Abstract
Objectives:
Peripherally inserted central catheters (PICCs) are increasingly recognized as an alternative to centrally inserted central catheters (CICCs) in critical care, yet the data regarding the safety and feasibility of this choice in septic shock management is growing but still lacking. In this study, we aimed to determine the feasibility, safety, and impact on outcomes of using dedicated vascular access specialist (VAS) teams to insert PICCs versus CICCs on patients admitted to the ICU with septic shock.
Design:
Retrospective cohort study.
Setting:
Mayo Clinic Rochester Medical ICU and Mayo Clinic Arizona Multidisciplinary ICU from 2013 to 2016.
Patients:
All adult patients hospitalized with diagnosis of septic shock excluding patients who declined authorization for review of their medical records, mixed shock states, and readmissions.
Interventions:
None.
Measurement and main results:
Comprehensive data regarding septic shock diagnosis and resuscitation were abstracted from electronic medical records. A total of 562 patients with septic shock were included in the study; 215 patients were resuscitated utilizing a PICC and 347 were resuscitated using a CICC. On univariate analysis, the time to central line insertion and time to vasopressor initiation were found to be reduced in those who received PICC at time of ICU admission versus CICC. Other favorable outcomes were also observed in those who received PICC versus CICC including shorter ICU length of stay and lower unadjusted hospital mortality. A multivariable analysis for hospital mortality showed that after adjusting for important covariates, neither the time to central line insertion nor the time to vasopressor initiation was associated with a lower hospital mortality.
Conclusions:
Across two tertiary referral centers within the same enterprise, use of a dedicated VAS team for insertion of PICCs for initial resuscitation in patients with septic shock was feasible and associated with shorter time to central venous access and initiation of vasopressors; however, adjusted hospital mortality was not different between the two groups.
Keywords
Introduction
Sepsis is one of the leading causes of mortality globally, yet strategies for improving mortality in the care of septic patients have only recently been a large focus of medical research over the past two decades. During this time, we have learned that early fluid resuscitation, early vasopressors, and early antibiotics improve mortality.1–3 It has also been demonstrated that excess fluid resuscitation can lead to undue harm. 4 What is not clear however, is the best route of central access for resuscitation in septic shock. Many physicians believe that PICCs are contraindicated in the ICU. 5 However, in the setting of a dedicated vascular access specialist (VAS) team, PICCs can be used effectively in the ICU. 6
We have previously shown that the implementation of a VAS team for insertion of peripherally inserted central catheters (PICC) within the ICU is both feasible and safe. 7 At Mayo Clinic Arizona, a dedicated ICU-specific critical care respiratory therapist team (VAS) trained in vascular access has been available since 1999 8 and is available to place PICC lines on demand. This has been favorably embraced by our staff due to a quicker time to central access when compared to centrally inserted central catheters (CICC). Compared to the Arizona hospital, the medical ICU (MICU) at Mayo Clinic St. Mary’s Hospital in Rochester, MN uses CICCs nearly exclusively in the treatment of septic shock as they do not have a dedicated vascular team in the ICU to expeditiously place PICC lines. PICCs are increasingly recognized as an alternative to CICCs in critical care, yet the data regarding the safety of this choice is growing but still lacking. 5 In this study, we aim to compare the feasibility, safety, and outcomes in septic shock patients resuscitated with a PICC placed by a dedicated VAS team versus a CICC line at two different ICUs of the same institution.
