Abstract
Bisphenol-A (BPA) is a synthetic chemical that has widely been used in the production of polycarbonate plastic and epoxy resins in the manufacture of consumer products. The most common path of human exposure to BPA is by oral intake that involves genotoxicity, oxidative stress, endocrine disruption, mutagenicity, and carcinogenicity in both in vitro and in vivo models. Melatonin is known as a free-radical scavenger and a powerful antioxidant agent. This study aimed to investigate the effects of melatonin on viability and genetic disorders of normal Human Gingival Fibroblasts (HGF), colon cancer (MKN45), and bone marrow stem cell (MSC) lines exposed to BPA. For this purpose, MTT and Comet assays were performed to evaluate the cytotoxicity and genotoxicity properties of BPA and the role of melatonin. The results showed that BPA exposure resulted in increased oxidative stress parameters including MDA and ROS, and decreased GSH content. The current study demonstrated the cytotoxicity and genotoxicity effects of BPA and the protective role of melatonin in preventing cytotoxicity and DNA damage are induced by BPA.
Introduction
Bisphenol-A (BPA: 2,2-bis(4-hydroxyphenyl) propane) is a synthetic chemical and a monomer, plasticizer, and phenolic compound. It has widely been used in the production of polycarbonate plastic and epoxy resins in the manufacture of consumer products, such as food cans, beverage containers, baby feeding bottles, medical tubing, toys, water pipes, dental sealants, eyeglass lenses, several paper products and adhesives, protective coatings, powder paints, automotive lenses, protective window glazing, building materials, compact disks, optical lenses, thermal paper, paper coatings, and encapsulation of electrical and electronic parts.1-7 BPA is a xenoestrogen that can trigger distinctive estrogen-signaling pathways with potential consequences on human health. Furthermore, the irreparable health effects of BPA are related to early exposure during gestational and neonatal periods. 8 The prevalent use of BPA in the manufacture of consumer products led to its detection in samples from dietary products, human fluids, water, dust, sewage, and indoor, outdoor air, and environmental media.6,9 BPA is released by food and beverage containers, especially when heated, thus human exposure has been widespread and continuous through the food chain.1,2,7 The major responsible factor for exposure to BPA in more than 90% of all humans at all age groups is through the ingestion of contaminated food. Therefore, tissues from the upper gastrointestinal tract are exposed to non-conjugated BPA (active form), 10 and the most common way of human BPA exposure has been supposed to be by oral intake, but exposure may occur through inhalation during production in the workplace. 7 Given its prevalent detection in human life, EPA ranks BPA at the third-highest Toxicological Priority Index (ToxPi) among the 309 environmental chemicals using the ToxCast program. 9 This matter resulted in the imposition of a ban on the use of BPA in child care products, including feeding bottles in the European Union, the United States, and Canada, and it has also been banned in thermal receipt papers in Japan.3,9 Demand for this chemical agent has steadily been growing in different industries over recent years. Thus there is a rising concern about BPA properties, including genotoxicity, oxidative stress, endocrine disruption, mutagenicity, and carcinogenicity, which have been figured out by in vitro and in vivo models. 11 In recent years, BPA has received a great deal of public and researchers’ attention because of its potential association with a variety of toxicities and adverse health effects that make the scientific society work on the prevention and protection paths of BPA adverse effects.1,3,11 BPA exposure increases the MDA concentration and decreases the GSH content, which are known as an indication of increased generation of reactive oxygen species (ROS), leading to lipid peroxidation. 1 Experiments on cultured cells and laboratory animals indicated the accumulation feature of BPA and its involvement in several vital organ functions, including the testis, brain, heart, liver, and pancreas. The findings also demonstrate the relation of oxidative stress and mitochondrial dysfunction to the adverse effects of BPA.12,13 Melatonin (N-acetyl-5-methoxytryptamine) has been known as an indoleamine that is mainly released by the pineal gland.4,5 Being a free radical scavenger is one of its important features along with significant antioxidant activity.5,11 The most toxic free radicals and the hydroxyl radical are promptly scavenged by this agent that directly scavenges the peroxynitrite anion, nitric oxide, singlet oxygen, and the peroxyl radical. Melatonin provokes the mRNA amount of antioxidant enzymes including superoxide dismutase. Other antioxidant enzymes, including glutathione peroxidase and glutathione reductase, are also agitated by melatonin. Other activities by melatonin involvement include the inhibition of pro-oxidative enzymes, nitric oxide synthase, chelating transition metal ions, prevention of cell membrane deterioration, and reduction of lipid peroxidation. 11 The protective feature of vitamin C and N-acetylcysteine against BPA-induced toxicity has been reported in a previous studies. 11 In this study, therefore, melatonin was chosen as a protective agent due to its potential antioxidant activity. This study aimed to investigate the melatonin protective role against BPA-induced cytotoxicity and genetic toxicity in three cell lines including colon cancer cell lines (MKN45), normal gingival cell lines (HGF), and bone marrow stem cell lines (MSC).
