Abstract
Background
Achromatopsia is an autosomal recessive cone dysfunction syndrome, characterized by absence of color discrimination, low visual acuity, photophobia, and nystagmus. Achromatopsia constitutes a common cause of visual impairment in children, with a prevalence of 1:30,000 worldwide.
Objective
To characterize the clinical characteristics of achromatopsia, the main genes causing the disease in our population and the clinical course of the disease, with an emphasis on visual function stability with increasing age.
Methods
Retrospective study based on medical charts of patients with achromatopsia. Patients were divided into two groups according to their age at last follow-up: older and younger than 10 years. A subset of patients with long term follow-up were analyzed separately, with patients being described in both age groups.
Results
Seventy-six patients were included in the study. The mean age was 14.28 years. Variants in the CNGA3 gene were the most common (73.6%). Clinical characteristics included photophobia (96.2%), nystagmus (93.6%), hypermetropia (72.3%) and strabismus (51.1%). In the large cohort there was no correlation of age with visual acuity (p = 0.129). In the separate subset cohort with long follow-up there was a relative improvement in visual acuity with age (p < 0.001).
Conclusions
CNGA3 is the main gene associated with achromatopsia in our population (around ∼ 73%), which is in contrast to the distribution worldwide (∼ 25%). Most achromats suffer from photophobia and nystagmus, and the main refractive error is hypermetropia. Achromatopsia's natural course seems to be stationary, and there may even be a slight improvement in visual acuity with time.
Keywords
Introduction
Achromatopsia (ACHM) is a rare congenital autosomal recessive disorder characterized by loss of cone function, 1 with an estimated prevalence of 1 in 30,000–50,000 worldwide. 2 However, the prevalence of achromatopsia is much higher in specific areas, with 1:10–1:16 achromats in Pingelapese island 3 and 1:5,000 achromats among Muslims in Jerusalem, Israel. 4 This may be related to founder mutations as well as consanguineous marriages in these populations.
Achromats suffer from various symptoms, including nystagmus and photophobia from early infancy, reduced visual acuity, hyperopia, and color vision defects.1,2,5–8 ACHM can be divided to complete (typical) or incomplete versions. 8 Patients with complete ACHM have total lack of cone function, whereas patients with the incomplete version retain some residual cone activity as not all three cone types are impaired.2,9 The symptoms of incomplete ACHM are similar to those of complete ACHM but are generally less severe. 6
The clinical diagnosis of ACHM is based on symptoms and supported by morphological and functional findings. 10 On electroretinography (ERG), cone-mediated responses are absent or markedly reduced, with normal or near normal rod responses.8,11 In addition, many ACHM patients show various degrees of morphological changes seen by spectral domain optical coherence tomography (SD-OCT) in the foveo-macular part of the retina, from loss of the outer segments of the cones to atrophy of the outer retina.8,12
The genetic basis of achromatopsia involves six currently known genes: CNGA3, CNGB3, GNAT2, PDE6C, PDE6H, ATF6. 12 Collectively, these genes account for approximately 90% of ACHM cases. 2 Mutation analysis in European subjects revealed that CNGB3 accounts for approximately 50% of the cases, 13 whereas CNGA3 accounts for only 25% of cases. 14
According to a recent study done in Jerusalem among Jewish and Palestinian populations, CNGA3 mutations are the leading cause of ACHM in Jerusalem (more than 50%).4,15
ACHM has been classically described as a stationary condition.11,16 However, several recent studies have suggested it may be a progressive condition and questions about the stability of the disease were raised.
According to several studies done in recent years, there is no correlation of age with deterioration in visual acuity or SD-OCT morphology, and patients generally remained stable.11,16,17 However, other studies have reported age-associated progression as measured by SD-OCT, with cone decay and retinal thinning.18,19 Thus, there is still an ongoing debate about disease progression and deterioration with age, and there is need for more research on the subject.
The objective of our work was to study the clinical characteristics of achromatopsia, the main genes causing the disease in our population and the clinical course of the disease, with an emphasis on visual function stability with age.
Methods
Subjects
Medical records of 53 patients with a diagnosis of ACHM (both complete and incomplete) who were followed at a national low vision center from January 2017 till December 2021 were retrospectivelly analyzed. The diagnosis was established by clinical examination and electrophysiology, and confirmed with molecular testing in all patients.