Methods
After approval by the Mayo Foundation Institutional Review Board (IRB #17-000273), this retrospective observational cohort study was conducted in two different ICUs at our institution: A 30-bed multidisciplinary ICU in Mayo Clinic Hospital, Phoenix, Arizona; and a 24-bed MICU at St. Mary’s Hospital in Rochester, MN. All consecutive patients who were admitted with septic shock to any of these two ICUs from 2013 to 2016 were included. The multidisciplinary ICU in Mayo Clinic Arizona has a dedicated team of critical care respiratory therapists with vascular access board certifications (VAS) located within the ICU; this team places arterial and PICC lines upon request of the ICU team. The VAS team uses an ultrasound guided microintroducer and Seldinger technique, selects the optimal vessel based on a goal vessel-to-catheter ratio of 3:1 and confirms tip position by using a vascular positioning system, along with electrocardiography and doppler for tip control technique, thus avoiding the delays of chest radiography for the majority of PICCs. 7 PICC lines were double-lumen open-ended catheter with staggered distal ports (Arrow®/Teleflex®, Wayne, USA) (18- and 20-gage lumina, 5F, 50-cm polyurethane catheter). Our group has previously published the safety and rapid acquisition of vascular access by the VAS team. 6 The patients from the MICU in Mayo Clinic Rochester nearly exclusively receive CICCs placed by the ICU team (attending and/or trainees) utilizing ultrasound guidance. CICC are 7F, 16-cm, noncuffed, triple-lumen, polyurethane-heparin-bonded CVCs impregnated with chlorhexidine gluconate and silver sulfadiazine (ARROWgard Blue; Arrow®/Teleflex®, Wayne, USA). Chest x-ray was used to confirm catheter position of CICCs. Our inclusion and exclusion criteria are described as follows:
Inclusion criteria
✓ Septic Shock as defined by the Sepsis-II criteria, standard at the time of the study period
✓ Central catheter inserted after admission to the ICU.
Exclusion criteria
Denied authorization for retrospective analysis
Pregnancy
Dialysis or “Introducer” catheters
Mixed shock states with clinically significant primary cardiogenic, hemorrhagic, or obstructive (e.g. cardiac tamponade) element
Definitive admitting diagnosis of acute coronary syndrome, myocardial infarction, or pulmonary embolism (concomitant with evidence of septic shock)
In cases where patients were readmitted to the ICU, only the first admission was analyzed
Study data were collected and managed using REDCap (Research Electronic Data Capture) electronic data capture tools hosted at Mayo Clinic Arizona. 9 Data collected included comprehensive clinical characteristics, type and site of central line placement, SOFA scores, fluid administered and fluid balance, vasopressor use, lactate levels, sepsis related interventions, and central line complications (mechanical complications, central line-associated bloodstream infection [CLABSI], and symptomatic deep venous thrombosis [DVT] associated with the line). The main outcome of the study was in-hospital mortality. Secondary outcomes included time to vasopressor initiation, time to central line insertion, and ICU and hospital length of stay. Time zero for each of the outcomes reported was time of ICU admission.
Statistical analysis
All data are summarized as median (interquartile range [IQR]) or percentages. Unpaired Student’s t-tests were used to compare continuous variables with normal distribution and Mann-Whitney U test for skewed distribution. For comparison of categorical variables, chi-square tests were used if the number of elements in each cell was ⩾5; Fisher’s exact test was used otherwise. In order to determine the independent impact of important covariates identified in the univariate analysis on hospital mortality, a multivariate logistic regression model using predictive variables collected within 24 h after ICU admission in the overall cohort were used. Covariates were entered in the model when significant differences (p < 0.1) were noted between those patients with septic shock resuscitated with a PICC versus a CICC. The model was refined with backwards stepwise regression, taking into account collinearity and interaction terms. When appropriate, the odds ratio (OR) and 95% confidence intervals (CI) were calculated. Model discrimination was assessed using receiving operator curves (ROC) curves. Model fit (calibration) was assessed using the Hosmer-Lemeshow goodness-of-fit test. P-values of <0.05 are considered statistically significant. JMP statistical software (version 14, SAS, Cary, NC) was used for all data analyses.
Results
A total of 352 patients with septic shock were initially identified in the Multidisciplinary ICU in Arizona with PICCs inserted as their only central intravenous access. Of these, 64 were excluded due to mixed shock states, 20 were excluded due to concomitant admitting diagnoses of acute coronary syndrome with septic shock, 26 patients were excluded due to readmission, and 27 were also excluded due to placement of PICC line before ICU admission. Of the 386 patients with septic shock admitted to the MICU at Rochester, MN with CICCs inserted, 5 were excluded due to mixed shock states and 15 were excluded due to readmissions, and 19 were excluded due to placement of the CICC before admission to the ICU. Therefore, a total of 562 patients with septic shock were included in the study including 215 patients that were resuscitated utilizing a PICC and 347 that were resuscitated using a CICC.