Materials and methods
Chemicals
BPA, Melatonin, Methanol, MTT dye, Potassium chloride (KCl), and Hydrochloric acid (HCl) were purchased from Merck-KGaA Chemical Company. DMSO, phenazine methosulfate (PMS), Penicillin/streptomycin, fetal bovine serum, L-glutamine, phosphate-buffered saline (PBS), and DMEM culture medium were procured from Sigma-Aldrich Chemical Company. All chemical reagents were of the highest and purest grade available.
Cell culture
In this study, three cell lines, including Normal Human Gingival Fibroblasts Cell Line (HGF), Colon Cancer Cell Line (MKN45), and Bone Marrow Stem Cell line (MSC) were obtained from the Pasteur Institute of Iran cell bank, Tehran. These cells were grown in the DMEM medium at 37 °C and 5% CO2.
Concentrations of the tested melatonin and BPA
Four concentrations of melatonin (12, 23, 46, and 93 µg/ml) and BPA (0.5, 5, 50, and 100 µg/ml) were prepared here.
Cytotoxicity assay
The MTT assay is one of the popular methods in evaluating cell viability. This assay is based on the reduction of MTT, a yellow water-soluble tetrazolium dye, primarily by mitochondrial dehydrogenases to purple-colored formazan crystals. The formazan product was dissolved in DMSO and then analyzed spectrophotometrically using an ELISA reader at 570 and 630 nm.14-17 The cell viability was studied through MTT test after 24 and 72 h exposure of the cell lines to BPA at the mentioned concentrations. Cisplatin and cell culture medium were considered as a positive and negative control groups, respectively. In the proses of melatonin effect investigation, BPA (50 µg/ml) was considered as a positive control. 50µg/ml of BPA was used for cytotoxicity induction followed melatonin exposure.
Analyses of genotoxicity induction by the comet assay
DNA strand break induction was assayed by the comet assay as described by von Bardeleben et al 18 . Comet assay is used to evaluate two dependent variables: global DNA damage and oxidative damage –a particular type of DNA damage caused by the oxidation of nucleotides. Comets were visualized by microscopy and quantified by the determination of the ‘Olive tail moment’ (OTM) using computer-based software (Komet4.02, Kinetics Imaging, UK). Fifty cells were analyzed per measurement for the calculation of the mean value.7,10,19 The genotoxicity of BPA was determined by the comet assay. cell culture medium was used as a control group for evaluating the cell genotoxicity of BPA. For evaluating of the melatonin effect, 50µg/ml of BPA was used for genotoxicity induction followed melatonin exposure. BPA (50 µg/ml) was considered as a control group. All experiments were performed in triplicate.
