Abstract
Introduction:
The most successful programme for secondary fracture prevention is the FLS (fracture liaison service) model. Our orthopaedic department carried out a prospective randomised study to measure the effectiveness of a 4-step intervention programme. The findings in this study reveal important additional clinical benefits to having an orthopaedic-based FLS programme and evaluates the usefulness of fracture risk tools.
Methods:
We carried out a prospective study to evaluate patients with a fragility fracture of the hip. There were 2 groups, intervention and control (each 100 patients). Of these, 20 were either removed from the study or dropped out, leaving 180 for analysis. In addition to routine preoperative blood tests, albumin and thyroid function levels were obtained and PTH (parathyroid hormone) levels when indicated.
The intervention group (83 patients) had a dual-energy x-ray absorptiometry (DEXA) scan performed and fracture risk (FRAX) was calculated.
Results:
12 patients (6.7%) had blood results which showed a potentially treatable cause for osteoporosis and 36 (20%) had blood results that changed their medical care.
FRAX scores (180 patients) showed that the major osteoporotic fracture score correctly predicted the hip fracture in only 49%. The hip fracture score correctly predicted the hip fracture in 83%.
DEXA scores (65 patients) showed osteoporosis in only 46% of hips and in only 26% of spines.
An abnormal FRAX score or DEXA scan would have predicted a fragility fracture 93% of the time.
Conclusions:
In addition to reducing secondary fractures, FLS programmes can provide fundamental benefits to the health of the patient. The intervention programme in this study identified patients with underlying treatable causes, correctable clinical conditions and patients with an unusually low bone density. When used together, FRAX and DEXA are more sensitive predictors for hip fracture risk than either are individually.
Trial registry:
201497CTIL (https://clinicaltrials.gov/ct2/show/NCT02239523)
Introduction
Patients who present with a fragility fracture are consistently under-evaluated and under-treated for underlying osteoporosis. 1 Orthopaedic surgeons treat the patients for their fracture and primary care physicians focus on general well-being, but no one addresses their bone health. Strategies to convince primary care to assume responsibility have not succeeded. 2 On the other hand, strategies where a fracture care coordinator is accountable have been successful. 3 A prospective 2-arm study evaluated the outcome of an attempt by an academic orthopaedic department to evaluate and treat osteoporosis.
The primary purpose of the study was to improve the initiation of treatment to prevent secondary osteoporotic fracture. This was accomplished by the introduction of an intervention programme by the orthopaedic department after a hip fragility fracture. However, the Fracture Liaison Service (FLS) programme provided additional clinical findings that directly benefitted the patients. In addition, the information collected could be used to further identify patients at risk of fragility fracture, information that could be useful for primary prevention.
Methods
The trial was registered at ClinicalTrials.gov prior to Helsinki approval (201497CTIL). All patients who had been hospitalised after a low-energy hip fracture at our Level I trauma centre were candidates for the study. After meeting inclusion criteria, patients were randomised into 1 of 2 levels of evaluation and treatment. Randomisation was done from a table provided by our statistician created by a randomisation programme. A research assistant assigned participants to 1 of the 2 interventions based on the sequence. All patients in the intervention group (83 patients) had laboratory evaluation that included the tests listed below.
The intervention group protocol included a dual-energy x-ray absorptiometry (DEXA) scan. In addition, fracture risk was assessed using the fracture risk assessment tool (FRAX). This is available free online through Sheffield University (https://www.sheffield.ac.uk/FRAX/) and is region-specific. The results of DEXA and FRAX were analysed individually and together to determine how well the fragility fracture could have been predicted.
Patients in the control group were given a summary with a recommendation for evaluation and treatment by their family physician at the time of discharge. The letter included a recommendation that the family doctor evaluate the patient for osteoporosis and that the patient should be started on appropriate medication. This was termed the Letter group.
Inclusion/exclusion criteria
All patients over the age of 50 years admitted with a fragility hip fracture were considered for inclusion. We defined a fragility fracture as a fracture resulting from a low-energy fall, typically occurring while standing or walking. Hip fractures were those in the subcapital, femoral neck, intertrochanteric or subtrochanteric region. Fractures of the trochanter alone, those involving the shaft or periprosthetic region were not included. Patients with a fracture sustained in a non-low energy fall, those with metastatic cancer or known metabolic bone disease or patients in end-of-life care were also excluded. Patients unable to consent because of dementia were excluded but were included if their dementia was mild and consent could be obtained.
