Abstract
Objective:
Proper documentation of the functionality and complications of peripheral intravenous catheters (PIVC) is the standard of care. This data can improve communication among team members about access concerns and highlight opportunities to improve PIVC care. Our objective is to determine if nursing personnel are compliant with institutional standards for documentation and documentation is reliable.
Methods:
This prospective observational analysis was conducted at a tertiary care academic center with 120,000 ED visits and 1100 hospital beds. Adults over 18 with a PIVC placed in the ED via palpation technique who were being admitted to regular medical/surgical wards were eligible. The primary outcome was compliance with PIVC documentation per institutional standards. Secondary outcomes included compliance subcategorized as insertion, daily assessment, and removal and reliability of assessments.
Results:
During July and August 2020, 77 patients were enrolled with a total of 1201 observations of PIVC compliance. PIVC documentation compliance was 86.0% (1033/1201). Compliance on insertion and removal was 93.3% (431/462) and 80.5% (186/231), respectively, with removal assessment being the least compliant at 49.4%. Daily catheter assessments were compliant 81.9% (416/508) of the time. PIVC documentation reliability was based on 693 total observations with 87.9% (609/693) reliability overall, and a reliability of 91.6% (423/462) and 74.9% (173/231) for insertion and removal, respectively. PIVC orientation had the highest reliability (98.7%) while post-removal assessment had the lowest reliability (45.5%).
Conclusions:
We observed moderate documentation compliance and reliability for PIVC assessments for catheters placed in the ED. Documentation of removal-related variables was the most deficient aspect of the assessments. Given the high rate of PIVC failure and its vast array of consequences, improvement of PIVC documentation of removal reasons is essential to better identify type and incidence of complications and help develop targeted solutions. Further larger studies are needed to survey PIVC documentation practices.
Keywords
Introduction
Functional vascular access is vital for delivering life-saving fluids, blood products, and medications during hospitalization. Nearly 90% of patients require peripheral intravenous catheter (PIVC) placement during their hospital stay, which results in placement of over 2 billion PIVCs worldwide every year.1,2 Unfortunately, PIVCs are prone to developing numerous complications that may result in premature catheter failure. In fact, up to 63% of PIVCs fail prior to completion of therapy.2 –4 Many patients experience significant harm related to vascular access dysfunction in the form of repeated insertion attempts and treatment delays. Additional downstream consequences of catheter dysfunction include venous depletion, increased rates of nosocomial infections, prolonged hospital stay, reductions in patient mobility and daily activities, and increased healthcare costs.5 –11 Despite these inadequacies, there have been limited interventions to solve this overwhelming problem from the healthcare community at large. 2
One obstacle to improving PIVC outcomes is the lack of precise data regarding complications. While Infusion Therapy Standards of Practice recommends assessment of catheter sites (every 4 to 6 h for inpatients and hourly among intensive care patients) in order to keep these essential devices functional, it is unclear if these evaluations are conducted regularly and reliably. 12 Several times a day, nursing staff must perform these assessments and are expected to document their findings in the electronic health record (EHR). As nurses generally have multiple patients of various acuity levels, compliance with the standard of frequent PIVC site assessments and documentation of findings may be a lower priority. Accurate and complete documentation, however, is a critical component of delivering quality patient care and should be prioritized. Practical documentation should reflect actual clinical practice and facilitate the best treatment and outcomes across the continuum of care by enhancing communication and transparency among team members. This would ultimately enable providers to anticipate patient vascular access needs and address complications in a timely manner. Furthermore, reliable documentation can help researchers establish the root causes of PIVC dysfunction and create tailored solutions. The majority of current literature relies on the EHR as the data source for PIVC complication rates, with evidence highlighting wide ranges in the frequency of complications, which may be due to inadequate documentation or assessment practices. 2 Therefore, the purpose of this study is to determine if clinicians are compliant with institutional standards for documentation and if the documentation is reliable.
Materials and methods
Study design, setting, and participants
This was a prospective observational exploratory study evaluating the reliability and compliance of PIVC assessments using medical record documentation as the data source. Study participants were a convenience sample of hospitalized emergency department (ED) patients within a large, 1100 bed, tertiary care center located in the Detroit metropolitan area of Michigan in the United States. The Institutional Review Board at our institution approved this study.