Visual acuity variation according to age (younger and older than 10 years) was analyzed in the whole group as well as in a sub-set of patients with repeated eye examinations with time. Subjects with long-term follow up who had exams in both age groups, were described separately in order to compare their visual acuity in two different time periods.
To further assess the effect of time on visual acuity, an additional sub-analysis of patients with multiple assessments was conducted. Visual acuity measurements were divided to 3 categories based on the patient's ages during the assessment: ages 0–5 years, 5–10 years, and 10–15 years, and the longitudinal analysis was performed among these age groups.
Patients with low vision secondary to retinal diseases other than ACHM were excluded from the study. The study was approved by the medical ethics committee of our tertiary hospital (approval number HMO-0046-22) and adhered to the tenets of the Declaration of Helsinki.
Clinical data
Clinical and genetic data was retrieved from the subject's medical charts. Available data of patients’ medical history, visual acuity, color vision, photophobia, strabismus, nystagmus, and refractive status were collected. Best correlated visual acuity (BCVA) was assessed with Snellen chart (LEA symbol in children), and the data of the best eye was taken.
Only pathogenic/likely pathogenic variants identified in known genes causing ACHM were included in the study.
Statistical analysis
Statistical analyses were performed using the Statistical Package for the Social Sciences software version 27 (SPSS Inc., Chicago, IL, USA). A P value of 0.05 or less was considered statistically significant. The comparison of a quantitative variable between two independent groups (eg. logMAR vs. two age groups) was performed by applying the two sample t-test or Mann-Whitney Test. The association between two categorical variables, (eg. gene type vs. nystagmus) was tested using the chi square test. The comparison of a quantitative variable between two dependent groups (eg. different visual acuity measurements of the same patient) was performed by applying the paired sample t-test or Friedman Test. In order to simultaneously assess the effect of time, gene type and the interaction between them on visual acuity the repeated measures Analysis of Variance model (ANOVA model) was applied.
