Abstract
Iron is an essential cofactor needed for normal functions of various enzymes and its depletion lead to increase DNA damage, genomic instability, deteriorate innate, adaptive immunity, and promote tumor development. It is also linked to tumorigenesis of breast cancer cells through enhancing mammary tumor growth and metastasis. There is insufficient data describing this association in Saudi Arabia. This study aims to determine the prevalence of iron deficiency and its association with breast cancer among premenopausal and postmenopausal women referred for breast cancer screening center in Al Ahsa, Eastern Province of Saudi Arabia. Age, hemoglobin level, iron level, history of anemia, or iron deficiency were collected from patients’ medical records. The included participants were grouped based on their age into premenopausal (<50 years) or postmenopausal (⩾50 years). The definition of low Hb implemented (Hb below 12 g/dL) and low total serum Iron levels (below 8 μmol/L). Logistic regression test was used to compute the association between having a positive cancer screening test (radiological or histocytological) and participant’s lab results. The results are presented as odds ratios and 95% confidence intervals. Thrree hundred fifty-seven women were included, 77% (n = 274) of them were premenopausal. This group cases had more history of iron deficiency (149 [60%] vs 25 (30%), P = .001) compared to those in the postmenopausal group. The risk of having a positive radiological cancer screening test was associated with age (OR = 1.04, 95% CI 1.02-1.06), but negatively was associated with iron level (OR = 0.9, 95% CI 0.86-0.97) among the entire cohort. This study is the first to propose an association between iron deficiency and breast cancer among Saudi young females. This could suggest iron level as a new risk factor that may be used by clinicians to assess breast cancer risk.
Introduction
Iron Deficiency (ID) is the most common micronutrient deficiency, affecting 24.8% of the general population globally resulting in Iron Deficiency Anemia (IDA),1,2 which accounts for 50% of all causes of anemia.3,4 In IDA, the individuals do not have enough iron to make red blood cells and therefore, hemoglobin levels and/or red blood cell volume reduces. 4 Child-bearing and premenopausal women particularly are more vulnerable to IDA due to loss of blood during menstrual cycles. IDA in elder postmenopausal women, may be related to malnutrition, chronic inflammation, kidney failure, malignant disorders that may induce blood loss and bone marrow (BM) diseases. 5
Recent studies have described an association between iron and cancer development6,7; however, whether iron association with carcinogenesis is more in iron deficiency or iron overload remains controversial.8-10 Iron imbalance may contribute to all aspects of cancer growth including initiation, microenvironment, and metastasis. 11 Remarkably, a large cohort of studies established that around 40% of all cancer patients were anemic when diagnosed with cancer. 12 Although this is surprisingly a high percentage, it might be large due to the typical association of anemia with chronic disease or BM involvement. Therefore, a contributing role for iron deficiency in tumorigenesis cannot be excluded. Breast cancer is the most common cancer type among Saudi Women 13 that is mostly diagnosed at the ages between 40 and 59 years. 14 Its diagnosis under the age of 45 years has a lower survival rate but a higher recurrence. 6 Its occurrence in young women is more likely to be in a higher histological grade and metastasize to other organs more readily with poor prognosis. 15 Premenopausal women usually suffer from iron deficiency due to repeated loss of blood during their menstrual cycles in addition to more aggressive forms of breast cancer. This suggests that ID may be a risk factor in breast cancer aggressiveness in young patients. 16 Although many studies have identified dietary iron as a potential factor that increases the risk of some cancers, its association with breast cancer is controversial. In addition, there is a scarce of data about iron biomarkers in breast cancer research. 17
We cannot address the differences in clinical outcomes between young and older women with breast cancer by estrogen status or family history.6,18 Previous research works suggest that elevated levels of estrogen are associated with increased systemic iron availability since it manipulates intracellular iron metabolism. 18 For future research, this issue requires more investigations to determine the associated factors.
In Saudi Arabia, specifically the Eastern Province, there is a high prevalence of sickle cell disease and IDA.19,20 However, there is no published research that describes any association between ID and breast cancer in this population. Such data could be crucial in identifying and assessing women at risk of breast cancer living in such area. Therefore, this retrospective pilot study aims to determine the prevalence of ID among referred young and old women for breast cancer screening test. The study also assesses the association between the risk of developing breast cancer and the iron serum level.