Eligible participants included adults (⩾18 years old) with a PIVC placed by a nurse or ED technician in the ED and requiring hospital admission to a regular medical or surgical ward. Exclusion criteria included voluntary withdrawal, cognitive impairment, and PIVCs inserted with ultrasound guidance or prior to arrival. Verbal informed consent was obtained from all subjects prior to enrollment in the study.
Study procedure
After patient enrollment, researchers assessed the functionality of the PIVC in the ED and recorded insertion-related data from the medical record, including the insertion date, insertion time, the individual that inserted the PIVC, size (18 gauge, 20 gauge, or 22 gauge), laterality (left vs right) and location of insertion (wrist, forearm, antecubital, upper arm). Researchers also recorded demographical data, which included age, gender, and BMI.
On a daily basis, researchers performed follow-up visual PIVC assessments to assess for complications and failure. Researchers also recorded documentation of routine catheter assessments in the EHR, including line status, dressing status, and site assessment, which is required every 8 h per institutional policy. After the catheter either failed or reached completion of therapy, researchers recorded EHR documentation pertaining to its removal, including removal date, removal time, and post-removal assessment.
Study definitions
On an individual documentation field level, compliance was defined as the presence of any value within a field regardless of accuracy, while noncompliance was defined as a missing or blank values from these same fields. Compliance was also calculated on a categorical level, looking specifically at insertion related variables, daily assessment variables, and removal related variables. To meet compliance standards within the category of insertion related variables, the following six fields needed to contain non-null values: insertion date, insertion time, inserted by, size, orientation, and location. To be categorized as compliant with daily assessment variables, the following fields needed to be a non-null value: line status, dressing status, or site assessment. Compliance with the institutional standard for frequency of daily assessments required PIVC evaluations every 8 h. Compliance with the removal related variables required a non-null value in the following three fields: removal date, removal time, and post-removal assessment. Post-removal assessment included whether or not PIVC reached completion of therapy and presence or absence of complications. Complications included: catheter damaged, infiltrated, leaking, occluded, site erythema, site pain, patient removed, and none. Overall compliance was the sum of all insertion, daily, and removal variables.
Reliability was defined as of accuracy of documentation variables when compared to assessment of the PIVC by research staff. We considered the gold standard assessment to be that which was performed by members of the research team. Missing fields were marked as incorrect. Reliability was assessed for insertion variables (insertion date, insertion time, inserted by, size, orientation, and location) and removal variables. Overall reliability on insertion and removal was the sum of all insertion and removal variables.
Outcome measures
The primary outcome was compliance of PIVC documentation with the institutional documentation standard. Secondary outcomes included compliance subcategorized as insertion, daily assessment, and removal and reliability of assessments.
Statistical analysis
All data were summarized through descriptive statistics. Categorical variables are reported as counts and percentages.
Results
This study took place from July thru August 2020. A total of 77 patients were enrolled and included in the final analysis.
Overall compliance of all PIVC documentation variables was 86.0% (1033/1201). Insertion-related compliance was 93.3% (431/462). Of the six insertion-related variables, the highest compliance (98.7%) was for orientation while the lowest compliance (80.5%) was for inserter details. Compliance for daily catheter assessments was 81.9% (416/508). Removal-related compliance was 80.5% (186/231). Of the three variables for removal compliance, removal date (98.7%) had the highest compliance while removal assessment (49.4%) had the lowest compliance (Table 1).
Compliance of peripheral intravenous catheter documentation.