Age and gender were similar between the two groups; however, BMI was significantly higher in patients resuscitated with CICC (Table 1). SOFA score upon ICU admission was significantly higher in the group treated with CICC. In both groups, most admissions to the ICU were from the ER (243), and the most common source of infection was the lung (215); however, PICC-resuscitated patients were more commonly admitted from the hospital ward and presented more intraabdominal source of infection as compared to the CICC group (Table 1).
General baseline characteristics of 562 patients admitted to the ICU with septic shock.
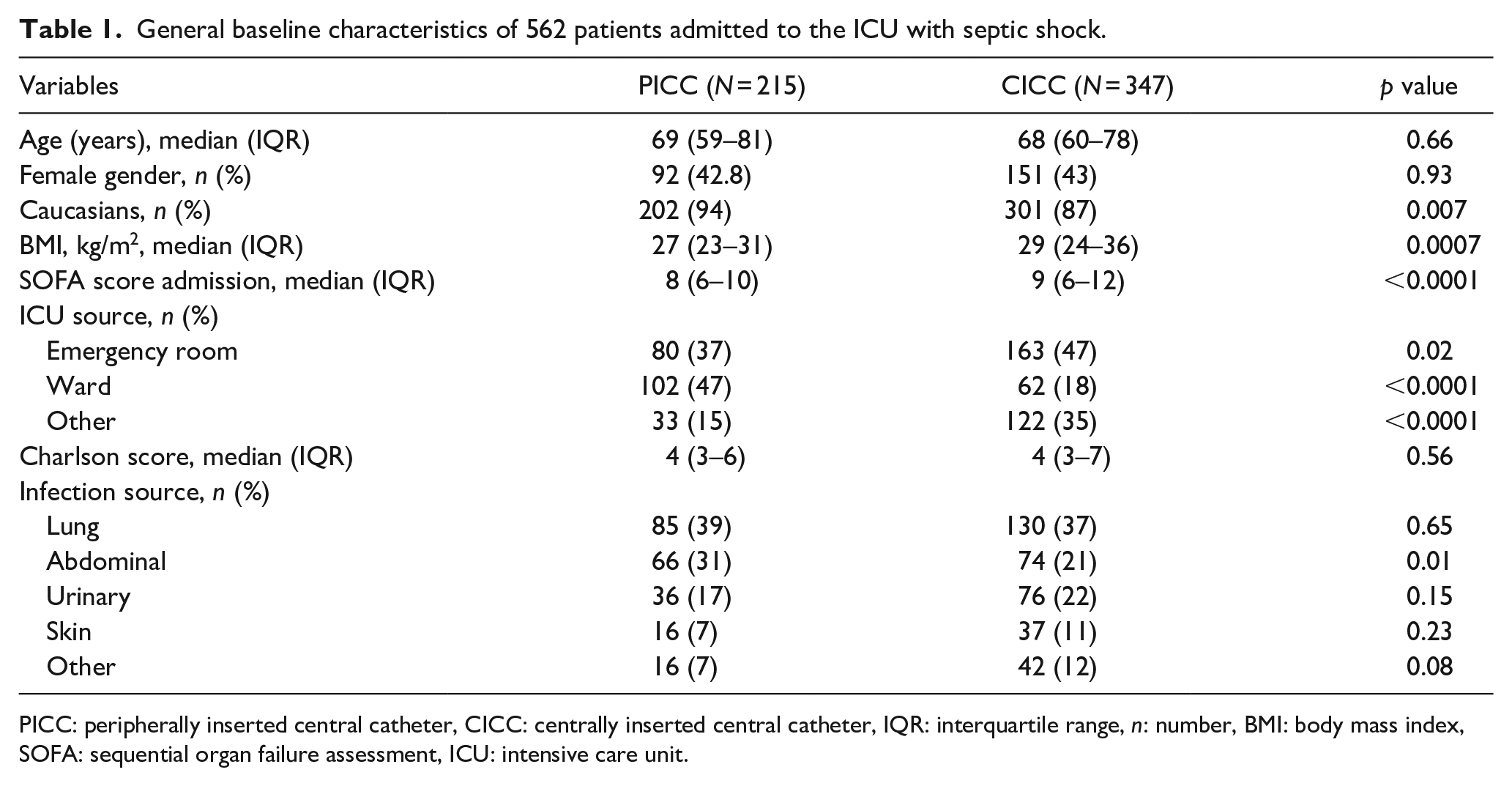
PICC: peripherally inserted central catheter, CICC: centrally inserted central catheter, IQR: interquartile range, n: number, BMI: body mass index, SOFA: sequential organ failure assessment, ICU: intensive care unit.
