Abstract
Background:
Our objective was to describe the hospital-based systemic treatment sequences in early stage HER2+ breast cancer patients treated with trastuzumab in France in 2016.
Methods:
This retrospective observational study was based on the national hospital discharge database (PMSI). Patients hospitalized for breast cancer in 2016 and administration of trastuzumab between 6 months prior and 1 year after surgery were included. The following treatments were identified: (1) trastuzumab ± chemotherapy; (2) chemotherapy alone; (3) q3w trastuzumab weekly chemotherapy. Hospital admissions for cardiac events before and after the surgery were investigated. An unsupervised machine learning technic called TAK (Time-sequence Analysis through K-clustering) was used to identify and visualize typical systemic treatment sequences.
Results:
Overall, 3531 patients were included: 2619 adjuvant cohort patients (74.2%) and 912 neoadjuvant cohort patients (25.8%). The mean age was 56.4 years (±12.3), 99.7% patients were female. Treatment initiation occurred within 6 weeks of the surgery in 58% and 92% of patients, and trastuzumab treatment lasted 12 months (±1 month) in 75% and 66% of patients in the adjuvant and neoadjuvant cohorts, respectively. Nevertheless, 12% and 22% of patients were treated with trastuzumab for <11 months in the adjuvant and neoadjuvant cohorts, respectively. There was not one standard sequence of treatments per cohort, but 4 and 3 typical treatment sequences in the adjuvant and the neoadjuvant cohorts, respectively, plus 2 treatment sequences with an early treatment withdrawal. The frequency of patients with ⩾1 hospital stay with a cardiac event was higher among patients with an early treatment withdrawal.
Conclusions:
The treatment sequences of most patients were in line with the recommendations in force. The machine learning approach provided a telling visual display of the results, thereby allowing healthcare professionals, health authorities, patients, and care givers to see the whole picture of the hospital-administered drug strategies.
Keywords
Introduction
In France, in 2018, with 58 459 new cases and an age-standardized rate of 100 new cases per 100 000, breast cancer is the most common cancer among women and the leading cause of cancer death (12 146 deaths and an age-standardized mortality rate of 14 per 100 000). 1 Breast cancer in men is rare, contributing to less than 1% of cases.2,3
Between 2009 and 2012, 88% of female breast cancers were diagnosed at early stage. 4 Treatment of early breast cancer requires combination of local procedures (surgery and possibly radiotherapy), systemic treatments, and supportive care.2,5 Primarily used to decrease tumor size prior to surgery and in patients with poor prognostic factors (eg, inflammatory tumor, or lymph nodes invasion), neoadjuvant therapies (chemotherapies, sometimes combined with an anti-hormonotherapy, and/or a targeted therapy prior to breast surgery) were used, in 2009 to 2012, in only 10% of breast cancer patients. 4
Breast cancer is no longer viewed as a single disease, but rather as a series of diseases defined by biologic characteristics based on hormone receptor status and human epithelial growth factor receptor 2 (HER2) status. In France, 10.3% of early breast cancer patients have HER2+ tumors. 6 As new drugs become available, the choice of treatment strategy is increasingly complex and patient-tailored. 7 To confirm and to complement clinical studies’ conclusions, it is essential to examine treatment sequences in the real-life setting. 8 Yet, in everyday clinical practice, due to the large number of patients and treatment combinations, as well as the possible small treatment protocol deviations, identifying patterns in treatment sequences is arduous. 9 To overcome these challenges, the use of machine learning is advantageous.
The objective of this study was to describe the hospital-based treatment sequences in early stage HER2 positive (HER2+) breast cancer patients treated with trastuzumab (Herceptin®) in 2016 in everyday clinical practice, using data from the hospital discharge database of the French National Health Data System (SNDS Système National des Données de Santé). 10 At the time of the study, trastuzumab was already part of the clinical practice, as it had been approved as a neoadjuvant treatment for early HER2+ breast cancer 5 years earlier. 11 Hence, HER2+ early breast cancer was inferred from trastuzumab administration. Hospital admissions for cardiac events were investigated in an exploratory analysis to fuel the interpretation of some treatment sequences.
Methods
Study design and data sources
This retrospective observational study was performed using data from one of the SNDS’ databases: the hospital discharge database (PMSI Programme de Médicalisation des Systèmes d’Information). 10 Specifically, claims data from the medicine, surgery, and obstetric departments (MCO Médecine Chirurgie Obstétrique) of all private and public hospitals as well as at home hospitalization (HAD Hospitalisation A Domicile) were analyzed.
All patients treated with trastuzumab—a drug that is only available at the hospital in France—are captured in the PMSI. Patients with early breast cancer are treated at the local hospital.
Study population and study period
Patients with at least 1 hospitalization with surgery for breast cancer (principal diagnosis or related diagnosis with ICD-10 code C50*) in 2016 and at least 1 administration of trastuzumab in the 6 months prior to surgery or within 1 year after surgery were included in the study. The index date was the first day of this hospitalization for surgery.
At the hospital, an administration of trastuzumab was detected by the combination of the drug code (UCD 9400037 and UCD 9220097 for sub-cutaneous and intravenous administration, respectively), a “cancer chemotherapy administration” code (DP Z511) with a related diagnosis (or significant associated diagnosis) of breast cancer. Among at home hospitalizations, only trastuzumab drug code was sought for.
