Abstract
Aims:
This study investigated the ability of soluble platelet selectin (sP-selectin) to identify infection and predict 30-day mortality in patients with a systemic inflammatory response syndrome (SIRS) on the intensive care unit.
Methods:
Soluble platelet selectin levels were measured daily in the first 48 hours in patients presenting with SIRS. The outcome, proven infection, was established using predefined definitions. The 30-day mortality was retrospectively assessed.
Results:
In a total of 313 patients with SIRS, sP-selectin levels were measured. Of these, 114 patients had proven infection on admission or developing during their intensive care unit (ICU) stay. Patients with proven infection had moderately higher levels of sP-selectin (147 ng/mL; interquartile range [IQR], 93.4-203 ng/mL) compared with noninfected patients (143.8 ng/mL; IQR, 89.6-194.7 ng/mL). This difference was not statistically significant (P = .072). However, in patients who were not admitted for infection (n = 235), sP-selectin levels were significantly related to the subsequent development of infection (P = .013). Soluble platelet selectin levels were particularly high in patients with abdominal sepsis and skin infections. Higher sP-selectin levels were associated with higher mortality (although not statistically significant, P = .08).
Conclusions:
This study shows that in patients with SIRS not admitted for infection, sP-selectin levels are significantly related to the subsequent development of infection. Furthermore, patients with higher sP-selectin levels in the first 2 days of admission had higher 30-day mortality, although this association is not statistically significant. Therefore, we conclude that sP-selectin is a potential future biomarker for both mortality and infection in patients with SIRS, but more research is needed to confirm its prognostic role.
Introduction
Patients react to an inflammatory stimulus with a variety of proinflammatory mediators. This phenotypically results in a clinical syndrome, characterized by fever, tachycardia, tachypnea, and leukocytosis: the so-called systemic inflammatory response syndrome (SIRS). SIRS is the final common pathway of inflammation. It is very difficult to distinguish SIRS caused by inflammation from SIRS resulting from true (bacterial) infection. 1 Sepsis is defined as SIRS caused by a true (bacterial) infection and is a life-threatening condition that requires antimicrobial chemotherapy immediately. 2 However, if SIRS results from noninfectious inflammation, antimicrobial therapy will not be beneficial and may even lead to an increase in resistant pathogens. Therefore, it is important to find tools that help discern infection from solely inflammation. 3
Endothelial activation is one of the hallmarks of SIRS (and therefore sepsis). The activated endothelium triggers leukocytes and platelets, which will subsequently express cell adhesion molecules on their surface. These cell adhesion molecules are transported to the cell surface in granules (the α-granules of platelets and the Weibel-Palade bodies of endothelial cells). One of the cell adhesion molecules is platelet selectin (P-selectin). P-selectin is vital for the innate immune system. It is present on the surface of the endothelium and platelets, where it decelerates the rolling of leukocytes and enables them to transmigrate through the blood vessel to the site of infection or inflammation.4,5 Soluble platelet selectin (sP-selectin) is the soluble form of P-selectin, which is released by the granules and can be measured in plasma.
Platelets and endothelial cells are one of the first responding cells during the development of sepsis.6,7 Activated platelets and endothelial cells release sP-selectin. Consequently, sP-selectin has been proposed as a useful biomarker for sepsis. 8 However, few studies have been conducted on the use of sP-selectin as a biomarker for sepsis. The studies that have been carried out show contradicting results (depending on the study design and investigated patient population).8–10
We hypothesized that sP-selectin is elevated in patients with a true (bacterial) infection compared with patients without infection (or rather, solely inflammatory SIRS). The aim of this study was to evaluate the ability of sP-selectin to identify infection in a mixed cohort of intensive care unit (ICU) patients who presented with SIRS.
Materials and Methods
Patient selection and exclusion criteria
The study design has been described previously.11,12 In short, a total of 313 consecutive patients with an anticipated ICU stay of >24 hours with at least 2 SIRS criteria at admission to the ICU were included. The SIRS criteria are temperature >38°C or <36.0°C, leukocyte count >12 × 109 L−1, tachypnea (respiratory rate >20/min or P
Blood collection and measurement of sP-selectin
Blood was sampled the first day (after establishing the presence of SIRS) and thereafter daily until discharge or up until day 10, whichever came first. The levels of sP-selectin were analyzed with an enzyme-linked immune sorbent assay (ELISA) with a TECAN Freedom EVO robot (Tecan, Männedorf, Switzerland). For the ELISA assays, 3 antibodies were used: R&D Mouse antihuman P-selectin (coating antibody), a biotin-labeled R&D sheep antihuman P-selectin, and a horseradish peroxidase–coupled DAKO streptavidin. Relative light units from horseradish peroxidase activity were measured with a SuperSignal West Pico Chemiluminescent Substrate by SpectraMax L Luminescence Microplate Reader. C-reactive protein (CRP), leukocytes, creatinine, and other blood values were measured as part of daily clinical routine. CRP was analyzed in heparin plasma on a DxC 800 routine chemistry system (Beckman Coulter, Brea, CA, USA), and leukocytes were counted in EDTA blood on the CD-Sapphire routine hematology analyzer (Abbott Diagnostics, Santa Clara, CA, USA).
