Abstract
Background:
Patients with Human Immunodeficiency Virus (HIV)have an atypical imaging pattern of lymphoma. There is paucity of literature on differences in tumor volume or burden of disease amongst HIV positive patients compared with HIV negative patients and how this correlates with clinicopathological parameters of aggressiveness and prognosis.
Methods:
This was a retrospective cross-sectional study of patients with non-Hodgkin lymphoma which were categorized into HIV positive and HIV negative. The tumor burden, disease sites, international prognostic score and Ki-67 index were recorded. Continuous variables were analyzed using the Kruskal Wallis test and categorical variables with Fisher’s Exact test.
Results:
Out of the 92 patients with non-Hodgkin lymphoma, 51.1% were HIV positive with a median age of 45.0 years. The median sum of product diameters used to measure tumor burden was 102.6 [IQR: 51.7, 173.1] with no significant difference seen between the 2 groups. The extranodal disease was significantly higher in the HIV positive group (85.1%) while exclusive nodal disease was seen predominantly in the non-HIV group (66.7%) (P < .001). Complete treatment response was higher in the non-HIV group 54.5% compared to 20.9% for the HIV group (P < .001). More HIV positive patients succumbed, 37.2% compared to the 4.5% for non-HIV patients (P < .001).
Conclusion:
HIV-related lymphoma remains a poorly understood subset. Although there was no significant difference in overall tumor burden between HIV positive and negative patients, extranodal disease was significantly higher in the HIV positive patients. Furthermore, the clinical prognostication score and Ki-67 which apply well for HIV-negative patients may not apply for HIV-related lymphoma.
Introduction
Non-Hodgkin lymphoma (NHL) accounts for approximately 3% of all cancers worldwide. 1 Infection with human immunodeficiency virus (HIV) is a known risk factor, increasing the likelihood of developing lymphoma by 5 to 15-fold. 2 There are conflicting findings on the influence of HIV on lymphoma outcomes. While some studies report no significant differences between HIV-positive and HIV-negative patients,3,4 others suggest a higher risk of cancer-related mortality among people living with HIV. 5 Recent data from southeastern Africa even suggest that HIV-positive patients may experience better outcomes under specific conditions. 6 These findings are particularly relevant in Kenya, where adult HIV prevalence in 2023 was estimated at 3.2%, with approximately 94% of people living with HIV receiving antiretroviral therapy (ART), according to UNAIDS. 7 Reported mortality rates for HIV-associated lymphomas vary widely, from 24.4% to 71.7% across the U.S. and Europe, 8 likely reflecting differences in staging, treatment access, antiretroviral use, and histological subtype.
Imaging plays a critical role in the diagnosis, staging, and treatment response evaluation of lymphoma. Fluorodeoxyglucose positron emission tomography-computed tomography (FDG PET/CT) is the gold standard for metabolic imaging, although contrast-enhanced CT remains an alternative in resource-limited settings or for non-FDG-avid tumors. 9 While some reports describe atypical imaging features of HIV-related lymphoma, 10 there is limited literature on how imaging-determined tumor burden differs between HIV-positive and HIV-negative patients. This distinction is clinically significant because higher baseline tumor burden on imaging is associated with poorer outcomes, including relapse and mortality.12 -14 For radiologists, understanding imaging patterns in HIV-associated lymphoma can refine interpretation and staging, particularly when disease presentation is atypical. For oncologists, tumor burden may inform therapeutic intensity, prognostication, and response monitoring.
Treatment response in lymphoma is typically assessed using interim and end-of-treatment imaging, categorized as complete response, partial response, stable disease, or progressive disease per the Lugano classification. 11 Higher tumor burden has been associated with increased risk of relapse and mortality.12 -14 Treatment response in lymphoma is typically evaluated using interim and end-of-treatment imaging per Lugano classification. 11 Yet, the relationship between pre-treatment metabolic tumor burden and clinical outcomes in HIV-positive versus HIV-negative individuals remains underexplored. The International Prognostic Index (IPI) remains the cornerstone for risk stratification in NHL, classifying patients into 4 risk groups based on clinical and laboratory features. 15 Several studies including Barta et al, have used IPI to evaluate outcomes. 16 Yet, data on how IPI scores relate to PET/CT-defined tumor burden in this population remain scarce. Similarly, while histological markers such as Ki-67, cell-of-origin (COO) classification, and MYC/BCL2 status are well-established indicators of lymphoma aggressiveness,17 -21 few studies have explored their relationship with metabolic tumor volume, particularly in the context of HIV.
