteristic of the presence of multiple positively charged amino acids in the complementarity-determining region (CDR3) of the variable region of the heavy (VH) chain.6,8 Anti-dsDNA mAbs with cell-penetrating activities are able to induce NF-κB activation; stimulate the secretion of pro-inflammatory cytokines, interleukin-6 (IL-6), interferon-α (IFN-α), tumor necrosis factor-α, IL-1β, IL-10, IL-33, and monocyte chemoattractant protein-1; and induce the activation of p38, RSK-1, Bcl-2, and ATF-2 molecules in mouse macrophages, mesangial cells, and human peripheral blood mononuclear cells.9–11
Serum levels of IL-6 and IFN-α have been implicated as important factors in the pathological process of LN/SLE. IL-6 and IFN-α correlate strongly with disease severity in patients as well as in murine models.12–16 Patients with SLE display overexpressed genes stimulated by type I IFNs, and the expression levels in the blood correlate with disease severity and active renal disease.17,18 Therefore, Abs against Type-1 IFN are a promising therapy for cutaneous lupus. 19 In LN/SLE and other autoimmune diseases, IFN-α shifts the balance between T-effector and T-regulatory cells in favor of T-effector cells by suppressing T-regulatory cell activation. 20 Moreover, other cytokines such as IL-8, IL-22, and monocyte chemoattractant protein-1 are implicated in the pathogenesis of LN/SLE.21,22
Spermatid nuclear transition protein 1 (TNP1), a positively charged protein consisting of 55 amino acids, was first reported as a novel autoantigen associated with LN by Onishi et al. 23 Here, we explored the possible role(s) of TNP1 in the pathogenesis of LN/SLE. We found that TNP1 plays an important role in inflammatory and pathogenic reactions mediated by G2-6 (highly penetrating auto-mAb in mesangial cells) and G5-8 (non-penetrating or low-penetrating auto-mAb in mesangial cells) in mouse macrophages. TNP1 enhanced the cell penetration efficiency of anti-dsDNA auto-mAb and increased the levels of transcripts and proteins of the pro-inflammatory cytokines, IFN-α and IL-6. Targeting pathogenic anti-dsDNA Abs and TNP1 in human could be a potential therapy for LN and SLE. This study aimed to explore the effect of the autoantigen, TNP1, and its complexes with anti-dsDNA auto-mAb on the levels of IL-6 and IFN-α in vitro.
Materials and methods
Cell line culture
The mouse macrophage cell line (RAW 264.7) was purchased from American Type Culture Collection (USA) and cultured in Dulbecco’s Modified Eagle Medium supplemented with 10% fetal bovine serum (FBS) and 1% penicillin/streptomycin at 37°C in a humidified incubator (Forma Scientific, USA) supplied with 5% CO2.
Anti-dsDNA mAbs
We previously produced mAb-secreting hybridoma cell lines, G2-6, and G5-8, from MRL-lpr/lpr mice.24,25 G2-6 and G5-8 IgGs (IgG2b, λ) were purified from hybridoma culture supernatants using affinity chromatography with a HiTrap IgG Sepharose 6FF or HisTrap FF column installed in purification module AKTA (GE Healthcare, USA) following the manufacturer’s instructions.
Spermatid TNP1-peptide
Intact mouse TNP1 comprises 55 amino acids (sequence number NP_033433 from NCBI -https://www.ncbi.nlm.nih.gov/protein/). The TNP1-peptide (1tsrklkthgm rrgknrap hk gvkrggskrk yrksvlksrk rgddasrnyr50), which is five amino acids (two of N-terminal and three of C-terminal) shorter, with a molecular weight of 6 kDa was synthesized and purified using high-performance liquid chromatography with >90% purity by PEPTRON Biotech (South Korea).