Most PICCs were placed in the basilic vein (200), and the rest (15) were placed in the cephalic vein. For CICCs, most were placed in the internal jugular veins (332), followed by the femoral veins (9), then the subclavian veins (6).
The median lactate on admission was similar between the two groups (3.3 mmoL/L [IQR, 2.6–4.8] and 3.7 mmoL/L [IQR 2.6–5.0] in PICC and CICC groups, respectively) (Table 2). After 24 h of admission, the PICC group demonstrated a larger rate of decline in plasma lactate levels (1.7 mmoL/L [IQR, 1.0–2.6]) compared to the CICC group (2.3 mmoL/L [IQR, 1.4–4.0]), which was statistically significant (p = 0.003). The PICC group also received less fluid resuscitation throughout the course of their first ICU day (2.8 L [IQR, 1.6–4.2] by 24 h), compared to the CICC group (4.4 L [IQR, 2.8–6.3] by 24 h), which was also statistically significant (p < 0.0001).
Resuscitation goals and line associate measures of 562 patients admitted to the ICU with septic shock.
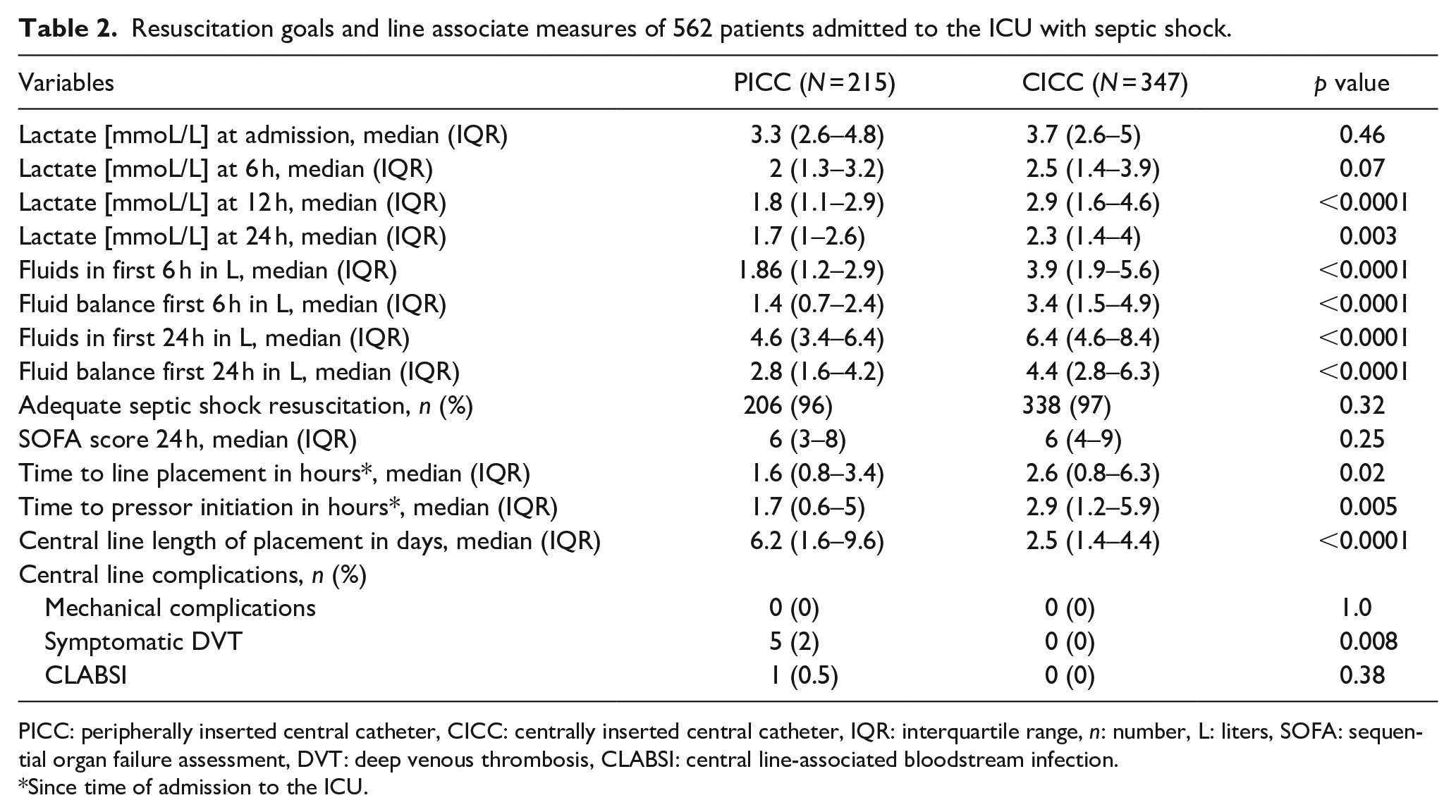
PICC: peripherally inserted central catheter, CICC: centrally inserted central catheter, IQR: interquartile range, n: number, L: liters, SOFA: sequential organ failure assessment, DVT: deep venous thrombosis, CLABSI: central line-associated bloodstream infection.
Since time of admission to the ICU.
The median time to insertion of PICCs (time zero being ICU admission time) was 1.6 h (IQR 0.8–3.4), compared to CICCs which was 2.6 h (IQR 0.8–6.3, p < 0.02). The median time to initiation of vasopressors in the PICC group was 1.7 h (IQR 0.6–5), and in the CICC group was 2.9 h (IQR 1.2–5.9, p = 0.005).
