Abstract
The overexpression of the Epidermal Growth Factor Receptor (EGFR) marks it as a pivotal target in cancer treatment, with the aim of reducing its proliferation and inducing apoptosis. This study aimed at the CADD of a new apoptotic EGFR inhibitor. The natural alkaloid, theobromine, was used as a starting point to obtain a new semisynthetic (di-ortho-chloro acetamide) derivative (
Introduction
Even with significant advancements, cancer treatment is still regarded as one of the most difficult medical issues. The burden of the cancer problem increasingly shifts to lower- and middle-income countries, reflecting the underlying socioeconomic relationship. 1 Cancer, which claimed 9.6 million lives in 2018, is the second largest cause of death globally, according to the WHO. Over the next 20 years, there will likely be a 70% increase in the number of impacted people. 2
The process of apoptosis is a vital cellular mechanism that responds to various oncogenic stresses, including uncontrolled proliferation or DNA damage, by eliminating damaged or potentially harmful cells. 3 This process is essential for preventing the development of cancer by removing cells that are at risk of transformation. 4 In addition, apoptosis can suppress tumorigenesis by clearing oncogenic proteins and modulating cellular signaling pathways. 5 EGFR is a significant member of the protein kinases family that plays a critical role in apoptosis and is linked to cancer cells’ elimination. 6 EGFR also contributes significantly to the development and progression of various types of carcinomas. 7 Overexpression of EGFR promotes cell proliferation, differentiation, and survival, and a high level of EGFR expression has been associated with a lower survival rate in many cancer types, making EGFR expression a powerful prognostic indicator. 8 One of the essential pathways that activated by the EGFR protein is the janus kinase, JAK, / signal transcription activators and transducers, STAT, pathway. 9 The JAK/STAT pathway has a pivotal role in mitosis simulation, causing cell proliferation and apoptosis inhibition. 10 Therefore, blocking its molecular function has significant antiproliferative and apoptotic effects 11 and could be considered a key approach in the development of novel treatments for cancer. 12
In the field of drug discovery and development, the study of structure-activity relationships is an important factor in the creation of effective drugs.13 -15 Computer Assisted Drug Discovery (CADD) depends on the relationships between a drug’s chemical structure and its biological activity that could be applied to optimize drug-likeness, pharmacokinetics, and pharmacodynamics. 16 CADD has become an increasingly important tool in the pharmaceutical industry, where it is used to speed up the drug discovery process and reduce costs. 17