TNP1 direct-binding enzyme-linked immunosorbent assay (ELISA)
The wells of 96-well ELISA plates (Nunc, Denmark) were coated with TNP1 (1 μg/mL) and incubated overnight at 4°C. Wells of the plates were blocked with 3% milk in phosphate-buffered saline with Tween 20 (PBS-T), followed by washing with PBS-T. Serially diluted (1:3) G2-6, G5-8, or normal mouse IgG (mIgG) were incubated in the wells for 2 h at 20°C. The binding of mAb with dsDNA was detected using a secondary antibody, horseradish peroxidase-conjugated goat anti-mouse IgG. A 3,3′,5,5′-tetramethylbenzidine (TMB) substrate was used for color development, and H2SO4 (2M) was used to quench the reaction. Absorbance at 450 nm was measured using a microplate reader (Bio-Rad, USA).
TNP1 competitive ELISA
ELISA plates were coated with TNP1 (1 μg/mL) and incubated overnight at 4°C. The wells of the plate were blocked with 3% milk in PBS-T. Various concentrations of TNP1 and mAb (10 μg/mL) were pre-incubated for 1 h at 4°C; the mixture was added to the TNP1-coated wells after washing the wells. Similar detection methods were used as in the direct-binding ELISA method.
Flow cytometry
Cells were grown to confluency and trypsinized using trypsin (0.05%) and Ethylenediaminetetraacetic acid (EDTA)(0.02%), and the reaction was stopped by adding the equivalent of two volumes of pre-warmed complete growth medium. The pellets were re-suspended in 5% FBS in phosphate-buffered saline (PBS) and treated with mAb or mAb+TNP1 complex (pre-incubated for 1 h) for 1.5 h at 37°C, followed by washing with PBS to remove the unbound mAb and subsequently fixed with paraformaldehyde (4%). To detect the penetration efficiency, cells were permeabilized with Triton-X 100 (0.1%) for 5 min, washed with PBS, and incubated with fluorescein isothiocyanate-conjugated anti-mouse goat IgG (BD Pharmingen, USA) for 1 h. Normal polyclonal mIgG was used as an isotype control, and only secondary Ab treatment was used as a negative control. Finally, the cells were washed with cold PBS. To analyze the effects of TNP1 on the cell penetration efficiency of mAb, analysis was performed with 10,000 cell counts on respective samples using a FACSCanto II (BD Biosciences, USA) and FlowJo-7 software.
Western blotting
Cells grown on the wells of 12-well plates were treated for 3 h with mAb (50 μg/mL), TNP1 (3 μg/mL) or mAb-TNP1 complex (1 h pre-incubated mixture), and the supernatants were collected. The cell pellets were treated using lysis buffer (25 mM Tris-HCl pH 7.6, 150 mM NaCl, 1% NP-40, 1% sodium deoxycholate, 0.1% SDS) and the lysates collected. SDS-PAGE gels (12%) were used to separate the proteins from the supernatant and lysate. The proteins were transferred on a polyvinylidene fluoride membrane. Respective bands were detected using anti-IL-6/IFN-α/α-tubulin as the primary antibody, HRP-conjugated anti-rabbit goat IgG as the secondary antibody, and the chemiluminescence substrate.