Definitions
The outcome “proven infection” was derived from the algorithms for proven infection of health care–associated infection from the Centers for Disease Control and Prevention (CDC). 14 This was recorded during the ICU stay. “Proven sepsis” was defined as more than 2 SIRS criteria (as described above) in combination with a proven infection. The infection site was defined as described in the CDC 2008 criteria. 14 Mortality was retrospectively added by searching the hospital database; there were no missing values.
Statistical analyses
Statistical analyses were performed with SPSS 21.0 (SPSS Inc., Armonk, NY, USA). Two sP-selectin variables have been computed. As a predictor of outcome, we used the maximum value of the first 2 samples of sP-selectin. As a correlator, the maximum value of sP-selectin was used (during admission on ICU). Both were assessed by the Shapiro-Wilk and Kolmogorov-Smirnov d statistic on normality. Group comparisons were made by 2-sided t test or Mann-Whitney U test, depending on the normality distribution.
sP-selectin and outcome
Cumulative survival was visualized by a Kaplan-Meier graph. The population was divided into 2 percentiles based on the maximum sP-selectin level of the first 2 days (the first 2 samples). A logistic regression, Cox regression analyses, and receiver operating characteristic (ROC) curve analyses were performed to investigate the discriminative value of sP-selectin on mortality and the additional value to the Acute Physiology and Chronic Health Evaluation IV (APACHE IV) score.
sP-selectin and (proven) infection
The relationship between sP-selectin and infection was investigated using the Mann-Whitney U test, both in the whole population and in the various subgroups. A 2-tailed P < .05 was considered statistically significant. A linear regression was conducted to compare sP-selectin with CRP.
Ethical approval
The study methods were submitted to and approved by the institutional review board of the University Medical Center Utrecht, and it waived the need for informed consent (University Medical Center Utrecht Institutional Review Board research protocol 108-188). The study was conducted in accordance with the Declaration of Helsinki.
Results
Baseline characteristics
A total of 313 consecutive patients were included in the study, of which 195 (62.3%) were men (Table 1). The median age was 61 years (interquartile range [IQR], 48-72 years), and 77 patients (24.3%) died within 30 days after inclusion. There were no missing data regarding sP-selectin, 30-day mortality, or infection status. On average, the patients stayed in the ICU for 14 days (median, 9 days; IQR, 5-15 days), giving a total of 2280 samples.