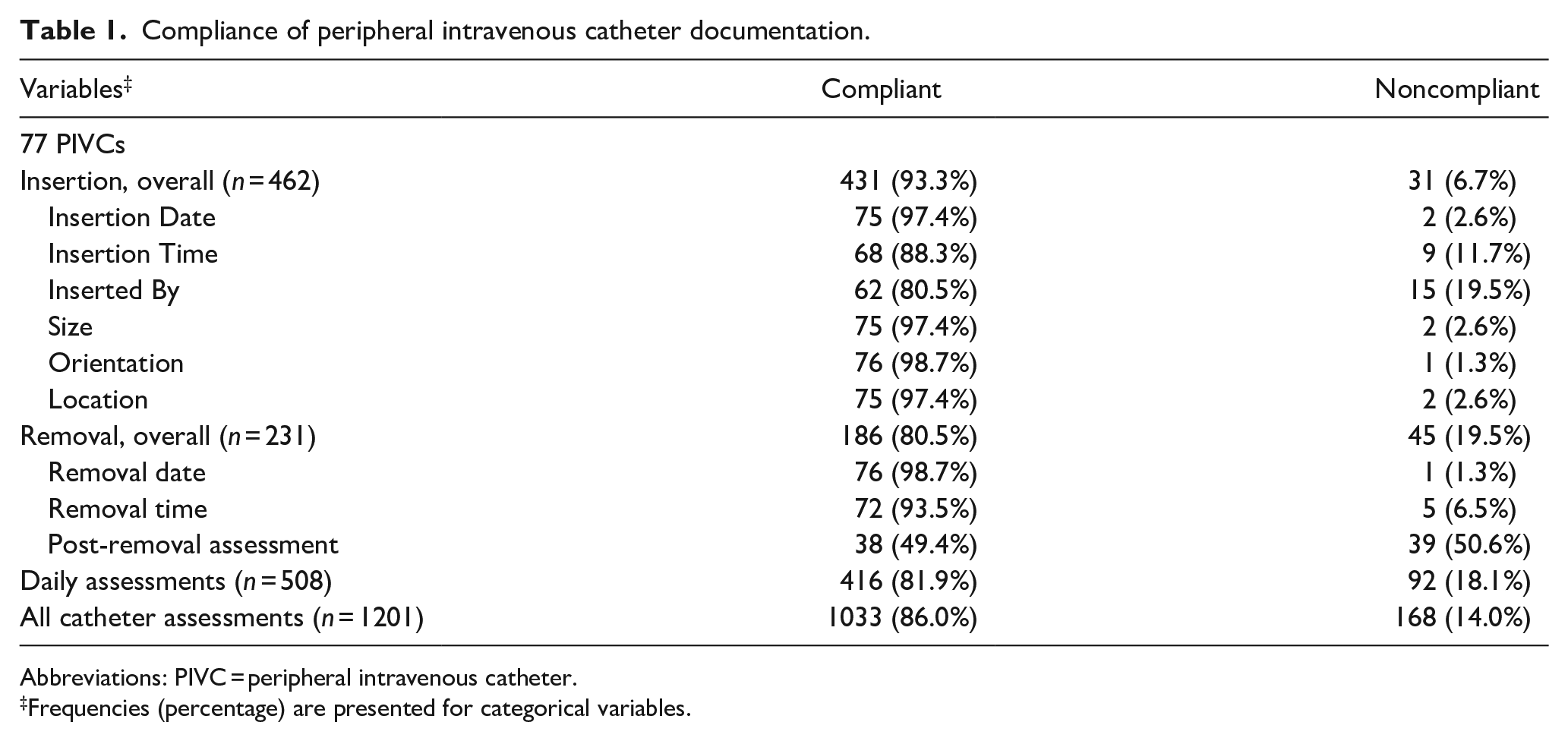
Abbreviations: PIVC = peripheral intravenous catheter.
Frequencies (percentage) are presented for categorical variables.
Overall reliability was 87.9% (609/693). Insertion-related reliability was 91.6% (423/462). For insertion related variables, the highest reliability (98.7%) was for orientation while the lowest reliability (80.5%) was for inserter details. Removal-related reliability was 74.9% (173/231). For removal-related variables, the highest reliability (90.9%) was for removal date while the lowest reliability (45.5%) was for post-removal assessment (Table 2).
Reliability of peripheral intravenous catheter documentation.
