Abstract
The follicular thyroid carcinoma (FTC) and follicular thyroid adenoma (FTA) are malignant and benign thyroid neoplasms, respectively. MicroRNA (miRNA) expressions have been touted as an indicator for prognostic outcome in thyroid cancer. The study objective was to explore genes suppressed by miRNA-21-3p and miRNA-21-5p for potential therapeutic insights. Differentially expressed genes and their functional enrichment were obtained from 25 FTA and 27 FTC gene microarray dataset GSE82208 using R and Bioconductor tools. The miRNA target sites were obtained from miR-TarBase database. A unique gene list of differentially expressed FTC and FTA were entered into miR-TarBase database to obtain target genes for both miRNA-21-3p and miRNA-21-5p. The result showed that miRNA-21-3p and miRNA-21-5p downregulated TIMP3, MAT2A, TGFBR2, and PLAT gene in FTC and FTA leading to significant expression of acute phase-response to metallothionein, metal ions, and unfolded protein response (UPR). The computational analysis suggests that the suppression of miRNA-21-3p and miRNA-21-5p could be an intervention strategy for therapeutically targeting FTC and FTA treatments.
Introduction
Cancer of the thyroid gland is the most common tumor of the endocrine system with a global increasing prevalence among all age groups. 1 Knowledge of thyroid cancer is still limited, and this is a concern for diagnosis and prognosis of the disease as it has several histology with different characteristics, prognoses, and cellular origin. 2 The parafollicular C cells and follicular thyroid cells are two types of endocrine thyroid cells from where thyroid cancers originate. 2 Generally, a proportion of lumps found within the thyroid (thyroid nodules) is established to be cancerous with in the global population, but the difficulty is in finding and therapeutically targeting them and in differentiating the cancerous ones from other tumor types. 3 For example, differentiating follicular thyroid carcinomas (FTC) from follicular thyroid adenomas (FTA) is not an exciting task because they are not very different genetically and histologically. 3 Moreover, the cause and origin of follicular thyroid neoplasms is limited, making accurate detection and distinguishing characteristics of the neoplasms challenging. 4
Studies have highlighted that classifying if a thyroid neoplasm is malignant or benign is still a challenge as several molecular markers are being investigated for accuracy in distinguishing the thyroid nodules into different tumor types. 5 FTA is a benign, oval, rubbery or firm encapsulated tumor of the thyroid gland; it is considered to be the thyroid neoplasm with tumor invasion that does not span the entire capsule. 6 FTC on the other hand is microscopically similar to FTA but tends to have more areas of necrosis, irregularly thick cellular capsule, and frequent mitoses; it accounts for between 25% to 40% of thyroid malignancies in iodine deficient areas. 6 The ratio of FTA to FTC in surgical specimens has been reported to be approximately 5 to 1. 6 In simple terms, FTA are benign thyroid tumors with similar structure to FTC, but unlike FTC, they lack vascular or capsular invasion and are typically encapsulated. 2
Studies invested in identifying differences between FTC and FTA for medical diagnosis are limited by the inability of histopathological sessions to distinguish between them, hence the need for other markers. 7 FTA and FTC have an imprecise classification, 8 and there is the problem of interpreting capsular invasion (i.e. the penetration of the tumor through the entire thick capsule). 7 The histopathological interpretation of capsular or vascular invasion varies and depends on the discretion of pathologists which leads to imprecise classification. 5 This led to calls that classification based on discretion should be discouraged to allow for careful and adequate clinical classification. 8
Consequently, a study to determine whether both FTC and FTA were slightly or absolutely different biological molecular entities using mRNA and miRNA microarray expression data to validate and compare with previously available data by Dom et al 3 found that there was an overlap between FTC and FTA mRNA expression data, indicating slight molecular and biological differences. Gene expression profiles (mRNA) have been largely studied to investigate FTC and FTA differentiation to discover features of thyroid tumors for therapeutic insights; however, the results lack consistent reproducibility. 7 Molecular differences between FTA and FTC have been suggested as a potential reason for this disparity. 7 Apart from mRNA, miRNAs are equally vital in investigating genes participating in FTC and FTA because of their involvement in many biological processes such as apoptosis, disease resistance and pathogenesis, cell differentiation, proliferation, and tumorigenesis. 9 The molecular mechanisms for FTA and FTC remain cloudy due to false-negative results and limited sampling. 9 Since both FTA and FTC are undesirable tumors, it would be beneficial to understand similar genes and molecular mechanisms underlying their interaction.