Despite the known prognostic value of tumor burden, IPI, and histological markers in lymphoma, there is limited data on how these parameters interact in HIV-infected versus uninfected patients, particularly in sub-Saharan African populations where both HIV and lymphoma are prevalent.
This study addresses this gap by comparing imaging-defined tumor burden, clinical risk indices, and histopathological markers between HIV-positive and HIV-negative lymphoma patients in Kenya, aiming to inform more tailored imaging interpretation and risk stratification in high-HIV-prevalence settings.
Methods
This was a single-center, retrospective cross-sectional analytical study conducted at Aga Khan University Hospital, Nairobi, a tertiary teaching and referral hospital in Kenya. This study was approved by the Institutional Ethics Review Committee (IERC) at Aga Khan University Hospital (Ref: 2020/IERC-133(vl)). The requirement for informed consent was waived due to the retrospective nature of the study and the use of de-identified data. The data collection period was from December 2020 to November 2021 and records from 1st January 2011 to 30th June 2020 were accessed. The start date coincided with the implementation of the Picture Archiving and Communication System (PACS) in the radiology department, which enabled consistent access to imaging data. Patients with incomplete imaging data, missing HIV status, or unavailable histopathology reports were excluded (Figure 1).

Patient selection process.
Patients diagnosed with lymphoma who had imaging performed during the study period were included. Patients were grouped based on HIV status. For diagnostic uniformity, only patients with diffuse large B-cell lymphoma (DLBCL) were included in the final analysis; patients with low-grade lymphomas and other histological subtypes were excluded due to their distinct prognostic and treatment profiles.
Lymphoma diagnoses were based on histological review of biopsy specimens. Immunohistochemistry (IHC) was routinely available and used for subtyping, with markers including CD20, CD3, CD10, BCL2, BCL6, MUM1, and Ki-67. The Ki-67 index was used as a surrogate marker of histological aggressiveness.
All patients underwent baseline CT or FDG PET/CT imaging. Bone marrow biopsy and cerebrospinal fluid (CSF) sampling were not routinely performed. Tumor burden was assessed on pre-treatment imaging based on the Lugano classification. Disease was categorized as measurable or non-measurable, and further as nodal or extranodal. Lesions were considered measurable if the long axis diameter was >1.5 cm for nodal disease or >1.0 cm for extranodal disease. Splenic involvement was defined as a spleen >13 cm. For measurable disease, up to 6 of the largest representative nodal or extranodal lesions were selected. The sum of product of diameters (SPD) was calculated by measuring the longest transverse diameter (LDi) and the short axis diameter (SDi) of each lesion, then summing all LDi × SDi values. Imaging interpretation and tumor burden assessment were performed by a single experienced radiologist. Inter-reader variability was not assessed.