Demographic and clinical baseline characteristics.
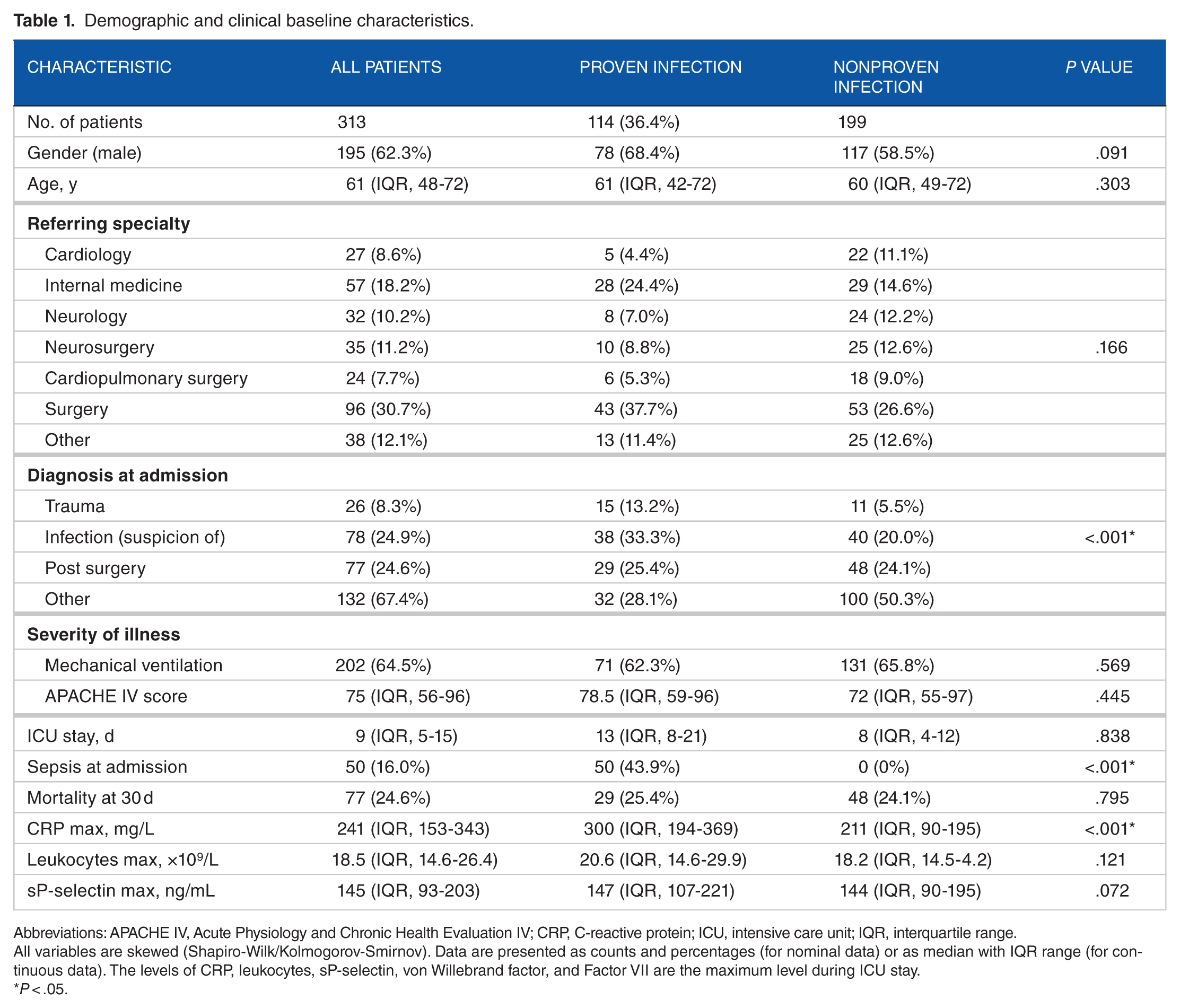
Abbreviations: APACHE IV, Acute Physiology and Chronic Health Evaluation IV; CRP, C-reactive protein; ICU, intensive care unit; IQR, interquartile range.
All variables are skewed (Shapiro-Wilk/Kolmogorov-Smirnov). Data are presented as counts and percentages (for nominal data) or as median with IQR range (for continuous data). The levels of CRP, leukocytes, sP-selectin, von Willebrand factor, and Factor VII are the maximum level during ICU stay.
P < .05.
Mortality
Mortality was comparable between patients who were and were not infected during their ICU stay (see Table 1). Mortality in patients with proven infection was 25.4% compared with 24.1% in the noninfected patient group (P = .795). Severity of illness was comparable between those 2 groups—APACHE IV: 78.5 (IQR, 58.75-96) (proven infection) versus 72 (IQR, 54.5-97) (no infection), P = .445. C-reactive protein was significantly higher in the proven infection group—299.5 mg/L (IQR, 194.3-368.5 mg/L) (proven infection) versus 211 mg/L (IQR, 89.7-194.7 mg/L) (no infection), P < .001. Leukocyte levels did not differ significantly in the 2 groups—20.6 × 109/L (IQR, 14.6-29.9 × 109/L) (proven infection) versus 18.2 × 109/L (IQR, 14.5-24.2 × 109/L) (no infection), P = .121.
sP-selectin and infection
Of the total 313 patients, 50 had a sepsis at admission (16.0%) and 64 developed an infection during ICU stay, which totals 114 patients (36.4%). The infection sites of the 114 patients are shown in Table 2. Patients who did have SIRS but did not develop a “proven infection” had lower, nonsignificant, median sP-selectin levels compared with patients who did—147 ng/mL (IQR, 93.4-203 ng/mL) (proven infection) versus 143.8 ng/mL (IQR, 89.6-194.7 ng/mL) (no infection), P = .072. However, in patients who were not admitted for infection (n = 235), sP-selectin levels were significantly related to the subsequent development of infection (P = .013; Figure 1). Skin infections and abdominal sepsis had significantly higher sP-selectin levels in comparison with patients without a skin infection/abdominal sepsis (189 ng/mL [IQR, 118-258 ng/mL] vs 143 ng/mL [IQR, 91-200 ng/mL], P < .05, and 229 ng/mL [IQR, 176-344 ng/mL] vs 142 ng/mL [IQR, 91-200 ng/mL], P = .002) with an area under the curve (AUC) of 0.633 (confidence interval [CI], 0.518-0.748) (skin infections) and 0.778 (CI, 0.614-0.942) (abdominal sepsis).
