Abstract
Objectives:
The current guidelines for managing patients with sepsis include the early cultures, administration of antibiotics, and fluid resuscitation. Several clinical trials have tried to determine whether or not the administration of corticosteroids improves outcomes in these patients. This study analyzed the characteristics of a large group of critically ill patients who either had cortisol levels drawn during their intensive care unit management or had hydrocortisone administered during their management.
Methods:
A list of patients who had cortisol levels measured or who had hydrocortisone administered empirically for the treatment of sepsis was identified by the medical record department at University Medical Center in Lubbock, Texas. The primary outcome was in-hospital mortality. Secondary outcomes included the need for mechanical ventilation, the need for renal replacement therapy, the need for vasopressors, length of stay, and the development of nosocomial infections.
Results:
This study included 351 patients, including 194 women (55.3%). The mean age was 62.9 ± 16.1 years. The mean admission SOFA score was 9.3 ± 3.63, the mean APACHE 2 score was 18.15 ± 7.7, and the mean lactic acid level was 3.8 ± 4.0 mmol/L. One hundred sixty-two patients required intubation, 262 required vasopressors, 215 developed acute kidney injury, and 319 had cortisol levels measured. The mean length of stay was 11.5 ± 13.7 days; the mortality rate was 32.2%. Multiple variable analysis demonstrated that higher cortisol levels were associated with increased mortality (44.1% if cortisol ⩾20 µg/dL versus 17.5% if cortisol <20 µg/dL). One hundred forty-five patients received corticosteroids, and multivariable analysis demonstrated that these patients had increased mortality (40.0% versus 26.7%).
Conclusion:
In this study, higher cortisol levels were associated with increased mortality. The administration of hydrocortisone was associated with increased mortality possibly reflecting the use of this medication in patients who had a higher likelihood of poor outcomes.
Introduction
Sepsis has complex effects on host defenses and organ function caused by a dysregulated host defense response. The Surviving Sepsis Campaign guidelines outlined the initial management priorities in these patients. 1 These include rapid assessment, collection of relevant cultures, the initiation of antibiotic therapy, and fluid resuscitation. Some patients with septic shock require vasopressor support. Important complications include acute respiratory failure requiring mechanical ventilation and acute kidney injury requiring renal replacement therapy. The Surviving Sepsis Campaign guidelines for 2021 make a weak recommendation for the addition of hydrocortisone at 200 mg/day based on moderate evidence in septic shock, but the optimal dose, timing of initiation, and duration of corticosteroids remain uncertain. 2
The use of hydrocortisone has several favorable features, including its low cost, simple administration, and ready availability. Studies have demonstrated that sepsis has a complex effect on the hypothalamus–pituitary–adrenal axis but do not identify a definite abnormality which has the potential for reversal by exogenous corticosteroids. Venkatesh et al. randomized 3800 patients with septic shock requiring mechanical ventilation with vasopressor dependence. 3 There was no difference in mortality between the group treated with hydrocortisone and the group treated with placebo. Annane et al. completed a complex study of 1241 patients; one treatment arm included hydrocortisone plus fludrocortisone. The patients in this arm had a lower 90-day mortality rate and had a higher number of vasopressor free-days and organ failure-free days by day 28 of hospitalization. 4 Rygård et al. did a meta-analysis of 22 randomized control trials that included 7297 patients. 5 Patients treated with corticosteroids had a shorter duration of shock, a shorter duration of mechanical ventilation, and a lower number of intensive care unit (ICU) days. However, there was no beneficial effect on either short-term or long-term mortality.
Randomized control trials provide important information and typically involve standardized protocols for patient management. Studies on the initial management steps of patients with sepsis have improved outcomes and reduced mortality. However, after the patient is admitted to the medical ICU, management defaults to the physicians working in that ICU. These patients usually have important differences in the underlying infection, acute organ dysfunction, prior comorbidity, and trajectory of clinical events. In this setting with heterogeneous patients, the use of corticosteroids likely has uncertain benefits. This study used real data and real evidence from a large cohort of critically ill patients and focused on relatively simple questions. What are the characteristics and outcomes of patients who had a cortisol level measurement and or received hydrocortisone during their ICU management for sepsis?