Results
Fifty-three patients with a confirmed clinical diagnosis of achromatopsia were included in the study. Twenty-eight patients were male (52.8%) and 25 were female (47.2%). The mean age of the cohort was 14.28 years (SD 10.18, range 10 months – 46 years). As previously described, patients were divided into two age groups, with 20 patients (37.7%) aged < 10 and 33 patients (62.3%) aged ≥ 10.
A total of 53 patients had complete genetic testing: variants were identified in 39 subjects (73.6%) in CNGA3 gene and in another 14 patients (26.4%) in CNGB3 gene. No variants in GNAT2, PDE6C, PDE6H, ATF6 were observed. Assuming the prevalence of CNGA3 in the world (as described in literature) is ∼25%, 19 the prevalence of CNGA3 in our Israeli-Palestinian cohort is significantly higher, with a P value <0. 001 (chi-square test).
Clinical features
Fifty patients had available verbal vision tests. Mean BCVA was 0.92 logMAR (range 0.18–1.7, SD 0.24). All studied patients had a normal retina appearance on fundoscopy examination, as well as a conserved retinal morphology on OCT (in selected patients who performed it). Young children were not able to perform OCT due to poor cooperation and/or the presence of nystagmus. (Figure 1)
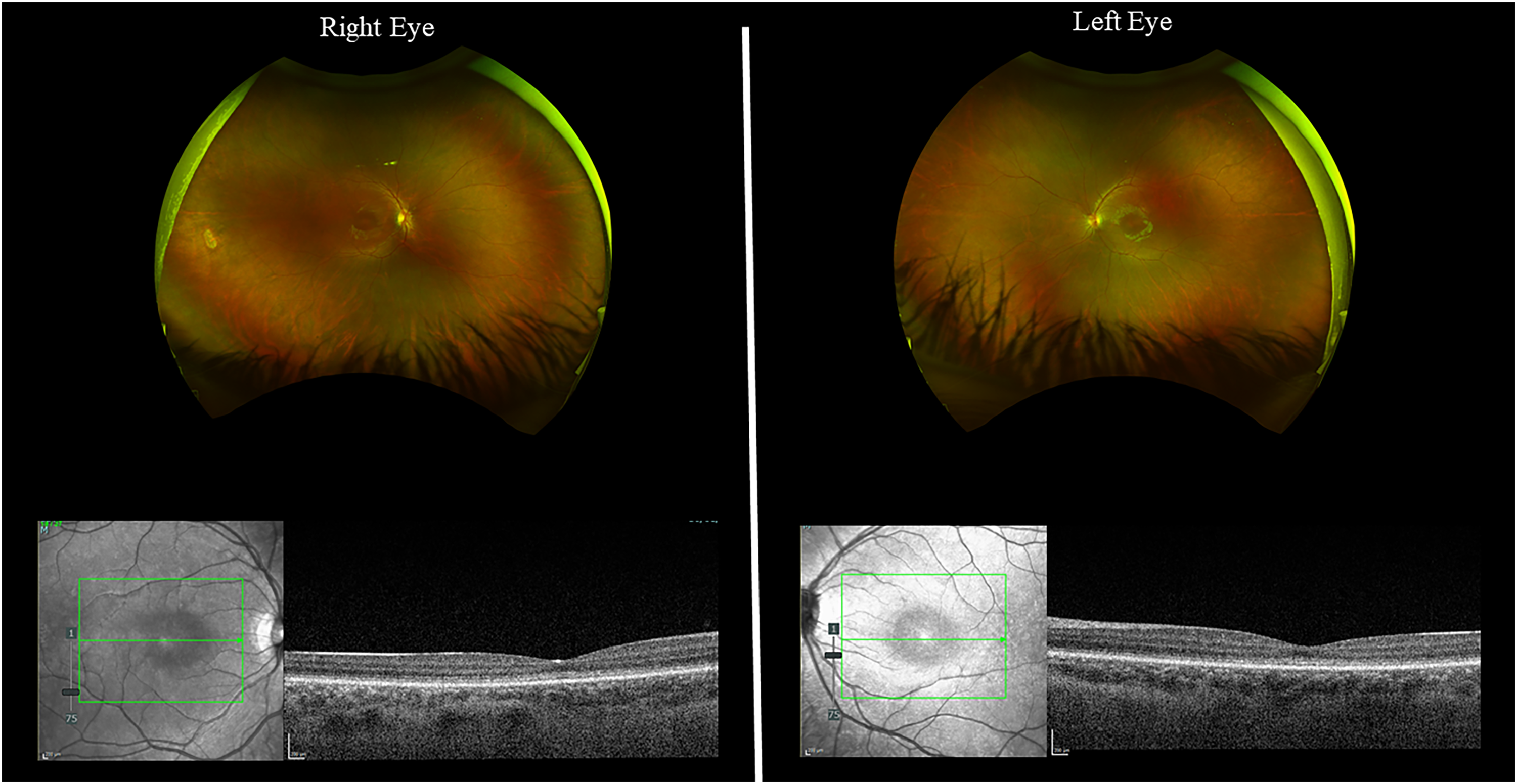
Color pictures of fundus and OCT of both eyes, showing normal retina appearance and morphology in one of the studied ACHM patients.
Most ACHM patients showed severe photophobia (96.2%) and nystagmus (93.6%). A total of 23 patients (51.1%) had strabismus, with exotropia (XT) being the most common (46.7%). Refractive errors ranged from high myopia to high hypermetropia (greater than + -4 diopters), with hypermetropia being the most common (72.3%). Three patients (5.7%) had high myopia (−4 or higher) and 15 patients (28.3%) had high hypermetropia (+4 or higher).
Phenotype of genes causing the disease
The mean BCVA in subjects with CNGA3 was 0.921 logMAR, and the mean BCVA in subjects with CNGB3 was 0.906 logMAR. There were no significant differences between the subjects with CNGA3 vs. CNGB3 mutations in terms of BCVA (p = 0.622), photophobia (p = 1.000), nystagmus (p = 1.000), strabismus (p = 0.739) and refractive error (p = 1.000) (Table 1).
Clinical phenotypes of the two main genes causing achromatopsia (CNGA3 & CNGB3).