Biochemical determinations
The protocol of Buege and Aust 20 was used to estimate lipid peroxidation. The lipid peroxidation product, malondialdehyde (MDA), was measured by the thiobarbituric acid assay, which is based on the MDA reaction with thiobarbituric acid to give thiobarbituric acid reactive substances (TBARS), a red species that is absorbed in 530 nm. The MDA level was reported in the values of moles/ mg protein.9,11,21,22 GSH was estimated with minor variations in the protocol by Jollow et al 23 . The supernatant was mixed with 0.1 M potassium phosphate buffer (pH 7.4), the reaction was initiated by the addition of DTNB, and the absorbance was measured at 412 nm. GSH levels were reported in the values of moles/mg protein. The calculations were made using the molar extinction coefficient of 1.36×104M−1 cm−1.9,24 DCFH-DA was used to assay the production of intracellular ROS. DCFH-DA is a non-fluorescent, non-polar, and electrically neutral compound and can easily enter the cell by passing through the membrane. The intracellular ROS oxidizes the DCFH to a highly fluorescent DCF. The treated PBMCs were incubated with 20 μM of DCFH-DA at 37 °C for 1 h. To determine the increase in ROS, the fluorescence was recorded at the emission wavelength of 530 nm using 485 nm as the excitation wavelength.9,25 During of the ROS, GSH and MDA estimation proses in the BPA-exposed cell lines, cell culture medium was used as a control group; and for the evaluating of melatonin effect on ROS, GSH and MDA amounts, BPA (50 µg/ml) was considered as a control group.
Statistical analysis
Data are presented as the mean ± SD (standard deviation). The statistical analyses were performed by one-way analysis of variance (ANOVA) followed by the Tukey’s test using SPSS 17 software at a statistical significance of P < 0.05.
Results
Cell viability
The effects of BPA on the viability of HGF, MKN45, and MSC cells were evaluated using the MTT assay. After 24 h exposure, BPA caused a decrease in the viability of the cell lines. As shown in Figure 1, there was a significant difference (P < 0.00001) in the cytotoxicity of BPA on cells at 5, 50 and 100 μg/ml in comparison with the cell culture medium as a control group. In spite of the BPA cytotoxicity on MKN45 cell line at 0.5 μg/ml concentration, there wasn’t significant difference between control group and BPA at 0.5 μg/ml concentration in HGF and MSC cells. The MTT assay was also used to evaluate the role of melatonin in BPA cytotoxicity on the cell lines. Melatonin at all concentrations (12, 23, 46, and 93 μg/ml) significantly (p < 0.0001) enhanced cell viability compared to the BPA group as a control (Figure 1). All cell lines were exposed to different melatonin concentrations and 50 µg/ml of BPA to study the effect of melatonin.

The cytotoxicity of BPA and melatonin effect on BPA-induced cytotoxicity on stem cells, HGF, and MKN45 cell lines.
Comet assay
Comet assay is a sensitive method for detecting DNA strand breakage at the level of an individual cell. The assay was run on HGF, MKN45, and MSC cells exposed to four concentrations of BPA and melatonin. Figure 2 shows the tail moment obtained through the comet assay on cells. A significant increase in the tail moment suggested DNA damage or fragmentation, which was significant (p < 0.0001) on all cells at 5, 50 and 100 μg/ml concentrations of BPA, indicating BPA genotoxicity. In spite of the increase in the tail moment of BPA on MKN45 cell line at 0.5 μg/ml concentration, there wasn’t significant difference between control group and BPA at 0.5 μg/ml concentration in HGF and MSC cells. The comet assay was also used to investigate the influence of melatonin on BPA-induced genotoxicity at 4 concentrations. According to Figure 2, melatonin at 23, 46, and 93 μg/ml could significantly (P < .0001) decrease the tail moment on all cell lines in compassion to the control (BPA 50 μg/ml). A significant (P < .01) decrease in the tail moment was also observed in the MKN45 and HGF exposed to 12 μg/ml of melatonin. A significant (P < .001) decrease in the tail moment was also observed in the MSC cells exposed to 12 μg/ml of melatonin. An increase in the concentration of melatonin increased its genotoxicity-decreasing effect.