Methods
We conducted an observational retrospective study by reviewing electronic medical records of patients aged 18–89 years old who were admitted to the medical ICU at University Medical Center, Lubbock, Texas, from 1 January 2016 to 31 August 2018. The inclusion criteria included any patient who had a cortisol level drawn during his or her ICU admission, or had hydrocortisone administered during the MICU admission, or both. The patients meeting these criteria were identified by requesting the Informational Technology department to produce a report. The initial list included 602 patients, and all medical records then were reviewed to identify patients with diagnosis of sepsis/septic shock with identifiable or suspected source of infection who were hemodynamically unstable (defined as systolic blood pressure (SBP) <90 mmHg or mean arterial pressure (MAP) <65 mmHg) or had organ failure. Patients who did not meet these criteria were not further characterized or analyzed.
Baseline characteristics, including age, gender, home medications (chronic corticosteroids or opioids), etiology of sepsis, co-morbidities, hemodynamic parameters (blood pressure, mean arterial pressure, and heart rate) on admission, length of hospital stay, SOFA score, 6 Glasgow coma scale, 7 APACHE II score, 8 adrenal response if tested, use of etomidate for intubation, dose of corticosteroids, duration of corticosteroid administration, and method of weaning (stopped abruptly, weaned off by reducing daily dosage, or kept at same dose for more than 10 days with no clear plan to decrease dose), were retrieved from medical records. The timing of cortisol measurements was based on physician judgment. The principal outcome was ICU mortality. Some patients were started on stress dose corticosteroids after adequate fluid resuscitation (defined as 30 mL/kg) and the subsequent use of vasopressors. These management decisions were made by the MICU team and were not based on any standard protocol. Other outcomes included the need for vasopressors and time on vasopressors, the development of acute kidney injury and the need for renal replacement therapy (RRT), the need for intubation and the days of mechanical ventilation, and the incidence of new infection.
Statistical analysis
Descriptive statistics were used to describe the characteristics of the study participants. Categorical variables were summarized as frequencies and percentages, and continuous variables were summarized using means and standard deviations or medians and ranges, as appropriate. Logistic regression models were used to evaluate the associations between serum cortisol level and the risks of mortality, RRT, acute kidney injury (AKI), and infection, while adjusting for other risk factors. Poisson regression models were used to evaluate the associations between serum cortisol level and days on mechanical ventilation, vasopressors, and length of hospital stay, while adjusting for other risk factors. The same analyses were also performed when serum cortisol was dichotomized by using a cutoff value of 20 µg/µdL. The statistically significant level was set at 0.05. Multiple testing adjustment was not performed. Analyses were performed using SAS (Windows version 9.4; SAS Institute, Cary, NC, USA) and the statistical program R version 4.0.2 (https://cran.r-project.org/).
This study was approved by the Institutional Review Board at Texas Tech University Health Sciences Center (L19-091) in Lubbock, Texas.
Results
This study included 351 patients, including 194 women (55.3%). The mean age was 62.9 ± 16.1 years. The mean admission SOFA score was 9.3 ± 3.63, the mean APACHE 2 score was 18.15 ± 7.7, and the mean lactic acid level was 3.8 ± 4.0 mmol/L (Table 1). Seventy-two patients had positive blood cultures, 34 patients had positive urine cultures, 24 patients had positive respiratory tract cultures, 16 patients had positive abdominal/GI cultures, and 8 patients had positive wound cultures. One hundred sixty-two patients required intubation, 262 required vasopressors, 215 developed AKI, and 32 developed hospital-acquired infection. The overall mean length of stay was 11.5 ± 13.7 days; the mortality rate was 32.2%.
Patient characteristics and clinical information based on steroid administration.

Median (25th, 75th percentiles).
These patients were split into two groups based on the receipt of corticosteroids and cortisol levels. One hundred forty-five patients (41.3%) received corticosteroids (Table 1), and 136 patients (42.6%) had cortisol levels >20 mcg/dL (Tables 2(a)). Cortisol was measured on hospital day 1.9 ± 4.4. Differences between the patients who received corticosteroids and the patients who did not receive corticosteroids are reported in Table 1. Patients who received corticosteroids had lower cortisol levels (18.5 ± 16.4 versus 23.7 ± 14.5,
(a) Patient characteristics and clinical information by cortisol level.

Numbers in bold represent statistically significant results at a p value less that 0.05.