Over the years, a range of CADD applications have been developed. These include molecular and drug design,18,19 docking simulations, 20 ligand-based approaches such as pharmacophore assessment, 21 structure similarity, 22 and ADMET 23 are also widely used. Other computational chemistry techniques include DFT, which is used to study the electronic structure of molecules. 24 Molecular dynamics (MD) simulations are a powerful CADD tool for investigating the dynamic behavior, energetic and structural changes of proteins at the atomic level. 25 These simulations provide an opportunity to explore protein-ligand interactions offering precise descriptions of the conformational changes induced by ligand binding in a protein. 26 Therefore, MD simulations are a valuable approach for studying protein dynamics and ligand binding mechanisms. 27 CADD involves the use of software employing a variety of techniques to study the interactions between potential drugs and biomolecules.
The need for new EGFR inhibitors arises from the challenge of overcoming resistance that cancer cells develop to current drugs. 28 This resistance often stems from mutations in the EGFR gene 29 or activation of alternative signaling pathways. 30 Additionally, novel inhibitors, particularly those derived from natural compounds like our lead drug, hold the promise of providing improved safety profiles with fewer side effects compared to existing medications.31 -33 Many EGFR inhibitors were discovered and reported to have promising activity in the clinical treatment of cancer. 34 There are 4 essential pharmacophoric features that EGFR inhibitors share, allowing them to effectively bind to diverse pockets within the ATP binding site. Firstly, these inhibitors possess a flat heteroaromatic ring/rings that occupy the adenine binding pocket. 35 Secondly, a terminal hydrophobic head is present, which is responsible for binding to hydrophobic region I. The third feature is an NH spacer that can form essential hydrogen bonding interactions with the amino acid residues inside the linker region. 36 Lastly, a hydrophobic tail is included, which is directed into the hydrophobic region II.37,38 The above-mentioned features, as shown in Figure 1, are crucial for the design of effective EGFR inhibitors and for understanding their binding mechanism.
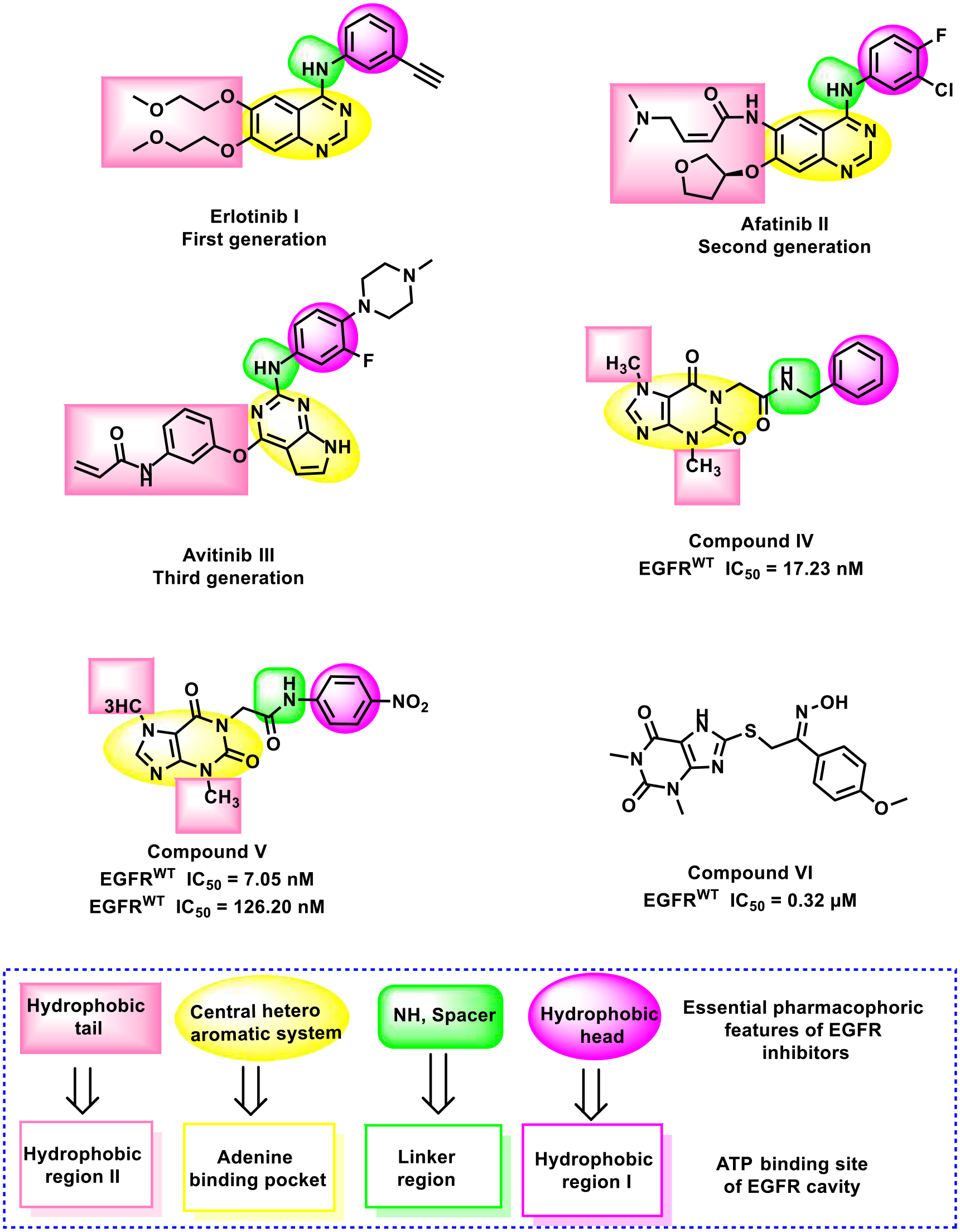
The crucial 4 pharmacophoric features of EGFR inhibitor drugs.
Figure 1 illustrates these features in several EGFR inhibitors. Erlotinib
Human beings have always depended on nature for their basic needs, including medicinal treatments, nourishing food, and cosmetic products, both in the past and in the present.43,44 Our team utilized the natural sources to present compound
Rationale
Compounds