While several studies have focused on distinguishing between FTA and FTC, it is beneficial to investigate common genes and pathways involved in their pathogenesis to possibly provide further insights on strategies for thyroid cancer intervention. 10 Analyzing specific miRNA in FTA and FTC tumor type is important because their dysregulation may contribute to tumorigenesis. 11 The miRNAs are single stranded short non-coding RNA (19–24 nucleotide long) transcribed from the genome. 12 They regulate the translation of protein-coding genes and can down-regulate expression of a gene by complementarily binding to 3pUTR of the gene and thus degrading the mRNA. 12 Molecular markers have highlighted upregulated and downregulated miRNAs in variants of thyroid carcinoma and differentiated clinical actions of various tumors. 3 Moreover, because miRNAs are highly conserved and have been reported to target up to a third of the human genome, 10 their role in biological processes and cancer is essential. For example, more than 50% of miRNAs genes have been reported in altered chromosomal sites in human tumors, and dysregulated miRNA-21 (also abbreviated as hsa-mir-21, MIRN21, and miR-21) is one of the types abundantly involved in thyroid tumorigenesis. 10 Highly expressed miRNA-21 is one of the earliest miRNAs associated with cancer proliferation and they are considered biomarkers of immune system activation. 13 They are very well expressed in mammalian miRNAs and have been found to negatively regulate tumor suppressor genes and promote cellular apoptosis. 14 Since it has been hypothesized that miRNA deregulation is associated with tumorigenesis, 10 identifying miRNA-21 targeted genes and pathways is fundamental in investigating the implications for thyroid tumors and potential targets for cancer treatments. 14
The aim of the study was to explore genes suppressed by miRNA-21-3p and miRNA-21-5p in FTC and FTA and if therapeutically targeting them could represent goldmines in thyroid cancer intervention strategies. The paper will discuss the methodological approach implemented. Next, bioinformatics analysis of FTA and FTC gene microarray dataset will be carried out to explore genes suppressed by miRNA-21-3p and miRNA-21-5p. The study will conclude with a discussion of the result, the implication, and possible intervention.
Method
Gene microarray dataset (GSE82208, Gene Expression Omnibus) for 25 FTA and 27 FTC patients were obtained from the NCBI database. 15 The microarray processing and statistical and bioinformatics analysis were done using R software and Bioconductor packages. The datasets were normalized and log2 transformation was implemented to reduce the skewness of the microarray distribution. MiRNA target sites were obtained from miR-TarBase database, 16 and the target genes for miRNA-21-3p and miRNA-21-5p were extracted by an intersection with the differentially expressed FTC and FTA unique gene list.
Statistical analysis
The pre-processed gene microarray dataset was fitted to a linear model. The log2 fold change, t-statistics, P-value, and the adjusted P-value was calculated using the Benjamini and Hochberg’s (BH) adjustment method and a threshold of 0.05 was considered as being significant. The log-odds that a gene is differentially expressed (B-statistic) was calculated with 1.5 being the value that a gene have 4.5 to 1 odds of being differentially expressed, calculated as exp.(1.5), and the value of 0 having a 50-50 chance that a gene is differentially expressed. The probe ids were mapped to gene names using the hgu133plus2.db Bioconductor package. Genes with a log fold change (logFC > 0) were upregulated while those with (logFC < 0) were considered downregulated.