Data management
Data was entered into the database created using the Research Electronic Data Capture (RedCAP) from Vanderbilt University and available through the research department provided by our medical centre.
DEXA study
While in hospital, a DEXA scan was carried out on those patients in the Intervention Group who had not had a DEXA scan in the previous 24 months or whose DEXA scan results were not available. We use the Hologic DXA system (Bedford, MA, USA) at our institution. The standard protocol is to scan the opposite non-fractured hip, the lumbar spine and the distal forearm. If the opposite hip has been fractured in the past, then only the spine and distal forearm are scanned. Bone mineral density used the T-score and was adjusted for the site of the scan.
Laboratory evaluation and medication treatment algorithm
Laboratory evaluation on admission included obtaining levels of calcium, albumin, creatinine, electrolytes (sodium, potassium, bicarbonate and chloride), full blood count (FBC), and thyroid stimulating hormone (TSH). Creatinine clearance was calculated. For the purpose of the medication algorithm, renal failure was defined as a creatinine clearance <30 ml per minute. If the calcium was elevated, or in those who were defined as having failed bisphosphonate therapy (see below), then the parathyroid hormone (PTH) level was also determined. If the calcium level was low, then in addition to giving vitamin D and calcium supplements, repeat levels of calcium were obtained every 2 weeks until the calcium was in the normal range. The medications for osteoporosis listed below, were given only after calcium had returned to within the normal range.
All patients in both groups with the exception of those with defined renal failure were given a loading dose of vitamin D (50,000 IU orally) then continued vitamin D at 1000 IU per day during and after their hospitalisation. If the patient had defined renal failure then no loading dose was given but vitamin D in the form of Alpha D3 0.25 micrograms orally was given instead and continued.
Statistical analysis: power of study
The study was initially powered for the intervention analysis. Specifically, we wanted to determine if the intervention group had a higher rate of starting medication compared to the control group. We assumed that 20% of patients in the Letter group would be on the recommended treatment by 4 months. We anticipated that we could improve treatment levels to 50% using the interventions described above. This required a minimum of 100 patients in each group. Overall, 118 patients in each group or 236 patients total were required to achieve 94% power, assuming 10% of patients were lost to follow-up at 4 months and a 5% mortality. As the study progressed, we had a lower mortality than predicted and no patients were lost to follow-up at 4 months so we stopped the study at 200 patients.
Baseline characteristics were compared using chi-square and Fisher’s exact tests for categorical data, depending on the distribution of the data. Body mass index (BMI) comparison was done with a Student’s t-test. Logistic regression was used to analyse differences in correct treatment rates. Fisher’s exact tests were conducted for differences between DEXA results and outcome.
Results
The enrolment period was from 21 February 2017 to 15 September 2018 when the target of 200 patients was reached. Their age range was 51–95 years with a mean of 79.2 (±9.2) years. 72% were women.
During the enrolment period, there were 618 patients with a low-energy hip fracture of whom 305 were eligible for enrolment (see flow diagram Figure 1). None of the patients in this series had bilateral fractures. Most of the ineligible patients were excluded on the grounds of moderate or severe dementia. 66% of those eligible agreed to participate in the study.