For patients who had post-treatment imaging, disease response was categorized per the Lugano criteria as: complete response, partial response, stable disease, or progressive disease. Vital status (alive or deceased) was recorded. Patients were followed retrospectively through medical records for a minimum of 2 years or until the last known clinical contact. Data collected included HIV status, age, sex, lactate dehydrogenase (LDH) levels, CD4 count, viral load, ECOG performance status, Ki-67 index, and site of disease (nodal vs extranodal). The IPI score was calculated using an established online tool MDCalc IPI Calculator for DLBCL, which automatically applies the standard 5 prognostic parameters: age >60 years, ECOG performance status ⩾2, Ann Arbor stage III/IV, elevated LDH, and more than 1 extranodal site of disease. The calculator generates both the traditional IPI score and corresponding IPI risk group classification prior to rituximab therapy, based on pre-rituximab survival estimates. In addition, the tool provides 2 prognostic outputs derived from validated models. The predicted 5-year overall survival rate prior to rituximab. The predicted 4-year progression-free survival rate, based on the Revised International Prognostic Index (R-IPI). Although the R-IPI traditionally classifies patients into 3 groups (Very Good: IPI = 0, Good: IPI = 1-2, Poor: IPI = 3-5), we categorized patients into 2 groups for analysis: Good (IPI 0-2) and Poor (IPI 3-5). This was due to the small number of patients with IPI = 0 (n = 4), which precluded statistically meaningful comparison as a separate group. Histological aggressiveness was uniformly assessed based on the Ki-67 proliferative index, as this was available for all patients in the histopathology reports. While cell of origin was documented in a few cases, the data were incomplete; hence, for consistency and comparability across the entire cohort, Ki-67 was used as the defining parameter. A Ki-67 cutoff of 70% was chosen based on a study by Broyde et al which demonstrated that, in patients with diffuse large B-cell lymphoma (DLBCL), a Ki-67 index ⩾ 70% was significantly associated with poor prognosis. 21 This cutoff has therefore been adopted as a threshold for defining aggressive disease in our study. Information on treatment regimens, response, and cause of death was extracted from available medical records, including physician notes, discharge summaries, and death certificates when present. Due to the retrospective nature of the study, detailed data on chemotherapy fidelity (eg, dosage, cycle completion, or dose intensity) and supportive treatments (such as use of granulocyte-colony stimulating factor, G-CSF) were not uniformly available and were therefore not analyzed. Baseline CD4 count and anti-retroviral treatment status were not available for all patients and were therefore not included in the multivariable analysis. For