Functional enrichment and annotation
The differentially expressed downregulated genes were used to identify activation pathways and genes involved in thyroid cancer from the KEGG pathway (pathway id: 05216) seen in Figure 3, while REACTOME (a pathway database) 17 was used to visualize the molecular pathway knowledge for the genome analysis (Figure 1). Gene ontology of the downregulated target genes in FTC and FTA was used to obtain functional gene annotation and pathway enrichment. The implementation script and steps are archived in GitHub repository. 18
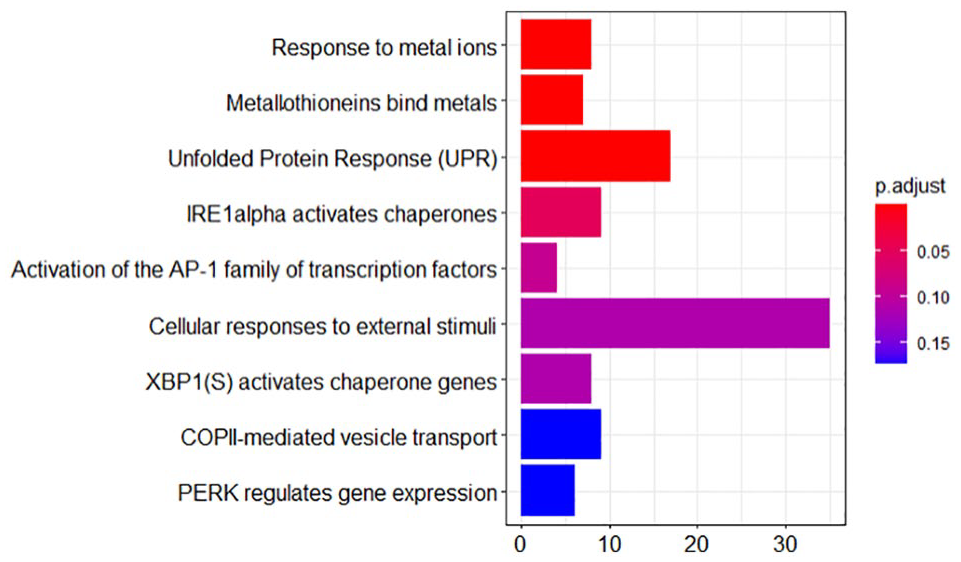
REACTOME pathway: Expression of acute phase-response to metallothionein, metal ions, and UPR in FTC and FTA with the red color indicating significant enriched pathways and blue indicating the least. FTA, follicular thyroid adenoma; FTC, follicular thyroid carcinoma; IRE1, inositol-requiring enzyme 1; UPR, unfolded protein response.
Results
From the analysis pipeline, miRNA-21-3p and miRNA-21-5p downregulated metalloproteinase-3 (TIMP3), methionine adenosyltransferase 2 (MAT2A), transforming growth factor beta receptor II (TGFBR2), and plasminogen activator (PLAT) genes in FTC and FTA leading to significant expression of acute phase-response to metallothionein, metal ions, and unfolded protein response (UPR) seen in Figure 1. The gene ontology (Molecular Function—GO:0005515) showed that the downregulated genes were involved in protein binding.
An overview of the result shows that TIMP3 is an oncogene suppressor that plays a role in inhibiting invasion, angiogenesis, and growth of several cancers. 19 While few studies have investigated TIMP3 in FTC and FTA, it has been reported to be downregulated in papillary thyroid cancer (PTC) but expressed in normal thyroid tissues. 19 In a study of TIMP3 regulation of migration, invasion, and in vivo tumorigenicity of thyroid tumor cells, Borrello et al 20 found that TIMP3 induced reduced metastatic activities of human thyroid cell line NIM1 by repressing angiogenesis and macrophage infiltration in PTC-derived NIM1 cell line, indicating that TIMP3 is involved in a negative regulatory role. 19 MAT2A mRNA level is post-transcriptionally regulated by methylated-HuR and non-methylated-HuR. 21 The non-methylated-HuR is an RNA binding protein that targets mRNAs stabilization and enhancement modulating their translation, thereby regulating gene expression; 22 on the other hand, methylated-HuR inhibits this action. 21 A change to non-methylated-HuR binding of the 3-UTR of MAT2A was reported to result in increased MAT2A mRNA levels in liver cancer. 21 TGFBR2 is a tumor suppressor and an immune-related cytokine, regulating T-cell development, tolerance, and homeostasis; mutation to this gene increases the probability of developing cancer. 23 The PLAT gene is a serine protease responsible for converting proenzyme plasminogen to plasmin. 24 They are expressed in thyroid tissue and are vital in promoting tissue modeling and cell migration. The suppression of their activity can lead to hypofibrinolysis resulting in reduced or obstructed flow of blood. 24
Changes in p53 were associated with FTC while mutations in RAS and RET were associated with FTA as seen in the KEGG pathway (Figure 3). The differentially expressed genes that were downregulated, namely, TIMP3, MAT2A, TGFBR2, and PLAT, are ranked in descending order of silenced expression from left to right. The gene ontology (Molecular Function—GO:0005515) showed that these genes are involved in protein binding.