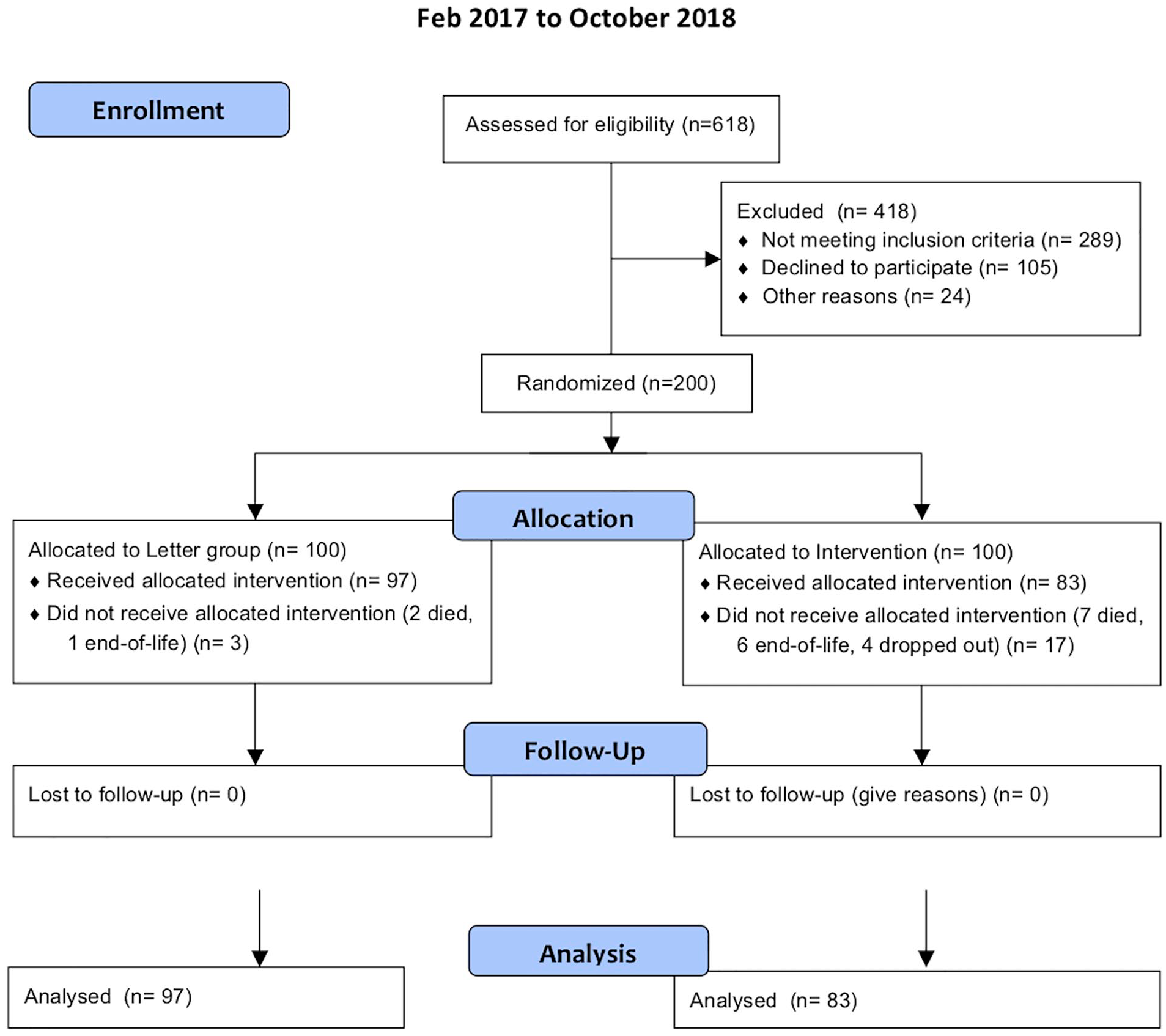
Fragility hip fractures flow diagram.
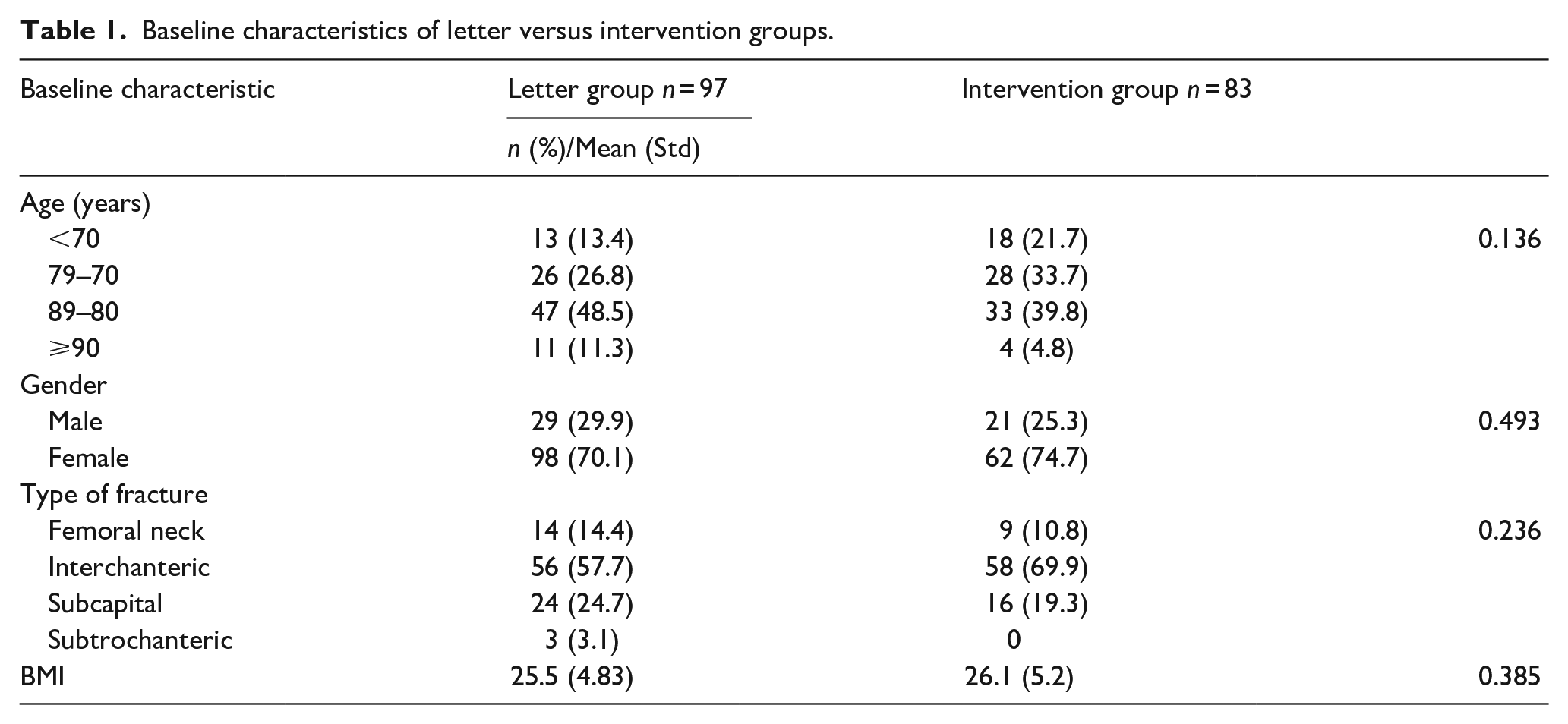
100 patients were enrolled into the Letter group. 3 of these were removed from the study (2 died and another was diagnosed as terminally ill before the 4-month outcome measure). 100 patients were enrolled into the intervention group. 13 were removed from the study, (7 died and 6 were diagnosed as terminally ill before the 4-month period) and 4 dropped out of the study. Baseline characteristics did not differ between groups (Table 1). The analysis is based on the remaining 180 patients in both the intervention group and control groups.
Baseline characteristics of letter versus intervention groups.
Laboratory results
Overall, 12 (6.7%) of the patients had a potentially treatable underlying cause identified (7 with high PTH and 5 with low TSH levels). An additional 36 (20%) patients had lab results that altered medical management. This included 4 with low calcium levels that needed correction, 17 with elevated TSH levels that required thyroid supplements and 15 with a new diagnosis of reduced kidney function. Overall, 27% of the patients had labs that were clinically important.
FRAX
FRAX was calculated for all 180 patients in the study. Knowing that the patient had sustained an osteoporotic fracture, the FRAX would ideally have predicted the fracture that had been sustained. There are 2 categories of risk identified, major osteoporotic risk and risk specifically of a hip fracture, both of which predict the 10-year probability of a fracture. The FRAX score is based on an algorithm that is geographically specific. The FRAX can be calculated with or without the results of the DEXA scan. The threshold for treatment is usually when the 10-year probability is 10% or more for a major osteoporotic fracture or >3% for a hip fracture. If this threshold was met, then we considered the FRAX score to have correctly predicted the fracture.
The hip fracture was predicted by the FRAX score in only 49% using the major osteoporotic fracture criteria. The hip fracture was predicted in 83% when using the hip fracture criteria. The hip fracture was better predicted by the hip score than by the major osteoporotic score (p < 0.0001).
DEXA
Although DEXA was part of the intervention protocol, only 65 (78.3%) of the patients in the intervention group had a DEXA scan carried out. Reasons for not for not doing it included scheduling issues during or after their hospitalisation, poor mobility, obesity and methicillin-resistant Staphylococcus aureus (MRSA) status.