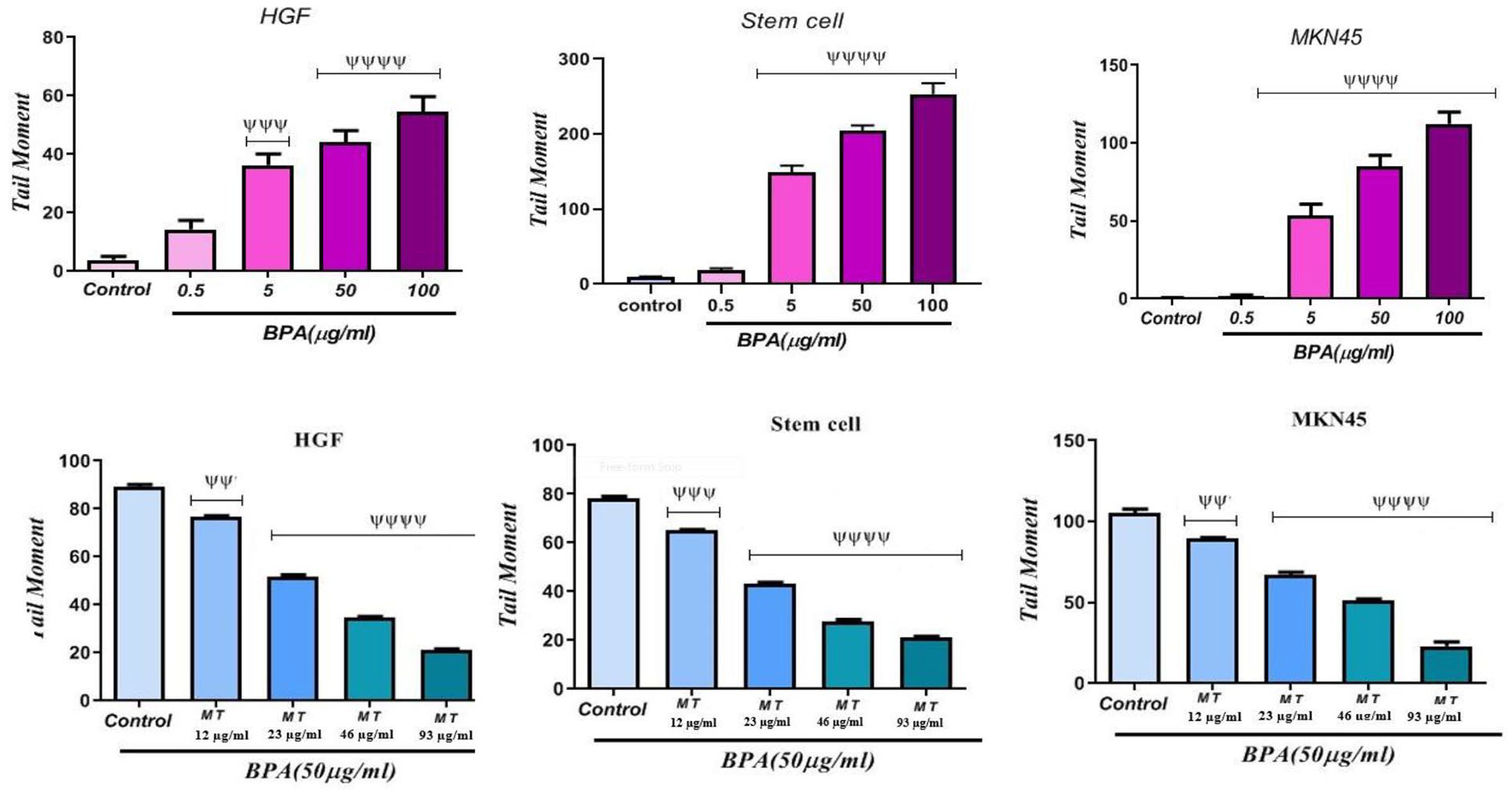
The genotoxicity of BPA and melatonin effect on BPA-induced genotoxicity on HGF, stem cells, and MKN45 cell lines.