Figure 2 shows rearrangements of the paired box 8–peroxisome proliferator-activated receptor gamma (PAX8/PPAR) fusion protein (PPFP) and alterations of the RAS gene in FTA, while alterations in p53 and its activating pathway are identified in FTC. This finding is in agreement with previous report by Nikiforov. 25 Figure 3 shows the significant expression of E-cadherin (ECAD) and PPFP in thyroid cancer. ECAD expression is a transcribed transmembrane glycoprotein from CDH1 gene; it is involved in metastasis and invasion in tumorigenesis and have been reported in a study by Zhou et al 26 examining its clinicopathological characteristics and susceptibility to thyroid cancer to be significantly associated with the predisposition to thyroid cancer. Moreover, ECAD expression has also been reported in breast cancer; and suggestions are that it may be a predictive factor in analyzing the clinical progression of thyroid cancer. 26 PPFP genes have been found to be less frequently methylated in benign tissues than in thyroid cancer. 27 They have been associated with increased anchorage-independent growth and invasiveness, and are estimated to be responsible for approximately 35% of FTC. 28

KEGG pathway showing expressed genes and activated pathways in FTA and FTC. FTA, follicular thyroid adenoma; FTC, follicular thyroid carcinoma.

KEGG pathway showing expression of ECAD and PPFP proteins in FTA and FTC. ECAD, E-cadherin expression; FTA, follicular thyroid adenoma; FTC, follicular thyroid carcinoma.
Discussion
MiR-21 inhibits metalloproteinases 3 (TIMP3) which suppresses malignancy and inhibits matrix metalloproteinases (MMPs). 29 The MMP is a proteolytic enzyme that promote tumor growth and vascularization and are involved in the degradation and remodeling of the extracellular matrix (ECM), in cell migration and metastasis. 30 From the results of the study, metalloproteinase-3 (TIMP3) gene was found to be downregulated in both FTC and FTA. TIMP3 have been previously reported to be downregulated in primary thyrocytes infected with RET/PTC1 retroviral vector 20 and hypermethylated in association with BRAF mutation. 31 Hypermethylation of TIMP3 leads to silenced expression and agrees with similar findings on papillary thyroid carcinoma (PTC)-derived tumor cell lines which reported increased reversal upon treatment with a demethylating agent (5-Aza-dC). 19 This suggests that the role of miRNA-21-3p and miRNA-21-5p in thyroid tumorigenesis is associated with TIMP3 silencing.
Methionine adenosyltransferase (MAT) was another downregulated gene and known target for miRNA-21 complementarily binding. The genes expressed by these human cells (MAT1A, MAT2A, and MAT2B) are responsible for encoding enzymes that biosynthesizes S-adenosylmethionine, a precursor for glutathione and polyamines and their dysregulation play a vital role in cancer development. 21 Moreover, they are associated with tumorigenesis—enhancing tumor migration and survival, increased oxidative stress, and genomic instability. 21 While there are few studies linking MAT2A with thyroid cancer, they have been implicated in other cancer types such as breast cancer, prostrate, liver, colon, and gastric cancers. 21 MiRNA 21-3p was reported to suppress MAT2A mRNA levels post-transcriptionally resulting in apoptosis of liver hepatocellular carcinoma (HepG2) cells after induction by the anticancer drug berberine. 21 S-adenosylmethionine and methionine adenosyltransferase protein-protein interactions are being studied for chemoprevention of cancers caused by MAT gene dysregulation. 21 As the result of the study showed that MAT2A was downregulated by miRNA-21-3p and miRNA-21-5p, suppression of miRNA-21-3p and miRNA-21-5p could provide further insight in FTC and FTA treatment.
The polymorphisms of TGFBR2, another differentially expressed gene (DEG) from the results of the study is linked to thyroid cancer as its mutation which leads to the development of papillary thyroid carcinoma (PTC) was reported by Choe et al 23 after finding significant differences in TGFBR2 polymorphism (rs2228048) in PTC conditions with and without extrathyroidal invasion. TGFBR2 loss or its single-nucleotide polymorphisms (SNPs) promote malignant transformation and tumor invasion. 23 Its expression affects cyclooxygenase and prostaglandin-endoperoxide synthase 2 (PTGS2); while its suppression promotes malignant transformation and tumorigenesis. 23 Cyclooxygenase (COX) is an enzyme that catalyzes arachidonic acid into prostanoids which modulate inflammatory activities contributing to inflammatory pain and many other pathological conditions.30,32 Two isoforms of COX exist (COX-1 and COX-2), and they play the same cyclooxygenase role but differ in cell inhibition and expression etcetera. 30 COX-2 is upregulated by p53, induced by tumorigenesis, cytokines, and largely undetectable in normal tissues. 30 Elevated MMP expression and COX-2 have been reported in cancerous tissues compared with normal samples, and COX-2 inhibitors have been touted as a strategy for cancer immunotherapy to reduce indoleamine 2,3-dioxygenase 1 (IDO1) overexpression. 30 IDO1 overexpression is a feature of many cancerous conditions and linked to unresponsive immunotherapy due to lack of infiltration of T-cells in the tumor. 33 Overall, it can be alluded that TGFBR2 downregulation promotes FTA and FTC tumorigeneses, COX-2 overexpression, and its prostanoid product.