The DEXA scan met the criteria for osteoporosis in only 46% of hips, and in only 26% of spines. The DEXA score indicated osteoporosis in the hip more often than in the spine (p < 0.023). DEXA identified 5 patients (7.7%) with an unexpected t-score of <−3.5.
Combination of FRAX and DEXA
The combination of FRAX and DEXA appeared to be most predictive of the ultimate fracture seen. Specifically, if 1 or the other or both scores had been abnormal, then a fragility fracture would have been predicted in 93% of the patients.
Discussion
Osteoporosis is a worldwide epidemic. Developed countries are not immune from the morbidity and mortality of weak bone. Even worse, for those who sustain a fragility fracture, the risk of a subsequent fragility fracture is 2–4 times higher. 4 Secondary fracture prevention programmes are critical in reducing this risk. Furthermore, in addition to reducing secondary fractures, the FLS programme is of fundamental benefit to the health of the patient. The intervention programme in this study identified patients with underlying treatable causes, correctable clinical conditions and those with unusually low bone density. We strongly encourage orthopaedic programmes to start their own FLS programme. We report separately on the compliance rate for starting treatment comparing the intervention to the letter groups. 5 The focus in this report is the additional health benefits for having an FLS programme.
Treatment for osteoporosis includes the use of vitamin D and calcium supplements. However, this alone is not enough to prevent further loss of bone. Treatment for osteoporosis needs to include either an agent that reduces bone turnover such as a bisphosphonate or an agent that actually increases bone density. Calcium needs to be corrected before such agents can be started and this protocol includes that requirement. Ideally, vitamin D level is measured and corrected. The Horizon study started patients on zoledronic acid after hip fractures without requiring vitamin D correction and showed an improvement in the rate of re-fracture and a reduced mortality. 6 We were concerned that any delay caused by the evaluation of vitamin D levels and their correction might lead to a delay in starting their medication. For that reason and given the benign nature of vitamin D, all patients in both control and intervention groups were simply started on supplements.
The primary limitation of our study is the lack of inclusion of patients with moderate or severe dementia. We made this an exclusion criterion since a prospective study with monthly follow-up requires a patient who is able to provide consent and to be in monthly communication.
DEXA scans were included in the protocol though not normally needed after a fragility fracture before starting treatment. There were 2 reasons for this. First it served as a baseline so that the effectiveness of treatment could later be evaluated. Second, we suspected, based on previous literature, that compliance rates might be higher in the group who had a DEXA scan carried out. 7 The scan also helped to identify patients with an unexpectedly low bone density who were then referred for further evaluation in our Department of Endocrinology. Specifically, 5 patients (7.7%) had t-scores <−3.5 and 1, aged 58, had a t-score of <−3.1.
Successful FLS programmes require orthopaedic involvement. Patients with low-energy fractures enter the health system through the orthopaedic department which has a unique opportunity to intervene. It would be both logistically unrealistic and financially prohibitive for all fragility fractures to be evaluated and treated by an endocrine department. Successful FLS programmes are designed with their guidance but managed by allied health providers using an algorithm to guide recommendations. 3
The first-year mortality after a hip fracture is approximately 20%. 3 It has a worse prognosis than most cancers, and is specifically comparable to those of thyroid and breast cancer. 8 For those that survive, over 80% never get back to their pre-injury level of function. 9 We owe it to our patients, our parents, our grandparents and 1 day to ourselves, to take responsibility for bone health. The primary goal of the FLS programme is to get patients started on medication to prevent further bone loss or actually strengthen their bone. However, in addition, intervention programmes as described here can identify patients with a possible underlying treatable cause for their osteoporosis. Some patients had additional health problems identified by the programme that were important to their overall health. Finally, a DEXA scan, though not required, identified some patients with an unusually low bone density.
Taken together, the FLS programme was not only effective at getting patients started on medication but provided additional clinical benefits to many of the patients enrolled. We believe that although the goal of compliance with medication protocols is already sufficient reason to have a FLS programme, we have shown that there are additional health benefits for enrolled patients. We hope the orthopaedic community takes the opportunity to implement such a programme.
Footnotes
Acknowledgements
We would like to acknowledge our research assistant, Bat-el Harris, and our statistician, Noga Fallach, for their hard work and expertise.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