Patients whose cancer evolved into metastatic cancer during the first 12 months of the follow-up period (ICD-10 C77-79*); those who were administered trastuzumab and pertuzumab during the same hospitalization over the first 12 months of the follow-up; and those with only isolated trastuzumab sessions (which aims to treat local cancer recurrences or a metastatic cancer but is not part of the adjuvant therapy, see definition in Supplemental Methods 1) were excluded. In 2016, trastuzumab administered in combination with pertuzumab was restricted to patients HER2+ metastatic breast cancer as in a first line therapy.
Patients’ data were analyzed from 6 months prior to the index date until 18 months after the index date or until death, whichever occurred first.
Patients were categorized into the adjuvant cohort (absence of chemotherapy and trastuzumab prior to surgery) or the neoadjuvant cohort (presence of chemotherapy or trastuzumab before, and most of the time after, surgery).
The PMSI is an exhaustive hospital database. All patients meeting the selection criteria during the inclusion period were included. No study size was computed. For billing purposes, all healthcare consumption is recorded in the PMSI. Thus, there were no missing data on the variables used in the study.
Variables
In the PMSI, besides breast cancer surgery, the following systemic anticancer treatment procedures were identified: (1) administration of trastuzumab, with or without administration of chemotherapy (hereafter designated as “trastuzumab ± chemotherapy”); (2) administration of a chemotherapy alone (in absence of trastuzumab administration); (3) administration every 3 weeks (q3w) of trastuzumab and weekly chemotherapy (alternating sessions of “trastuzumab ± chemotherapy” and “chemotherapy alone”).
The “chemotherapy alone” modalities correspond to standard chemotherapies (such as anthracyclines or taxanes) and are included in the total cost of the hospitalization (ie, it is not possible to capture the compounds of each chemotherapy). In contrast, trastuzumab is identifiable from the list of expensive drugs dispensed in hospitals and budgeted separately from the cost of a standard hospitalization.
In the at home hospitalization dataset, only trastuzumab could be captured and not standard chemotherapies.
By definition, a systemic treatment cycle (trastuzumab ± chemotherapy or chemotherapy alone) covered a patient for 21 days following the first intravenous infusion, unless another administration (of the same treatment or of another one) occurred within this period. A “grace period” was a period of less than 21 days not covered by a treatment (21 days without treatment corresponds to missing one infusion). Conversely, a “treatment-free period” started 21 days after the last treatment administration if the administration is not followed by a grace period.
To explore short treatments, early treatment withdrawals, and long treatment-free periods, as trastuzumab is associated with cardiac dysfunction, 2 hospital admissions for cardiac events between 6 months prior to the index date and 18 months after the index date were investigated. Codes in chapter IX “Diseases of the circulatory system” of the ICD-10, reviewed by a clinical expert, recorded as the principal or related diagnosis on the hospital discharge form were used (Supplemental Table 1).
Analytical process
Statistical methods
Continuous data were summarized by their mean, standard deviation (SD), median, and first (Q1) and third (Q3) quartiles. Categorical data were summarized by the frequency and percentage of patients per category. Duration of each systemic treatment and the median cumulative treatment duration for each patient were computed.
TAK method
An innovative Machine Learning technic called TAK (Time-sequence Analysis through K-clustering) was used to identify typical systemic treatment sequences. The TAK is a 3-step analytical process which has been detailed elsewhere 12 and is briefly described here.
Step 1: Each treatment for each patient was represented by a time vector. Then, each patient was replaced by a succession of his/her time vectors;
Step 2: The most similar sequences of vectors (ie, treatment pathways) were ordered using an unsupervised hierarchical agglomerative clustering method. 13 This was carried out by first computing a matrix of distances between all possible pairs of patients. The distance used was the hamming distance. 14 Then the Ward’s linkage method 15 was used to combine the closest pairs of patients into groups (clusters);
Step 3: Finally, a noise filtering technique (a modal filter) was applied to the groups to retain key treatment sequences and improve visual outputs. The filter is parametrized by a kernel size, adapted to the study population size and the follow-up duration. In a nutshell, each pixel in the kernel is replaced by the most frequent value found in the kernel.
Outputs with 2 to 10 clusters were examined and the optimal number of clusters was determined by a clinician, based on observed clinical practices.
On the TAK output graphs, time is shown on the x-axis while all study patients are stacked-up up on the y-axis. To each patient corresponds 1 line; the color of the line indicates a treatment type; the length of the line indicates the treatment duration. A TAK graph reads from left to right, where each patient starts on the most left-hand side of the graph (6 months before surgery) on the vertical axis and moves horizontally toward the right-hand side of the graph over the course of the follow-up.
Ethics approval and patient consent
Access to the data fell under the 006 framework (MR-006) developed by the French data protection authority (Commission Nationale de l’Informatique et des Libertés, CNIL). Institutional review board is not required under the 006 framework. The data controller’s certificate was approved by the CNIL on September 11, 2018 (No. 2206508 v0). The data controller and the Technical Agency for information on hospitalization (Agence technique de l’Information sur l’Hospitalisation, ATIH) signed an agreement to use the PMSI data on October 2, 2018. In accordance with the regulations in force, patient consent was not necessary because this study used secondary data, there was a public interest in assessing, for the first time to our knowledge, cancer treatment sequences in the population of HER2 positive early breast cancer patients treated with trastuzumab in France in a real-life study, and the protection of patients’ rights and freedom were guaranteed at all times.