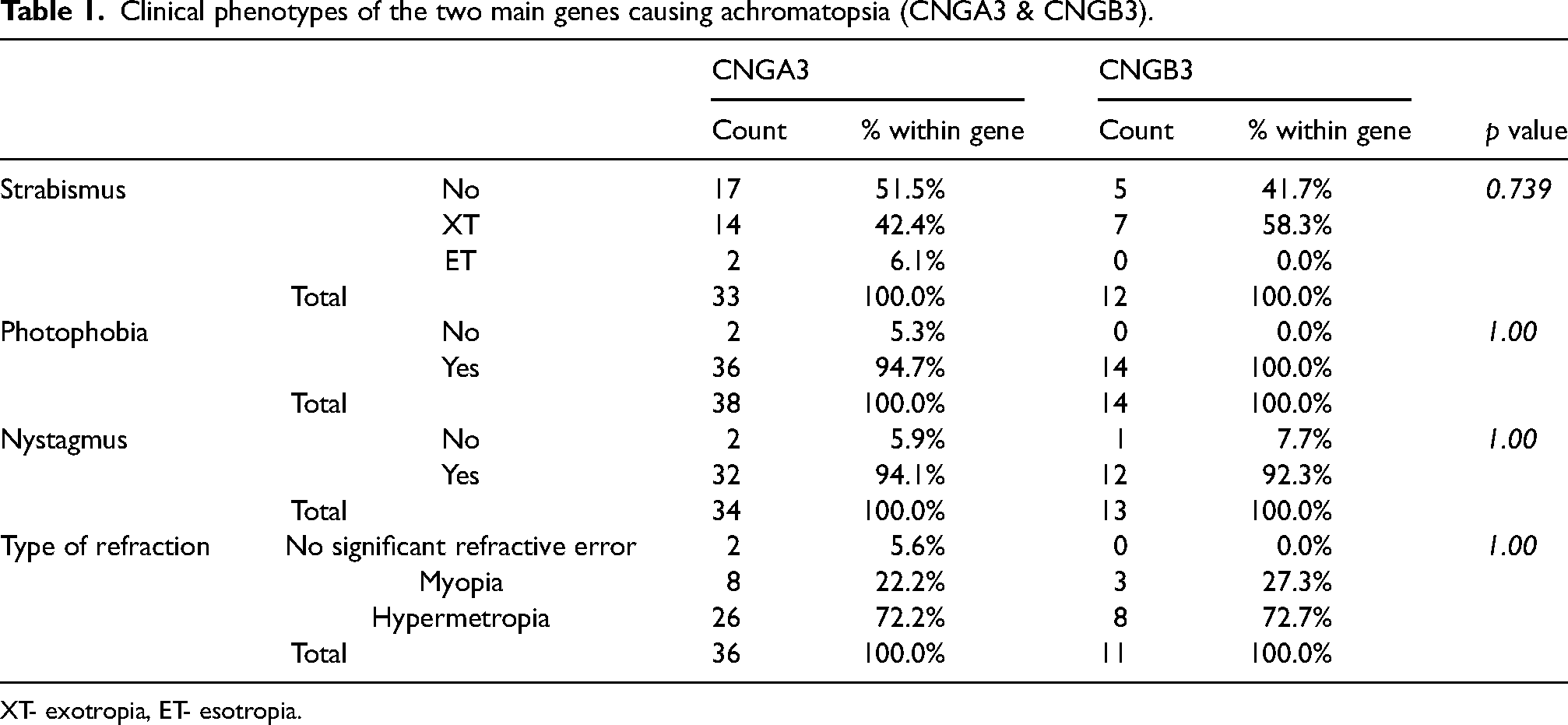
XT- exotropia, ET- esotropia.
Visual acuity according to age groups
Among young children, only those that were able to cooperate with vision tests (LEA symbols) with verbal visual acuities, were included. When comparing visual acuity of all 50 patients in two main age groups (younger or older than 10 years of age) using the t-test for independent samples, the differences were not statistically significant (p = 0.129) (Table 2).
BCVA measurements of all patients (50) at two age groups: age < 10 and age ≥10.

BCVA- visual acuity measurement of the best eye, (logMAR).
Table 3 shows further
BCVA measurements at age <10 and age >10, within the n = 18 long term follow-up cohort.