ROS detection
ROS production increases more than their intracellular threshold concentrations due to external stimuli resulting in oxidative stress. To understand whether the cytotoxicity of BPA was due to the induction of oxidative stress in HGF, MKN45, and MSC cells upon their exposure, intracellular ROS detection was carried out using DCFH-DA as a probe. According to the data (Figure 3), ROS content significantly (p < 0.0001) rose in the cell lines exposed to BPA at 5, 50, and 100 µg/ml compared to the control group, indicating its cytotoxic effect. BPA at 0.5 µg/ml increased significantly (p < 0.01) the amount of ROS in comparison with the control group, suggesting that BPA can exert its cytotoxicity through oxidative stress. Figure 3 also shows the decrease in the percentage fluorescence intensity of DCF upon treatment with melatonin on cells exposed to BPA. It demonstrated a steady decrease in intensity and a significant (p < 0.0001) decrease at different concentrations (23, 46, and 93 µg/ml) of melatonin compared to the control group that only exposed to BPA (50 µg/ml). Melatonin at 12 µg/ml also decreased significantly (p < 0.01) ROS levels in comparison with the control group, which proves its protective effect against BPA-induced cytotoxicity.

The effects of BPA and melatonin on ROS content in the stem cell line, HGF, and MKN45 cell lines.
Estimation of GSH and lipid peroxidation
Glutathione is known as an important free radical scavenger that is mainly involved in the detoxification process, including suppressing ROS; a decrease in GSH content and an increase in MDA levels indicate an oxidative stress induction. A major point concerning ROS is its reaction with vital macromolecules (e.g. lipids) that results in their oxidation to MDA leading to damage to membrane lipids. 8 GSH and MDA levels were estimated to confirm whether BPA was able to induce oxidative stress. Figures 4 and 5 show the effect of BPA on the status of GSH and MDA content of BPA-exposed cell lines. There was a significant depletion in GSH content and a rise in MDA level in the BPA-exposed cells at 5, 50, and 100 µg/ml (p < 0.0001). These data suggest that BPA-induced ROS-mediated oxidative stress was responsible for the mitigation of GSH content and an increase in the levels of MDA. 8

The effects of BPA and melatonin on GSH content in the stem cell line, HGF, and MKN45 cell lines.

The effects of BPA and melatonin on MDA levels in the stem cell, MKN45, and HGF cell lines.
Figures 4 and 5 depict the influence of melatonin on the depletion of GSH content and the rise of MDA levels caused by BPA. It was found that GSH content significantly (p < 0.0001) increased in cell lines exposed to 23, 46, and 93 µg/ml of melatonin compared to the control (BPA) group. Melatonin at 12 µg/ml also increased significantly (p < 0.05) the GSH content in comparison with the control group. Results of melatonin evaluation on MDA levels showed a significant (p < 0.0001) decrease in cell lines exposed to 23, 46, and 93 µg/ml of melatonin in comparison with the control group. The melatonin effect at 12 µg/ml was not significantly different in comparison to the control group.
Discussion
BPA as an endocrine disruptor is a monomer of polycarbonate plastic and a constituent of epoxy and polystyrene resins. 11 Human exposure to BPA is widespread due to various uses in consumer products. 2 BPA is released by food and beverage containers thus human exposure is prevalent and continuous through the food chain. 1 Factors involving in BPA toxicity include lipid peroxidation and the generation of free radicals causing oxidative stress. 11 The unique features of melatonin, including low toxicity and the ability to cross biological barriers, enable it to prevent oxidative damage to DNA hence it is known as a free-radical scavenger that can neutralize diverse threatening factors.1,11,26 This study investigated melatonin effects on BPA-induced genotoxicity and cytotoxicity on MKN54, HGF, and stem cell lines. Here, melatonin was used as a protective agent against BPA-induced genotoxicity and cytotoxicity considering potential antioxidant activity and protective role of melatonin.