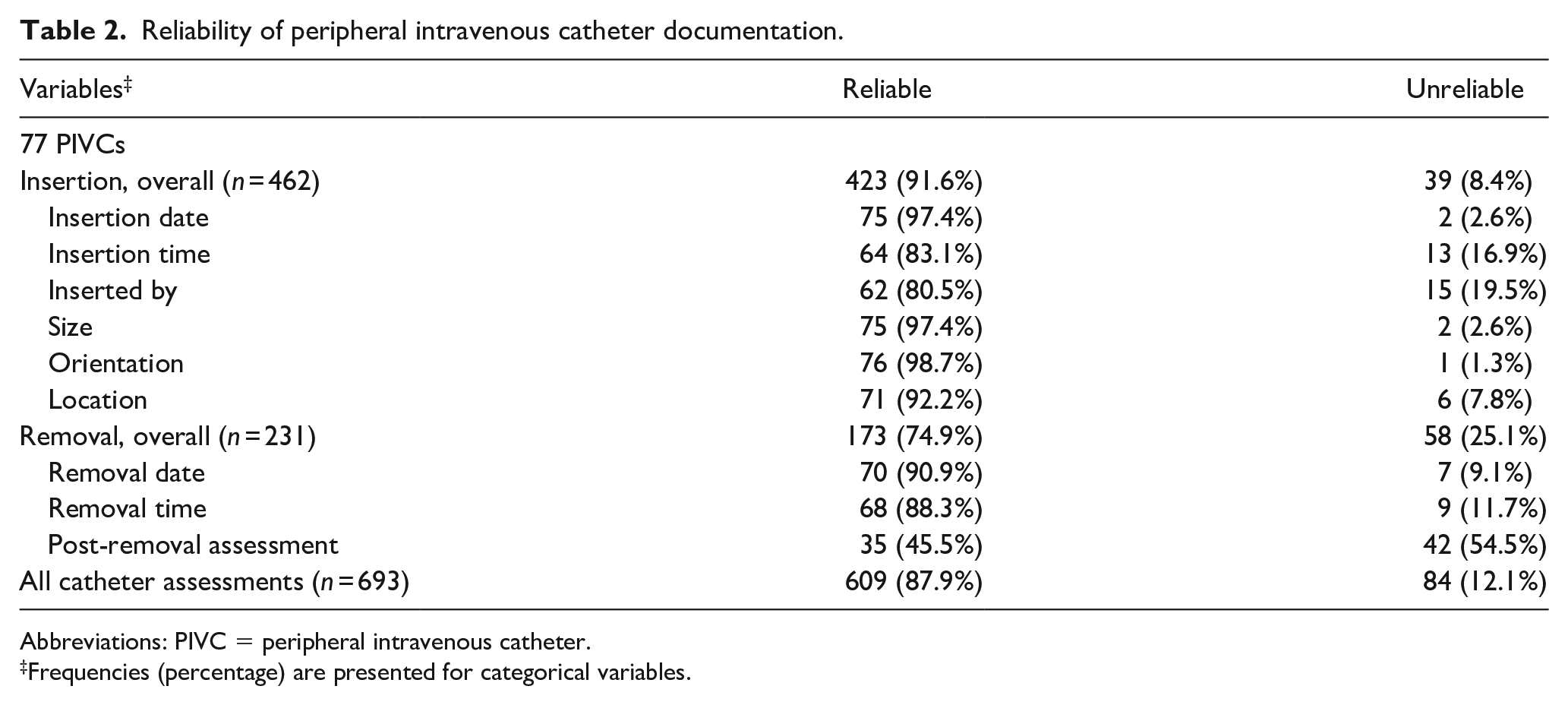
Abbreviations: PIVC = peripheral intravenous catheter.
Frequencies (percentage) are presented for categorical variables.
Limitations
Our study had some limitations. First, this was a small sample size of inpatients at a single tertiary care center and it is possible that results may not precisely capture the true incidence of the PIVC documentation problem. Further, because the study was exploratory, no sample size estimation or power analysis was performed during the design phase. Second, compliance assessed documentation practice, not clinical practice. It is likely that some assessments may have been completed that were never documented. It is also plausible that some assessments were never done but marked as complete. Third, the reliability of daily assessments could not be adequately assessed. Standard institutional policy requires assessments to be performed every 8 h and include visual site and functionality assessments. As research assessments were performed once daily and were limited to visual site assessments, no meaningful or objective comparison could be made. Fourth, as there is inherent subjectivity to PIVC evaluation particularly in daily and removal assessments, there are some limitations to the accuracy component of the results. Finally, there is a selection bias due to convenience sampling and likely Hawthorne Effect as nurses may have been aware that patients were being enrolled into a research study regarding PIVCs.