(b). Patient characteristics and clinical information, by cortisol level quantiles.

Important differences between patients who had cortisol levels ⩾20 mcg/dL and patients who had cortisol levels <20 mcg/dL are reported in Table 2(a). Patients who had cortisol levels ⩾20 mcg/dL had higher mortality rates (44.1% versus 17.5%,
Multivariable analysis indicated that patients with higher cortisol levels (entered as a continuous variable) had an increased odds ratio for mortality (adjusted odds ratio (AOR) = 1.04 95% confidence interval (CI) = 1.02–1.07); patients with higher admission SOFA scores also had increased odds ratios for mortality (AOR = 1.32, 95% CI = 1.15–1.52) (Table 3). When the analysis was repeated using cortisol as a binary variable, patients with cortisol levels of ⩾ 20 mcg/dL and higher admission SOFA scores had an increased odds ratio for mortality (Table 4). Patients with increased APACHE 2 scores had increased odds ratios for the development of AKI (AOR = 1.09, 95% CI = 1.03–1.16) (Table 4). Patients who received corticosteroids required mechanical ventilation (AOR = 1.66, 95% CI = 1.02–2.27) and vasopressors (AOR = 1.56, 95% CI = 1.10–2.22) for longer periods (Tables 5 and 6). Patients with increased admission SOFA scores required vasopressors for longer periods (AOR = 1.08, 95% CI = 102 versus 1.15) (Tables 5 and 6).
Clinical outcomes and risk factors.

Presented as adjusted odds ratios. Risk factors adjusted include serum cortisol, steroid use, age, gender, admission SBP, DBP, SOFA, APACHE II score, and lactic acid. *Due to missing values. A 1 μg/dL increase in serum cortisol is associated with a 4% increase in the odds of in-hospital mortality, adjusting for other risk factors. A one unit increase in admission SOFA is associated with a 32% increase in the odds of in-hospital mortality.
Clinical outcomes and risk factors with binary serum cortisol levels.

Presented as adjusted odds ratios. Risk factors adjusted include serum cortisol, steroid use, age, gender, admission SBP, DBP, SOFA, APACHE II score, and lactic acid.
Due to missing values. Patients with ⩾20 μg/dL serum cortisol had more than 300% increase in mortality compared to those with serum cortisol <20 μg/dL.
Numbers in bold represent statistically significant results at a p value less that 0.05.
Days on ventilation, vasopressors, LOS, and risk factors.
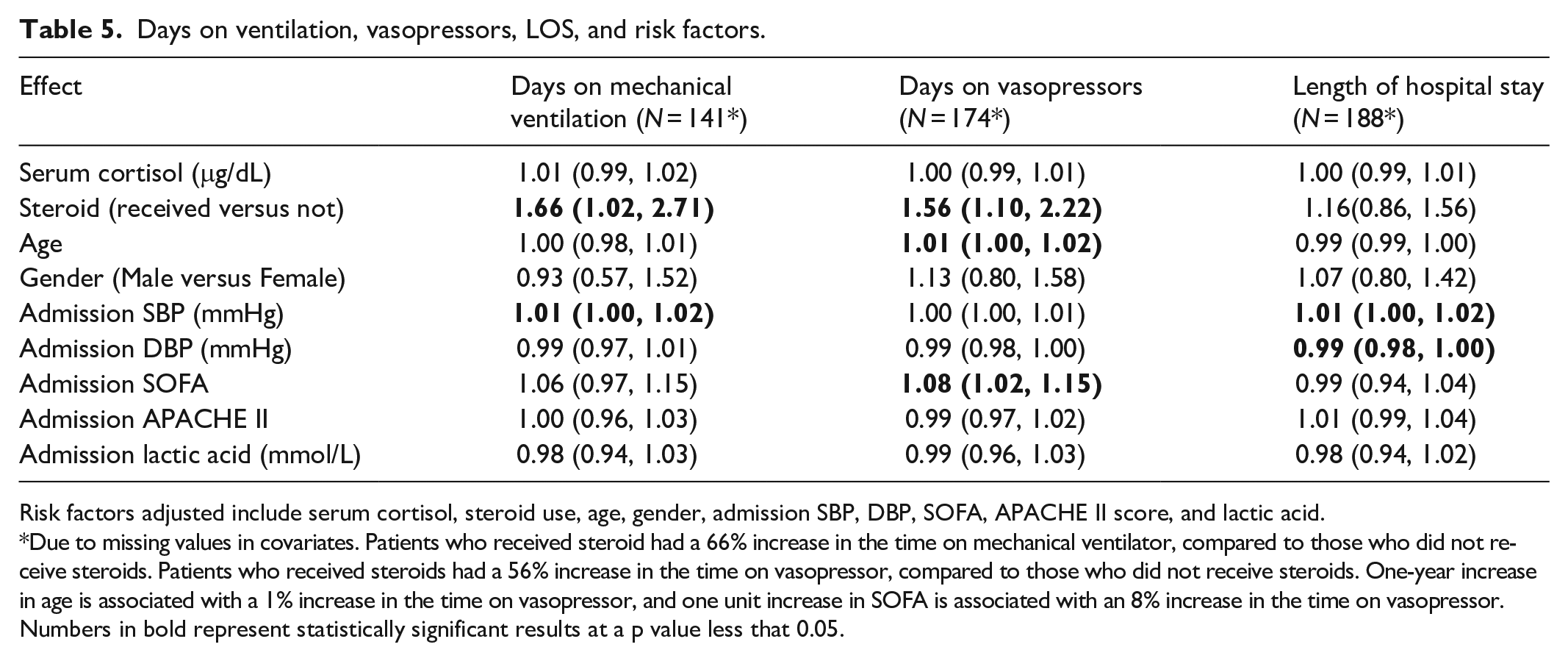
Risk factors adjusted include serum cortisol, steroid use, age, gender, admission SBP, DBP, SOFA, APACHE II score, and lactic acid.
Due to missing values in covariates. Patients who received steroid had a 66% increase in the time on mechanical ventilator, compared to those who did not receive steroids. Patients who received steroids had a 56% increase in the time on vasopressor, compared to those who did not receive steroids. One-year increase in age is associated with a 1% increase in the time on vasopressor, and one unit increase in SOFA is associated with an 8% increase in the time on vasopressor.