Results
Study population
In 2016, in France, 60 160 patients were hospitalized for breast cancer surgery. After examining patients for eligibility, 3531 patients were included in the study: 2619 patients (74.2%) received trastuzumab after surgery only (adjuvant cohort) and 912 patients (25.8%) received trastuzumab prior to (and most of the time after) surgery (neoadjuvant cohort) (Figure 1).

Study population selection process.
On average, patients were followed for 1.99 years (±0.03), corresponding to a total of 7042 patient-years.
Ten patients died at the hospital during the study follow-up (no information is available in the database regarding deaths occurring outside of the hospital). Four patients died within 12 months of the surgery and 6 died later.
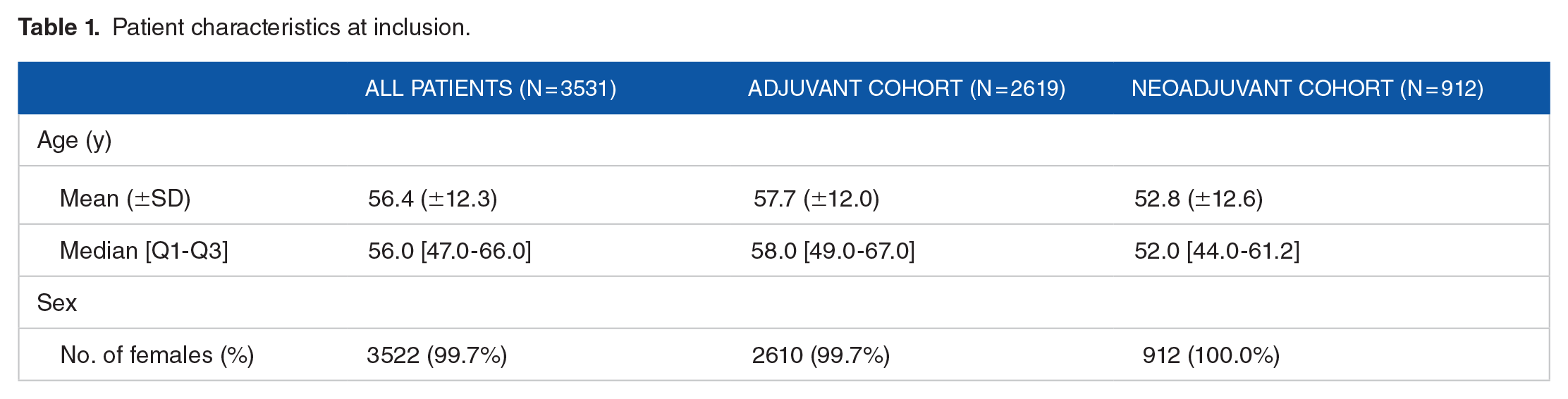
Patients’ characteristics at inclusion
At inclusion, the patients’ ages ranged from 20 to 95, and the mean age was 56.4 years (±12.3) (Table 1). Almost all patients were women (99.7%). Patients in the adjuvant and neoadjuvant cohorts were, on average, 57.7 (±12.0) and 52.8 (±12.6) years old, respectively. All 9 male patients received trastuzumab ± chemotherapy in the adjuvant setting.
Patient characteristics at inclusion.
Treatment sequences
All study patients
Over the study period, 73% of patients were treated for 12 months (±1 month) with trastuzumab (regardless of the treatment protocol). On average, patients were treated with trastuzumab for 11.7 months (±2.3) (Supplemental Table 2). Overall, 67% and 94% of study patients were treated within 6 weeks and 2 months after surgery, respectively.
All 9 male patients were part of the adjuvant cohort. Five of them underwent an early and definitive trastuzumab ± chemotherapy withdrawal.
Adjuvant cohort
Figure 2 presents the proportion of patients under hospital-administered treatment over time in the adjuvant cohort. By 6 weeks and 2 months after surgery, 58% and 94% of the 2619 patients had started their treatment, respectively. The mean time between surgery and initiation of chemotherapy alone was 1.6 month (±1.2), whereas the mean time between surgery and initiation of trastuzumab ± chemotherapy was 2.6 months (±1.2). Three months after surgery, more patients switched to trastuzumab ± chemotherapy or q3w trastuzumab with weekly chemotherapy. Trastuzumab ± chemotherapy became the most frequent type of treatment protocol 3.3 months after surgery and was eventually administered to 89% of patients 6 months after surgery.