Discussion
This prospective observational study is one of the first investigations to our knowledge that explores the compliance and reliability of PIVC documentation. Aggregate compliance and reliability was moderate at 86.0% and 87.9%, respectively but there were notable deficiencies in the documentation of removal variables with only 49.4% compliance. As there is limited existing data on this subject, these results serve as a baseline evaluation of this important topic. In fact, we only identified two other studies that evaluated the compliance of PIVC documentation. New et al. observed a 75% compliance rate with daily PIVC assessments, which was similar to our findings of 82% for this component. 13 Their study, however, did not address the likely more important questions of whether key outcome data was being documented, such as post-removal assessment and if this documentation was accurate. The other study reported a much lower rate of location documentation (32%) compared to this study (97%). 14
In our analysis we demonstrated that while insertion-related variables had a high rate of compliance and accuracy, the variables tied to removal outcomes had very low compliance and even lower reliability. Given that EHR data is the surveillance mechanism to characterize PIVC failures and complications, it is imperative that we clarify the limitations of this data. Our analysis highlights that we are likely unable to draw any meaningful conclusions about the causes of PIVC failure and complications from EHR data given the extent of the documentation problem. Thus, it is not surprising that existing evidence informs immensely broad ranges of the most common complications. For instance, catheter-related phlebitis, infiltration and occlusion are reported to occur 0.1%–63.3%, 15.7%–33.8%, and 2.5%–32.7%, respectively. 2
The prospective nature of this study enabled the research team to monitor the reliability of PIVC documentation. Similar to compliance, the reliability of post-removal assessments was most deficient, with 54.5% of assessments being unreliable. As subjectivity in PIVC assessments limits characterization of inaccuracies, research staff only considered an assessment unreliable for missing data or grossly inaccurate documentation highlighting that performance may be even worse. For instance, in one case, research staff reported kinking and dislodgment of a catheter upon assessment, but the documentation reported a functional catheter for 3 days until patient discharge. In another scenario, research staff witnessed infiltration after flushing that led to catheter removal. This catheter was also noted to be functional for the next 4 days with a post-removal assessment indicating the catheter reached completion of therapy without complication. While the spectrum of reliability ranged from incomplete fields to gross inaccuracy, it was clear that the documentation of removal variables represents a substantial opportunity for improvement. Because the documentation of complications and failure is essential to understanding the root causes of PIVC dysfunction and developing strategies to improve these outcomes, PIVC documentation needs prioritization. Even though PIVC complications occur regularly, its inaccurate and inconsistent documentation may be obscuring the true impact of PIVC failure on patients. Increasing clinician awareness of the value of these documentation elements via education may help curb this deficiency and lead to improvements in patient care.
Conclusions
Compliance and reliability of PIVC documentation in the medical record for catheters inserted in the ED was moderate. Insertion-related variables had the highest compliance and reliability, whereas removal-centered variables had substantial deficiencies. As PIVCs have high complication and failure rates, accurate and complete documentation, specifically for removal variables, is essential for the best quality of care and a target for improvement. Further larger studies are needed to survey PIVC documentation practices.
Footnotes
Author contributions
AB, NM, and SJ designed the study, had full access to the data, and take responsibility for the integrity and accuracy of the data analysis. AB, NM, and SJ contributed to subject enrollment, data collection, and statistical analysis. All authors contributed to the writing and editing of the manuscript. All authors contributed to data acquisition, analysis and interpretation, and all reviewed and approved the final version of the manuscript. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Within the past 36 months, AB has had research grant support from Teleflex, Becton Dickinson, B. Braun, Adhezion, 3M, Access Vascular, and Medline.
AB is a paid consultant for Teleflex and B.Braun.
All other authors declare no relevant conflicts of interest relevant to this work.
Data sharing statement
The data that support the findings of this study are available via a data access agreement. Please contact the corresponding author (AB) for this request.
Ethics committee approval
This study was approved by the Beaumont Health Institutional Review Board.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