Total RNA extraction, cDNA synthesis, and quantitative real-time PCR (qRT-PCR)
Cells were grown to confluence in a 60-mm Petri dish and stimulated with mIgG, TNP1, mAb, or mAb-TNP1 complex (pre-incubated for 1 h). Cell pellets were obtained to extract total RNA and synthesize cDNA. MiniBEST universal RNA extraction kit and PrimeScript™ 1st strand cDNA synthesis kit (Takara, Japan) were used for RNA extraction and cDNA synthesis, and instructions were followed as described in the manufacturer’s manuals (Takara, Japan). RNA samples were quantified using a Nanodrop Lite spectrophotometer (Thermo Fisher Scientific, South Korea). DNA was amplified using a PCR machine (C1000 Touch Thermal Cycler, Bio-Rad, USA); qRT-PCR was performed to determine the relative change in mRNA cytokine expression levels with respect to GAPDH as an internal control using cytokine primers and kits (Takara, Japan). Amplification was performed using the ‘7500 Real Time PCR System’. Analysis was conducted using the
IL-6
Forward: 5′- CCA GAA ACC GCT ATG AAG TTC C -3′
Reverse: 5′- TCA CCA GCA TCA GTC CCA AG -3′
IFN-α
Forward: 5 ′ - TTT CCC CTG ACC CAG GAA GAT G -3′
Reverse: 5′- CTC TCA GTC TTC CCA GCA CAT T -3′
GAPDH
Forward: 5′- TGT CCG TCG TGG ATC TGA C -3′
Reverse: 5′- CCT GCT TCA CCA CCT TCT TG -3′
Statistical analysis
The results are presented as means ± standard deviations. Student’s t-test tool within the Microsoft Excel Software was used for Statistical Data Analysis, and the p values obtained were assigned as follows: not significant (NS), p > .05, *p < .05, **p < .005, and ***p < .001.
Results
The characteristics of anti-dsDNA auto-mAbs, G2-6 and G5-8, with respect to their pathogenic roles in vitro are quite different in mesangial cells. We analyzed the properties of G2-6 and G5-8, as well as the role of autoantigen TNP1 in RAW 264.7 macrophages treated with the mAbs.
Autoantigen TNP1 binds specifically to G2-6 and G5-8
First, direct-binding ELISA was performed to determine the affinity of purified G2-6 and G5-8 for TNP1. G2-6 and G5-8 bound to TNP1 in a dose-dependent manner with mAbs concentrations ranging from 0.04 to 10 μg/mL (Figure 1(a)). The full-length recombinant TNP1 protein could bind to the mAbs in a similar way (data not shown). G5-8 had a higher affinity for TNP1 than G2-6. Significant non-specific binding affinity was not observed for the mIgG-negative control. Binding of TNP1 to each Ab was confirmed using inhibition ELISA. Pre-incubation of serially diluted TNP1 (0.625–10 μg/mL) with 10 μg/mL G2-6 or G5-8 specifically inhibited the binding of mAb to TNP1 coated on the bottom of plate wells in a dose-dependent manner (Figure 1(b)). Specific binding of G5-8 was higher than that of G2-6 in the TNP1 range of 2.5–10 μg/mL. TNP1 direct-binding (a) and competitive enzyme-linked immunosorbent assay (ELISA) (b) of