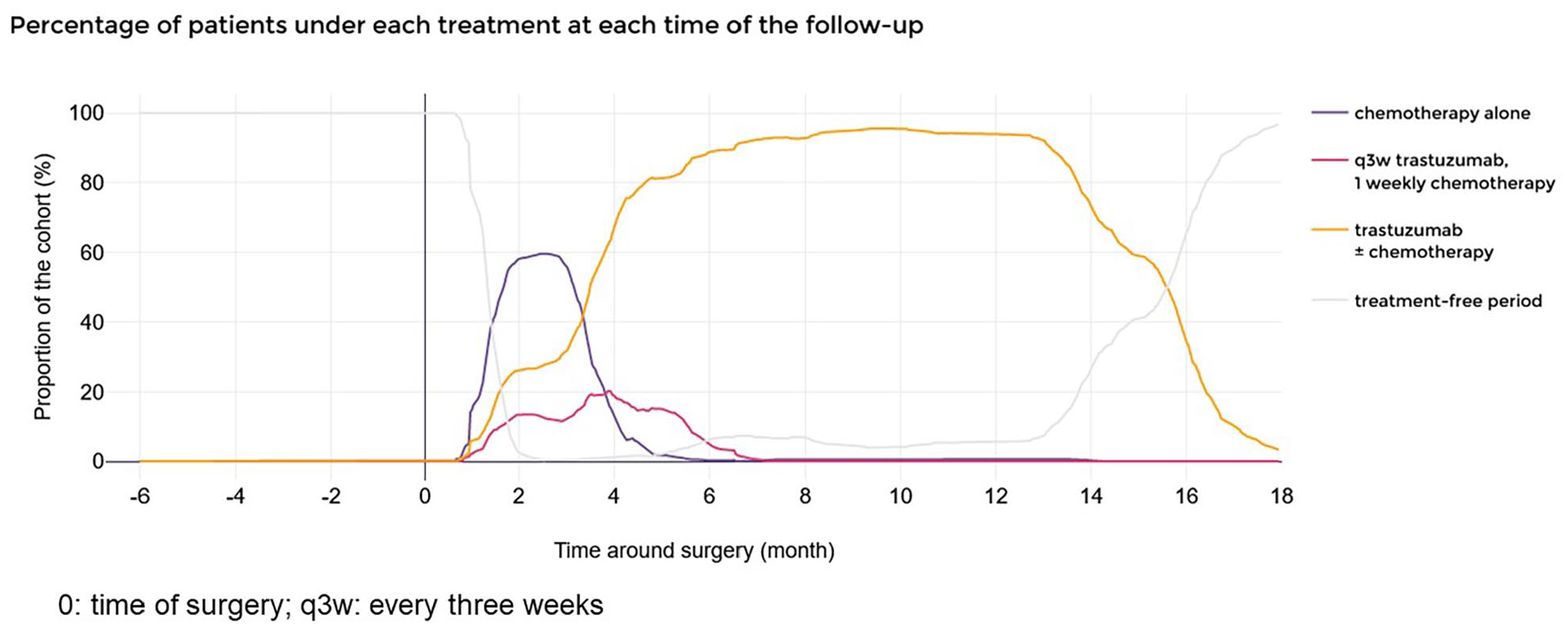
Hospital-dispensed treatment proportions in the adjuvant cohort of early breast cancer patients, over time.
In the adjuvant cohort, patients were under drug treatment for, on average, 13.4 months (±2.4) in total (Supplemental Table 2), 75% of the patients were treated for 12 months (±1 month) with trastuzumab (regardless of the treatment protocol). Nevertheless, 12% of patients were treated with trastuzumab for <11 months.
Figure 3 shows the results of the TAK analysis.

TAK (Time-sequence Analysis through K-clustering) analysis of treatment sequences in the adjuvant cohort of early breast cancer.
Six groups of patients in the adjuvant cohort were identified, each corresponding to a treatment sequence:
Group adjA (965 patients, 37% of the adjuvant cohort patients): a short period of chemotherapy (alone mean duration of 2.4 months, ±0.7), followed by a long trastuzumab treatment ± chemotherapy (mean duration of 12.1 months, ±1.4)
Group adjB (415 patients, 16% of patients): a short chemotherapy (mean duration of 2.5 months, ±0.6), followed by a short period under q3w trastuzumab weekly chemotherapy (mean duration of 1.9 months, ±0.7), and finally a long trastuzumab treatment ± chemotherapy (mean duration of 10.3 months, ±0.8)
Group adjC (340 patients, 13% of patients): a short period under q3w trastuzumab weekly chemotherapy, then a long trastuzumab treatment ± chemotherapy
Group adjD (680 patients, 26% of patients): only a long period of trastuzumab treatment ± chemotherapy
The last 2 groups gathered patients who followed a treatment sequence like Group adjA and adjD but stopped all treatments of interest early
Group adjE (115 patients, 4% of patients): initial chemotherapy or directly under trastuzumab treatment ± chemotherapy. The striking feature in this group’s treatment sequence was the early trastuzumab treatment ± chemotherapy withdrawal followed by the restart of trastuzumab ± chemotherapy. The whole treatment sequence (chemotherapy or trastuzumab ± chemotherapy) lasted, on average, 14.1 months (±1.1) and the treatment free period lasted, on average 2.3 months.
Group adjF (104 patients, 4% of patients): As in Group adjE, the patients start their treatment with a chemotherapy or trastuzumab ± chemotherapy, were then treated with trastuzumab ± chemotherapy and finally underwent an extremely early trastuzumab treatment ± chemotherapy withdraw. The difference between groups adjE and adjF lays in the absence of trastuzumab ± chemotherapy re-challenge in Group adjF patients. The whole treatment sequence (chemotherapy or trastuzumab ± chemotherapy) lasted, on average, 4.7 months (±2.3).
Altogether, 4% of patients withdrew early from the adjuvant trastuzumab therapy.