Sites of infection in patients with proven infection defined by the CDC 2008 criteria.

Abbreviations: CDC, Centers for Disease Control and Prevention; IQR, interquartile range; sP, soluble platelet.
Cumulatively there were 126 infections in 114 patients with “proven infection.”
In patients with no infection on the particular infection site.
P < .05.

Boxplot of difference in the development of proven infection in high and low sP-selectin levels: patients not suspected of infection at admission (the dotted lines represent the mean value). Mann-Whitney U is significantly different with a P value of .034. The dots represent outliers.
The characteristics of sP-selectin to predict “proven sepsis” were comparable to that of CRP. The AUC was 0.603 (CI, 0.514-0.691) for sP-selectin and 0.611 (CI, 0.522-0.700, P = .90) for CRP. In a subpopulation of patients with an abdominal sepsis (n = 11), the AUC was 0.778 (CI, 0.614-0.942) for sP-selectin and 0.615 (CI, 0.435-0.795) for CRP (P = .189).
sP-selectin and mortality
The patients were divided into 2 equal-sized groups based on their sP-selectin level. We observed that patients with a higher sP-selectin level in the first 2 days of admission (sP-selectin >104 µg/mL) had a worse survival than patients with a lower sP-selectin level (P = .079; Figure 2), which was not statistically significant. The hazard ratio of sP-selectin >104 µg/mL compared with a lower sP-selectin level was 1.5 (CI, 0.95-2.35, P = .083). The AUC of sP-selectin for 30-day mortality was 0.547 (CI, 0.471-0.624). We created a logistic regression model combining sP-selectin levels and the APACHE IV score. When comparing the ROC curve of APACHE IV alone to the curve of APACHE IV combined with the sP-selectin levels, we found no significant difference in the AUC (0.777 vs 0.784, P = .859) for 30-day mortality.

Kaplan-Meier estimates of survival of patients enrolled in the study cohort stratified into 2 strata of sP-selectin: the 50% upper percentile (>104 ng/mL, dashed line) and the 50% lower percentile (<104 ng/mL, continuous line); P = .079.
Discussion
Our data show that sP-selectin levels are significantly related to infection in patients who had no infection on admission. Soluble platelet selectin levels were particularly high in patients with abdominal sepsis or skin infections. Moreover, higher sP-selectin levels were associated with a higher risk of dying (although not statistically significant, P = .08).
Prior to our study, only 2 other studies analyzed sP-selectin as a biomarker for infection in the ICU.8,10 The review of Zonneveld et al. showed that sP-selectin was higher in sepsis patients compared with healthy controls. However, this study only included patients with an infection. In our study, we included all kinds of patients with SIRS. The a priori chance of infection in our population was much lower than that of Zonneveld et al. Hence, the a posteriori chance (predictability) of “proven sepsis” was lower in our study.
Vassilou et al. showed that elevated sP-selectin levels differentiate between septic and nonseptic patients in an ICU setting. In contrast to our study, the study of Vassilou et al. showed a higher prognostic accuracy of sP-selectin compared with CRP and procalcitonin. However, our patients were more severely ill with a higher predicted mortality compared with those of Vassiliou et al. (The predicted hospital mortality in our cohort was 30%, whereas the predicted mortality in the study cohort of Vassilou et al. was 16%.) This might have contributed to the higher sP-selectin levels in our patient population compared with the sP-selectin levels of Vassilou et al. (sepsis-negative patients: 83 ng/mL; IQR, 49-118; sepsis-positive patients: 142 ng/mL; IQR, 100-188). However, differences in laboratory procedures cannot be ruled out as a cause of different sP-selectin levels. In addition, in some populations, the relationship between severity of illness (and therefore the height of the biomarker at admission) and outcome is not straightforward. For example, in abdominal sepsis, mortality is often the resultant of the inability to reach surgical source control rather than the severity of illness at admission. Including such patients in our study attenuates the association between sP-selectin and outcome.
Despite the fact that we did not reach statistical significance, the direction of the association between sP-selectin and the presence of “proven infection” was similar to that of the previously published report. Levels of sP-selectin were higher in SIRS patients with an infection than those without. This can be explained by increased endothelial, platelet, and leukocyte activation, a hallmark of sepsis. 6 The interaction between leukocytes and the endothelium and platelets leads to activation of the endothelial cells and platelets, which on activation express higher levels of P-selectin. 5 This could lead to an increased release of sP-selectin from the granules of both the endothelium and the platelets. This hypothesis is supported by the relation we found between sP-selectin levels and leukocyte levels plus the endothelial activation in the proven infection group.