Numbers in bold represent statistically significant results at a p value less that 0.05.
Days on ventilation, vasopressors, LOS, and risk factors with binary serum cortisol levels.

Risk factors adjusted include serum cortisol, steroid use, age, gender, admission SBP, DBP, SOFA, APACHE II score, and lactic acid.
Due to missing values in covariates.
Numbers in bold represent statistically significant results at a p value less that 0.05.
Discussion
This study provides information on a group of patients hospitalized in a medical ICU with sepsis and septic shock. Cortisol levels were measured in the subset of patients based on decisions made by the clinicians managing these patients. Hydrocortisone was added to the therapeutic regimen in these patients also based on clinical decisions. There was no protocol dictating when to collect cortisol levels or when to start hydrocortisone. Consequently, this represents “a real world” study that provides us with information on outcomes in a heterogeneous group of patients. Higher cortisol levels were associated with increased mortality and increased AKI. Administration of hydrocortisone was associated with increased acute respiratory failure, increased vasopressor use, and increased mortality. This study suggests that high cortisol levels in critically ill patients indicates that these patients are at increased risk for complications and for mortality. Cortisol levels seem to represent a parameter of current levels of stress. The use of hydrocortisone did not improve outcomes, and its use was either associated with worse outcomes or was primarily used in patients who are at risk for worse outcomes, except possibly in the patients with cortisol levels <10 micrograms per deciliter (based on unadjusted comparisons).
Randomized control trials have provided mixed results on the use of hydrocortisone in patients with septic shock. Venkatesh and coworkers randomized 3800 patients with septic shock requiring mechanical ventilation into one cohort receiving 200 mg/day of hydrocortisone by continuous infusion and into one cohort receiving placebo. 3 They had been treated with vasopressors for a minimum of 4 h up to the time of randomization. The median APACHE 2 score was 24.0, the highest lactate level was 34.2 mg/dL, the lowest PaO2/FiO2 ratio was 64.6 mmHg. At 90 days, there was no difference in mortality between the hydrocortisone group and the placebo group. In addition, there was no significant difference in mortality at 28 days, the rate of recurrence of shock, the number of days alive and out of the ICU, the number of days alive and out of the hospital, the recurrent need for mechanical ventilation, the rate of RRT, and the incidence of new onset bacteremia or fungemia. The percentage of patients with adverse events was higher in the hydrocortisone group than the placebo group. Important events include hyperglycemia, hyponatremia, and myopathy.