The total duration of placement of the PICCs was 6.2 days (IQR 1.6–9.6), compared to that of CICCs was 2.5 days (IQR 1.4–4.4, p ⩽ 0.0001). There were no mechanical complications during line placement between the two groups. There were five symptomatic DVTs in the PICC group, and zero DVTs in the CICC group (p = 0.008). There was one CLABSI in the PICC group, and zero in the CICC group, although this difference was not statistically significant (p = 0.38)
There was a statistically significant reduction in ICU length of stay (LOS) with a median LOS of 2.2 days (IQR 0.5–4.5) in the PICC group, and 2.8 days (IQR 1.8–4.9, p = 0.002) in the CICC group. (Table 3). Renal replacement therapy (RRT) was not different between the groups, it was needed in 34 patients in the PICC group (15.8%; 20 CRRT, 12 IHD, and 2 SLED), and 57 in the CICC group (16.4%; all CRRT). Mechanical ventilation also showed no difference between the groups, it was needed in 82 patients in the PICC group, and 154 in the CICC group (p = 0.15). The unadjusted ICU mortality for the PICC group was 9%, compared with 15% in the CICC group, which was statistically significant (p = 0.04). The unadjusted hospital mortality in the PICC group was 13%, compared with 24% in the CICC group, which was also statistically significant (p = 0.0007). A multivariable analysis for hospital mortality (ROC 0.8) showed that after adjusting for important covariates, only the ICU admission SOFA score and the hospital ward source of ICU admission were independently associated with hospital mortality (Table 4).