Materials and Methods
Study population, data collection, measures, and definitions
A total of 357 Saudi women who are referred by primary health care centers to the breast cancer pathway at Abdulaziz AlAfaliq Tumor Early Detection and Diagnosis Centre (ATEDDC). The study period was from July 2018 to December 2019. The included participants were initially examined with radiology test for any abnormal breast masses, distortion, or calcification. Those with a positive radiological screening test were referred to a histocytology investigation.
We collected clinical and laboratory data from the participants’ medical records. The data included age, results of cancer screening test (radiological or histocytological), hemoglobin level, serum iron level, previous history of anemia or ID. The definition of anemia and low iron level implemented in this study was (hemoglobin below 12 g/dL for anemia and iron level below 8 µmol/L for iron deficiency). 21
History of anemia was coded as general anemia if the patients’ hemoglobin was below 12 g/dL with no specific type of anemia reported in their medical records, or ID if iron level was below 8 µmol/L.
Participants were categorized based on their age group into premenopausal (below 50 years), postmenopausal (50 years or more) and based on their screening results into positive or negative results.
Statistical analysis
Demographic variables, hemoglobin levels, iron levels, history of anemia or ID and positive cancer screening test were reported using mean and standard deviation (SD) for continuous parametric variables. Median/interquartile range (IQR) used for continuous non-parametric variables and number/frequency for binary variables. For comparisons of continuous variables, the student-T-test for parametric or the Mann Whitney U-test for non-parametric variables were used. The Chi square test was used to compare the frequency of categorical variables between pre-versus postmenopausal and between patients with positive versus negative screening tests (radiological or histocytological) in both age groups. Binary logistic regression was performed to compute unadjusted and adjusted odds ratios (OR) and 95% confidence interval (95% CI) to describe the association between age, hemoglobin levels or iron serum level and risk of having a positive cancer screening test (radiological or histocytological). Statistical analysis was performed using the Statistical Package for the Social Sciences software version 26 (SPSS, Inc., Chicago, IL) and P ⩽ .05 was considered statistically significant. The figures were constructed using GraphPad prism software.
Ethical consideration
This study was approved by the Institutional Review Board (IRB) research committee at King Fahad Hospital in Hoffuf (approval number. 30-24-2020).
Results
The total number of participants was 357; 77% of them (n = 274) were below the age of 50 years (premenopausal) (Figure 1). 87 women had a positive radiological breast cancer screening test showing an abnormality and were referred for a histocytology breast cancer test, 70 % of them (n = 61) were from premenopausal group. Twenty-two of those referred for the histocytology test had a positive test, whereas 55% (n = 12) of them were in postmenopausal group.

Total number of included participants classified based on menopausal age, positive radiological screening service, and positive histocytology screening service.
For the entire cohort, 174 participants presented with a history of anemia and ninety-six participants had a known history of ID (Figure 2). The premenopausal group had more cases with a known history of anemia (149 [60%] vs 25 [30%], P = .001) and more cases with a known history of ID (84 [31%] vs 12 [15%], P = .004) compared to those in postmenopausal group.
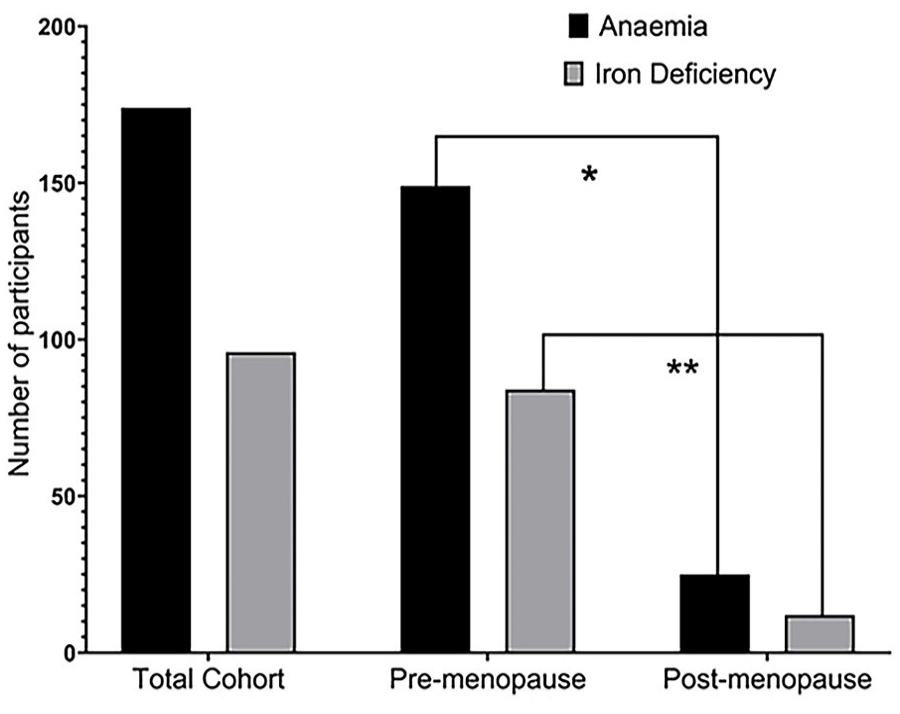
Difference between pre versus postmenopausal groups in terms of anemia or iron deficiency.