The design rationale of
Moreover, the theobromine moiety in
This article reports on the ongoing research of our team to discover potential anticancer agents that target EGFR protein.45,48
-52 The focus of this paper is to introduce a newly synthesized compound,
Results and Discussions
Dft
Geometry optimization and Mulliken charge
To clarify the correlation between the chemical system’s structure, reactivity, and selectivity of active sites of

T-1-DOCA’s fully optimized chemical structure (A), and the Mulliken charge (B) at B3LYB/6-311G++(d,p) level.
The Mulliken charge analysis is another sign of charge transfer and polarizability. It is shown in color scale in Figure 3B and the direction of the dipole moment is illustrated. The charge distribution within
Frontier molecular orbital (FMO) analysis
Figure 4 shows the results of the FMOs study for

T-1-DOCA’s FMO and energy levels at B3LYB/6-311G++(d,p) level.
T-1-DOCA’s reactivity indices and energetic parameters.

Descriptors of chemical reactivity and total density of state (TDOS)
In FMO analysis, Koopman’s theorem was applied to determine the molecular electronic properties and global reactivity receptors of
Figure 5 displays the relative spectrum after analysis of the “total density distribution function, TDOS.” In the situation that the FMO analysis is unable to provide a complete description, the TDOS spectrum is a crucial tool for locating the maximum density. Virtual orbitals above the LUMO orbital, according to the TDOS study in Figure 5, have the highest electronic density.