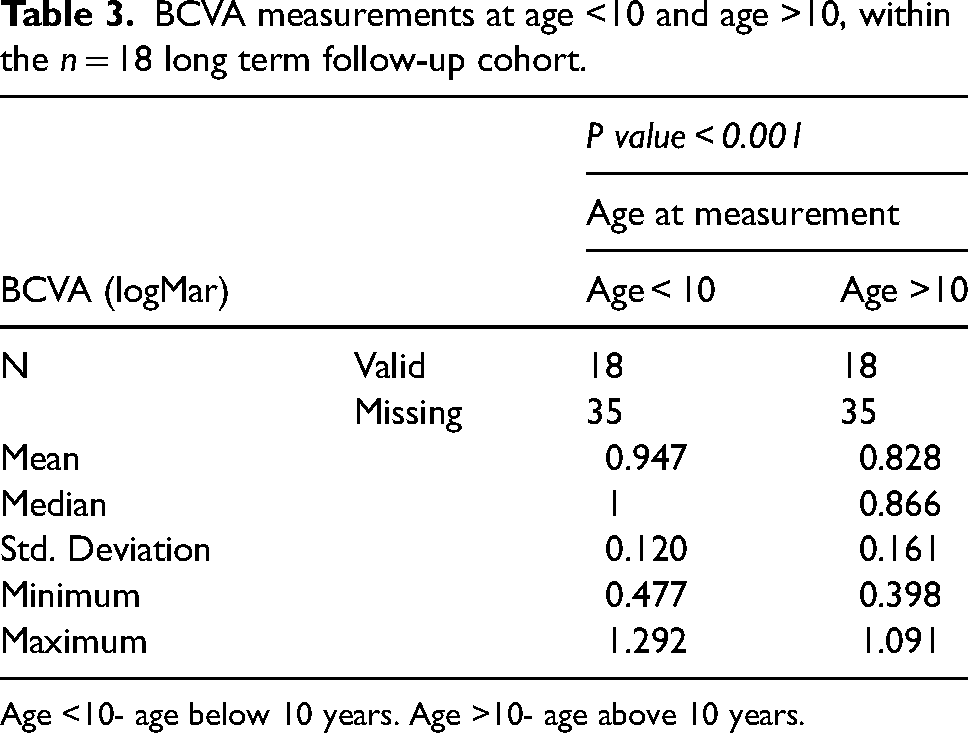
Age <10- age below 10 years. Age >10- age above 10 years.
Table 4 displays the longitudinal analysis of another sub-set of 10/18 patients with multiple BCVA measurements over three time periods (ages 0–5, > 5–10, and >10–15 years), with an average follow up of 8.83 years. Once more, among young children, only those that cooperated with vision tests (LEA symbols) were included. There was a statistically significant improvement of visual acuity over time (p = 0.001), with mean BCVA being 1.05 at age 0–5, 1.010 at age >5–10 and 0.831 at age >10–15. (Figure 2)