Neoadjuvant cohort
Prior to surgery, the first treatment initiated in the patients of the neoadjuvant cohort was chemotherapy alone (starting as early as 5.7 months prior to surgery for few patients) (Figure 4). Then, about 3 months prior to surgery, the proportion of patients treated with a chemotherapy alone declined and the proportion of patients treated with trastuzumab ± chemotherapy increased. Exactly 3 months prior to surgery, 98% of patients were under some type of treatment, whether chemotherapy (50%), trastuzumab ± chemotherapy (38%), or q3w trastuzumab weekly chemotherapy (10%). The mean time between the last trastuzumab ± chemotherapy administration or chemotherapy administration and surgery was 0.5 month (±0.5) or 2.7 months (±1.3), respectively.
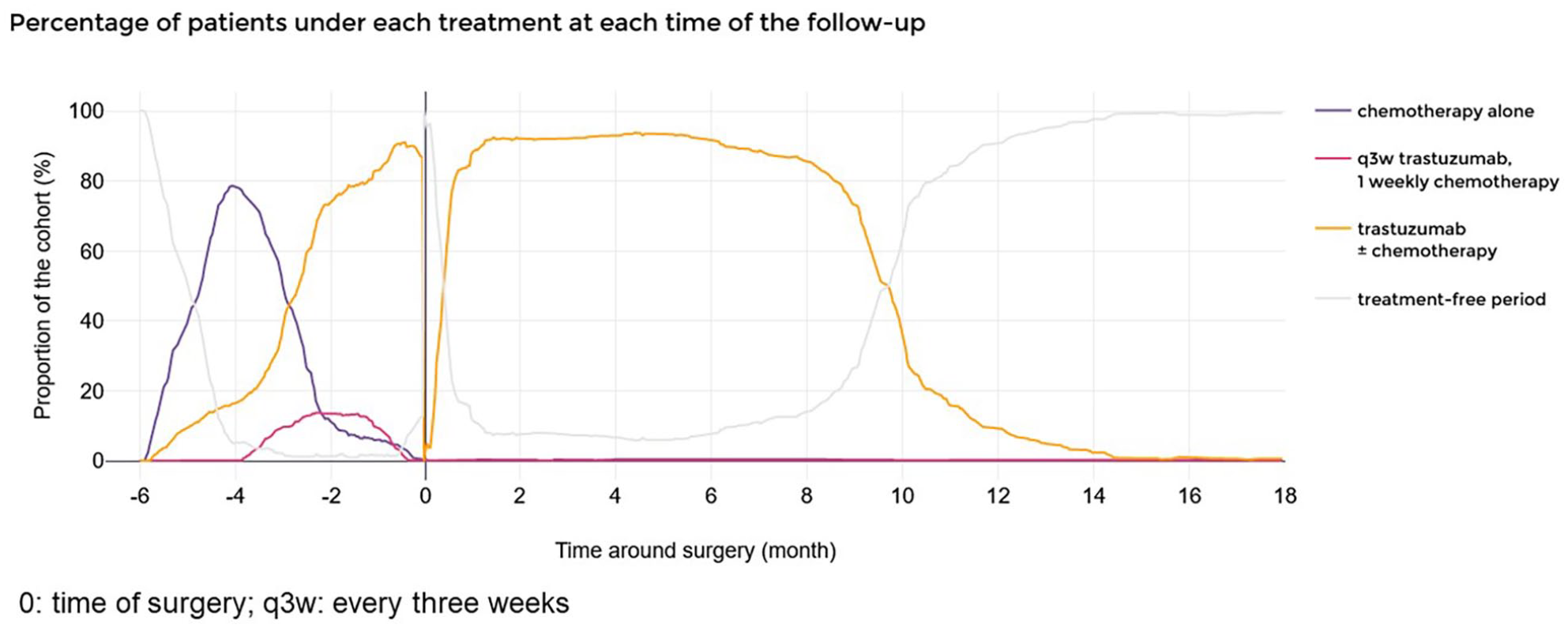
Hospital-dispensed treatment proportions in the neoadjuvant cohort of early breast cancer patients, over time.
After surgery, just over 90% of patients were under trastuzumab treatment ± chemotherapy and about 10% of patients were not treated with any of the drugs which were identified in the PMSI. The mean time between surgery and the restart of the trastuzumab treatment ± chemotherapy was 0.6 month (±0.8). Overall, 92% and 95% of study patients were treated within 6 weeks and 2 months after surgery, respectively. One year after surgery, 9% of patients were still being treated with trastuzumab ± chemotherapy.
As for patients in the adjuvant cohort, patients in the neoadjuvant cohort were under drug treatment for, on average, 13.6 months (±2.5) in total (Supplemental Table 2). Two thirds of the patients were treated for 12 months (±1 month) with trastuzumab (regardless of the treatment protocol). Nonetheless, 22% of patients were treated for <11 months with trastuzumab and 3% of patients were treated for more than 15 months.
Five groups of patients in the neoadjuvant cohort were identified, each corresponding to a treatment sequence (Figure 5). The main treatment difference between the groups lay in the treatment modality prior to surgery; after surgery, all patients were treated with trastuzumab ± chemotherapy (although 1 group started its treatment later and 2 groups stopped earlier).
Group neoA (568 patients, 62% of patients): a short neoadjuvant chemotherapy (mean duration of 2.5 months, ±1.0), followed by a long trastuzumab treatment ± chemotherapy period (mean duration of 11.9 months, ±1.1) shortly interrupted around the time of the surgery.