patients with diffuse large B-cell lymphoma (DLBCL)the institutional standard treatment was CHOP or R-CHOP, depending on HIV status, comorbidities, and immune function. HIV-positive patients were co-managed by hematology-oncology and infectious disease teams, with antiretroviral therapy optimized prior to chemotherapy initiation where possible. Cause of death was recorded based on the final clinical or death notes. Deaths attributed to opportunistic infections (OIs) were defined as those with documented fungal, mycobacterial, or viral infections considered secondary to immunosuppression. In several cases, it was not possible to clearly distinguish whether the OIs preceded treatment or were related to chemotherapy-induced neutropenia. Descriptive statistics were used to summarize patient demographics and clinical characteristics. The Kruskal–Wallis test was used to compare median SPD values between HIV-positive and HIV-negative groups. Associations between HIV status and categorical variables (eg, extranodal disease, IPI score, treatment response) were tested using Fisher’s Exact Test. For the effect sizes, we used logistic regression and presented as odds ratios and 95% confidence intervals. A P-value < .05 was considered statistically significant. All analyses were conducted using IBM SPSS Statistics (Version 20). The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines for observational studies. 22
Results
A total of 192 patients with non-Hodgkin lymphoma were identified during the study period (January 2011 to June 2020). A 100 patients were excluded due to lack of HIV status or lack of imaging studies in our PACS system and 92 patients met the inclusion criteria and were included in the study for the final analysis.
Baseline Characteristics
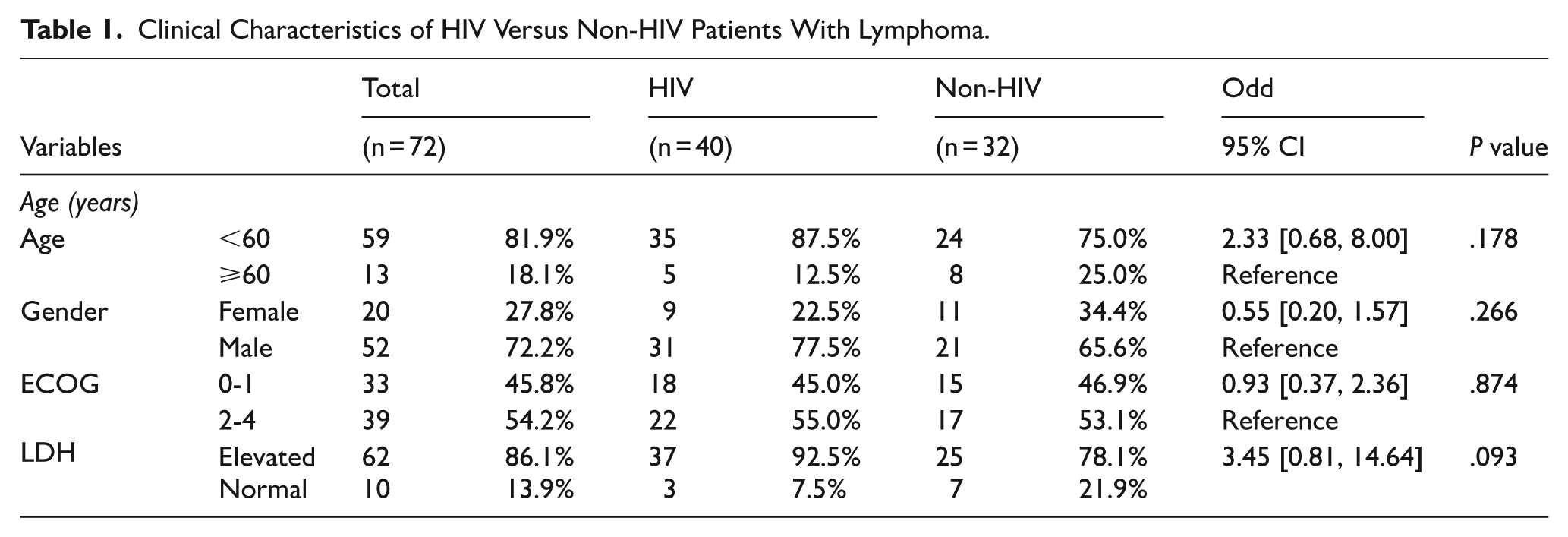
Out of the 72 patients with diffuse large B cell lymphoma that were enrolled, 87.5% (n = 35) were HIV positive while 75.0%% (n = 24) were HIV negative. The median age of patients was 45.0 years (IQR: 36.5, 55.0) where the median age for HIV patients was lower as compared to non-HIV patients but not statistically significant. The majority were male patients comprising 72.2%. For the HIV positive patients, the median CD4 count was 147 [IQR: 67.0, 211.5] with a viral load of 19 032.0 [IQR: 34.0, 255193.0]. In terms of the ECOG status, the 2 groups were similar with majority (58%) having a good ECOG status of 0-1. Most patients, 87% presented with an elevated LDH status. The baseline characteristics are summarized in Table 1.
Clinical Characteristics of HIV Versus Non-HIV Patients With Lymphoma.
Disease Burden on Imaging
As shown in the Table 2, the sum of product diameters which denotes the tumor burden was similar in the 2 groups. The median sum of product diameters was 104.9 [IQR: 49.5, 168.9]. However, extranodal disease was significantly higher in the HIV positive group at 85% as compared to non-HIV at 31.2% (P < .001) Figure 2. Although not statistically significant, a higher Ann Arbor stage of disease that is, either stage III or IV was seen in HIV positive patients (77.5%) compared to the non-HIV patients (59.4%).