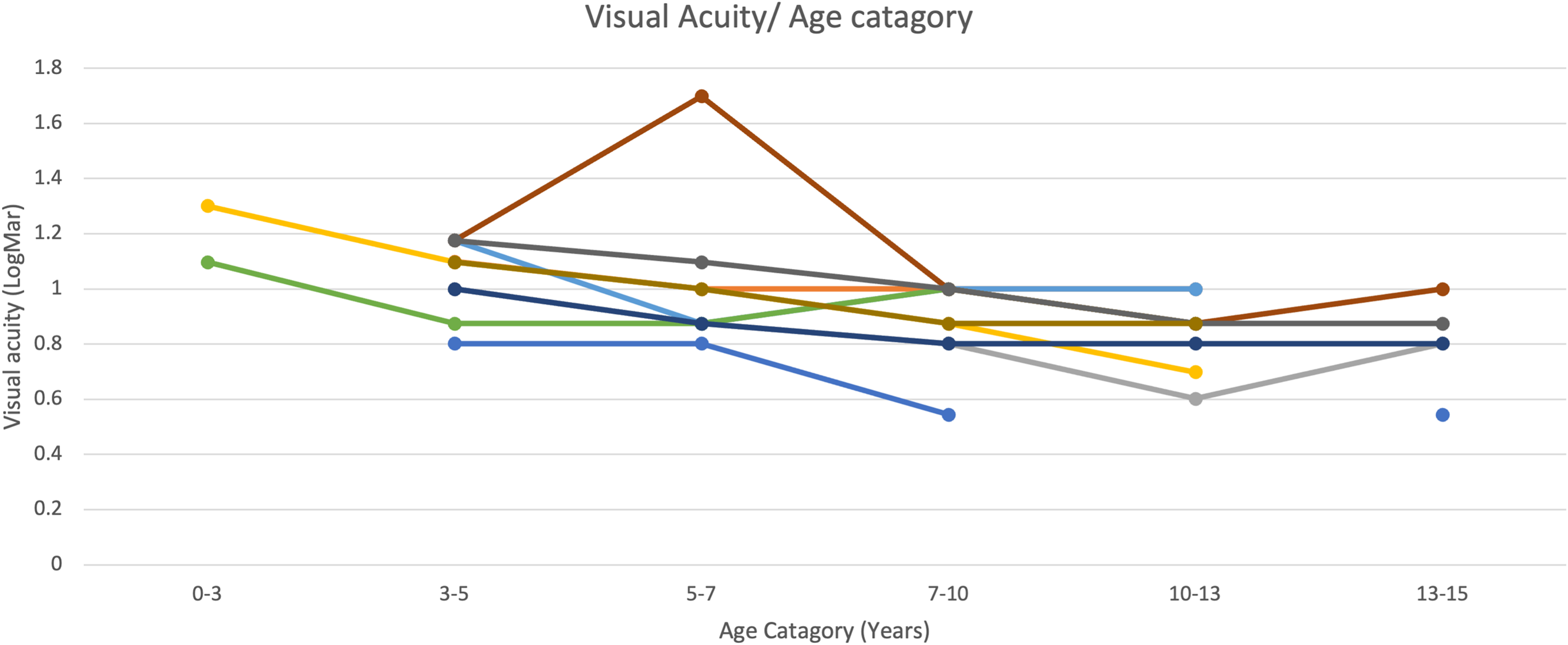
Longitudinal analysis of patient's visual acuity over time.
BCVA measurements at age 0–5, age >5–10, age >10–15, within the n = 10 cohort.
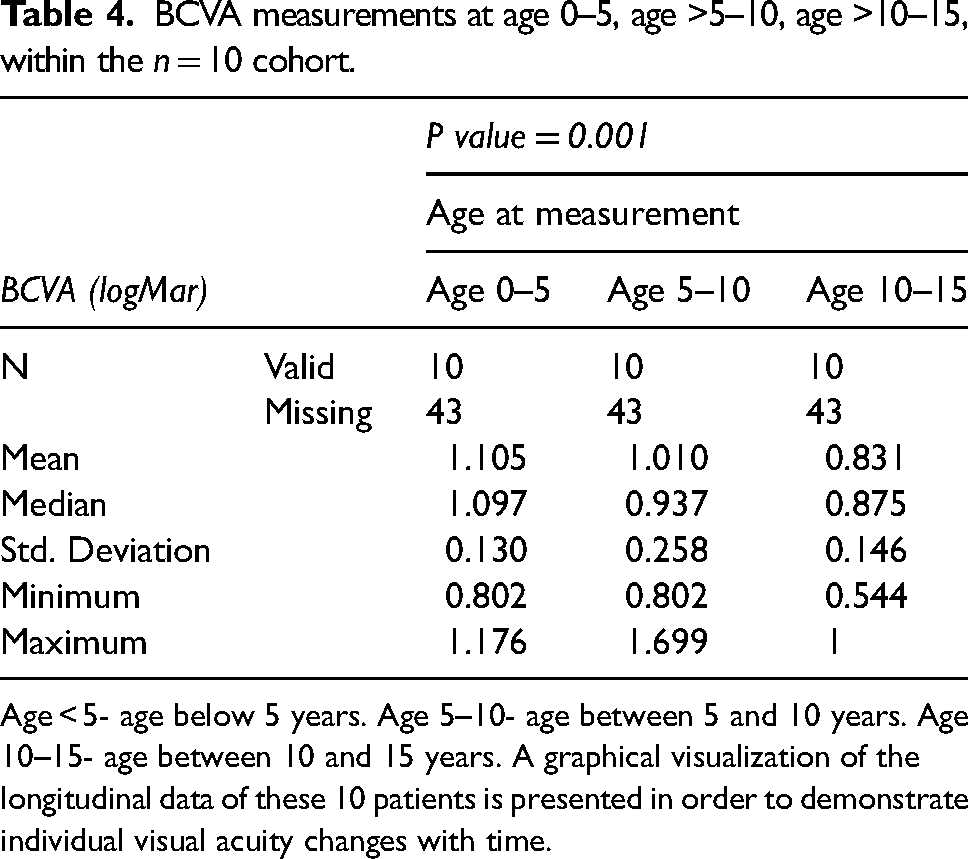
Age < 5- age below 5 years. Age 5–10- age between 5 and 10 years. Age 10–15- age between 10 and 15 years. A graphical visualization of the longitudinal data of these 10 patients is presented in order to demonstrate individual visual acuity changes with time.