In agreement with findings from the computational analysis, PLAT gene was demonstrated to be differentially expressed in human FTC tissues compared with normal tissues in the study by Rudzińska et al 34 on the role of Transcription Factor Prospero Homeobox 1 (PROX1) on angiogenesis in FTC and squamous cell carcinoma of the thyroid gland after PROX1 knockdown. The expression of TIMP3 and PLAT gene transcript have been reported to be significantly suppressed after PROX1 silencing 34 which is consistent with result of this study that showed downregulation of the gene expression with respect to miRNA-21-3p and miRNA-21-5p complementary binding. The PLAT gene is a vital trigger for proteinase cascade resulting in plasmin concentrations and MMPs that are inactivated by TIMPs. 34 In addition, the MMPs are regulated by metallothionein, a metal-binding protein and antioxidant known to be upregulated under oxidative stress. 35 This corroborates with results from the REACTOME pathway that shows significant expression of metallothionein in FTC and FTA gene expression profiles seen in Figure 1. Metallothioneins (MTs) have been reported to induce resistance apoptosis and some chemotherapies as well as cancer cells proliferation suggesting that its targeting could be used in aggressive management of both FTC and FTA. 36 While higher expression of MTs in FTC compared with FTA in differential nuclear cytoplasmic expression has been alluded as a possible clinical distinguishing feature, 36 the study was unable to distinguish between the expression levels which could be as a result of the limitation of a computational analysis.
The UPR was shown to be significantly increased in both FTC and FTA gene expression profiles as seen in the REACTOME pathway. UPR is activated in cells when endoplasmic reticulum (ER) transmembrane protein sensors such as activating transcription factor 6 (ATF6), inositol-requiring enzyme 1 (IRE1), and RNA-like ER kinase (PERK) detects accumulated unfolded proteins. 37 The ER is a vital component of cells for maturation, synthesis, and protein folding; ER stressors play a role in maintaining cell homeostasis. 37 The UPR responds to lower the accumulation of misfolded protein, however, sustained ER stress culminates in a shift in its signaling towards apoptosis as its components/signal transducers (such as PERK, IRE1, and ATF6) have been reported to play a role in ER-mediated apoptosis. 37 The IRE1 catalyzes the induction of genes related to protein folding and is involved in pro-apoptotic factors, while PERK causes translational arrest after undergoing oligomerization and autophosphorylation. 37 PERK also induces eukaryotic initiation factor 2 (eIF2) phosphorylation that activates translation of ATF4 which upregulates pro-apoptotic proteins responsible for DNA damage. 37 Additionally, the gene ontology (Molecular Function—GO:0005515) showed that the downregulated genes in FTC and FTA were involved in protein binding which further correlates with results from the REATOME pathway that showed significant expression of UPR giving further evidence to the presence of misfolded proteins and ER stress in both FTC and FTA.
Conclusion
Dysregulated miRNA-21-3p and miRNA-21-5p are implicated in thyroid tumorigenesis so identifying their target genes and pathways is fundamental in investigating strategies for treating thyroid tumors. MiRNA-21-3p and miRNA-21-5p overexpression was found to downregulate TIMP3, MAT2A, TGFBR2, and PLAT gene in FTC and FTA leading to significant acute phase-response to metallothionein, metal ions, and UPR. TGFBR2 downregulation promotes COX-2 overexpression and its prostanoid product, MAT2A downregulation inhibits S-adenosylmethionine biosynthesis, while PLAT and TIMP3 silencing is linked with significant expression of metallothionein and MMP activation. The analysis suggests that miRNA-21-3p and miRNA-21-5p, UPR, metallothionein, and metal ions could represent vital prognostic and diagnostic biomarker for thyroid tumor treatment. The suppression of miRNA-21-3p and miRNA-21-5p, and the inhibition of COX-2 may be an intervention strategy for therapeutically targeting FTC and FTA for beneficial outcome.
Footnotes
Funding:
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
MN devised the conceptual ideas and wrote the manuscript.