Imaging Findings of HIV Versus Non-HIV Patients With Lymphoma.

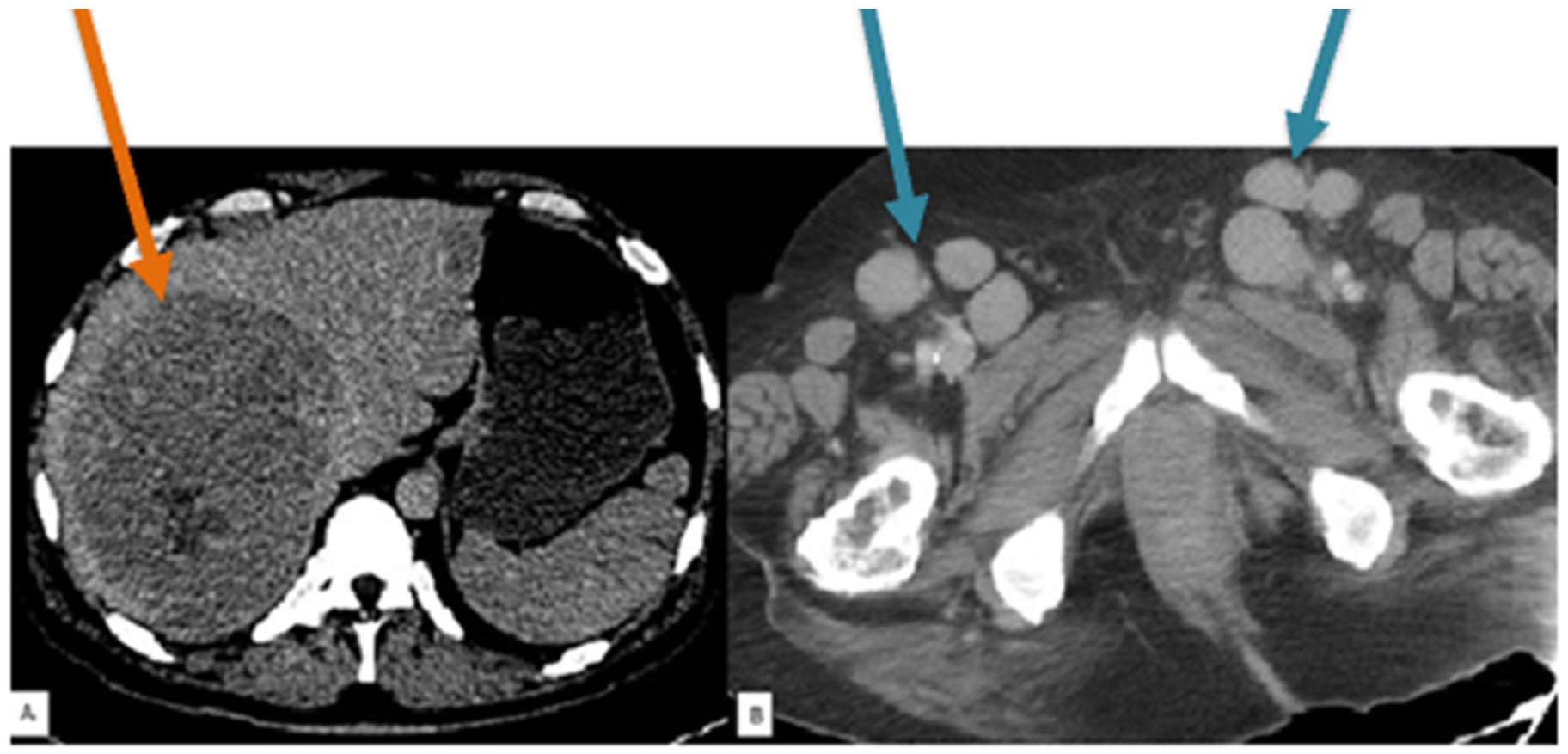
Represents an axial CT image of an HIV patient with extranodal disease in the liver (orange arrow) compared to the CT image of a non-HIV patient with bilateral inguinal lymphadenopathy (blue arrow).
Clinical IPI Score
There was no statistical difference in the IPI score between the HIV and non-HIV groups (Table 3). There was also no difference in the predicted 4-year survival rate between the 2 groups. However, when comparing the IPI score with the volume of disease (SPD), the patients who had a higher burden of disease on imaging also had a High IPI score and vice versa. This finding was however only statistically significant for the non-HIV group. Table 4 summarizes the associations with Clinical IPI scores and Tumor burden.
Comparing the Clinical Characteristics and Outcomes With HIV Versus Non-HIV Patients.

Comparing of SPD and Ki-67 With the Clinical IPI Score Between HIV and Non-HIV Patients.

Histological Aggressiveness
Although not statistically significant, there were more patients with HIV (78.8%) with histologically aggressive tumor with Ki-67 of 70%. There was no association between the histological aggressiveness and the clinical IPI score in the 2 groups as reported in Table 4.
Clinical Outcome
Out of 72 patients,50 patients had follow-up imaging. Complete treatment response was higher in the non-HIV group 54.5% compared to 20.9% for the HIV group. More HIV positive patients succumbed (37.2% compared to the 4.5% for non-HIV patients). Out of the 16 patients that died in the HIV group, 68.8% (n = 11) had opportunistic infections at the time of death. Out of the 2 patients that succumbed in the non-HIV group, one succumbed due to cardiac arrest and the other patient succumbed due to an opportunistic infection.
Discussion
Patients with HIV are known to have an increased predisposition to developing lymphoma with lymphoma being the most common AIDS related cancer. At the onset of the HIV epidemic, lymphoma was up to 100 times more prevalent in HIV patients, but this has significantly reduced since the advent of anti-retroviral drugs. However, the risk remains threefold higher compared to the non-HIV population. 20
Contrary to previous literature where AIDS related lymphoma has been shown to be associated with the more aggressive histological subtypes, we observed no difference in the 2 subgroups, although this may be due to selection bias. These results are similar to other studies that have shown black population not only present at a younger age, but also have more aggressive disease. In addition, they have more symptomatic disease, more B symptoms and higher ECOG status >2.23 -25 This may also explain why we found no difference between the tumor burden as measured by the sum of product diameters between the HIV and the non-HIV groups.