T-1-DOCA’s FMO and TDOS at B3LYP/6-311++G(d,p).
Electrostatic potential maps (ESP)
The active binding sites and hydrogen bonds (H-Bs) with the target are determined by the molecular electrostatic potential (ESP), which is a representation of the 3D surface charge distribution (both positive and negative) over the chemical structure of
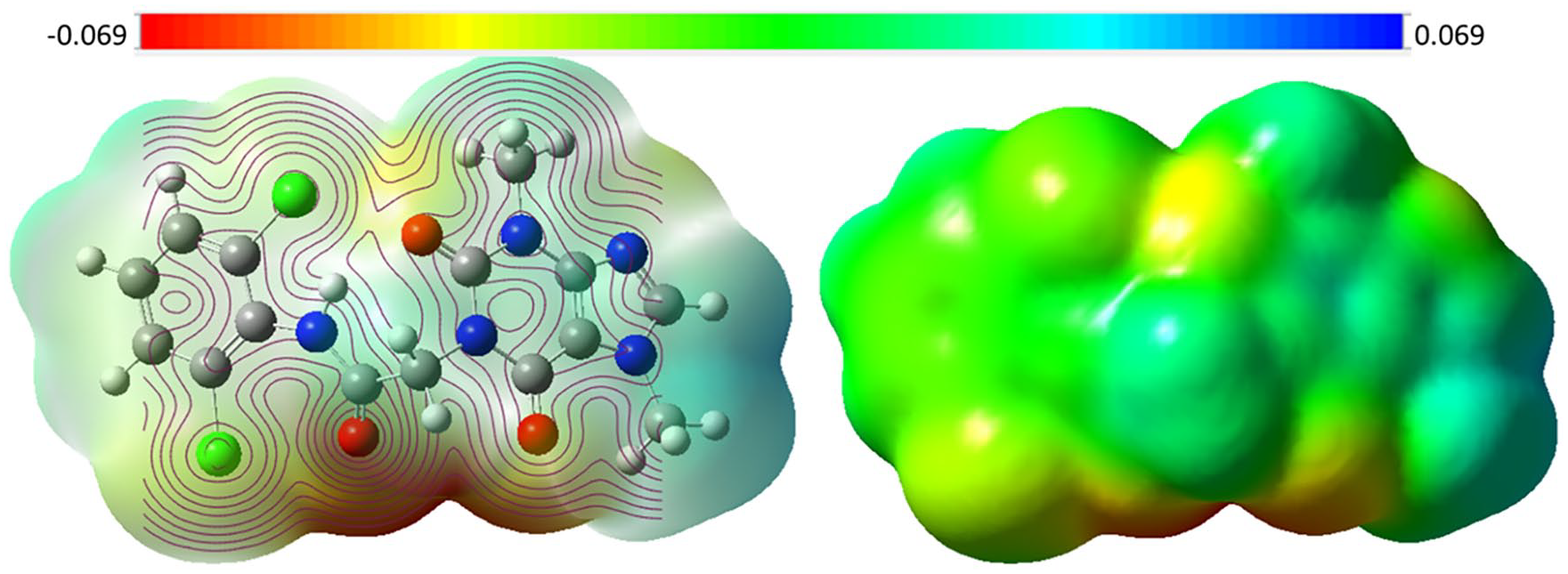
T-1-DOCA’s ESP at the 6-311G++(d,p) level.
Molecular docking against EGFR proteins
MOE protocol was applied for
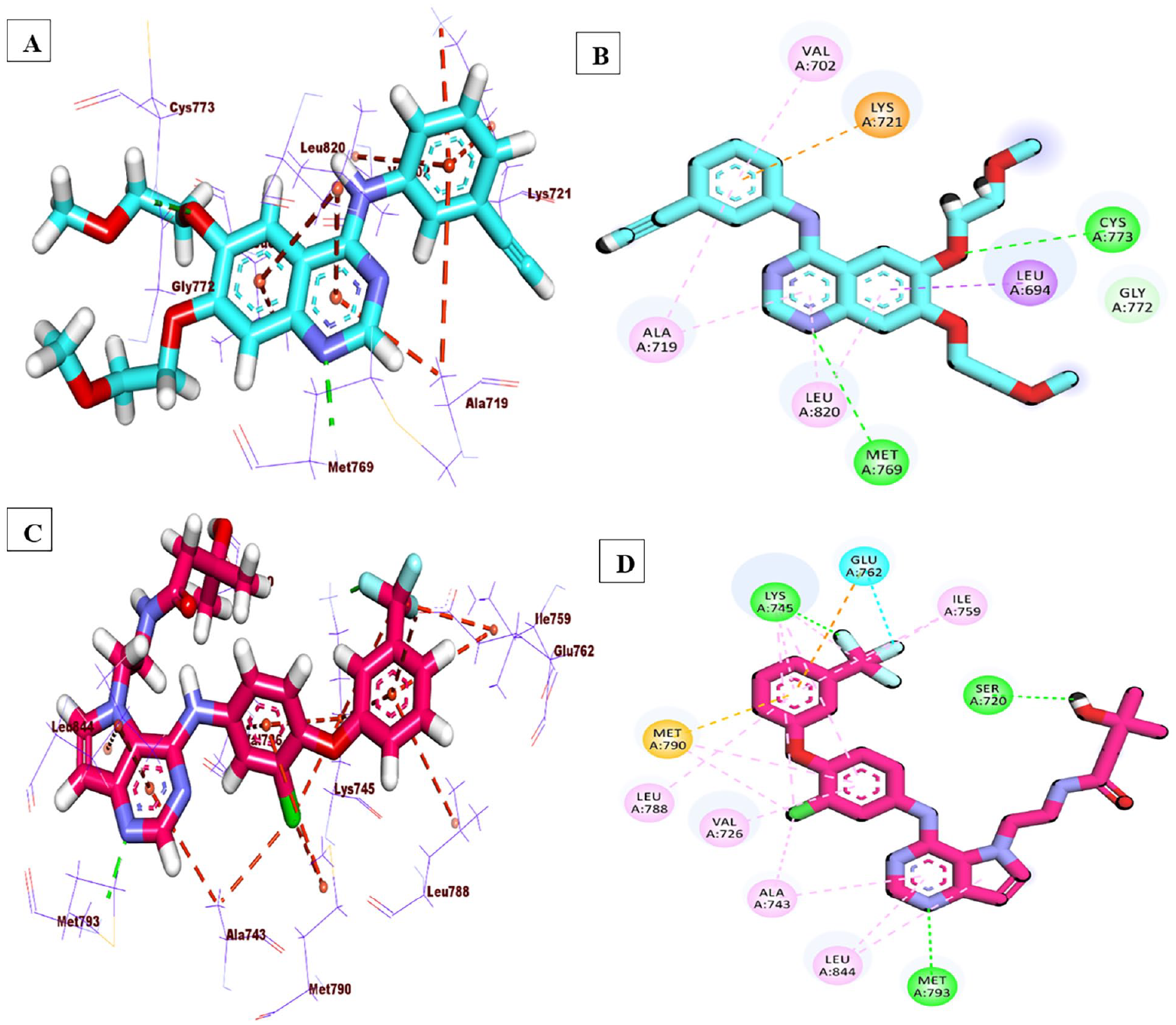
(A and B) 3D & 2D representations of erlotinib, (C and D) 3D & 2D representations of TAK-285.