The mean age for the included participants was 40 years, the average hemoglobin reading was 12 g/dL, and the average iron serum level was 12.2 µmol/L. The postmenopausal group had more cases with positive histological screening test (10 [4%] vs 12 [14%], P < .001) compared to premenopausal group (Table 1). The premenopausal group had a lower hemoglobin level (11.9 vs 12.7 g/dL, P-value < .001) and a lower iron level (12.1 vs 12.8 g/dL, P-value = 0.03) compared to those in postmenopausal group. The postmenopausal group had more cases with positive histological screening test (10 [4%] vs 12 [14%], P < .001) compared to premenopausal group.
Characteristics of recruited patients into the study.
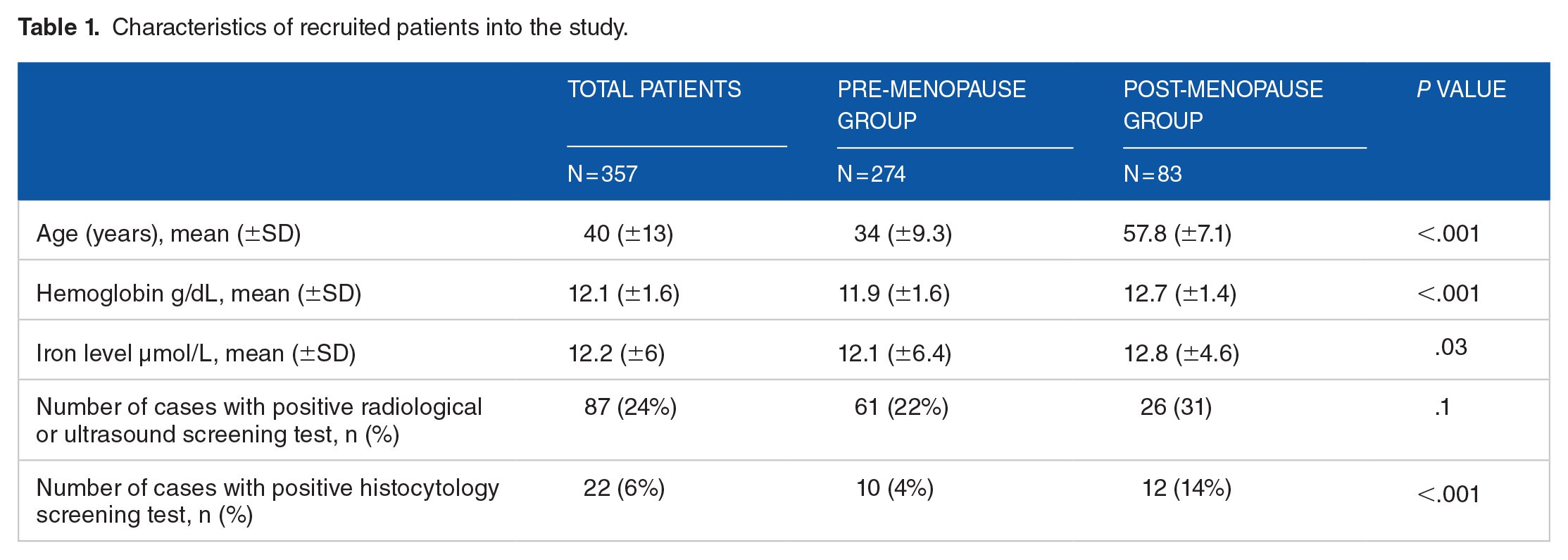
Table 2 shows that those presented with a positive radiological screening test among premenopausal group were older (33.8 vs 37.4 years, P = .01), had a lower hemoglobin level (12.1 vs 11.2 g/dL, P < .0001) and had a lower iron level (13 vs 9 µmol/L, P < .0001) compared to those with a negative screening test.