Extranodal disease was significantly higher in the HIV group compared to the non-HIV group. This is similar to previous reports by Nganga et al 2020 which demonstrated HIV patients present with atypical imaging patterns with predominant extranodal disease sites. Although, the exact mechanism of this is unknown, there are studies that have demonstrated HIV virus has transforming properties resulting in B-cell immortalization, dysregulation of MYC, and activation of EBV. 26 Certain HIV gene products such as Tat have oncogenic properties and may interfere with cell cycle control by interaction with other regulatory proteins such as Rb2/p130 and plays a role in pathogenesis of HIV Burkitt lymphoma.27,28 Other indirect mechanisms for HIV oncogenesis which may also influence the subtype of HIV lymphoma include duration and degree of immunosuppression, induction of cytokines leading to B-cell proliferation, and opportunistic infections with oncogenic herpesviruses such as EBV and HHV8. 29 Other contributing factors include duration of HIV infection, level of immunosuppression, and chronic immune activation.
The clinical IPI score has been extensively validated for prognostication of patients with lymphoma. There was no statistically significant difference in the age at presentation of the 2 patient groups, with the median age in the being 45 years in the HIV group and 48 years in the non-HIV group. This translates to a younger population being diagnosed with non-Hodgkin’s lymphoma in the current population. These results are similar to those of a study from the region which showed an early age of onset of lymphoma in Africans compared to the West. 14 The LDH levels were high in most of the patients which is in tandem with other studies that have found that genetic make-up and ethnic differences may explain why African Americans have a significantly lower age of onset, elevated LDH levels, and more B symptoms compared to the white population. 23 Although Nganga et al 2020 30 described most African Americans with lymphoma as having a higher ECOG status of >2 the ECOG status for most of the patients in the current study was between 0 and 2. This has led to questions whether the clinical IPI score needs to be revised in this population. Furthermore, the utility of clinical IPI score in the setting of HIV remains questionable. There has also been work to develop new IPI scores for HIV related lymphoma, however these remains controversial. While there are studies that have identified a number of prognostication factors including older age > 60, advanced stage, low serum albumin and treatment with ART as relevant prognostic factors, these are few and a consensus is yet to be arrived.14,29 Barta et al 16 suggested a separate IPI score for HIV patients which should include a HIV score including CD4 count, viral load and prior AIDs, number of sites of extranodal disease in addition to the parameters included in the original IPI score. This appears a plausible approach as we observed HIV patients with a greater burden of extranodal disease had a worse prognosis. Similar to prior studies, we did not find an association between the clinical IPI score and the histological aggressiveness. 21 The Ki-67 index is also a subjective index and may not be the ideal marker of histological aggressiveness.
The median CD4 count in the HIV group was 147 [with interquartile range 67.0, 211.5] with a viral load of 19032.0[interquartile range 34.0, 255193.0] which compared closely to a study by Barta et al where the median CD4 was 173 and viral load of 23 801. 16 The mortality rate in patients with HIV related lymphoma was higher compared to the Non-HIV group. Although, this study was not designed to ascertain the cause of mortality, it was observed that the 16 HIV positive patients who died had opportunistic infections at the time of death. This differs from a previous study which observed the main cause of death as progressive or relapsed disease. 16 This may be due to the patients in the current study having poorly controlled HIV infection with lowCD4 count and a high viral load.
Although, there is paucity of literature in this field, there are a few studies that have observed a similar trend with some recommending incorporating the stage of HIV in prognostication. 16 A study by Spina et al, found the 3-year overall survival was 37% among patients with HIV-related lymphoma and 74% among HIV-negative patients with non-Hodgkin lymphoma. 25 This has therefore led to recommendation of optimization of the HIV disease prior to chemotherapy regimen initiation with some advocating for cotrimoxazole prophylaxis for all patients irrespective of their CD4 count. 31 The latter is done in the current setting as part of the National Guidelines for management of HIV. The cut-off for institution of immunoprophylaxis has not yet been established but there are studies which have recommended prophylaxis for patients with low CD4 counts and patients with profound neutropenia. 32
In view of the poor prognosis in patients with HIV related lymphoma, some studies have suggested modifying the chemotherapy regimen in this subgroup with some advocating for cyclophosphamide, vincristine, doxorubicin, high-dose methotrexate/ifosfamide, etoposide, and high-dose cytarabine(CODOX-M/IVAC) with addition of rituximab rather than the conventional Rituximab-cyclophosphamide, doxorubicin hydrochloride, Vincristine sulfate and prednisone (R-CHOP) regimen. 33 Although larger studies are needed to validate such regimens in our setting, the preliminary findings from this study do not support this since the leading cause of mortality was opportunistic infections rather than progressive or recurrent disease.