(A and B) 3D & 2D of
MD simulations
The analysis of 100 ns of the MD production run shows that the EGFR

(A) RMSD, (B) Ro G, (C) SASA, (D) H-Bs, (E) RMSF, and (F) Center of Mass distance through the EGFR
MM-GBSA of the T-1-DOCA _EGFR complex
The binding free energy analysis conducted using MM-GBSA (depicted in Figure 10) showcases the various factors involved in the binding process. For

MM-GBSA of the
Additionally, Figure 11 presents a decomposition analysis that identifies the specific amino acids in close proximity to

Binding energy decomposition of the
The amino acids that contribute with a value greater (less) than −1 kcal.mol−1are Leu694 (−1.11 kcal.mol−1), Phe699 (−1.29 kcal.mol−1), Val702 (−3.26 kcal.mol−1), Lys721 (−5.64 kcal.mol−1), and Leu834 (−1.11 kcal.mol−1). Interestingly, there is one amino acid (Asp831) that has a positive binding contribution with an average of (+1.03 kcal.mol−1).
PLIP of the T-1-DOCA _EGFR complex
To obtain representative frames for each cluster, the trajectory of the
PLIP analysis of
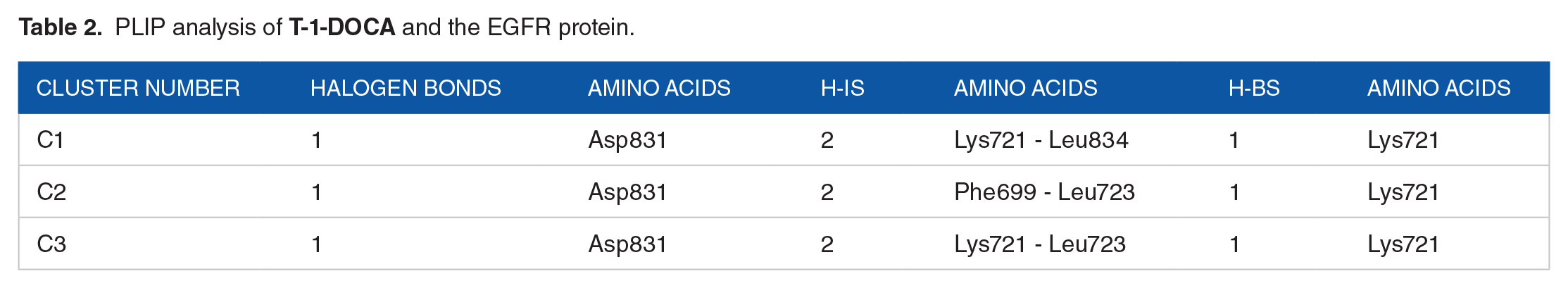
Among the 3 representative frames, the amino acids Lys721 and Asp831 were found to be the most frequently occurring. Lys721 formed a hydrogen bond (H-B) while Asp831 formed a halogen bond with the chloride atom. Additionally, two hydrogen-ion interactions (H-Is) were observed in each of the 3 cluster representatives. These findings are in line with the minor difference between the electrostatic and van der Waals energy values obtained from the MM-GBSA analysis.
Furthermore, the PLIP analysis not only provided information on the types and numbers of interactions, but also generated a .pse file, enabling visualization of the three-dimensional conformation of