Annane et al. included 1241 patients in a randomized control trial that compared outcomes in patients who received hydrocortisone plus fludrocortisone, drotrecogin alfa, a combination of these three drugs, or placebo. 4 These patients were entered into this trial if they had septic shock for less than 24 h, a clinically or microbiologically documented infection, an increased SOFA score, and receipt of a vasopressor for at least 6 h. Patients received hydrocortisone 50 mg IV every 6 h and fludrocortisone 50 mcg daily by tablet. The 90-day mortality rate was lower in the hydrocortisone plus fludrocortisone group. The number of vasopressor-free days at day 28 and the number of organ failure-free days were higher in the patients treated with active drug. The risk of hyperglycemia was significantly higher in the hydrocortisone group, but the risks for GI bleeding and superinfection were not higher. The difference in outcomes between these two randomized control trials may reflect the effect of fludrocortisone on outcomes or it may reflect the fact that patients in the Annane trial had to have shock for <24 h before entry. The Surviving Sepsis Campaign guidelines published in 2021 concluded that corticosteroids improved shock resolution and increased vasopressor-free days. This resulted in a weak recommendation of using low-dose corticosteroids and septic shock in patients with an ongoing requirement for vasopressors. 1 Our study would not support this conclusion but has the limitations of a retrospective study with patient management by several clinicians who may have different perspectives about the use of corticosteroids.
The hypothalamus–pituitary–adrenal axis has complex responses during acute stressful disorders such as sepsis.9,10 The hypothalamus produces corticotropin releasing hormone and arginine vasopressin that increase the release of ACTH. This leads to increased adrenal hormone production and increased cortisol levels. Cortisol metabolism is decreased in both the liver and kidney during sepsis, and this increases the half-life. The cellular effects of cortisol are mediated by binding to the glucocorticoid receptor, which allows entry into the nucleus and mitochondria. This results in either increased or decreased transcription of pro-inflammatory genes. However, some studies suggest that tissues are resistant to glucocorticoids during sepsis, and even though levels are high there is not necessarily an increase in transcriptional activity. Studies have not identified an optimal level of cortisol associated with improved outcomes.
Sam et al. measured cortisol levels in 100 patients with sepsis. 11 These levels were measured within the first 48 h of the development of severe sepsis or septic shock. Typically, the levels were obtained in the early morning. There was a significant spread in the cortisol levels in these patients, and they ranged from less than 345 nmol/L (12.5 mcg/dL) to greater than 1242 nmol/L (45.2 mcg/dL). They classified these patients into four groups. Patients in the group with the highest cortisol level had increased mortality (81%). In three groups there was no correlation between the APACHE 2 scores and the cortisol levels. In addition, there was no significant difference in the glucose levels in the four cortisol groups. There was a significant variability in the APACHE 2 scores and glucose levels in each cortisol group. These authors concluded that high cortisol levels in patients with septic shock were associated with increased mortality. Goodman et al. 12 measured unstimulated and ACTH-stimulated cortisol levels in 34 patients with sepsis on days 1, 2, and 28. Cortisol levels ranged from 221 nmol/L (8.01 mcg/dL) to 1545 nmol/L (56 mcg/dL) on day 1. Eight patients had unstimulated cortisol levels of less than 15 mcg/dL on days 1 and 2, a result consistent with adrenal insufficiency. Patients with the lower unstimulated cortisol levels had longer ICU and hospital lengths of stay.
Vassiliadi and co-authors measured baseline cortisol, stimulated cortisol levels, and ACTH every 3–4 days from 30 days levels in 51 consecutive mechanically ventilated patients with sepsis. 13 They did not find significant variation in either baseline or stimulated cortisol levels. ACTH levels were low on day 1 and then increased after day 10. Depending on the definition used, there was significant patient variability in the diagnosis of adrenal dysfunction at different time points. Many patients changed classification between having and not having adrenal dysfunction depending on the times they were tested. These authors suggested that ACTH stimulating tests in patients with sepsis may not have much utility. Venkatesh et al. briefly discussed 10 misconceptions about cortisol in critically ill patients. 14
Important comments relevant to this discussion include the observation that higher random cortisol levels are associated with greater mortality and that there is a substantial variation in cortisol levels in these patients. In our study, higher cortisol levels were associated with increased mortality and other adverse clinical events. Therefore, high cortisol levels should lead to a review of the clinical status and management plans for that particular patient. Levels below 10 mcg/dL suggests that the patient may have adrenal insufficiency and may warrant replacement therapy, but not necessarily pharmacologic doses.