anti-double stranded DNA (dsDNA) Abs. (a) Wells of enzyme-linked immunosorbent assay (ELISA) plates coated with TNP1 (1 μg/mL). Serially diluted (1:3) G2-6, G5-8, or normal mouse IgGs were incubated with the coated TNP1. Mouse IgG was used as a negative control. (b) The binding of G2-6 or G5-8 was inhibited using various concentrations of TNP1. Various concentrations of competitor, TNP1, were incubated with 10 μg/mL of G2-6 or G5-8 in solution and added to the wells of the ELISA plates coated with TNP1 (1 μg/mL). The results shown are the average of triplicate measurements. The t-test was performed to determine the respective p values (*p < .05, **p < .005, ***p < .001 and NS, p > .05). TNP1 = Spermatid nuclear transition protein 1.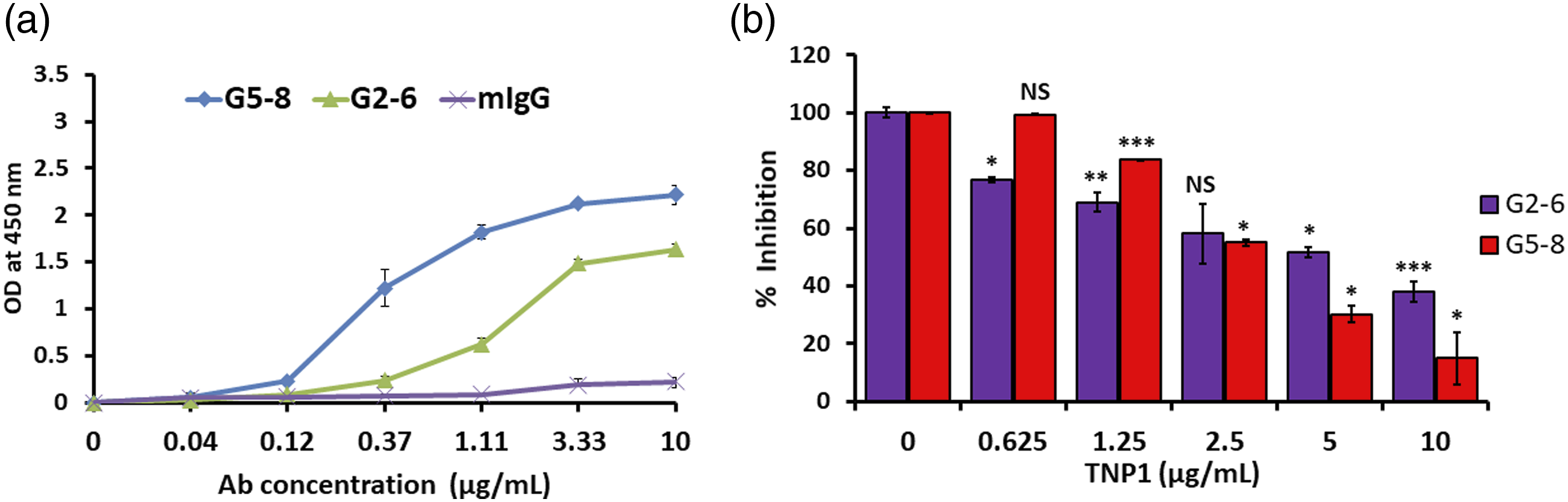
TNP1 enhanced cell-penetrating activities of G2-6 and G5-8 in macrophages
We performed flow cytometry analysis to determine the effect of TNP1 on the penetrating activities of G2-6 and G5-8 into macrophages at least three times (Figure 2). The level of cell penetration by G2-6 without TNP1 (63.9 ± 3.2%) was similar to that previously reported for mesangial cells.
6
G5-8, however, showed a much higher level of penetration (41.6 ± 0.9%) in macrophages than the activity previously observed in mesangial cells,
6
where the level was extremely low. Pre-incubation of G2-6 or G5-8 with TNP1 enhanced the cell-penetrating activity of each mAb. An increase in the concentration of TNP1 increased the penetrating ability of the mAb. Compared to G2-6 only, G2-6 with 1 and 10 μg/mL of TNP1 enhanced the penetration level by 33.4 ± 1.9% (p < .005) and 35.7±1.5% (p < .05), respectively. Similarly, TNP1 (1 and 10 μg/mL) increased the penetration efficiency of G5-8 by 11.5±1.1% (p < .001) and 31.1 ± 3.1% (p < .005), respectively, compared to G5-8 only. Interestingly, enhanced cell penetration of G2-6 by TNP1 was not observed in mesangial cells (data not shown). The cell penetration efficiency of Abs and the enhancement of cell penetration by TNP1 may differ depending on the type and origin of the cells. Analysis of the penetration efficiency of anti-double stranded DNA (dsDNA) Abs, G2-6 (a) and G5-8 (b) in RAW 264.7 cells by flow cytometry. Cells were incubated with 25–50 μg/mL of Ab alone (G2-6 or G5-8) or Ab-TNP1 complex pre-incubated for 1 h at 37°C for 1.5 h. Two different concentrations of TNP1 (1 and 10 μg/mL) were used. Fluorescein isothiocyanate (FITC)-conjugated anti-mouse goat IgG (10 μg/mL) was used as the detecting Ab. Mouse IgG was used as γ-isotype control, and only the secondary Ab was treated as a negative control. The results are the representative data of at least three different experiments. Statistical numbers including analysis of t-test are presented in “Results” section of the text. TNP1 = Spermatid nuclear transition protein 1.