Outcomes of 562 patients admitted to the ICU with septic shock.
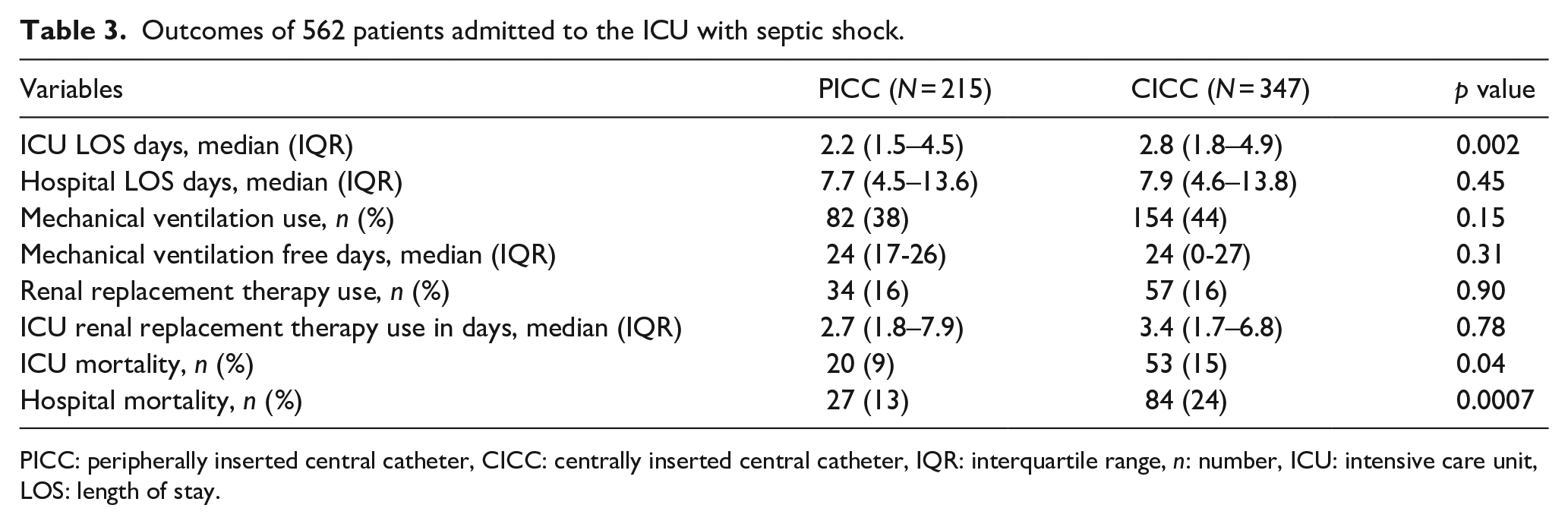
PICC: peripherally inserted central catheter, CICC: centrally inserted central catheter, IQR: interquartile range, n: number, ICU: intensive care unit, LOS: length of stay.
Multivariate analysis for hospital mortality (ROC 0.8).