Data obtained from the present study showed that BPA diminished cell viability percentage due to its cytotoxic effect. Melatonin was also able to increase the viability percentage of cell lines exposed to BPA. Results of the comet assay showed that there was a significant increase in the tail moment suggesting DNA damage on cell lines exposed to BPA. On the other hand, the influence of melatonin on BPA-induced genotoxicity showed that it could significantly decrease the tail moment on cell lines exposed to BPA. The determination of biochemical factors illustrated that BPA disrupted the biochemical agents as evidenced by elevated lipid peroxidation and ROS, and decreased GSH content. In the current study, melatonin significantly modulated the biochemical factors in the cell lines exposed to melatonin + BPA. This was verified by a decrease in the levels of MDA and ROS and an increase in the levels of GSH in the melatonin+ BPA-exposed cell lines compared to the BPA-exposed groups. The anti-oxidative and scavenging properties of melatonin enable it to protect DNA and lipids from oxidative agents such as BPA. 1
An in vitro study by Ikhlas et al 9 approves our results. They used the MTT test to assess cytotoxicity by measuring a reduction in cell viability due to apoptosis or necrosis . Our results are also in line with a previous report of Hercog et al. 17 They showed the BPA genotoxic potential and its analogs, such as BPS, BPF, and BPAF, as well as their mixtures, their effects on the expression of selected genes involved in xenobiotic metabolism, response to oxidative stress, and DNA damage on HepG2 cells.
Consistent with our cytotoxicity and comet assay results, another study was done by Ramosa et al 10 on cytotoxic and genotoxic effects of environmentally relevant concentrations of BPA and interactions with doxorubicin. Their comet assay results emonstrated DNA damage as a consequence of Hep-2 cells exposure to BPA.
The results of determining biochemical factors are consistent with a previous report of El-Missiry et al 1 They claimed that melatonin ameliorated oxidative stress, modulated death receptor pathway proteins, and protected the rat cerebrum against BPA-induced apoptosis. Their results suggest that melatonin owns unique physical and chemical properties among several antioxidants that enable it to cross easily the biological membranes and reach the cytosol, nucleus, and mitochondria.
Similar to our results, Kobroob et al 12 conducted a study entitled Damaging Effects of Bisphenol A on the Kidney and the Protection by Melatonin: Emerging Evidence from in Vivo and in Vitro Studies. They figured out that BPA exposure not only increased the oxidant molecule nitric oxide (NO) but also decreased the levels of antioxidants glutathione (GSH) and superoxide dismutase enzyme (SOD) in the kidney tissues. Consistent with the current study, BPA has been shown to stimulate oxidative injury in a variety of cells and organs both in vitro and in vivo experiments . According to Anjum et al 11 melatonin ameliorates bisphenol A-induced biochemical toxicity in testicular mitochondria of the mouse, which approves our findings. It was observed that BPA treatment significantly affected GSH as a vital agent in cell viability. It is expressed that GSH either alone or in conjugation with other proteins can protect the cell against lipid peroxidation. The results revealed that melatonin as a potent antioxidant could ameliorate the BPA-induced oxidative stress. They also showed that the melatonin function as a free radical scavenger might be related to its electron-donating ability. It has been reported that long-term melatonin intake leads to an increase in the number of mitochondria in cells.