Groups neoB (140 patients, 15% of patients): a short neoadjuvant chemotherapy followed by q3w trastuzumab weekly chemotherapy, completed with trastuzumab ± chemotherapy, shortly interrupted around the time of the surgery.
Group neoC (96 patients, 11% of patients): the patients were only treated with trastuzumab ± chemotherapy, both before and after surgery, for a mean total duration of 13.5 months (±2.3).

TAK (Time-sequence Analysis through K-clustering) analysis of the treatment sequences in the neoadjuvant cohort of early breast cancer patients.
The last 2 groups gathered patients who followed a treatment sequence like Group neoC and neoA but stopped all treatments of interest early:
Group neoD (66 patients, 7% of patients): regardless of the treatment modality prior to surgery, the main feature of this group was the “early” permanent trastuzumab treatment ± chemotherapy withdrawal after surgery (mean duration of treatment of 10.3 months, ±2.0).
Group neoE (42 patients, 5% of patients) the patients with hardly any treatment after surgery (mean duration of treatment of 6.7 months, ±3.5).
Of note, at the very top of Figure 5, in the Group neoA, 29 patients (3%) did not receive any trastuzumab prior to surgery and did receive some after the surgery.
Patients hospitalized for cardiac events
From 6 month before surgery to 18 months after surgery, 62 patients (1.8%) were hospitalized for cardiac events (81 hospital stays), including 47 in the adjuvant cohort (1.8% of the adjuvant cohort) and 15 in the neoadjuvant cohort (1.6%) (Supplemental Figure 1). The most frequent diagnoses were left ventricular failure (12 patients), congestive heart failure (10 patients), paroxysmal atrial fibrillation (9 patients), atherosclerotic heart disease (6 patients), silent myocardial ischemia (5 patients), and unstable angina (4 patients).
Four adjuvant patients were admitted to the hospital for a cardiac event within the 6 months prior to their breast cancer surgery (3 Group adjD patients and 1 Group adjF patients with early definitive trastuzumab treatment withdrawal). Patients with at least 1 hospital stay for a cardiac event were more frequent in the groups with early treatment withdrawal 9% of group adjF) and hardly any treatment after surgery (7% of group neoE patients).
Discussion
In 2016, in this national, hospital-based, French real-life study, 3531 early HER2+ breast cancer patients treated with trastuzumab were included and followed for 2 years. The mean age at inclusion was 56.4 years old and all but 9 patients were women. Trastuzumab was administered as an adjuvant therapy in almost three-fourth of patients and as a neoadjuvant therapy in a one-fourth of patients. The systemic therapy of most patients met the guidelines (treatment initiation within 6 weeks of the surgery in 58% and 92% of patients and trastuzumab treatment for 12 months (±1 month) in 75% and 66% of patients in the adjuvant and neoadjuvant cohorts, respectively). The machine learning analysis revealed that there was not a single sequence of treatments but 4 and 3 typical treatment sequences in the adjuvant and the neoadjuvant cohorts, respectively, plus 2 treatment sequences in each cohort characterized by a temporary or early treatment withdrawal. The frequency of patients with at least 1 hospital stay with a cardiac event was higher in the groups with an early treatment withdrawal.
Clinical interpretation of the findings
With all due precautions, the groups identified by the machine learning analysis seemed to match the routine clinical management with systemic treatments of early HER2+ breast cancer patients treated with trastuzumab around 2016, from 6 months prior to surgery to eighteen months after. The advantages of this analysis are the ability to quantify the proportion of patients with each treatment sequence, to observe to treatment switches over time, and to evaluate their duration. Moreover, the TAK offered an easily interpretable data visualization of the treatment sequences
In the adjuvant cohort, it can be inferred that in the first group, the treatment corresponded to 3 anthracyclines cycles followed by q3w taxanes-based chemotherapy (Docetaxel) in combination with trastuzumab and followed by trastuzumab monotherapy; in the second group, the treatment was 3 cycles of fluorouracil + epiribucin + cyclophosphamide (FEC) and 3 cycles of trastuzumab + taxanes (Paclitaxel); in the third group, the treatment was 3 cycles of trastuzumab + taxanes (Paclitaxel); in the fourth group, the treatment was q3w taxane-based chemotherapy (Docetaxel) in combination with trastuzumab and followed by trastuzumab monotherapy; all first 4 groups ended with a long trastuzumab treatment; finally, the fifth group had a 2- to 3-month treatment-free interval, and the sixth group had an extremely early treatment withdrawal.
In the neoadjuvant cohort, it can be inferred that, in the first group, the treatment corresponded to 3 anthracyclines cycles followed by 3 cycles of trastuzumab + taxanes (Docetaxel or Paclitaxel); in the second group, the treatment was 3 cycles of FEC and 3 cycles of trastuzumab + taxanes (Paclitaxel); in the third group, the treatment was 3 cycles of trastuzumab + taxanes (Docetaxel); all first 3 groups ended with a long trastuzumab treatment; finally, the fourth group (respectively the fifth) gathered patients with an early (respectively extremely early) treatment withdrawal after surgery, regardless of the administered drug.
All in all, prior to the Katherine clinical trial comparing trastuzumab emtansine to trastuzumab alone among patients who have residual invasive breast cancer after treatment, 16 3 quarters of patients were treated in the adjuvant setting, half followed an anthracyclines/taxanes-based treatment protocol and about 40% received no anthracyclines at all. More than half and more than 3 quarters of patients in the adjuvant and neoadjuvant cohorts were sequentially treated with chemotherapy alone then trastuzumab ± chemotherapy, respectively.