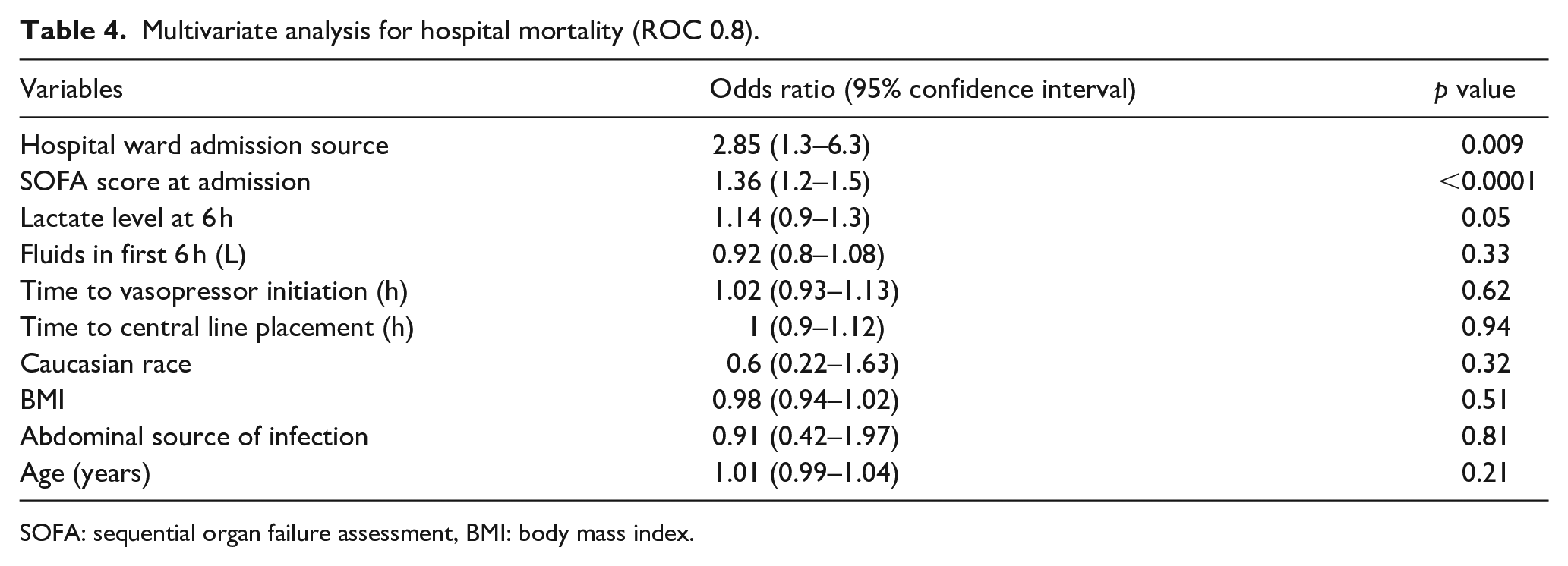
SOFA: sequential organ failure assessment, BMI: body mass index.
Discussion
In two ICUs of the same institution, the use of central lines in septic shock resuscitation was common and expected according to current septic shock treatment standards. In particular, the use of PICCs was very common in the Arizona hospital ICU due to its dedicated team of respiratory therapists with vascular access board specialty certification (VAS Team). Our study shows that the use of a VAS team with their preferential use of PICCs for initial resuscitation in patients with septic shock in the ICU is feasible and associated with shorter time to central venous access and shorter time to initiation of vasopressor drugs as compared to not using a dedicated VAS team in the ICU and instead placing CICCs. Other favorable outcomes were also observed in the PICC group including shorter ICU length of stay and lower hospital mortality. However, after adjustment for important covariates, only the source of admission (hospital ward) and the initial admission SOFA score were independently associated with in-hospital mortality reflecting more likely that mortality was mainly affected by the severity of disease. Although PICCs were associated with more complications than CICCs in our study, they also had a much longer duration of usage compared to CICCs, which may explain the increased associated complications. There were no pulmonary emboli in the PICC group. However, as noted, there were significantly more DVTs in the PICC group (n = 5) and zero DVTs in the CICC group.
DVTs were identified using compression ultrasonography. Notably, the longer duration of usage is likely explained by the better tolerability and design of the PICCs as compared to CICC whilst affording the same broad array of clinical endpoints including but not limited to: cardiovascular monitoring, vasopressor administration, fluid resuscitation, long term antibiotic administration, parenteral nutrition, and blood transfusions. Notably, there was only one CLABSI within the PICC group, which was not significantly different than the CICC group (p = 0.38). There is often a fear of placing PICCs in septic patients due to concern over line seeding. Our data do not support this fear, nor is there sufficient literature to justify this concern.