Complexes of TNP1 and G2-6 or G5-8 enhance transcription and protein expression of IFN-α and IL-6 in macrophages
We analyzed the effect of TNP1 on the levels of pro-inflammatory cytokines, IL-6 and IFN-α, at mRNA and protein levels in macrophages treated with G2-6 or G5-8.
The cytokine transcripts produced by G2-6 or G5-8 alone were measured for 3 h. G2-6 stimulated the production of IL-6 transcripts at a high level at 0.5 h (∼18-fold compared with the GAPDH control), at 1.5 h (∼17-fold), and the level subsequently declined at 3 h (∼3-fold) (Figure 3(a)). The transcript level of G2-6 was higher in IFN-α than in IL-6. Compared with the control, the transcript level at 0.5–1.5 h was approximately 23–25-fold and was reduced ∼2-fold at 3 h) (Figure 3(b)). Furthermore, G5-8 enhanced the transcript levels but much less than G2-6 (Figures 3(c) and (d)); the transcript levels of IL-6 and IFN-α were increased ∼6-fold at 0.5 h and decreased 2–3-folds at 1.5–3 h. Analysis of transcripts of pro-inflammatory cytokines, IL-6 and IFN-α, in RAW 264.7 cells stimulated by G2-6 or G5-8, using quantitative real-time PCR. G2-6 or G5-8 of 50 μg/mL were treated at varying times (0, 0.5, 1.5, and 3 h/s), and the measured levels were normalized by those of glyceraldehyde 3-phosphate dehydrogenase (GAPDH). (a) IL-6 expression after stimulation by G2-6. (b) IFN-α expression by G2-6. (c) IL-6 expression by G5-8. d) IFN-α expression by G5-8. The experiments were performed at least thrice, and the t-test was performed to determine the respective p values (*p < .05, **p < .005, ***p < .001 and NS, p > .05).
The effects of TNP1 on cytokine transcript levels were analyzed. When cells were pre-treated with TNP1 for 1 h, followed by treatment with G2-6 or G5-8 for 3 h, the expression of IFN-α increased remarkably (Figure 4). The transcript level of IL-6 was enhanced 8–9-fold by the addition of TNP1 with the treatment of G2-6 (Figure 4(a)) or G5-8 (Figure 4(c)). The level of IFN-α in cells stimulated by G2-6 or G5-8 was enhanced ∼51–53-fold by the addition of TNP1 (Figures 4(b) and (d)). Additionally, the effects of TNP1 on cytokine stimulation were analyzed at the protein level by western blotting (Figure 5). IL-6 and IFN-α were detected in the supernatant, but not in the cell lysate (data not shown), suggesting the extracellular secretion of the cytokines soon after translation by macrophages stimulated by G2-6 and G5-8 as one of the possible reasons. α-tubulin from cell lysates was analyzed as a loading control. Addition of TNP1 increased the protein levels of IFN-α and IL-6, although the increased levels were relatively low compared to the transcript levels. The effect of TNP1 was more distinct in G5-8 than in G2-6. Cells treated by TNP1 alone tended to stimulate IL-6 expression to some extent. Figure 5 presents the supportive results of Figure 4, although the statistical analysis to compare the protein levels of IL-6 and IFN-α among different groups is not shown. Figures 4 and 5 suggest that complexes of TNP1 and G2-6 or G5-8 enhance transcription and protein expression of IFN-α and IL-6 in macrophages. Analysis of transcripts of pro-inflammatory cytokines, IL-6 and IFN-α, in RAW 264.7 cells stimulated by G2-6 or G5-8 with and without TNP1 using quantitative real-time PCR. Treatment with TNP1 (3.3 μg/mL) and 50 μg/mL of G2-6 or G5-8 for 3 h. For the complexes (G2-6/G5-8-TNP1), TNP1 and G2-6 or G5-8 