While managing HIV-associated lymphoma requires a careful approach, current NCCN guidelines recommend that cancer treatment should not be delayed for HIV workup or ART initiation. Prompt treatment of opportunistic infections and consideration of drug–drug interactions remain important, but delaying chemotherapy for immune optimization may lead to worse outcomes in aggressive disease. Future research should assess whether regional data support any modifications to this approach, especially in low-resource settings.
Limitations
This study has several limitations. Its retrospective design and small sample size may limit generalizability, and selection bias is possible given the exclusion of patients without HIV status or available imaging. Missing clinical and treatment data also limited our ability to assess the impact of therapy intensity or supportive care. Differences in mortality between HIV-positive and negative patients may have been confounded by unmeasured factors such as baseline CD4 count, ART status, or infection-related complications. Additionally, imaging assessments were performed by a single radiologist without evaluation of inter-reader variability, introducing potential measurement bias. Despite these limitations, the study provides novel insights into the imaging and clinical features of HIV-associated lymphoma in a sub-Saharan African population. These findings have important implications for local clinical practice and risk stratification protocols. Despite similar overall tumor burden and IPI scores between HIV-positive and HIV-negative patients, the significantly higher rate of extranodal disease and poorer treatment outcomes observed in the HIV-positive group suggest a need to refine prognostic assessment in this population. In particular, the current reliance on IPI alone may be insufficient in HIV-positive patients, where opportunistic infections and atypical disease distribution are common and contribute to increased mortality. Incorporating imaging features including the presence of extranodal disease into baseline risk stratification may help guide more aggressive staging, closer follow-up, or earlier initiation of supportive interventions. Additionally, our findings support the development of modified prognostic models tailored to immunosuppressed populations. For radiologists, heightened vigilance in identifying extranodal involvement on baseline imaging in HIV-positive patients may facilitate timely biopsies and influence staging accuracy. For oncologists, these findings underscore the importance of integrated HIV care and infection prophylaxis alongside lymphoma treatment in regions with high HIV burden. Future prospective, multicenter studies are needed to explore the relationship between tumor burden, ART status, HIV biomarkers, and lymphoma outcomes, and to inform evidence-based prognostication and treatment guidelines tailored to this population.
Conclusion
HIV-related lymphoma remains a poorly understood subset of lymphoproliferative disease, and imaging plays a critical role in its diagnosis, staging, and prognostication. In this study, we found no statistically significant difference in total tumor burden between HIV-positive and HIV-negative patients. However, extranodal disease was more frequently observed in the HIV-positive group, although this finding was not adjusted for other components of the IPI and should be interpreted cautiously.
Our findings suggest that traditional clinical prognostic markers, including the IPI score and histological markers of aggressiveness, may not apply as reliably to HIV-associated lymphoma. Based on these observations, we propose the hypothesis that a separate prognostic index tailored for HIV-related lymphoma—potentially incorporating disease stage and placing greater emphasis on extranodal involvement—may be beneficial. Further prospective studies are needed to develop and validate such an index, and to explore how the timing, type of HAART, and specific chemotherapy regimens influence prognosis in this unique patient population.
Footnotes
Acknowledgements
The authors would like to acknowledge Dr. Jillo Bika and Dr. Hanika Patel for proof reading and constructive criticism.
List of Abbreviations
COO Cells of Origin
CT Computed Tomography
ECOG Eastern Cooperative Oncology Group
FDG Fluorodeoxyglucose
HIV Human Immuno-Deficiency Virus
IPI International Prognostic Score
LDi Long Axis Diameter
PET CT Positron Emission Tomography – Computed Tomography
R-CHOP Rituximab – cyclophosphamide, doxorubicin hydrochloride, vincristine sulfate and prednisone
SPD Sum of Product Diameters
SDi Short Axis Diameter
IQR Interquartile Ranges
CI Confidence Intervals
AKUHN Aga Khan University Hospital, Nairobi
Ethical Considerations
This retrospective study was approved by the Institutional Ethics Review Committee (IERC) of Aga Khan University Hospital, Nairobi (Ref: 2020/IERC-133(vl)).
Consent to Participate
The requirement for informed consent was waived as the study involved analysis of de-identified patient data.
Authors’ Contributions
P.L.: Conceptualization, data curation, image analysis, manuscript drafting. J.S.: Statistical analysis, interpretation of results, manuscript review. E.C.: Supervision, study design guidance. A.M.: Histopathology and IHC review, clinical input. SNG.: Supervision, study design guidance, critical revision. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