Comparison between included participants with positive versus negative radiology cancer screening test among pre-menopause group.
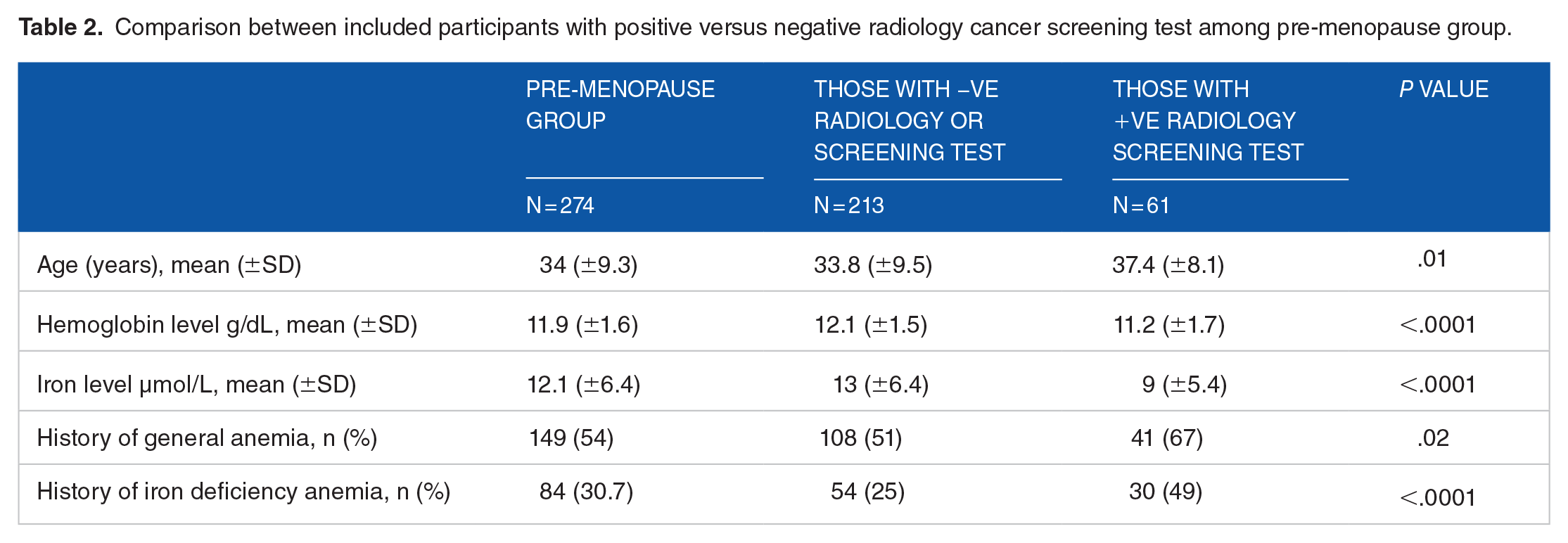
Interestingly, those with positive radiological screening test had more cases with known history of anemia (108 [51%] vs 41 [67%], P = .02) and history of ID (54 [25%] vs 30 [49%], P < .0001) compared to those with a negative screening test among premenopausal group.
Those with a positive histcytology breast cancer screening test were older (43.8 vs 36.2 years, P = .002), had a higher hemoglobin level (12.6 vs 10.9 g/dL, P = .002) and had less cases with known history of anemia (37 [73%] vs 4 [40%], P = .045) compared to those with a negative screening test among those presented with a positive radiological screening test within the premenopausal group (Table 3).
Comparing participants between positive and negative histocytology cancer screening among those presented with a positive radiological screening test within the pre-menopause group.

However, there was no significant difference between those who presented with a positive versus the ones with a negative radiological screening test among postmenopausal group (Table 4). Similarly, no differences were observed between patient with a positive versus negative histology screening test among those presented with a positive radiological screening test within postmenopausal group (Table 5).