The relationship between sP-selectin and infection is more profound in the group without infection at admission. In patients with a proven infection at admission, the infection could already be on its return, and therefore, sP-selectin levels could be already decreased.
Second, our study shows that higher sP-selectin levels are associated with higher mortality. An explanation of the trend between sP-selectin and mortality could be the role of sP-selectin in disseminated intravascular coagulation (a systemic activation of the coagulation system). Disseminated intravascular coagulation is strongly associated with end-organ failure and death in septic patients and, hypothetically, could contribute to the relation between sP-selectin and mortality. 15 The only other study that researched the relationship between sP-selectin and mortality is the study by Tang et al. In comparison with our analysis, this study did not show a relation or trend between mortality and sP-selectin levels. 16 However, as this study researched patients with trauma, infection was seldom seen. Other selectins (such as soluble E-selectin and soluble L-selectin) have been researched more often. Those studies discovered that these selectins are an independent predictor of mortality.17–19 However, extrapolation of these results to our study population is difficult because of the different selectins measured in these studies. 20
One of the strong features of our study is the selection of a relevant patient population: patients with a suspicion of infection. In many patients, SIRS is the first presenting syndrome triggering physicians to start antibiotics. However, not all patients with SIRS turn out to have “proven infection.” A biomarker that can discriminate between “merely SIRS” and “proven bacterial infection” would be welcomed. Our findings show the limited potential of sP-selectin as such a new biomarker for infection.
Some limitations of our study need to be addressed. First, the sensitivity of the SIRS criteria for infection has been criticized: Using the SIRS criteria, one out of every 8 patients with severe sepsis is missed. 1 This study was conceived in the era when the “old” sepsis criteria still prevailed. We would recommend using the new sepsis criteria as published by Singer et al. in future studies. 21 These newer sepsis definitions overcome some of the low specificity disadvantages of the old SIRS criteria.
Second, one of the major limitations of our study is the lack of a gold standard for infection. Cultures might turn out to be false-negative because of the prior use of antibiotics or false-positive because of colonization or contamination. To compensate for this absence, we adhered to generally accepted definitions of infections. 14 Still, we cannot exclude that in a more experimental setting of sepsis, sP-selectin might turn out to correlate with the presence of bacterial infection.
Third, to overcome the limitations of adjudicating “true bacterial infection,” we also looked at the association with 30-day mortality. However, in many patients, the mortality beyond the first week is no longer directly associated with the severity of infection at admission. Complications during ICU stay determine the long-term outcome. The relationship between a biomarker at admission and 30-day outcome might be obscured by ICU-acquired complications.
Fourth, despite the sample size of this study, the associations between P-selectin and mortality differences are not statistically significant. A future larger study, preferentially including all ICU patients rather than a subgroup of SIRS, may find a statistically significant effect.
To our knowledge, this study is the largest of a very limited number of studies conducted on sP-selectin as a biomarker for infection. This study confirms the potential relationship between sP-selectin and infection and underlines the importance of sP-selectin kinetics in patients with abdominal sepsis and skin infections.
Conclusions
This study shows that in patients with SIRS not admitted for infection, sP-selectin levels are significantly related to the subsequent development of infection. Soluble platelet selectin levels were particularly high in patients with abdominal sepsis or skin infections. Furthermore, patients with higher sP-selectin levels in the first 2 days of admission had higher 30-day mortality, although this association is not statistically significant. Therefore, we conclude that sP-selectin is a potential future biomarker for both mortality and infection in patients with SIRS, but more research is needed to confirm its prognostic role.
Footnotes
Peer review:
Four peer reviewers contributed to the peer review report. Reviewers’ reports totaled 1075 words, excluding any confidential comments to the academic editor.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author Contributions
MR, HK, JK, and DWdL conceived and designed the experiments. ITS and DWdL analyzed the data, and ITS wrote the first draft of the manuscript. DWdL contributed to the writing of the manuscript. MR, HK, and JK agree with manuscript results and conclusions. ITS and DWdL jointly developed the structure and arguments for the paper. MR, HK, and JK made critical revisions and approved the final version. All authors reviewed and approved of the final manuscript.
Disclosures and Ethics
The authors have read and confirmed their agreement with the International Committee of Medical Journal Editors (ICMJE) authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication. Publication of this manuscript is approved by all authors and the responsible authorities. There are no conflicts of interest. We declare that none of the contents of this manuscript has been published previously and that none of this material is currently under consideration for publication or in press elsewhere.