PLIP for the obtained 3 cluster representatives. Halogen bonds: green solid lines, H-I: dashed gray line, H-B: Blue solid line, amino acids: blue sticks, and
The ATP binding site of EGFR is part of the tyrosine kinase domain of the EGFR, which is responsible for the receptor’s intracellular signaling activity.
While the ATP binding site is naturally competitive for any molecule, a molecule’s higher affinity primarily hinges on how well each of its pharmacophores can interact with specific amino acids within designated sub-pockets. In our study, we crafted
Interestingly, the docking studies affirmed
ADMET profiling study
The approval of a new medication relies on assessing both its pharmacokinetic properties and biological activity.
57
Thus, it is essential to evaluate the pharmacokinetic characteristics of a new compound early in the drug development process to prevent any potential delays in approval or late withdrawal.
58
To achieve this, ADMET parameters of

Computationally predicted ADMET parameters for
ADMET profile (computational) for

Lipinski’s and Veber’s rules violation
If a molecule adheres to at least 3 of Lipinski’s rules (H bond donors ⩽ 5, H bond acceptors ⩽ 10, molecular weight < 500, and logP < 5), it is more likely to have improved oral absorption. Compounds that deviate from more than one of these criteria are unlikely to exhibit high bioavailability. Additionally, reduced molecular flexibility, as indicated by a lower number of rotatable bonds, along with a smaller polar surface area, are key indicators of favorable oral bioavailability.
59
Also, compounds featuring 10 or fewer rotatable bonds and a polar surface area of 140 Å or less tend to have a heightened likelihood of good oral bioavailability.57,59 In the case of
Physicochemical properties of

In silico toxicity studies
Ensuring adequate toxicity estimation in the early stages of drug development is critical to minimizing drug approval failures.
60
However, using in vitro and in vivo methods can be ethically restricted, expensive, and time-consuming.
61
Therefore, the use of in silico techniques in toxicity prediction is necessary to avoid these challenges. In this study, the toxicity of
In silico toxicity studies of

Chemistry
Semi-synthesis of T-1-DOCA
The procedure outlined in

The semi-synthetic procedure of
Absorption bands at 3249 for NH, 1719, and 1660 cm-1 for C=O have appeared in
Biological evaluation
In vitro EGFR inhibition
In order to validate the proposed design and computational findings, we examined the potential of
Cytotoxicity
Based on the inhibitory potentials demonstrated by
The results of assessing the anti-proliferative properties of

Flow cytometric analysis of apoptosis
Apoptosis is a highly regulated process of programed cell death that helps to maintain a balance between the production and death of cells, which is essential for normal cellular homeostasis. Any disturbance in this balance can lead to a variety of pathological conditions such as autoimmune diseases and abnormal cell proliferation.
62
Apoptosis plays a vital role in various developmental processes, tissue regeneration, and eliminating inflammatory cells. In the 1980s, the induction of apoptosis became an important therapeutic strategy after the discovery of DNA breakage in thymocytes following exposure to glucocorticoids.5,63 To evaluate the apoptotic potential of
The impact of


The impact of
Reverse transcription-polymerase chain reaction (RT-PCR)
The present study aimed to investigate the impact of
T-1-DOCA’s potentials on apoptotic proteins.