were pre-incubated for 1 h. The results were normalized with those of glyceraldehyde 3-phosphate dehydrogenase (GAPDH). Untreated cells were used as control to determine the significance. (a) IL-6 expression levels stimulated by G2-6, TNP1-peptide, and its complex. (b) IFN-α expression levels stimulated by G2-6, TNP1-peptide, and its complex. (c) IL-6 expression levels stimulated by G5-8, TNP1-peptide, and its complex. (d) IFN-α expression levels stimulated by G5-8, TNP1-peptide, and its complex. The experiments were performed at least thrice, and the t-test was performed to determine the respective p values (*p < .05, **p < .005, ***p < .001 and NS, p > .05). Analysis of pro-inflammatory cytokines, IL-6 and IFN-α, in RAW 264.7 cells stimulated by G2-6 or G5-8 with and without TNP1, using western blotting. Cells were treated with 50 μg/mL mAb, 3 μg/mL TNP1, or mAb-TNP1 complex (1 h pre-incubated mixture) for 3 h. Culture supernatant or cell lysates in radioimmunoprecipitation assay (RIPA) buffer were loaded in 12% SDS-PAGE. After transferring the proteins on a membrane, IL-6 (a) or IFN-α (b) and α-tubulin were detected using specific antibodies and the secondary antibody. The results are the representative data of at least three different experiments. TNP1 = Spermatid nuclear transition protein 1.

Discussion
Anti-dsDNA Abs are able to penetrate several different cell types, including cancer cell lines and normal cells, and trigger the activation of pro-inflammatory cytokine production and signaling molecules.6,9–11 Here, we demonstrate that the LN autoantigen TNP1 could amplify the inflammatory reaction in macrophages. TNP1 enhanced the levels of IFN-α and IL-6 triggered by anti-dsDNA auto-mAbs, G2-6 and G5-8, in a macrophage cell line, suggesting a possible involvement of the anti-dsDNA-TNP1 complex in the provocation of the inflammatory reaction. We further showed that the degree of cell-penetrating activity of anti-dsDNA auto-mAbs could differ depending on the cell type.
Renal mesangial, epithelial, endothelial cells as well as macrophages are critical targets for renal damage in LN/SLE.26,27 Previously, we published evidence indicating that G2-6, which has multiple arginine residues in CDR3-VH, is an anti-dsDNA auto-mAb with pathogenic properties in SLE and exhibits efficient cell-penetrating activity in renal mesangial cells, SV40 MES. 6 In contrast, G5-8, which has a single arginine residue in CDR3-VH, displayed either non-penetrating or penetrating properties with very low efficiency in SV40 MES cells. 6 Here, we report that G2-6 could efficiently penetrate RAW 264.7 macrophages. Moreover, it was observed that G2-6 effectively penetrated the kidney epithelial cells (Renca) (data not shown). Interestingly, G5-8 could penetrate RAW 264.7 cells more effectively than it could penetrate SV40 MES cells. Therefore, it suggests that the extent of the pathogenic reaction triggered by cell-penetrating Abs could vary depending on the cell type that is involved with LN or SLE. Macrophages might be profoundly involved in the pathogenesis of SLE or LN triggered by pathogenic anti-dsDNA mAb at the same level or to a greater extent than kidney-resident cells in certain environments. Treatment of macrophages with a pre-incubated complex of the auto-mAb and TNP1 enhanced the cell penetration ability, suggesting the evoking role of TNP1 in the pathogenesis of anti-dsDNA auto-mAbs.