ANOVA test was used to simultaneously assess the effect of both gene type and age on visual acuity. Eight patients had mutation in CNGA3 and 2 patietnts had mutations in CNGB3.There was no correlation with gene type (p = 0.314), but there was a statistically significant improvement of visual acuity with age (p = 0.011).
Discussion
Our study characterizes the clinical phenotype of ACHM in the Israeli-Palestinian population. According to our findings, ACHM in our population is mainly caused by a CNGA3 mutation, with a distribution of approximately 75%. This is significantly greater than the distribution of CNGA3 in the world, which is about 25%. 14
Main clinical characteristics found associated with ACHM were photophobia (96.2%) and nystagmus (93.6%). Strabismus was very common in the studied population reaching 51%, with exotropia being the most common type seen (46.7%). The main refractive error was hypermetropia (72.3%) with a significant rate of high hypermetropia (44.1%). The mean BCVA of the cohort was 0.92.
We also found no correlation between the main two genes causing disease and the clinical phenotype, including visual acuity, photophobia, strabismus, and nystagmus. These findings correlate with other studies that came to similar conclusions. 9
These characteristics are crucial for determination of long term visual function prognosis. Moreover, understanding the unique genetic distribution in our population may play an important key in genetic counseling and testing, as well as future family planning..
Our study is based on one of the largest cohorts so far for genetically confirmed ACHM clinical data, with an average follow-up of 8.49 years. Limitations of our study include the retrospective design and the investigation of solely functional biomarkers. We do not have enough retrospective data on OCT in order to compare findings on retina structure during the long-term follow up. The reason for the lack of OCTs is due to the combination of poor cooperation in young patients as well as the presence of nystagmus, that makes it very difficult to perform high quality OCT in these patients.
Questions about the progressive vs. stationary nature of achromatopsia have been raised throughout the years, with an ongoing debate about the disease's natural course. According to our findings, there is no deterioration of visual acuity with age. In fact, slight improvement over time was seen, as demonstrated in our sub- analysis with extended patient follow- up. This strengthens the presiding notion that ACHM is not a progressive disease. The observed marginal enhancement in visual acuity over time may be explained by the increased cooperation for subjective refraction in older patients reaching higher test accuracy and thus improved vision in adulthood. A study conducted by pan et al, 20 which examined 1722 healthy subjects, showed that visual acuity test performance improves with age in normal preschool children. They attributed this phenomenon to factors such as the maturation of attention, general cognitive abilities, or even the maturation of visual function itself. This trend corresponds closely with our own results, which also indicate a slight increase in visual acuity over time. Therefore, it is essential to emphasize that our key findings may not necessarily signify a consistent improvement but rather the absence of significant deterioration.
We acknowledge that the methods used to measure visual acuity between younger and older children were different (Lea in young children and Snellen in older children), but we believe this fact could not have affected our results based in the literature that shows good correlation of visual acuities values between these tests. A study conducted by Inal at el. 21 that compared visual acuity measurements in preschool children (including Lea symbols and Snellen) showed that there is no statistically significant difference between the two methods strengthening the validity of our results.
Gene therapy in ACHM is currently under investigation and has the potential to be a key treatment in the future. 12 The stationary nature of the disease could have an impact on a possible wider therapeutic window in which this treatment may be relevant and its efficacy.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Data was analyzed anonymously.
Submission
All listed authors have authorized the submission of this manuscript.