Experimental
Docking studies
Docking studies have been administered for
M D simulations
M D simulations studies have been administered for the EGFR-
MM-GBSA
MM-GBSA has been administered for EGFR-
ED analysis
Principal Component Analysis (PCA) have been employed to EGFR-
Bi-dimensional assays
Bi-dimensional assays have been administered for EGFR-
DFT
DFT computations have been administered for
ADMET studies
ADMET profiling has been computationally administered for
Toxicity studies
Toxicity profiling has been computationally administered for
Chemistry
General procedure for the synthesis of T-1-DOCA
The potassium 3,7-dimethyl-3,7-dihydro-1H-purine-2,6-dione

Off-white powder (yield, 70%); m. p. = 222–224°C; IR (KBr) ν cm−1: 3249 (NH), 2966, 2926 (CH aliphatic), 1719, 1660 (C=O); 1H NMR (400 MHz, DMSO-d6) δ 10.05 (s, 1H), 8.08 (s, 1H), 7.88 (m, 1H), 7.55 (m, 1H), 7.25 (m, 1H), 4.80 (s, 2H), 3.90 (s, 3H), 3.45 (s, 3H); 13C NMR (101 MHz, DMSO-d6) δ 167.26, 154.59, 151.32, 149.00, 143.76, 136.27, 132.01, 131.41, 126.16, 124.78, 124.38, 107.02, 43.82, 33.69, 29.93; Mass (m/z): 382 (M+, 43%), and 267 (100%, base peak); Anal. Calcd. For C15H13Cl2N5O3 (382.20): C, 47.14; H, 3.43; N, 18.32; Found: C, 47.30; H, 3.57; N, 18.49%.
In vitro EGFR inhibition
The In vitro EGFR inhibition assay has been administered for
In vitro antiproliferative and safety activities
The In vitro antiproliferative activities have been administered for
Flow cytometry of apoptosis
The Apoptosis incidence has been evaluated for
Rt-PCR assay
Leveles of BAX, Bcl-2, caspase-3, and caspase-9 were investigated by RT-PCR using the kit (Qiagen RNA extraction/BioRad syber green PCR MMX). The Supplemental section includes an additional explanation and details about this study.
Conclusion
In conclusion, this study aimed to design and evaluate a novel apoptotic EGFR inhibitor,
Supplemental Material
sj-pdf-1-evb-10.1177_11769343231217916 – Supplemental material for Computer-Assisted Drug Discovery of a Novel Theobromine Derivative as an EGFR Protein-Targeted Apoptosis Inducer
Supplemental material, sj-pdf-1-evb-10.1177_11769343231217916 for Computer-Assisted Drug Discovery of a Novel Theobromine Derivative as an EGFR Protein-Targeted Apoptosis Inducer by Ibrahim H Eissa, Reda G Yousef, Eslam B Elkaeed, Aisha A Alsfouk, Dalal Z Husein, Ibrahim M Ibrahim, Hesham A El-Mahdy, Hazem Elkady and Ahmed M Metwaly in Evolutionary Bioinformatics
Footnotes
Author Contributions
The study was conceptualized and designed by IHE and AMM. AMM, HE, and EBK supervised the work. The semisynthesis was performed by RGY and HE, while DZH, and IMI conducted the DFT and the MD studies, respectively. Biological investigations were carried out by HAE. The funding for the study was obtained by EBE and AAA, who also contributed to writing the manuscript. All authors have thoroughly reviewed and approved the final manuscript.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Princess Nourah bint Abdulrahman University Researchers Supporting Project number (PNURSP2023R116), Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia. The authors extend their appreciation to the Research Center at AlMaarefa University for funding this work.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Upon request, the corresponding authors have access to the data and can provide it.
Sample Availability
Upon request,
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