Sepsis has been associated with a remarkable growing cost of healthcare.10,11 Septicemia has been the singular most expensive hospital diagnosis, accounting for 23.6 billion dollars of the expense of U.S. hospitals in 2013. 12 These astronomical cost burden to our already expensive healthcare demands that efforts be made to provide more cost-effective care in general. The Rivers trial and the often practiced EGDT protocol has been shown to lead to over usage of hospital resources, currently with either no significant benefit in mortality or sometimes even worse outcomes as described earlier. Our data suggest that the use of a VAS team for sepsis resuscitation is feasible and associated with favorable outcomes that can potentially reduced costs such as lower ICU LOS and decreased volume of fluids used for resuscitation. Dedicated vascular access teams are an affordable, mobilizable resource that can potentially both reduce costs and improve outcomes. 13 Early vasopressor therapy allows for earlier, titratable MAP control with concurrent fluid as needed on an individual patient basis. This study demonstrates that PICCs are a viable option for the management of septic shock.
It should also be considered that sepsis management within the ICU represents a very contextual management practice, which may not be generalizable to those triaging septic patients on the front lines. In light of a dogmatic approach to sepsis management being steeped into training and professional expectations and management practices, we believe that PICCs might afford an avenue toward individualized therapy. Further, we believe that in addition to favorable outcomes observed in our study, PICCs may be associated with potential added benefit in comfort, externalization of workload to vascular access teams, and cost reduction, all of which should be assessed prospectively in a clinical trial.
Several limitations of our study must be addressed. First, in light of the retrospective nature of the study, it may have been difficult to ensure the accuracy of certain data points such as time of diagnosis of septic shock and time of vasopressor initiation. It is not entirely clear if the times charted into the system are the precise times in which they occurred, which may alter the results, though this is an understood limitation in analyzing medical record data. There are also limitations in our recording of severity of illness, using SOFA rather than APACHE owing to the data that was clinically available. It is also important to recognize that there is a subset of patients who truly do need large volume resuscitation. In this context, the law of Poiseuille, in conjunction with clinical experience tells us that PICCs are not appropriate. Further, despite both study sites being tertiary referral centers within the US, it is not clear how comparable the two study populations were beyond their SOFA score. Moreover, it is important to emphasize that some patients should not get a PICC line, specifically those with CKD stage 3b or higher and a life expectancy of >6 months, as this may limit future HD access sites. Of note, having a dedicated VAS team is not common practice and may limit the generalizability of our study to other hospitals. Additionally, having a dedicated VAS team placing PICCs as opposed to a wide breadth of providers within the ICU placing CICCs likely contributes to the timing of line placement. An important variable that was not accounted for within our dataset is that the VAS team also placed arterial lines for many patients, allowing for closer hemodynamic monitoring. As such, having a dedicated VAS team rather than the type of access could be viewed as the primary driver of the outcomes we observed, though this is difficult to account for in a retrospective manner. Perhaps the greatest limitation of our study however is that an ICU with a dedicated VAS team, different patient populations, and differing management strategies is likely not the ideal comparator against an ICU without a VAS team. However, several important outcomes tended to be better or similar in the group of patients that underwent PICC placement by the VAS team as compared to the CICC group which highlights the importance of addressing this matter in a prospective manner. Finally, our “time zero” was defined as ICU admission time which is an easy identifiable variable; however, ideally we should have used the time of sepsis diagnosis as “time zero” but unfortunately this variable is difficult to determine in a retrospective manner.
Conclusions
In conclusion, our study is unique in that it shows PICC placement by a dedicated VAS team may be a viable and relatively safe alternative to CICC in septic shock management. However, the results of this study should be interpreted with caution due to the study limitations. Despite its limitations, our study suggests that PICC placement with the use of a dedicated VAS team has potential benefits in the management of septic shock patients. Based on our experience, we would recommend a VAT for PICC placement in the ICU based on timely line placement. Although, the decision for mode of line placement should be individualized based on patient characteristics and available staff in a given ICU. Future studies are needed to better clarify which patients are the ideal candidates for PICC over CICC in septic shock management.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