Comparison between the included participants with positive versus negative cancer screening test among post-menopause group.

Comparing participants with positive versus negative histocytology cancer screening among those presented with a positive radiological screening test within the post-menopause group.

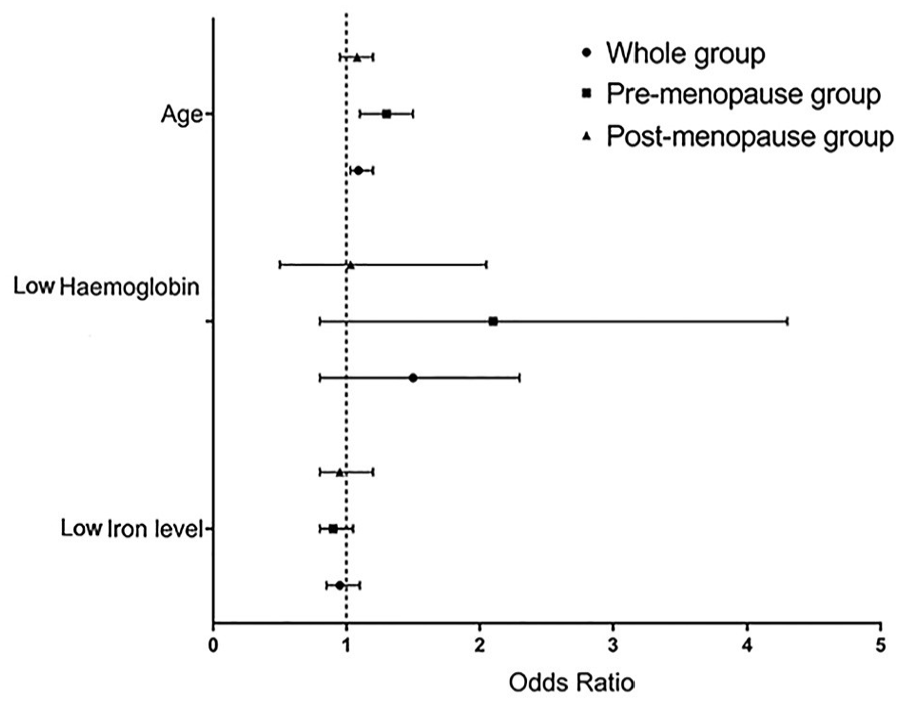
The binary logistic models represent the ones having positive radiological cancer screening tests was associated with aging among the whole cohort (OR = 1.04, 95% CI 1.02-1.06) and among the premenopausal group (OR = 1.04, 95% CI 1.01-1.08) (Figure 3). Interestingly, the models also demonstrate a negative association between having a positive radiological cancer screening and increasing iron serum level among whole cohort (OR = 0.9, 95% CI 0.86-0.86) and among premenopausal group (OR = 0.9, 95% CI 0.8-0.96). However, the models show a positive association between aging and having a positive histocytology cancer screening test among the whole cohort (OR = 1.08, 95% CI 1.04-1.12) and among the premenopausal group (OR = 1.2, 95% CI 1.1-1.4) (Figure 4).

Binary logistic models demonstrate the association between age, hemoglobin level, and iron level with risk of having a positive radiological cancer screening test among the whole cohort.
Binary logistic models demonstrate the association between age, hemoglobin level, and iron level with risk of having a positive histological cancer screening test among those with positive radiological screening test.