Auto-mAb G2-6 with its efficient cell-penetrating mechanism effectively stimulated IL-6 and IFN-α transcription. With the addition of TNP1, stimulation of their transcription by G2-6 was remarkably enhanced, particularly that of IFN-α. The low-penetrating G5-8 Ab by itself did not remarkably stimulate IL-6 and IFN-α transcription, but in the presence of TNP1, there was a tremendous increase in the stimulation level, particularly for IFN-α. TNP1 stimulated the transcription of IL-6 at low levels. TNP1 could amplify the inflammatory effects exerted by G2-6 in macrophages. Although the inflammatory reaction was low in cells treated with G5-8 alone, there was a remarkable increase in the presence of TNP1. The increase of IFN-α transcription by G5-8-TNP1 complex was observed to some extent, although stimulation of IL-6 and IFN-α and their enhancements by TNP1 at protein levels were not significant. Furthermore, we showed the cross-reaction of G2-6 and G5-8 anti-dsDNA auto-Abs to TNP1 autoantigen, in contrast to the non-binding of anti-TNP1 Ab to DNA. 23 The specific binding of G2-6 or G5-8 to TNP1 with high affinity could possibly be one of the important factors leading to the upregulated pro-inflammatory reaction observed in macrophages.
The importance of IL-6 and/or type I IFN has been frequently highlighted in the pathogenesis and therapeutic targets of LN/SLE in human and murine models. IL-6 production within the glomerular basal membrane is specific to particular cell types, such as the macrophages in a mouse model with chronic LN. Preventing IL-6 activity could therefore be beneficial for LN. 28 IL-6 is involved in mesangium pathological irregularities and is implicated in LN conditions; it is, therefore, a reliable therapeutic target. 29 Type I IFN could be produced by resident kidney cells, epithelial cells, and mesangial cells, and mediates the pathogenesis of SLE in patients.30,31 Auto-Abs, including anti-dsDNA, are strongly associated with type I IFN in LN/SLE patients.32,33 IFN-α alone is sufficient to trigger SLE in non-autoimmune-prone mice, and sustained SLE is caused by type I IFN.34,35 IFN-α is one of the factors that may predict SLE activity in patients. 36
In LN/SLE, auto-Ab production, inflammation, and organ damage resulted from immune dysregulation caused by the interaction of various factors and the breakdown of tolerance towards self-antigens. Induction of inflammation by a subset of anti-DNA auto-Abs, including cell-penetrating Abs could be in the pathogenesis of SLE and LN. We demonstrate that inflammation caused by anti-dsDNA auto-Abs can be exaggerated by the enhancing role of TNP1, supporting the theory of the pathogenic role of auto-Abs and immune complexes (ICs).6,9,37 The ICs of pathogenic and inflammatory anti-dsDNA auto-Ab with TNP1 autoantigen may deposit on the glomerulus and may penetrate renal resident cells and macrophages. The IC could possibly display enhanced penetration activity and increase the level of pro-inflammatory cytokines through gene expression. Further, the IC may activate the complement cascade in kidney parenchymal cells and be involved in inducing the local production of chemokines, the recruiting of inflammatory cells, and T- and B-cells, as suggested in previous reports.17,38
Our study has some limitations such as a preliminary data in vitro without animal experiments and extensive analysis of various cytokines. Further in vitro and in vivo studies need to be conducted to explore more clearly the mechanism(s) of action underlying the activity of anti-dsDNA auto-Ab-TNP1 complex in macrophage and kidney-resident cells.
Conclusion
The results of the present study showed that TNP1 enhanced the cell-penetrating activities of anti-dsDNA auto-Abs, G2-6 and G5-8, and remarkably increased the levels of IL-6 and IFN-α in macrophages. Our results suggest that the autoantigen TNP1 and cell-penetrating pathogenic anti-dsDNA auto-Abs might be potential targets for promising future therapeutic approaches to treat LN/SLE.
Footnotes
Acknowledgements
Author Contributions
Y-JJ contributed in the study design, interpretation of results, and writing manuscript. LPU and YHP conducted experiments and participated in interpretation of results. All authors reviewed and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea [grant number NRF-2017R1D1A1B03031689].
Ethics approval
Ethics approval was waived by Institutional Review Board of Ajou Medical Center. The waiver number is AJIRB-BMR-EXP-22-203.