The results of the present study are consistent with those of Ikhlas et al 9 on evaluating the cytotoxicity of BPA analogs based on their oxidative and genotoxic potential using human peripheral blood cells. Although BPA genotoxicity is conflicting in nature, some studies report that BPA is able to induce DNA damage in mouse cell lymphoma cells, MCF-7 cells, and chicken DT40 cells while some reports indicate its non-genotoxic function on Chinese hamster ovary cells, intestinal cell lines (LS174T), hepatoma cell lines (HepG2), and renal cell lines (ACHN). Our findings are also in agreement with those of Wu et al, 7 who studied DNA damage by the effect of BPA and melatonin treatments on DNA damage in male germ cells. They observed a significant increase in the comet parameters in the pachytene spermatocytes of rats exposed to BPA. Moreover, they suggested that metabolites of BPA might react with DNA, especially when the GSH level is low in the cell. In another study, the histomorphological, immunohistochemistry, and TUNEL analyses of the cultured tests were investigated by Zhang et al. 27 ordered al. to examine whether the addition of melatonin to the culture medium would prevent the adverse effects of BPA on the testis. Their results showed that 10 μM of melatonin exerted a protective function against BPA, which is in agreement with our result about the melatonin protective effect on cell lines exposed to BPA. Likewise, Park et al 28 investigated the anti-oxidative effects of melatonin against BPA-derived superoxide on oocyte maturation in pigs. The melatonin protective effect was certified concerning superoxide production upon BPA exposure during oocyte maturation . Our results are also supported by those of Abdullah and Rashid. 29 on evaluating BPA-induced oxidative stress in human RBCs in vitro and its amelioration by melatonin. They figured out that BPA exposure of red blood cells caused an increase in oxidative stress, while melatonin decreased the BPA-induced oxidative stress in these cells.
Findings of Neslihan Akçay et al 30 on the effects of melatonin on possible damage to adipocytokines and liver tissue by coadministration of fructose and bisphenol a (BPA) are in agreement with our data. They stated that co-administration of BPA and fructose caused an increase in lipid peroxidation level due to the increase of oxidative stress. On the other hand, melatonin treatment induced antioxidant enzyme activity and reduced lipid peroxidation levels. In the study on alterations of Kiss 1 receptor, GnRH receptor and nuclear receptors of the hypothalamo-pituitary-ovarian axis following low dose bisphenol-A exposure in Wistar rats which was done by Kadir et al 31 ; it is figured out that melatonin antioxidant effects give some promising outturns against bisphenol-A induced toxicities which is in agreement with our result about the melatonin protective effect.
Our finding is also in agreement with Ishtiaq et al, 32 who suggested that melatonin alleviated oxidative stress and mitochondrial dysfunction–linked apoptosis, rendering its neuroprotective potential against BPA-induced toxicity, through investigation on Melatonin abated Bisphenol A–induced neurotoxicity via p53/PUMA/Drp-1 signaling.
Conclusion
The present study provides convincing evidence to prove that BPA has a detrimental impact on the stem cell lines, HGF, and MKN45 cells. The current study confirmed that BPA exposure resulted in increased oxidative stress factors, including MDA and ROS, and decreased GSH content. BPA exposure also induced lipid peroxidation demonstrating the vulnerability of the mitochondrial membrane. BPA clearly showed cytotoxicity in the three cell lines. Investigation of certain oxidative stress parameters showed that it induced ROS overproduction and lipid peroxidation and caused depletion of GSH levels. Therefore, we conclude that BPA can induce cytotoxicity via oxidative stress and genotoxicity. This study demonstrated the melatonin protective effect on BPA-induced cytotoxicity and genotoxicity. Melatonin could also abate BPA-induced ROS and MDA increase and relieve DNA damage. On other hand, melatonin could compensate for the GSH decrease induced by BPA exposure. In summary, we emphasize the importance of further investigations on the beneficial qualities of melatonin due to its unique features, including being a very nontoxic, inexpensive, readily available, and potentially protective pharmacologic agent for preventing cytotoxicity and DNA damage induced by toxic chemicals, especially BPA.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration Of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MS designed and directed the project; NGB and RE performed the experiments; RE wrote the article.