It was observed that 4% of adjuvant patients experienced a temporary trastuzumab treatment withdrawal, and 4% and 5% of adjuvant and neoadjuvant patients experienced an extremely early permanent withdrawal, respectively. Temporary and definitive trastuzumab withdrawal probably have different causes. Trastuzumab, in combination with taxanes, may have cardiotoxic effects and should be avoided in patients whose cardiac function deteriorates during therapy. 5 Hence hospital admissions with cardiac events in the study population were further investigated. Indeed, patients with at least 1 hospital admission for a cardiac event were more frequent in the groups with temporary or early permanent treatment withdrawal and the most frequent diagnoses were left ventricular failure and congestive heart failure. Beyond cardiac toxicity, other reasons for an early withdrawal may include omission, HER2-status overturn following the laboratory examination of the resected tumor, patient preference, and loss of follow-up (when a patient moves out of the country).
Three percent of neoadjuvant patients did not receive any trastuzumab prior to surgery and did receive some after the surgery. The HER2-status of the tumor might have been reconsidered following its analysis.
Implications of the findings
The treatment sequences were in line with international treatment guidelines in force in 2016. 5 For instance, it is recommended to initiate chemotherapy within 6 weeks after surgery. 17 In our study, 58% and 92% of patients of the adjuvant and neoadjuvant cohorts had started their treatment within this timeline, respectively. Likewise, it is recommended to administer trastuzumab for 12 months and the mean trastuzumab treatment duration was 11.7 months. Nevertheless, some patients were treated with trastuzumab for <11 months even though adjuvant trastuzumab lowers the hazard of death by about one-sixth to one-third, 5 uncovering possible deviations to the recommendations. Conversely, 3% of neoadjuvant patients were administered trastuzumab for more than 15 months. These patients might be treated for a relapse, or for 12 months after surgery and had already been treated prior to surgery, or this could be the consequence of the grace period set to 21 days (ie, patients treated every 28 days instead of 21 days).
Less than 2% of patients were hospitalized with cardiac events. As seen in a pooled analysis of 3 trials with trastuzumab used as an adjuvant, 18 those events did not systematically prevent completing the 12-month trastuzumab treatment.
The number of male breast cancer cases is low.2,3 As a matter of fact, in our study, only 0.3% of the patients were males. All of them were in the adjuvant cohort, which is consistent with the fact that male patients have a total mastectomy and do not need trastuzumab treatment to decrease the tumor size prior to the surgical procedure.
Strengths
Our study has several strengths. First, an exhaustive, national, hospital-based database was used, allowing to present robust results, in the real-world setting. Furthermore, the large study population made analyses by sub-groups (adjuvant and neoadjuvant) possible and included male breast cancer patients (often disregarded due to the low number of cases). Second, in the database, trastuzumab is captured in the dedicated list of expensive drugs administered by hospitals, granting an accurate identification of the HER2+ breast cancer population. Third, the retrospective data collection prevents patient selection bias. Then, the long follow-up (18 months after surgery) permitted to study the patients’ hospital-based therapy until the end of their trastuzumab treatment. Last but not least, the machine learning enabled to unambiguously identify treatment sequences which were consistent with the clinical practice and offered a comprehensive, understandable, and precise picture of the treatment sequences. It further offers a visual and educational support for the patients to take ownership of their treatment.
Limitations
However, the results also must be interpreted bearing in mind the study limitations. Some limitations pertain to the database. First, the PMSI—a claims database—contains healthcare consumption but few medical information. In particular, it contains no clinical outcomes, imaging tests’ results, reason for treatment change or withdrawal, all of which would have enabled to confirm the HER2+ early breast cancer status and would have supported the clinical interpretation of our results. However, cardiac events were investigated to aid the interpretation of early treatment withdrawal. Next, cardiovascular death was not accounted for because cause of death is not available. As stage at diagnosis is not recorded, it was not possible to know why patients were allocated to neoadjuvant or to adjuvant treatment. Then, outpatient treatments (anti-hormonotherapy, oral treatments)—all missing in the PMSI—and radiotherapies could not be examined. Drug dispensing (subcutaneous or intravenous) was not examined. Therefore, this study only offers a partial view of the global therapeutic management of early HER2+ breast cancer patients treated with trastuzumab. Given that standard chemotherapies are not reported, their naming is based on the examination of the frequency of their dispensing, names of the drugs administered concomitantly or consecutively, and expert knowledge. When a patient leaves the country or is transferred to a nursing home, the information is not recorded in the PMSI. Consequently, patients are interpreted as having stopped their treatment in our study while they might have continued their treatment abroad or at the nursing home. Only deaths occurring at the hospital are reported in the PMSI. Therefore the number of deaths is underestimated in this study. Although death due to early HER2+ breast cancer is rare (18-month survival of early breast cancer HER2+ patients is close to 100% 6 ), 70% of breast cancer deaths (all stages) occur at the hospital, 19 and 85% of breast cancer patients who die are admitted to the hospital during the last month of their life, it is possible that some treatment withdrawals are due to uncaptured death. In addition, other limitations are specific to this study. First, patients with a cancer progressing to metastatic cancer within 12 months of the surgery were excluded from the study. Thus, short trastuzumab treatments are underrepresented in our results. Conversely, patients starting trastuzumab as late as 63 days after surgery were excluded if there was no chemotherapy in-between (“isolated session”). Consequently, the time to treatment initiation after surgery is slightly underestimated. At the time of the study, pertuzumab (in combination with trastuzumab) was restricted to metastatic breast cancer patients. Consequently, we excluded patients treated with pertuzumab. If early breast cancer patients were treated with pertuzumab for an early breast cancer—off-label use—they were not analyzed. Then, although based on clinical expertise, some parameters in the machine learning method are subjective (eg, the size of the TAK filter and the number of clusters) and inevitably impact the typology of treatment sequences. Lastly, the results only apply to clinical practices around 2016, new treatments have since then been released (notably trastuzumab emtansine).