Discussion
This pilot study is the first to suggest an association between ID and the risk of developing breast cancer among women presenting for a breast cancer screening test in Al Ahsa, Saudi Arabia. The prevalence of ID was more prominent among premenopausal women compared to postmenopausal group. The risk of having a positive radiological cancer screening is positively associated with aging, but negatively with increasing iron serum level. Significantly, this association was present among the whole cohort and premenopausal group, indicating to the importance of iron serum level and the risk of having breast cancer. This could support the negative association between increasing iron serum level and the risk of having a positive histocytology breast cancer screening test. These associations were not statistically significant which could be a result of the limited number of participants with a positive hisotcyology cancer screening included in the study. However, the narrow 95% confidence interval range implies that this negative association is important to consider. Previous studies reported that incidence ratio of cancer increased by two folds in patients with ID, whereas patients with iron deficiency could be at risk of poor cancer prognosis.1,6
In this study, there was a high prevalence of ID, which is expected, as the study population is known for high prevalence of sickle cell diseases. 19 This study revealed that the proportion is higher in premenopausal group compared to postmenopausal group. This difference can be due to menstruation or malnutrition. 22 Such a fact is important to consider as a recent study reported that there was a high prevalence of IDA among Saudi University female students as a reason of malnutrition or poor eating habits. In contrast, another study revealed that the high prevalence of iron deficiency and IDA in Saudi Arabia is due to sickle cell or thalassemia diseases.20,23
Findings from the current study support the idea of utilizing iron serum level as an early risk measure for assessing possibility of breast cancer especially in young women. Previous study reported that iron deficiency and high estrogen levels in premenopausal women can lead to breast cancer due to stimulated production of vascular endothelial growth factor (VEGF). 24 Hypoxia-inducible factor-1α (HIF-1α) is a transcription factor that induces angiogenesis in cancer cells under low oxygen conditions. Iron is a cofactor of prolyl-4hydroxylase enzyme that degrades HIF-1α and limits the formation of angiogenesis and metastasis. Therefore, iron deficiency can lead to increased HIF-1α expression levels in breast cancer patients, which in return increases VEGF concentration and subsequently angiogenesis.25,26
Another significant finding revealed by this study is the positive association between aging and having positive radiological screening results. Similar associations have well established and reported in previous reports. 27 It was apparent that postmenopausal group had more cases with positive histocytology cancer screening test compared to premenopausal group in our study. As cancer requires time to develop and establish, early diagnosis of ID could be a good predictive measure to assess risk of breast cancer and treating it could reduce such risk.
One strength of the current study is that it examined diverse groups of women (pre vs postmenopausal) and it investigated the association between positive cancer screening (radiological or histocytological) test with age, hemoglobin level, and serum iron level. In-depth knowledge of these associations and how it can be applied for better assessment of breast cancer risk in these patients is required.
Findings from this study could be crucial for enlightening the current practice on a novel approach to assess breast cancer risk. Clinicians should pay close attention to low iron serum level and treat any ID within their patients to limit their risk of developing breast cancer with poor prognosis. Also, it could help them to deliver a clearer message while educating their patients about risks associated with ID. Such information is necessary when caring for a society known for its high prevalence of sickle cell diseases.
However, clinicians should be cautious when treating their patients’ low iron serum level, as exceeding the recommended iron level could be harmful for their patients. A strong association exists between increased iron concentration and breast cancer incidence in postmenopausal women through oxidative stress pathways. 28 The increased iron levels in cancer cells and their surrounding microenvironments, protect them from the cytolysis by natural killer cells (NK) as the iron antagonize the effects of nitric oxide and tumor necrosis factor alpha (TNFα). 28
Future studies with a larger cohort of patients are needed to support better statistical results with narrower deviation range. In addition, collecting more information about the patients, such as family history of previous cancer, history of chronic anemia diseases, previous pregnancies, lactation, ongoing hormonal, supplement, or replacement therapy to treat low hemoglobin or low iron serum level are required to investigate more factors might be associated with the risk of breast cancer in such a cohort.
A major limitation of this study is its nature. Being a pilot study has an impact on full interpretation of the finding due to the low level of statistical significance. Specifically, for postmenopausal group or the total number of positive histocytology cancer screening tests. The unavailability of patients’ complete iron profile and dependence on serum iron is another point to be considered as this parameter could be influenced by diet and food supplement. The limited number of participants had also affected the overall statistical test causing a wide range in standard deviation or 95% confidence interval values.
Conclusions
Thi
Footnotes
Acknowledgements
The authors would like to thank Dr. Omar Ba Yameen, Dr. Ali Al Dandan, Sherifah Al Zamil, Haider Haj Mohammed and Abdulrahman Alyousef for their early support in data collection phase. Also, would like to thank and acknowledge the contribution of Dr. Jenan Al Matog from Mohammed Al Mana College for Medical Sciences.
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
Mohammad AlKhamees. First author, Research planning, data collection, data coding and analysis, reviewing the manuscript and approving the final draft.
Ethical Approval
Consent was waived by all participants in this study. King Fahad Hospital in Hoffuf issued IRB 30-24-2020.
Payment/Services info
All authors have declared that no financial support was received from any organization for the submitted work.
Other Relationships
All authors have declared that there are no other relationships or activities that could appear to have influenced the submitted work.