Sepsis causes complex pathophysiologic responses in patients, including the activation phagocytes, the production of cytokines, the activation of complement, and alterations in mitochondrial function resulting in the production of reactive oxygen and nitrogen species.15,16 There is no uniform pattern for the development and trajectory of these responses. Consequently, the introduction of additional treatment in patients with prolonged and refractory sepsis has a high likelihood of failure unless an easily identifiable subgroup of patients qualifies for this add-on treatment. For example, Andrades et al. reviewed redox pathways in patients with sepsis and found that these patients often have reduced antioxidant levels and increased oxidant levels. 17 Jensen et al. reported that patients with sepsis have prolonged reactive oxygen species production from monocytes up to 28 days. 18 Studies on the use of vitamin C as an antioxidant illustrate the difficulties in using a single drug in heterogeneous group of patients.19–21 Hydrocortisone might be beneficial in some patients, but we do not know which patients and how much and when it should be started. This drug even in pharmacologic doses is unlikely to benefit patients with glucocorticoid resistance. 22 Recent systematic reviews and meta-analyses on the use of hydrocortisone in patients with sepsis and septic shock have provided disparate results.5,23,24
This study has the usual limitations often associated with retrospective studies. Cortisol levels were collected based on clinical decisions made during patient management. In general, they were collected early in the hospital course but not in all cases. Patients in whom cortisol levels were measured later in the course likely have worse outcomes because they had been sick for a longer period of time. Consequently, there was no consistent protocol for measuring cortisol levels. In this study hydrocortisone was started based on clinical judgment and not based on either protocol or on a particular cortisol level. Most of these patients likely had persistent vasopressor-dependent shock with no other obvious management option. There was no protocol used for starting hydrocortisone in these patients. The study protocol did not have an
Conclusion
In summary, patients with sepsis and septic shock have a wide range of cortisol levels. In this study, higher levels were associated with increased mortality and presumably reflect the overall level of clinical stress in these patients. The use of hydrocortisone did not improve outcomes and, in fact, was associated with increased mortality except possibly in patients with low cortisol levels. This could reflect adverse effects of the drug or the use of the drug in patients who had a high likelihood of an adverse outcome. Consequently, clinicians could consider measuring cortisol levels as an additional parameter that has the potential to predict outcomes. The use of hydrocortisone in patients with refractory shock has a relatively low likelihood of improving outcomes and can potentially increase the number of adverse events. Therefore, the use of this drug in critically ill patient requires careful consideration of the potential advantages and disadvantages; in particular, the best dose remains uncertain.
Footnotes
Author contributions
Marcella Rivas – study design, data collection, literature review, manuscript drafting, final approval of manuscript. Arunee Motes – data collection, data review, literature review, final approval of manuscript. Amr Ismail – data collection, data analysis, final approval of manuscript. Shengping Yang – data analysis and final approval manuscript. David Sotello – study design, data collection, literature review, final approval of manuscript. Meily Arevalo – data collection, data review, final approval of manuscript. Wasawat Vutthikraivit – data collection, data review, final approval of manuscript. Sakolwan Suchartlikitwong – data collection, data review, final approval of manuscript. Cynthia Carrasco – data collection, data review, final approval of manuscript. Kenneth Iwuji – data collection, data review, final approval of manuscript. Pavida Pachariyanon – data collection, data review, final approval of manuscript. Sarah Jaroudi – data collection, data review, final approval of manuscript. Subhanudh Thavaraputta – data collection, data review, final approval of manuscript. Kenneth Nugent – study design, data review, literature review, manuscript drafting, final approval of manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from the Institutional Review Board at Texas Tech University Health Sciences Center (L19-091) in Lubbock, Texas.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was not sought for the present study because this was a retrospective study approved by the Institutional Review Board at our university and this requirement was waived.