Conclusion
The analysis of the complete population of early stage HER2+ breast cancer patients treated with trastuzumab in France in 2016 indicated that the treatment sequences of most patients were in line with the recommendations in force at that time. Nevertheless, few patients withdrew early from trastuzumab treatment and some patients were treated with trastuzumab for <11 or >15 months. The proportions of patients with at least 1 hospital stay for a cardiac event were higher in the groups with early treatment withdrawal.
The unsupervised machine learning approach enabled the identification of 4 and 3 typical treatment sequences in the adjuvant and the neoadjuvant cohorts, respectively and 2 treatment sequences similar to the typical treatment sequences but with a treatment withdrawal. This methodological approach successfully offered a telling visual display of the results, on a large and exhaustive patient population, albeit making no compromise on the precision of the treatments’ durations. It provides a meaningful snapshot of treatment sequences, thereby allowing healthcare professionals, health authorities as well as patients and care givers to see the whole picture of the hospital-administered drug strategies.
Supplemental Material
sj-docx-1-cix-10.1177_11769351221135134 – Supplemental material for Innovative Approach for a Typology of Treatment Sequences in Early Stage HER2 Positive Breast Cancer Patients Treated With Trastuzumab in the French National Hospital Database
Supplemental material, sj-docx-1-cix-10.1177_11769351221135134 for Innovative Approach for a Typology of Treatment Sequences in Early Stage HER2 Positive Breast Cancer Patients Treated With Trastuzumab in the French National Hospital Database by Olivier Tredan, Marie Laurent, Melina Gilberg, Rim Ghorbal, Alexandre Vainchtock, Joannie Lortet-Tieulent, Martin Prodel and Julien Dupin in Cancer Informatics
Footnotes
Acknowledgements
We thank Majda Le Foll-Elfounini, who works for Aixial on behalf of Roche, for project coordination. We thank Gwendoline Poinsot for extracting and curating the data and Baptiste Jouaneton for supervising the data management. Both Ms. Poinsot and Mr. Jouaneton work for HEVA, the company hired by Roche to carry out the study.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Roche.
Declaration Of Conflicting Interests:
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: OT reports grants and personal fees from Roche and MSD-Merck; grants from BMS; personal fees from Novartis-Sandoz, Pfizer, Lilly, Astra-Zeneca, Daiichi Sankyo, Seagen, Pierre Fabre, and Eisai, outside of the submitted work. JD, MG, and RG are Roche employees. AV, JLT, MP, and ML are HEVA employees, the company hired by Roche to carry out the study.
Author Contributions
The study was initiated by AV, JD and MG. OT and RG were members of the Steering Committee which oversaw the implementation of the study. Data extraction and analyses were performed by ML and MP. All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data. ML, JD and MP designed the statistical analysis plan in accordance.
All eight authors contributed to the interpretation of the study and to the decision to publish the results. Preparation of the study manuscript was coordinated by JLT and the writing was shared and revised among authors based on their field. All authors were involved in drafting the article or revising it critically for important intellectual content and gave final approval of the version to be published. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Data Availability
The data supporting the study findings are part of the National health data system (SNDS, Système national des Données de Santé) and are available from the HDH (Health Data Hub ![]() ). Restrictions apply to the availability of these data, which were used after agreements signed with the French data protection authority (Comité National de l’Informatique et des Libertés, CNIL) and the Technical Agency for information on hospitalization (Agence technique de l’Information sur l’Hospitalisation, ATIH) for this study.
). Restrictions apply to the availability of these data, which were used after agreements signed with the French data protection authority (Comité National de l’Informatique et des Libertés, CNIL) and the Technical Agency for information on hospitalization (Agence technique de l’Information sur l’Hospitalisation, ATIH) for this study.
Ethics Approval and Patient Consent
Access to the data fell under the 006 framework (MR-006) developed by the French data protection authority (Commission Nationale de l’Informatique et des Libertés, CNIL). Institutional review board is not required under the 006 framework. The data controller’s certificate was approved by the CNIL on September 11, 2018 (No. 2206508 v0). The data controller and the Technical Agency for information on hospitalization (Agence technique de l’Information sur l’Hospitalisation, ATIH) signed an agreement to use the PMSI data on October 2, 2018. In accordance with the regulations in force, patient consent was not necessary because this study used secondary data, there was a public interest in assessing, for the first time to our knowledge, cancer treatment sequences in the population of HER2 positive early breast cancer patients treated with trastuzumab in France in a real-life study, and the protection of patients’ rights and freedom were guaranteed at all times.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
